Class: Antihistamine
Dosage Forms. Tablet: 30 mg, 60 mg, 180 mg; Disintegrating Tablet: 30 mg; Suspension: 30 mg/5 mL
Common FDA Label Indication, Dosing, and Titration.
1. Seasonal allergic rhinitis: Children 2-11 y, 30 mg po bid; Children 12 y and older and Adults, 60 mg po bid or 180 mg po daily
2. Idiopathic urticaria: Children 6 mo-2 y, 15 mg po bid; Children 2-11 y, 30 mg po bid; Children 12 y of age and older and Adults, 60 mg po bid or 180 mg po daily
Off-Label Uses.
1. Perennial allergic rhinitis: Children 2-11 y, 30 mg po bid; Children 12 y and older and Adults, 60 mg po bid or 180 mg po daily
MOA. Fexofenadine, the major active metabolite of terfenadine, is an antihistamine with selective peripheral H1-receptor antagonist activity. Both enantiomers of fexofenadine displayed approximately equipotent antihistaminic effects.
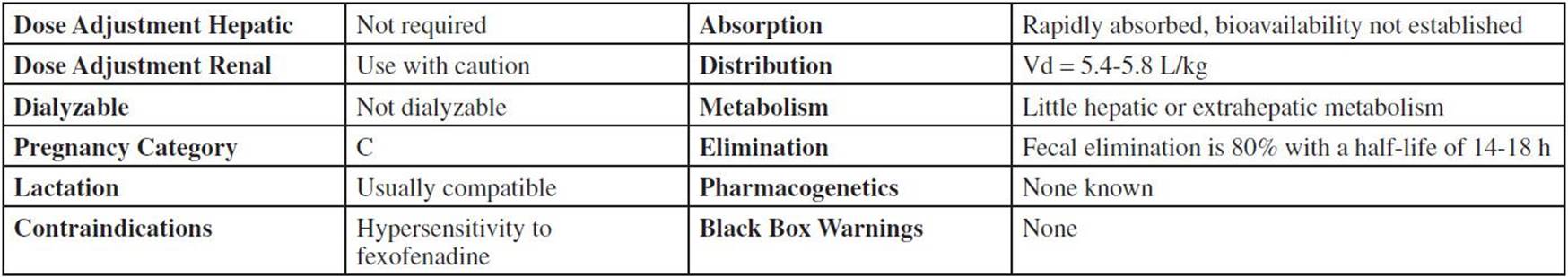
Drug Characteristics: Fexofenadine
Medication Safety Issues: Fexofenadine

Drug Interactions: Fexofenadine

Adverse Reactions: Fexofenadine

Efficacy Monitoring Parameters. Improvement in rhinitis or urticaria symptoms.
Toxicity Monitoring Parameters. Seek medical attention for signs of severe CNS toxicity.
Key Patient Counseling Points. Patients should avoid activities requiring mental alertness or coordination until drug effects are known, as drug may cause dizziness or sedative effects.
Clinical Pearls. Product is available OTC in several additional dosage forms.