The hypothalamic hormones and their related drugs are discussed in relation to the anterior pituitary hormones that they modulate.
Table 16.1 lists the second messengers used by the hormones discussed in this unit.
|
Table 16.1 |
||
|
Hormones |
Second Messenger |
|
|
CRH, ACTH, LH, FSH, TSH, PTH, calcitonin, glucagon, ADH (V2 receptors), hCG |
↑ Cyclic AMP |
|
|
Prolactin |
↓ Cyclic AMP |
|
|
GHRH, GnRH, TRH, ADH (V1 receptors), oxytocin |
DAG and IP3 |
|
|
Cortisol, aldosterone, testosterone, estrogen, progesterone, calcitriol (vitamin D), thyroid hormones (T3 and T4) |
Steroid mechanism |
|
|
NO |
Cyclic GMP |
|
|
Insulin and IGF-1 |
Tyrosine kinase |
|
|
Abbreviations: ACTH, adrenocorticotropic hormone; ADH, antidiuretic hormone; AMP, adenosine monophosphate; CRH, corticotropin-releasing hormone; DAG, diacylglycerol; FSH, follicle-stimulating hormone; GHRH, growth hormone-releasing hormone; GMP, guanosine monophosphate; GnRH, gonadotropin-releasing hormone; hCG, human chorionic gonadotropin; IGF-1, insulinlike growth factor; IP, inositol triphosphate; LH, luteinizing hormone; NO, nitric oxide; PTH, parathyroid hormone; TRH, thyrotropin-releasing hormone; TSH, thyroid-stimulating hormone. |
||
16.1 Pituitary Hormones
Anterior pituitary hormones are secreted or inhibited in response to the action of releasing hormones or inhibiting hormones from the hypothalamus. Posterior pituitary hormones are synthesized in the hypothalamus, stored in the posterior pituitary, and secreted in response to direct neural stimulation (Fig. 16.1). Table 16.2 summarizes the hormonal cascade from the hypothalamus to target organs or the posterior pituitary.
Hormone receptors
Hormones that are hydrophilic cannot penetrate the plasma membrane; they therefore interact with membrane receptors and exert their effects via second messenger molecules (e.g., cyclic adenosine mono-phosphate [cAMP], phospholipase C, and Ca2+. Examples of hydrophilic hormones are insulin and adrenocorticotropic hormone (ACTH). Conversely, hormones that are lipophilic (e.g., cortisol and estrogen) are able to pass through the cell membrane and interact with receptors in the cytoplasm or nucleus. In this case, receptor binding alters the gene transcription of proteins to exert the hormones’ physiological effects.
|
Table 16.2 |
||
|
Hypothalamic Hormone |
Anterior Pituitary Hormone |
Target Organ(s) |
|
GHRH |
↑ GH |
Liver, skeletal muscle, and bone |
|
Somatostatin |
↓ GH |
Liver, GI tract, and pancreas |
|
CRH |
ACTH |
Adrenal cortex |
|
GnRH |
LH and FSH |
Gonads |
|
TRH |
TSH |
Thyroid gland |
|
PIH (dopamine) |
Prolactin |
Mammary glands and gonads |
|
Posterior Pituitary Hormones |
||
|
Oxytocin |
Uterine and other smooth muscle |
|
|
ADH (vasopressin) |
Kidney tubules (mainly); also vascular smooth muscle, liver, and anterior pituitary gland |
|
|
Abbreviations: ACTH, adrenocorticotropic hormone; ADH, antidiuretic hormone; CRH, corticotropin-releasing hormone; FSH, follicle-stimulating hormone; GH, growth hormone; GHRH, growth hormone–releasing hormone; GI, gastrointestinal; GnRH, gonadotropin-releasing hormone; LH, luteinizing hormone; PIH, prolactin-inhibiting hormone; TRH, thyrotropin-releasing hormone; TSH, thyroid-stimulating hormone. |
||
Fig. 16.1 ![]() Hypothalamic and pituitary hormones.
Hypothalamic and pituitary hormones.
Hormones released from the hypothalamus stimulate or inhibit the release of hormones from the anterior pituitary. The pituitary hormones then travel to target tissues, where they exert their physiological effects. Posterior pituitary hormones are synthesized in the hypothalamus and stored in the posterior pituitary until release is stimulated by an action potential. These hormones then travel in the blood to their target tissues. (ACTH, adrenocorticotropic hormone; ADH, antidiuretic hormone; CRH, corticotropin-releasing hormone; FSH, follicle-stimulating hormone; GH, growth hormone; GnRH, gonadotropin-releasing hormone; GHRH, growth hormone–releasing hormone; GRIH, growth hormone-inhibiting hormone (somatostatin); LH, luteinizing hormone; PIH, prolactin inhibiting hormone; TRH, thyrotropin-releasing hormone; TSH, thyroid-stimulating hormone.)

Negative feedback
Negative feedback is when the output of a pathway inhibits inputs to a pathway. This physiological phenomenon is particularly evident in the endocrine system and is an important mechanism in homeostasis.
Growth Hormone
Hypothalamic regulation. Growth hormone release from the anterior pituitary is controlled by two hormones released from the hypothalamus:
– Growth hormone–releasing hormone (GHRH) increases the synthesis and secretion of growth hormone (GH).
– Somatostatin (growth hormone–inhibiting hormone [GHIH]) decreases the sensitivity of the anterior pituitary to GHRH, thus decreasing levels of GH.
Growth hormone receptors
The receptors for GH are found in peripheral tissues and belong to the class 1 cytokine receptor superfamily. These are receptor-associated tyrosine kinase receptors that, upon binding of GH, dimerize and activate the JAK–STAT (Janus kinase–signal transducer and activator of transcription) pathway. The JAK–STAT pathway is a signaling alternative to the second messenger systems. Its activation ultimately causes DNA transcription within the cell.
Effects. See Table 16.3.
|
Table 16.3 |
||
|
Effects Mediated by GH |
Effects Mediated by Somatomedins |
|
|
↑ somatomedin synthesis* ↑ gluconeogenesis ↑ lipolysis ↑ protein synthesis ↑ amino acid uptake in the gut ↓ insulin (causing ↓ glucose uptake into cells) |
↑ protein synthesis resulting in the following effects: ↑ muscle mass ↑ cartilage growth (this causes linear growth) ↑ growth of the internal organs |
|
|
* Somatomedins are insulinlike growth factors that are intermediaries for some GH actions. Many of the actions of GH occur in association with cortisol. |
||
Factors affecting growth hormone secretion
The major stimuli for GH secretion are deep sleep, hypoglycemia, stress, GHRH, and metabolites (e.g., amino acids and free fatty acids). Secondary stimuli include exercise, glucagon, antidiuretic hormone (ADH), opioids, and pyrogens (fever-inducing substances). GH secretion is inhibited by hyperglycemia and cortisol secretion.
Growth hormone and nitrogen balance
GH creates a positive nitrogen balance in the body. This is mainly due to an increased rate of lipolysis, which provides the energy the body needs while sparing proteins and glucose. Diseases in which there is a negative nitrogen balance, such as acquired immunodeficiency syndrome (AIDS), cachexia (loss of lean body mass that cannot be corrected with increased calorific intake), trauma, and severe burns, can be treated with GH to improve lean body mass and wound healing.
Growth at epiphyseal plates
The epiphyseal plates consist of hyaline cartilage at the end of long bones. Chondrocytes in the epiphyseal plates are constantly undergoing mitosis throughout childhood and adolescence, but ceasing in adulthood. The older cells (at the diaphysis end) are then ossified by osteoblasts. This progressive laying down of bone leads to longitudinal growth. GH acts to increase the mitosis of chondrocytes in the epiphyseal plates..
Disorders
– Deficiency of GH causes short stature with normal body proportions.
– Excess GH leads to gigantism in children and acromegaly in adults.
Somatotropin
Mechanism of action. Somatotropin is a recombinant form of GH.
