Class: Oral Contraceptive
Product Contents: Oral Contraceptive—Monophasic
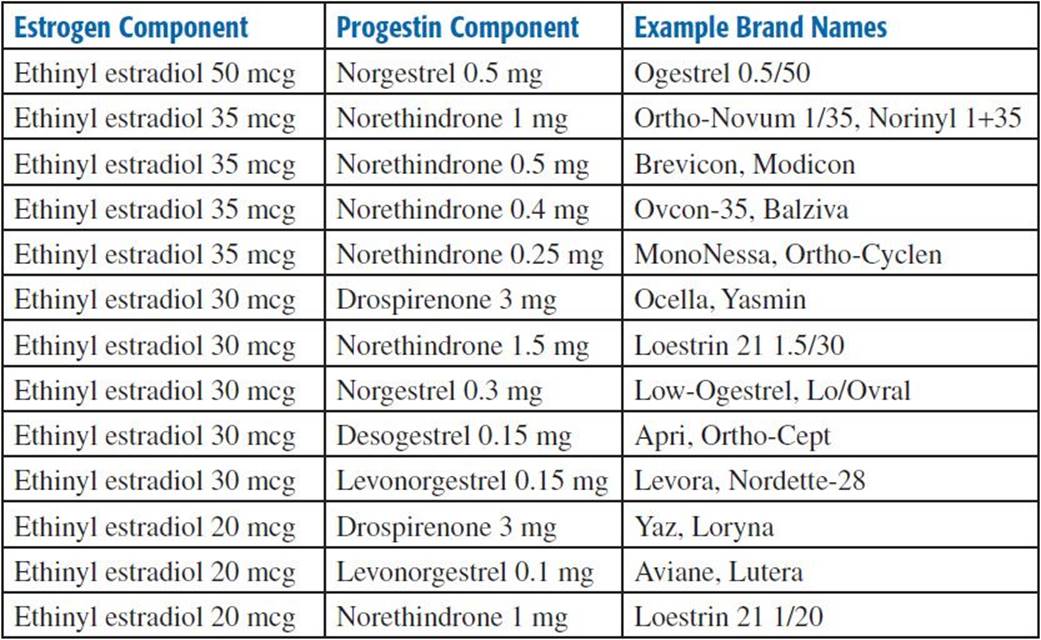
Dosage Forms. Tablet: Monophasic products include tablets that each contains the same dose of an estrogen and progestin agent; products are either in 21-d or in 28-d cycles; may also include inert tablets containing either plain lactose or iron supplements, generally as 75-mg ferrous fumarate
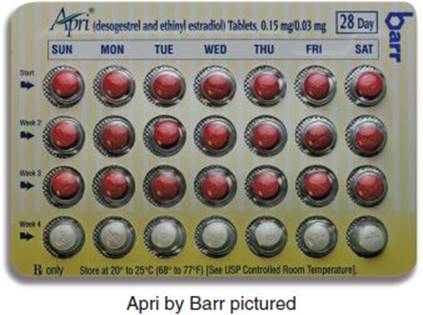
Common FDA Label Indication, Dosing, and Titration.
1. Contraception: 1 tablet po daily beginning either on the first Sunday after menstruation begins (“Sunday start”) or on the first day of menstruation (“day 1 start”); tablets are taken sequentially, following the arrows marked on the dispenser
Off-Label Uses. None
MOA. See Preface C Card: General Content Related to All Oral Contraceptives.
Drug Characteristics: Oral Contraceptive—Monophasica
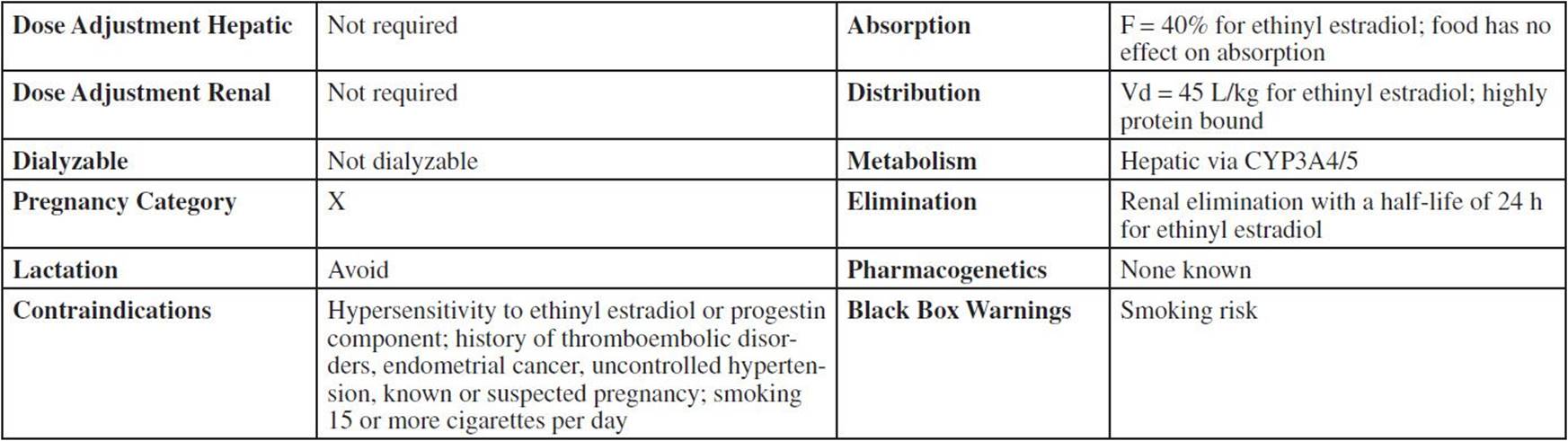
aSee Preface C Card: General Content Related to All Oral Contraceptives for ADME data on progestins.
Medication Safety Issues: Oral Contraceptive—Monophasic

Drug Interactions and Adverse Reactions: Oral Contraceptive—Monophasic. See Preface C Card: General Content Related to All Oral Contraceptives.
Efficacy Monitoring Parameters. Lack of pregnancy.
Toxicity Monitoring Parameters. Annual physical examination including cervical cytology (Pap smear) and breast exam.
Key Patient Counseling Points. See Preface C Card: General Content Related to All Oral Contraceptives.
Clinical Pearls. Patients should not smoke during therapy, as this increases the risk of serious cardiovascular side effects. Noncontraceptive benefits (non-FDA approved) of oral contraceptive use include treatment of dysmenorrhea; acne; menstrual migraine; pelvic pain due to endometriosis; and decreased risk of endometrial, ovarian, and colorectal cancer. Multiphasic products have a lower total steroid dose than monophasic products and may have lower adverse effect rates, but treatment is usually initiated with monophasic products. Multiple nonoral hormonal contraceptive products also available.