Uses
– GH deficiencies in children prior to epiphyseal plate closure (complete ossification)
Side Effects
– Peripheral edema
– Localized muscle pain and weakness
– Carpal tunnel syndrome
Contraindications
– Closed epiphyses
– Active neoplasia
Carpal tunnel syndrome
Carpal tunnel syndrome is a condition caused by compression of the median nerve within the carpal tunnel of the wrist. It may be caused by anything that produces soft tissue swelling, e.g., pregnancy and rheumatoid arthritis. Symptoms include burning pain, tingling, and numbness in the hand, especially in the thumb, index, middle, and radial half of the ring finger but not the little finger. This pain may be relieved by placing the hand in cold water or by patients “shaking out” their hands. There may also be muscle weakness and wasting. Treatment includes wrist splints, nonsteroidal antiinflammatory drugs (NSAIDs), and corticosteroids (hydrocortisone). Surgical decompression may be necessary if other treatments are ineffective.
Octreotide and Lanreotide
Mechanism of action. Octreotide and lanreotide are cyclic peptide analogues of the biologically active portion of somatostatin.
Effects
– These agents mimic the actions of somatostatin to inhibit GH secretion from the anterior pituitary.
– They also inhibit GH secretion from tumors.
Uses
– Acromegaly
– Carcinoid syndrome and vasoactive intestinal polypeptide (VIP)–secreting tumors (octreotide).
Side effects. Gastrointestinal (GI) side effects, including nausea, diarrhea, and steatorrhea (excess fat in stools), are seen in a majority of patients with acromegaly treated with octreotide. Cholelithiasis (gallstones) is observed in one third of patients with use of octreotide for 6 months or more.
Vasoactive intestinal peptide
Vasoactive intestinal peptide (VIP) is a neurocrine peptide secreted by neurons in the mucosa and smooth muscle of the GI tract in response to distention of the stomach and small intestines and vagal activity. It acts to reduce lower esophageal sphincter tone, relax the proximal muscles of the stomach, allowing for entrance of food (“r eceptive relaxation”), and to increase water and electrolyte secretion in intestine.
Pegvisomant
Mechanism of action. Pegvisomant is a GH receptor antagonist that blocks the effects of endogenous GH.
Uses
– Acromegaly in patients unresponsive to surgery, radiation, or octreotide
Adrenocorticotropic Hormone
Hypothalamic regulation. Corticotropin-releasing hormone (CRH) from the hypothalamus stimulates the release of adrenocorticotropic hormone (ACTH) from the anterior pituitary.
Effects. The primary target of ACTH is the MC2 subtype of melanocortin receptor, a G protein– coupled receptor expressed primarily by cells of the adrenal cortex. The MC2 receptor activates Gs to increase intracellular levels of cyclic AMP, which stimulates the synthesis of corticosteroids, including glucocorticoids, mineralocorticoids, and androgens.
Disorders
– ACTH deficiency causes secondary adrenal insufficiency. This is characterized by fatigue, weakness, anorexia, nausea, and vomiting.
– ACTH excess leads to Cushing syndrome.
Cushing syndrome
Cushing syndrome is a group of signs and symptoms that occur due to high levels of cortisol in the blood. It can be caused by corticosteroid (and ACTH) administration, pituitary adenomas, adrenal gland adenomas/carcinomas, and by excessive intake of alcohol. Signs and symptoms typically include weight gain, particularly to the trunk, with sparing of the limbs; moon face (or moon facies); “buffalo hump” (due to fat deposition on the back); purple striae, especially on the abdomen; sweating; thin skin; and hirsuitism. Treatment depends on the cause.
Corticorelin
Mechanism of action. Corticorelin is bovine CRH that acts like the natural hormone.
Uses. Corticorelin is used to differentiate pituitary ACTH-dependent Cushing disease from ectopic ACTH-secreting tumors (Cushing syndrome). Patients with Cushing disease show normal to increased plasma ACTH and cortisol response, whereas ectopic tumors do not.
Adrenocorticotropic Hormone and Cosyntropin
Mechanism of action. Cosyntropin is a synthetic peptide consisting of the first 24 amino acids of ACTH. It acts like the natural hormone and is used similarly.
Uses. These agents are used for the differential diagnosis of primary versus secondary adrenal insufficiency. If there is primary adrenal insufficiency, there will be no response to ACTH; however, if there is secondary adrenal insufficiency due to inadequate ACTH release from the pituitary, administered ACTH will increase plasma glucocorticoids.
Primary and secondary adrenal insufficiency
Primary adrenal insufficiency occurs when there is impairment or destruction of the adrenal glands. The cause of this may be idiopathic (unknown) or it may be due to autoimmune disease (e.g., Addison's disease), adrenal hyperplasia, or adenoma. Secondary adrenal insufficiency occurs when there is inadequate ACTH secretion from the pituitary to stimulate adrenal hormone production.
Gonadotropins (Follicle-stimulating Hormone and Luteinizing Hormone)
Hypothalamic regulation. Gonadotropin-releasing hormone (GnRH) released from the hypothalamus stimulates the release of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) from the anterior pituitary.
Menstrual cycle
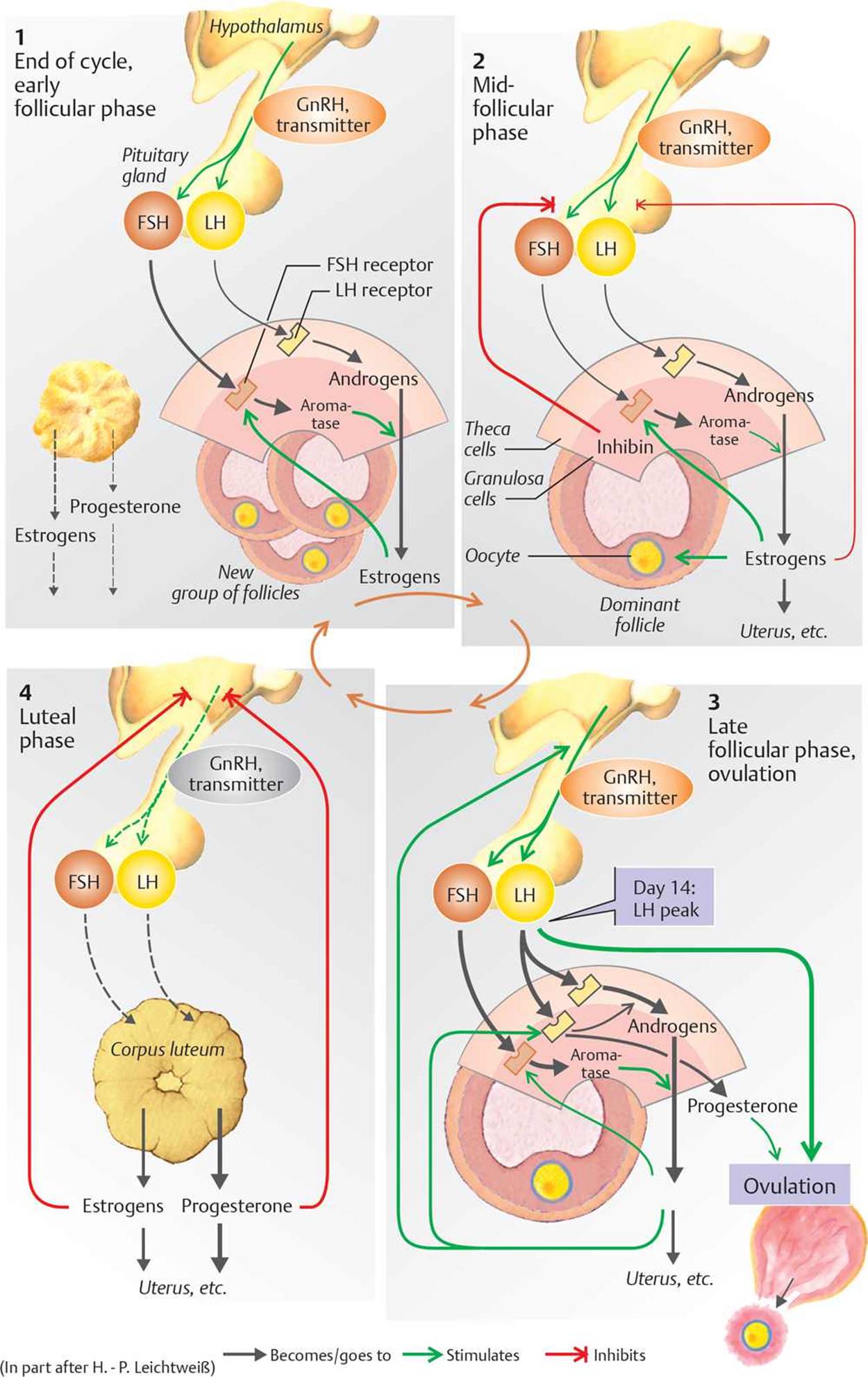
The menstrual cycle varies in length from 21 to 35 days. The first half of the cycle is the follicular phase, which begins with menstruation. Following menstruation, in the early follicular phase, FSH induces the production of ~20 follicles. Small amounts of LH are also secreted. Both FSH and LH stimulate enzymes that catalyze the production of androgens that are needed for estrogen synthesis. Estrogens released from follicles cause upregulation of FSH receptors, so the follicle with the highest estrogen content is most sensitive to FSH. This follicle becomes the dominant (graafian) follicle in which an ovum develops. The remaining follicles containing oocytes undergo atresia. In the midfollicular phase, the follicular cells also start to produce progesterone, which causes progressive thickening of the endometrium. In the late follicular phase, increased quantities of FSH and LH are secreted once again. This causes more androgen and estrogen production, which positively feeds back to the hypothalamus, causing an increase in LH. This rapid rise in LH concentration (LH surge) induces ovulation. Just after ovulation, the basal body temperature rises and stays elevated until the end of the cycle. The second half of the cycle is the luteal phase. LH, FSH, and estrogen transform the follicle into a corpus luteum, which secretes large quantities of progesterone, causing further endometrial thickening. If fertilization of the ovum has not occurred, estrogen and progesterone now inhibit FSH and LH both directly and via negative feedback on the hypothalamus and anterior pituitary. This causes a marked drop in plasma estrogen and progesterone concentration, causing constriction of endometrial blood vessels and discharge of the endometrium (menses).
Effects
– In men, LH stimulates testosterone production by the Leydig cells of the testes. FSH stimulates the Sertoli cells and is critical for maturation of spermatozoa.
– In women, LH stimulates estrogen and progesterone production by the ovaries. FSH stimulates the development of the ovarian follicle (Fig. 16.2).
Fig. 16.2 ![]() Hormonal control of the menstrual cycle.
Hormonal control of the menstrual cycle.
Follicle-stimulating hormone (FSH) and luteinizing hormone (LH) act in the follicular phase to cause follicular growth, estrogen production (causing the selection of a dominant follicle in which the ovum develops), progesterone production (which is primarily responsible for endometrial thickening), and ovulation. Estrogens and progesterone produced in the luteal phase induce endometrial thickening necessary for implantation of the ovum. If this does not occur, feedback inhibition of FSH and LH triggers menstruation.
Leuprolide, Goserelin, and Buserelin
Mechanism of action. Leuprolide, goserelin, and buserelin are GnRH agonist analogues. Chronic treatment with one of these agents produces desensitization of the normal response to GnRH. After an initial surge of LH and FSH, the secretion of these hormones decreases.
Pharmacokinetics. These agents are administered intramuscularly in depot form or inhaled intranasally.
Uses
– Endometriosis in women
– Prostatic carcinoma in men
Side effects. These agents may cause bone loss through a prolonged hypoestrogenic state.
Endometriosis
Endometriosis is a condition in which endometrial tissue that normally lines the uterus grows outside the uterus. The displaced endometrial tissue (or “implants”) responds to FSH and LH during the menstrual cycle as normal endometrial tissue does, causing thickening and then breakdown and bleeding. Surrounding tissue eventually becomes inflamed leading to fibrosis. Symptoms include dysmenorrhea (painful periods); pain during intercourse, bowel movements, or urination; and infertility (usually due to obstruction of the fallopian tubes by scar tissue). Treatment involves the use of NSAIDs for pain, contraceptive hormones to control endometrial buildup, gonadotropin-releasing hormone agonists and antagonists to block the production of FSH and LH, and aromatase inhibitors that block estrogen production. Surgery may be required to remove endometrial implants or hysterectomy in cases of severe endometriosis.
Ganirelix and Cetrorelix
Mechanism of action. Ganirelix and cetrorelix are competitive antagonists of GnRH.
Uses. These agents are used to suppress the LH surge and prevent premature follicular luteinization in ovarian-stimulation protocols.
Human Chorionic Gonadotropin and Human Menopausal Gonadotropin (hMG)
Human chorionic gonadotropin (hCG) and human menopausal gonadotropin (hMG) are discussed further in the ovulatory agents section on pages 165 and 166.
Mechanism of action. Both hCG and hMG mimic the effects of gonadotropins.
Pharmacokinetics
– Administered parenterally
Uses
– Used to promote fertility
Ovarian hyperstimulation syndrome
Ovarian hyperstimulation syndrome occurs when gonadotropins, such as hCG, are given to stimulate ovulation. In ~10 to 25% of patients who are given these drugs parenterally, the ovaries are overstimulated, and fluid leaks from them into the belly and chest following ovulation. Most of the time this produces mild symptoms, such as nausea, vomiting, diarrhea, mild abdominal pain, bloating, and weight gain. However, in a small percentage of cases, these symptoms can be more severe, and there may be additional symptoms, such as shortness of breath (dyspnea), blood clots, electrolyte disturbance, and kidney failure. No treatment may be required if mild, but hospitalization for fluid replacement and management of any serious complications may be necessary for severe cases.
Thyroid-stimulating Hormone (Thyrotropin)
Hypothalamic control. Thyrotropin-releasing hormone (TRH) is a tripeptide released from the hypothalamus that stimulates thyroid-stimulating hormone (TSH) synthesis and release in the anterior pituitary.
Origins of the thyroid gland
The thyroid gland is formed from the pharyngeal arches. During embryonic development, it descends from the foramen cecum of the forming tongue, through the thyroglossal duct, until it reaches its final location in the neck, surrounding the trachea.
Effects. TSH stimulates the release of thyroid hormones via several mechanisms (see Table 16.4).
|
Table 16.4 |
||
|
↑ sensitivity of TSH receptors to TSH ↑ thyroglobulin synthesis ↑ thyroid peroxidase and glucose oxidase levels, which increase the iodination of thyroglobulin ↑ activity of the iodide pump ↑ Na+-K+ ATPase activity, which increases the capacity for iodide intake ↑ T3 formation relative to T4 under acute increases in metabolic demand |
||
Disorders
– TSH deficiency causes secondary hypothyroidism. Symptoms generally mimic those of primary hypothyroidism but are less severe.
– Excess TSH secretion is characterized by goiter and hyperthyroidism.
Protirelin and Thyrotropin
Mechanism of action. Protirelin is a synthetic peptide identical to TSH. Protirelin and thyrotropin act like the natural hormone.
Uses
– Diagnosis of hypothyroid states (protirelin)
– Given intramuscularly to stimulate iodine 131 (I131) uptake in the treatment of metastatic thyroid carcinoma, but its diagnostic use has largely been replaced by TRH.
Prolactin
Hypothalmic regulation. Dopamine (prolactin-inhibiting hormone [PIH]) released from the hypothalamus binds to the D2 subtype of dopamine receptor in the anterior pituitary, which is coupled to Gi and leads to inhibition of adenylate cyclase. This, in turn, causes tonic inhibition of the release of prolactin from the anterior pituitary.
Effects. Prolactin stimulates the mammary glands to produce milk in the postpartum period.
Disorders. Loss of PIH after hypothalamic destruction is associated with hypersecretion of prolactin. This causes amenorrhea or galactorrhea.
Cabergoline and Bromocriptine
Mechanism of action. Cabergoline and bromocriptine are dopamine (PIH) analogue agonists at the D2 dopamine receptor.
Pharmacokinetics. Cabergoline has a much longer duration of action (7–14 days) compared with bromocriptine (1–2 days).
Uses
– Prolactin-secreting adenomas
– Amenorrhea or galactorrhea
– Suppression of physiological lactation
Antidiuretic Hormone and Oxytocin
Antidiuretic hormone ([ADH], or vasopressin) and oxytocin are synthesized in the hypothalamus and are transported to the posterior pituitary, where they are stored. Both are peptide hormones consisting of nine amino acids. They differ only in the amino acids at positions 3 and 8, and both have a short half-life (15−30 min) once released into the systemic circulation. Because of their chemical similarities, ADH has slight oxytocic activity, and oxytocin has slight antidiuretic activity. However, oxytocin has no vasoconstricting activity.
See Chapter 17 for a further discussion of oxytocin and Chapter 19 for a discussion of ADH.
Stimuli for ADH synthesis and secretion
ADH is synthesized and released in response to increased plasma osmolality (detected by hypothalamic osmoreceptors), e.g., dehydration; decreased plasma volume (detected by peripheral mechanoreceptors), e.g., hemorrhage (hypovolemia); and decreased blood pressure (detected by baroreceptors). Its release is most sensitive to plasma osmolality, yet larger quantities of ADH are released in response to changes in blood pressure and blood volume.
Table 16.5 summarizes the drugs affecting hypothalamic and pituitary hormone levels.
|
Table 16.5 |
||
|
Drug |
Mechanism |
|
|
Agonists |
||
|
Octreotide, lantreotide |
Somatostatin analogues |
|
|
Somatotropin |
GH analogue |
|
|
Corticorelin |
CRH analogue |
|
|
ACTH, Cosyntropin |
ACTH analogue |
|
|
hCG, hMG |
Mimics gonadotropins |
|
|
Protirelin, thyrotropin |
TSH analogues |
|
|
Cabergoline, bromocriptine |
Dopamine (PIH) analogues |
|
|
Antagonists |
||
|
Pegvisomant |
GH receptor antagonist |
|
|
Ganirelix, cetrorelix |
Competitive inhibitors of GnRH |
|
|
Abbreviations: ACTH, adrenocorticotropic hormone; hCG, human chorionic gonadotropin; hMG, human menopausal gonadotropin. |
||
16.2 Thyroid Hormones
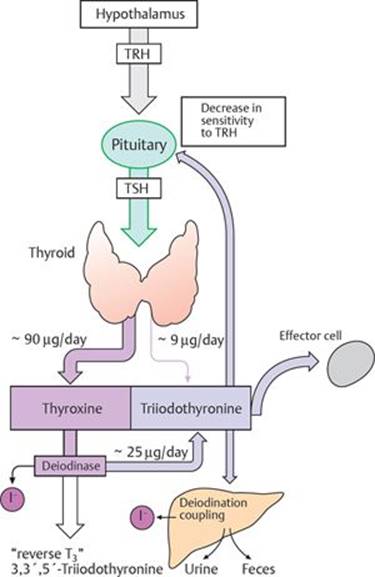
The natural thyroid hormones produced by the thyroid gland are thyroxine (T4) and triiodothyronine (T3). The mechanism of their release and degradation is shown in Fig. 16.3.
Fig. 16.3 ![]() Thyroid hormones: release, effects, and degradation.
Thyroid hormones: release, effects, and degradation.
TRH from the hypothalamus causes TSH release from the anterior pituitary. The thyroid secretes the hormones thyroxine (T4) and triiodothyronine (T3). T4 is produced in higher quantities than T3, but T3 is the more active form. T4 is converted to T3 in tissues. T3 attaches to effector cell receptors, producing its physiological effects. It also causes feedback inhibition of TSH production.
Thyroid hormone synthesis
The follicles of the thyroid gland synthesize and store thyroglobulin (TG), a glycoprotein with tyrosine residues. These tyrosine residues are conjugated with iodine (from dietary sources), under the influence of the enzyme thyroid peroxidase, to form monoiodotyrosine (MIT) or diiodotyrosine (DIT). These then undergo a coupling reaction, while still attached to TG, to produce T3 (MIT + DIT) or T4 (DIT + DIT). Proteolysis of TG releases free T3 and T4 into the circulation.
Mechanism of action. Most cells in the body are responsive to thyroid hormones. Within cells, T4 is converted to T3, the active form. T3 binds to its receptor in the cell nucleus to affect gene transcription.
Effects. See Table 16.6.
|
Table 16.6 |
||
|
Category |
Effects |
|
|
Development |
Essential for CNS development |
|
|
Growth |
↑ protein synthesis ↑ bone formation (with GH and somatomedins) ↑ ossification and fusion of growth plates |
|
|
Metabolic |
↑ intestinal absorption of glucose ↑ gluconeogenesis, glycogenolysis, and glucose oxidation ↑ lipolysis ↑ cholesterol turnover and plasma clearance of cholesterol T3 potentiates the “hypoglycemic” actions of insulin by increasing glucose uptake into muscle and adipose tissue. |
|
|
Basal metabolic rate (BMR) |
↑ BMR ↑ ATP hydrolysis (↑ O2 consumption and heat as a consequence of the above) |
|
|
Systemic |
Stimulation of adrenergic β-receptors leading to increased heart rate, cardiac output, and ventilation and decreased peripheral vascular resistance. These actions support increased oxygen demand in tissues. T3 facilitates the actions of cortisol, glucagon, and GH. |
|
|
Abbreviations: ATP, adenosine triphosphate; CNS, central nervous system; GH, growth hormone. |
||
Pharmacokinetics
– T4 has a slower onset, is more extensively bound to plasma proteins, and has a longer duration of action than T3.
– T3 is four times more potent than T4.
Treatment of Hypothyroidism
Levothyroxine
Levothyroxine, a synthetic form of the thyroxine (T4), is the drug of choice for replacement therapy.
Uses
– Hypothyroidism, regardless of etiology, including congenital (cretinism)
– Autoimmune thyroiditis (e.g., Hashimoto thyroiditis)
– Pregnancy and postpartum hypothyroidism
– Thyroid carcinoma (to suppress TSH)
Toxicity. It causes tachycardia, palpitations, restlessness, tremor, and cardiac arrhythmias.
Hypothyroidism (myxedema)
Hypothyroidism occurs due to decreased levels of plasma T3 and T4. Metabolism is slowed producing symptoms such as weight gain, constipation, cold intolerance, lethargy, depression, and dementia. Signs of hypothyroidism include bradycardia, dry skin and face, goiter, congestive heart failure, and edema. This condition may be spontaneously acquired, or it may occur after a thyroidectomy, radioiodine treatment, or following drug therapy (e.g., amiodarone and lithium). It is treated by replacement therapy with levothyroxine. More severe, life-threatening hypothyroidism is called myxedema crisis. This can lead to impaired cognition, somnolence (sleepiness), and coma (myx edema coma). Myxedema can be treated with IV levothyroxine or with liothyronine to achieve a more rapid response.
Treatment of Hyperthyroidism and Related Disorders
Propylthiouracil and Methimazole
Mechanism of action. Propylthiouracil and methimazole inhibit thyroid hormone synthesis by inhibiting the peroxidase enzyme that catalyzes the iodination of tyrosine residues in thyroglobulin and couples iodotyrosines to form T3and T4. Effects are not apparent until the thyroid reserve is depleted.
Pharmacokinetics
– Methimazole is more potent and has a longer duration than propylthiouracil.
– These drugs cross the placenta and are excreted into milk.
– Babies who are exposed to these agents should have thyroid function monitored.
Uses
– Hyperthyroidism
Side effects
– Rash is common.
– Agranulocytosis (acute low white blood cell count) is rare but serious.
Hyperthyroidism (thyrotoxicosis)
Hyperthyroidism occurs when there are elevated levels of T3 and T4 in the blood. Patients may develop hyperthyroidism due to adenomas/carcinomas of the thyroid gland or thyroiditis (inflammation of the thyroid), or it may be autoimmune in origin. Signs and symptoms are reflective of a hyper-metabolic state and include weight loss, increased appetite, frequent stools, tremor, heat intolerance, increased sweating, tachycardia, tremor, ptosis (lid lag) and thyroid enlargement. Severe hyperthyroidism (thyrotoxic storm) is a medical emergency. Treatment depends on the cause.
Radioiodine (Sodium Iodide, I131)
Mechanism of action. Radioiodine accumulates in the thyroid gland and destroys parenchymal cells. Clinical improvement may take 2 to 3 months.
Uses. It is the preferred treatment for most patients with hyperthyroidism. Subsequent hypothyroidism occurs in 20 to 80% of patients.
Iodine (Lugol Solution, Potassium Iodide)
Mechanism of action. Iodine (supraphysiological dose) inhibits thyroid hormone release, but the effect is not sustained (Wolff-Chaikoff effect); therefore, it only produces a temporary remission of symptoms.
Uses
– Thyrotoxicosis
– Prior to thyroid surgery (to decrease vascularity of the gland)
– Following radioiodine therapy
Na+- I– Pump
Iodine (from ingested food) is necessary for thyroid hormone synthesis. Because dietary intake inevitably varies, the thyroid gland must sequester iodine so that adequate amounts are always available for thyroid hormone synthesis. It does this via the Na+-I− pump on the cell membrane. The pump symports two Na+ ions into the cytoplasm for every one I− ion. It is driven by the low intracellular [Na+], via facilitated diffusion, that is maintained by the Na+-K+-ATPase pump. TSH is the major physiological stimulator of the iodide pump. High intracellular levels of iodide inhibit the activity of the pump.
Wolff–Chaikoff effect
The Wolff–Chaikoff effect is a reduction in the synthesis and release of thyroid hormones caused by a large amount of iodine. This effect lasts ~10 days, after which iodine incorporation into TG and thyroid peroxidase function returns to normal. It is widely believed that the resumption of normal functioning is due to downregulation of the iodide pump on the follicular cell membrane. The Wolff–Chaikoff effect is the principle behind the use of iodine for the treatment of hyperthyroidism. Amiodarone may also elicit this side effect, given that it contains iodine.
Propranolol, Atenolol, Esmolol, and Metoprolol
The pharmacology of these adrenergic blocking agents was discussed in Chapter 6.
Uses. These agents are used as adjuncts to treat or prevent thyrotoxicosis.
16.3 Parathyroid Hormone and Other Factors Affecting Bone Metabolism
Components of bone
The three major components of bone are osteogenic cells (osteoblasts, osteocytes, and osteoclasts), organic matrix, and mineral. The matrix consists of collagen and proteoglycans and accounts for approximately one third of bone mass. The mineral component of bone is calcium phosphate crystals deposited as hydroxyapatite (two thirds of bone mass).
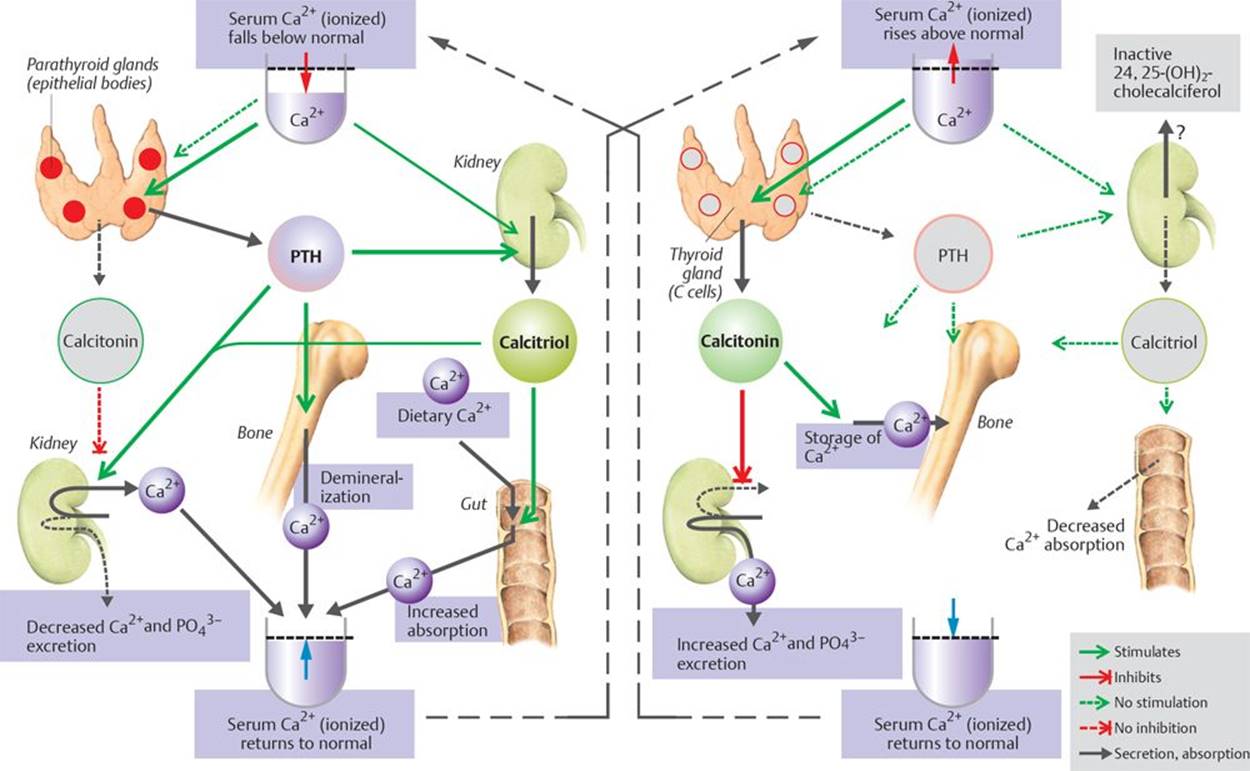
The major hormones involved in bone mineral homeostasis are parathyroid hormone (PTH) and vitamin D (Fig. 16.4). Other endogenous regulators of bone metabolism are calcitonin, glucocorticoids, and estrogens. Numerous exogenous agents are available that affect bone and mineral homeostasis.
Fig. 16.4. ![]() Hormonal regulation of the blood Ca2+ concentration.
Hormonal regulation of the blood Ca2+ concentration.
Ca2+ homeostasis is achieved by three main hormones: parathyroid hormone (PTH, from parathyroid gland), calcitonin (from parafollicular cells of the thyroid gland), and calcitriol (mainly produced in the kidney). In low serum Ca2+ states, the actions of parathyroid hormone and calcitriol predominate, causing increased Ca2+ uptake from the gut and bone and decreased renal excretion. In high serum Ca2+ states, the action of calcitonin predominates, causing decreased Ca2+ uptake from the gut, increased renal excretion, and storage of excess Ca2+ in bone.
Parathyroid Hormone
Regulation of secretion. PTH is released from the parathyroid glands in response to low plasma Ca2+ concentrations.
Multiple endocrine neoplasia type I
Multiple endocrine neoplasia type 1 (MEN1) is a rare, inherited disorder that causes multiple tumors (usually benign) in the endocrine glands and duodenum. It affects both genders equally and is usually not detected until adulthood, when tumors start growing. The parathyroid glands are most commonly affected. Tumors in the parathyroid gland cause hyperparathyroidism, which leads to hypercalcemia and its associated symptoms. It also commonly affects the pancreas, causing gastrinomas (from excess gastrin secretion), which, in turn, causes ulcers. These ulcers are more sinister than normal gastric ulcers and are highly prone to perforate. Multiple gastrinomas causing ulcers is referred to as Zollinger–Ellison syndrome. Pancreatic tumors may also cause insulinomas, leading to hypoglycemia; glucagon excess, leading to diabetes; or vasoactive intestinal peptide (VIP), leading to watery diarrhea. Pituitary tumors may also occur, leading to derangement of its hormones. Patients with MEN1 are more likely to develop cancerous tumors in later life. MEN1 can be detected early by gene testing, and individuals affected have a 50% chance of passing the disease to their children. There is no cure for MEN1, but there are various drugs and surgical options to treat the effects.
Effects. See Table 16.7.
|
Table 16.7 |
||
|
Organ/System |
Effects |
|
|
Bone |
Mobilization of Ca2+ and PO43- from bone In the longer term, PTH increases the number of both osteoblasts and osteoclasts and increases the remodeling of bone (Fig. 16.10).* |
|
|
Kidney |
↑ Ca2+ reabsorption ↑ formation of calcitriol which is the active form of vitamin D |
|
|
GI tract |
↑ absorption of Ca2+ (effect mediated via calcitriol) |
|
|
* Daily, intermittent administration of PTH for 1 to 2 hours per day leads to a net stimulation of bone formation. C ontinuous exposure to elevated PTH leads to bone resorption. Abbreviation: GI, gastrointestinal. |
||
Physiological roles of calcium
Calcium is vital to normal body functioning, and its levels must be maintained within tight limits. Calcium is the major structural element of bones and teeth, where it is stored in the form of hydroxyapatite crystals [Ca10(PO4)6(OH)2]. It is also involved in neural transmission, muscle contraction, vasodilation and vasoconstriction of blood vessels, activation of vitamin K–dependent clotting factors (II, VII, IX, and X), and secretion of hormones (e.g., insulin).
Teriparatide
Mechanism of action. Teriparatide is a synthetic polypeptide PTH analogue; it therefore affects calcium homeostasis in the same way as PTH.
Pharmacokinetics
– Given by daily subcutaneous injection
Uses
– Osteoporosis in postmenopausal women at high risk of fracture
– Hypogonadal osteoporosis in men at high risk of fracture
Magnesium and bone metabolism
Magnesium is the fourth most abundant mineral in the body. About half of the body's magnesium is stored in the hydroxyapatite crystals of bone, and the other half is intracellular. The levels of magnesium in the blood are well regulated and tend to follow those of calcium and phosphate. Like calcium and phosphate, magnesium has a role in bone turnover (but to a much lesser extent). Magnesium deficiency may result from severe diarrhea, alcohol abuse, drugs (e.g., diuretics), and diabetic ketoacidosis. It causes parethesias, seizures, arrhythmias, and tetany (due to accompanying hypocalcemia and hypokalemia). Treatment is by replacement of magnesium.
Cinacalcet
Mechanism of action
Cinacalcet activates the Ca2+-sensing receptor of the parathyroid gland, which leads to decreased PTH secretion.
Uses
– Hypercalcemia
– Primary hyperparathyroidism
Vitamin D
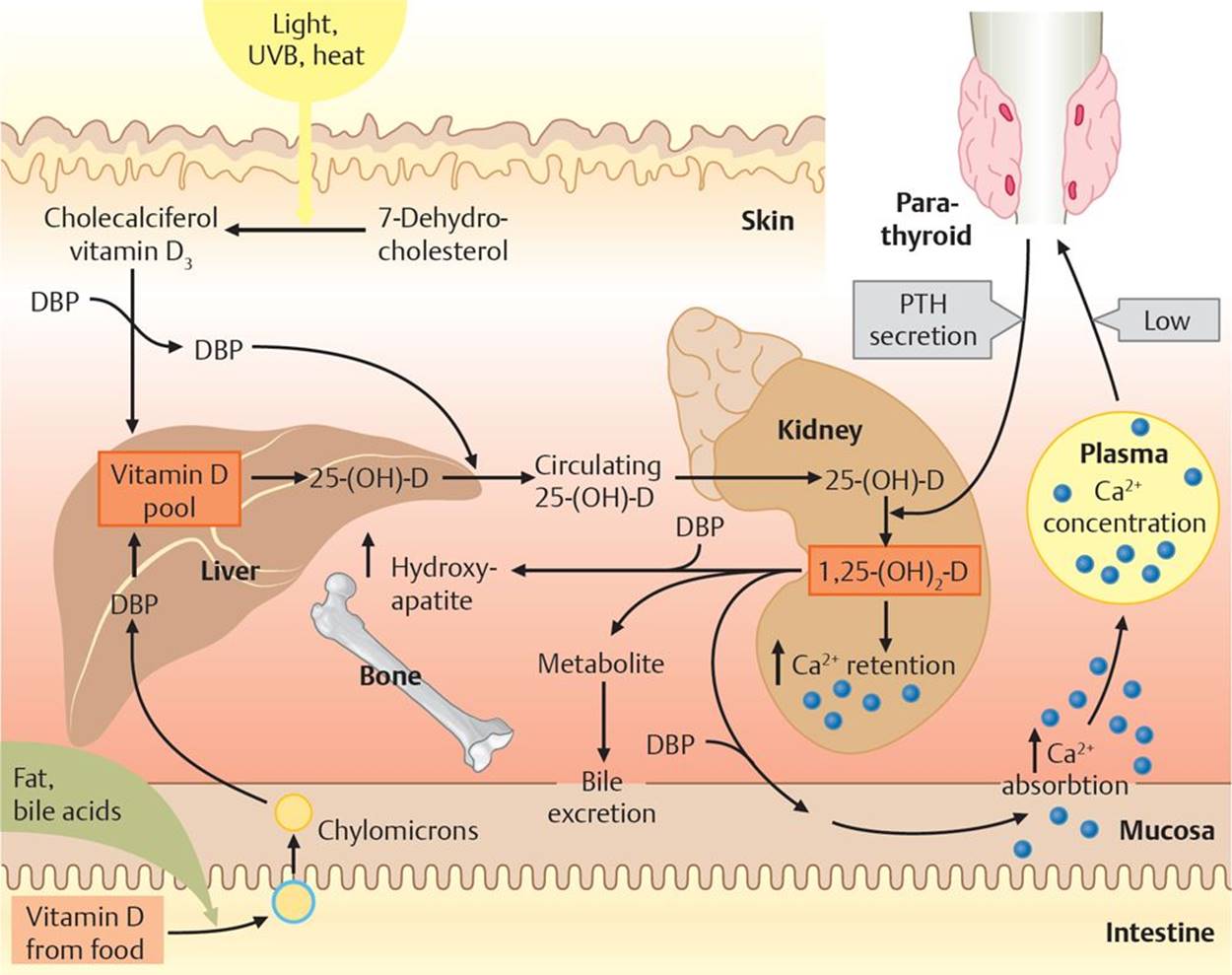
The term vitamin D refers to cholecalciferol (vitamin D3) and ergocalciferol (vitamin D2), which are interchangeable with respect to clinical use. Ergocalciferol is the prescription form of vitamin D and is also used as a food additive. Cholecalciferol is usually used for vitamin D–fortified milk and foods; it is also available in drug combination products. The metabolism of vitamin D is shown in Fig. 16.5.
Fig. 16.5 ![]() Vitamin D metabolism.
Vitamin D metabolism.
Ultraviolet light B (UVB) converts 7-dehydrocholesterol to cholecalciferol (vitamin D3). Ingested vitamin D is fat soluble and is transported to the liver in chylomicrons. All free vitamin D is transported in the blood and liver by a specific vitamin D–binding protein (DBP). The liver converts vitamin D to 25-hydroxycholecalciferol 25-(OH)-D, which is then transported to the kidneys, where it is converted to its active form 1,25-(OH)2-D, under the influence of parathyroid hormone. The effects of this are increased mineralization of bone, increased calcium and phosphate reabsorption in the kidneys, and increased calcium absorption in the gut. Excess vitamin D is excreted into bile.
Chronic renal failure and renal osteodystrophy
In chronic renal failure, the failing kidneys are unable to perform the necessary 1-αhydroxylation reactions to produce calcitriol, and they have a reduced capacity to excrete phosphate. This leads to hyper-parathyroidism due to hypocalcemia and hyperphosphatemia. Derangement of bone remodeling occurs, which is referred to as renal osteodystrophy. The symptoms of renal osteodystrophy include bone and joint pain, bone deformation, and increased likelihood of bone fractures. Chronic renal failure requires hemodialysis several times per week until renal transplantation can occur. Renal osteodystrophy is treated by calcium and calcitriol, restricting dietary intake of phosphate, and by the administration of medications that bind phosphate, such as calcium carbonate and calcium acetate.
Effects
– Bone: The effects of vitamin D on bone are a result of its actions that provide the proper balance of Ca2+ and PO43– to support bone mineralization.
– Kidney: Increased reabsorption of Ca2+ and PO43–
– Gastrointestinal tract: Increased absorption of Ca2+
Ergocalciferol
Uses
– Hypoparathyroidism
– Prophylaxis of vitamin D deficiency
Calcitonin
Calcitonin is produced by the parafollicular cells (C cells) of the thyroid gland. It is released when there is an elevated level of Ca2+ in the blood.
Effects
– Bone: Decreases bone resorption by inhibiting osteoclast activity (Fig. 16.6)
– Kidney: Decreases reabsorption of Ca2+ and PO43–, thus increasing their excretion
Calcitonin (exogenous)
Pharmacokinetics
– Available as a nasal spray
Uses
– Paget disease
– Hypercalcemia
– Osteoporosis
Paget disease
Paget disease is a metabolic bone disease which affects 2 to 3% of the population over age 60. It consists of increased bone resorption and new bone formation; however, the newly formed bone is disordered, leading to bowing, stress fractures and arthritis. Additional symptoms may include enlargement of the skull, femur, and clavicle. Nerve compression may occur due to bony overgrowth causing pain, paresthesias, or numbness. Complications include congestive heart failure (due to increased work of the heart) and sarcomas (rarely). Blood biochemistry results show that Ca2+and PO43– are usually normal but alkaline phosphatase (ALP) is markedly increased. Because ALP is a by-product of osteoblastic activity, levels of ALP are raised during periods of rapid bone growth (puberty), in bone diseases that cause bone turnover (e.g., Paget disease and osteomalacia), and during calcium derangement (e.g., hyperparathyroidism). Treatment is with a bisphosphonate and/or calcitonin.
Glucocorticoids
Cortisol
Mechanism of action. Glucocorticoids, e.g., cortisol, enhance bone loss by decreasing Ca2+ absorption, increasing Ca2+ excretion, and blocking bone formation.
Uses
– Hypercalcemia of malignancy
– Vitamin D poisoning
Side effects. Prolonged administration leads to osteoporosis.
Estrogens and Selective Estrogen Receptor Modulators (SERMs)
Estrogens: Estradiol, Premarin™, Ethinyl Estradiol, Mestranol, and Diethylstilbesterol
– Estradiol esters are administered intramuscularly and by a transdermal patch.
– Premarin™ is a conjugated estrogen that contains estrone and equilin. It is administered orally.
– Ethinyl estradiol and mestranol are synthetic steroid estrogens that are administered orally.
– Diethylstilbestrol is a nonsteroidal synthetic estrogen that is administered orally or parenterally.
Mechanism of action. Estrogens decrease osteoclast activity. The mechanism by which this occurs is unclear, but it may be that the estrogens cause an increase in cytokines (small cell-signaling protein molecules) that support osteoclast formation, or they increase the rate of apoptosis of osteoclasts (Fig. 16.6). Estrogens may also decrease other cytokines that decrease osteoblasts.
SERM: Raloxifene
See page 163 and Fig. 17.3 for a further discussion of SERMs.
Uses. Estrogens and SERM agents are used (in the context of bone regulation) to treat post-menopausal osteoporosis. However, estrogens are now only used with caution in patients for whom nonestrogen therapies are not appropriate.
Bisphosphonates
Alendronate, Etidronate, Ibendronate, Pamidronate, Risedronate, Tiludronate, and Zoledronate
Mechanism of action. Bisphosphonates are exogenous regulators of bone metabolism. They are analogues of pyrophosphate that accumulate in bone and prevent bone resorption by inhibiting osteoclast activity (Fig. 16.4).
Uses
– Prevention and treatment of osteoporosis
Fig. 16.6 ![]() Regulation of bone remodeling.
Regulation of bone remodeling.
Bone remodeling is complex and is initiated by osteoblasts upon stimulation by parathyroid hormone. Osteoblasts interact directly with osteoclast precursors (that form from progenitor cells) via RANKL. Estrogens (and OPG secreted by osteoblasts) block RANKL, thus inhibiting the fusion of osteoclast precursors and their activation to osteoclasts. They also promote osteoclast apoptosis. Bisphosphonates inhibit bone resorption by inhibiting osteoclast activity and promoting apoptosis. Calcitonin inhibits bone resorption by transferring active osteoclasts into a resting state.
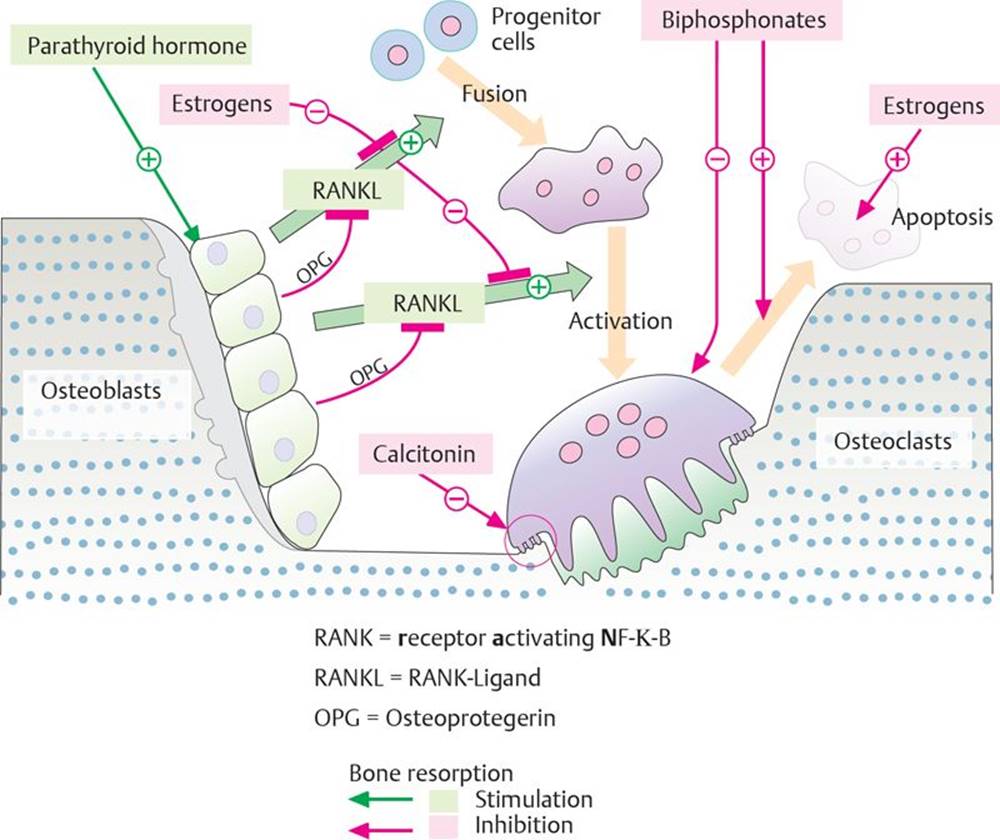
Bisphosphonates and osteonecrosis of the jaw
Osteonecrosis is death of bone tissue due to a lack of blood supply. Bisphosphonate usage puts patients at risk of developing osteonecrosis of the jaw, which can present following dental extractions. It produces symptoms such as bone pain, swelling, infections, exposed bone, numbness, and loosening of the teeth. This condition usually occurs with IV bisphosphonates, but it can occur when they are taken orally. Patients who are at risk of developing osteonecrosis should have any sources of infection treated before bisphosphonate therapy is initiated to prevent the condition from developing. However, if osteonecrosis does occur, surgical débridement of necrotic bone and areas of infection is necessary.
Fluoride
Mechanism of action. Fluoride accumulates in bone and teeth where it slows resorption and promotes calcification.
Uses
– It is added to drinking water to prevent dental caries
– May have a positive effect in osteoporosis
16.4 Adrenocortical Hormones
The adrenocortical steroids, or corticosteroids, are steroid hormones produced by the adrenal cortex (Fig. 16.7). They include glucocorticoids, mineralocorticoids, and androgens. The glucocorticoids regulate metabolism and stress responses, and the mineralocorticoids regulate sodium reabsorption. Androgens will be covered on pages 166–168, Chapter 17.
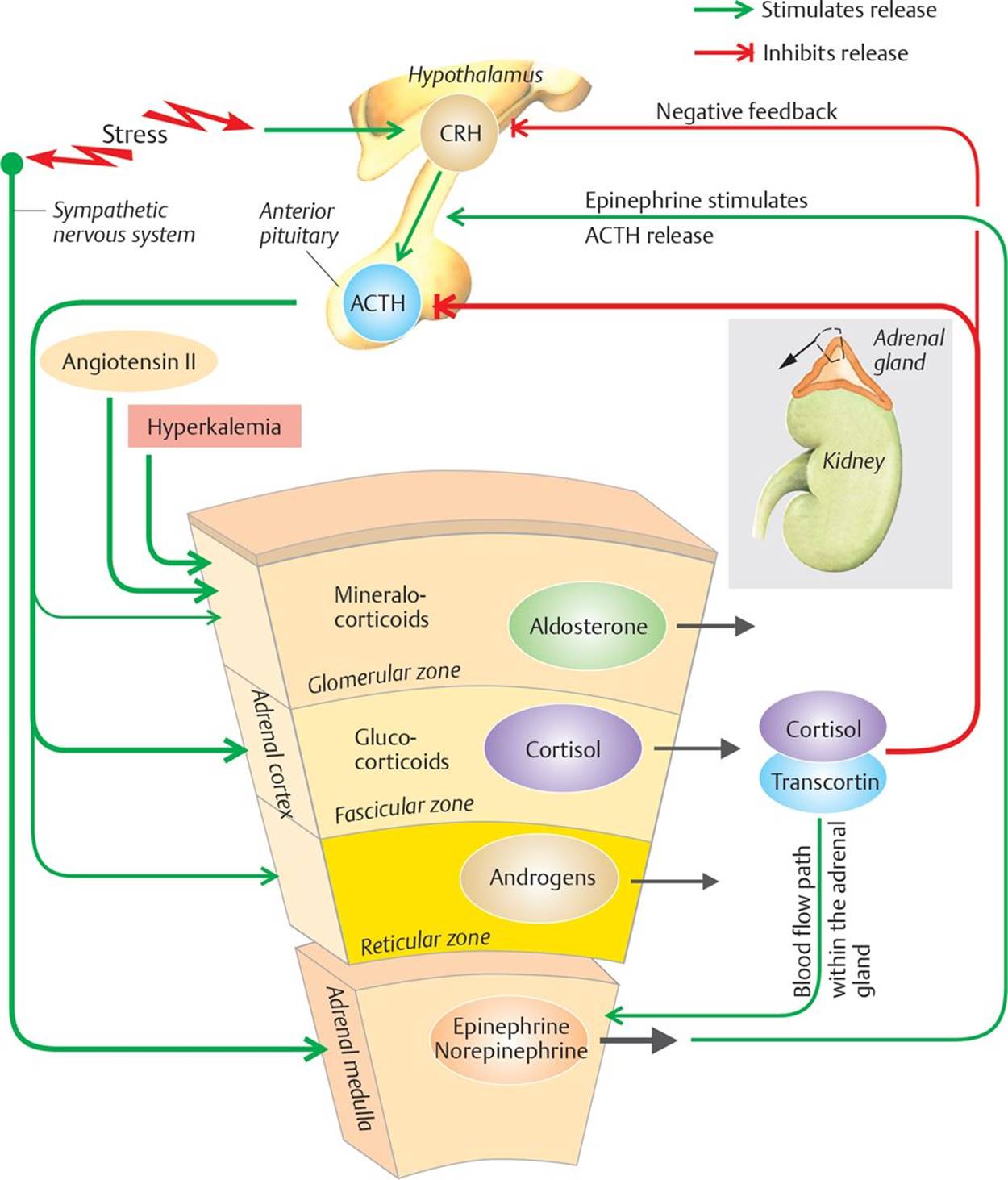
Fig. 16.7 ![]() Hormonal secretions of the adrenal gland.
Hormonal secretions of the adrenal gland.
Stress causes the release of corticotropin-releasing hormone (CRH) from the hypothalamus, which then stimulates adrenocorticotropic hormone (ACTH) release from the anterior pituitary. ACTH acts on all zones of the adrenal cortex, but it predominantly causes the release of cortisol from the fascicular zone. One of the local actions of cortisol is to stimulate the release of norepinephrine and epinephrine from the adrenal medulla, although the main stimulus for this is sympathetic system activation. Cortisol causes negative feedback inhibition of ACTH (predominantly) and CRH.
Glucocorticoids
Regulation of secretion. The natural glucocorticoids, cortisol and corticosterone, are synthesized and released in response to ACTH from the anterior pituitary, which is released in response to CRH from the hypothalamus.
Mechanism of action. Glucocorticoids bind to receptors in the cytosol. These receptors then dissociate from heat shock protein complexes, translocate to the nucleus, and bind to specific sites on DNA within the nucleus, altering gene transcription.
Fig. 16.8 ![]() Glucocorticoids: receptors and second messengers.
Glucocorticoids: receptors and second messengers.
Glucocorticoids act to alter gene transcription, the effects of which are to reduce inflammation. At higher concentrations, they may also act on the cell membrane to reduce the activation capacity of the cell. (ACE, angiotensin-converting enzyme; COX-2, cyclooxygenase-2; hsp, heat shock protein; mRNA, messenger RNA. mRNA mRNA
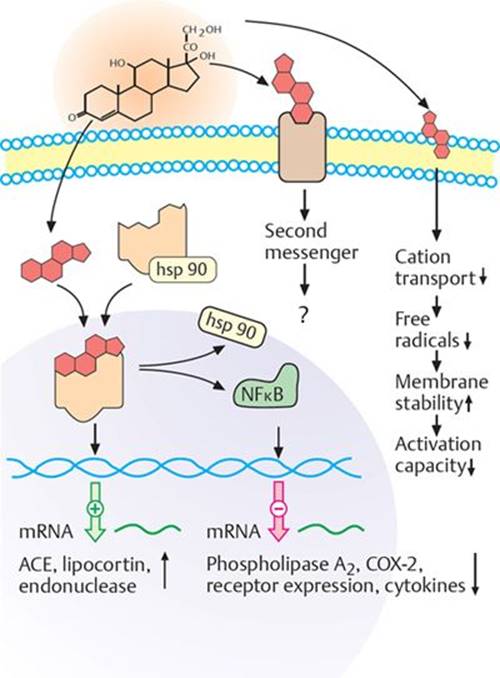
At higher concentrations, glucocorticoids are thought to be integrated into the cell membrane, which alters the physiochemical properties of the cell. The ultimate effect of these changes is that the activation capacity of the cell is reduced (Fig. 16.8).
Effects. Table 16.8 lists the effects of glucocorticoids. These are further illustrated in Figs. 16.9 and 16.10.
The effect of corticosteroids on the respiratory burst
The respiratory burst is the rapid release of reactive oxygen species (superoxide radical and hydrogen peroxide) from phagocytes (neutrophils and monocytes) when a microbe is encountered and phagocytosed. It is one of the mechanisms by which phagocytes exert their microbicidal effects and is an important immune defense. The reactive oxygen species are generated by the partial reduction of oxygen in the respiratory chain (electron transport chain). They combine with Cl– to form hypochlorous acid, which dissociates to form hypochlorite ions, which kill the microbes. Cortisol (and exogenous corticosteroids) inhibits the respiratory burst and may predispose an individual to infection.
Hydrocortisone (Cortisol), Prednisone, Methylprednisolone, Triamcinolone, Dexamethasone, Betamethasone, Beclomethasone, and Fluocinonide
Mechanism of action. These agents are synthetic glucocorticoids that act like the endogenous hormones.
|
Table 16.8 |
||
|
Category |
Effects |
|
|
Metabolic |
↑ hepatic gluconeogenesis ↑ glycogen synthesis ↑ proteolysis ↑ release of amino acids (key enzymes) for gluconeogenesis ↑ lipolysis, which increases fatty acid and ketone body formation ↓ peripheral glucose utilization |
|
|
Antiinflammatory (see also Fig. 16.10) |
↓ transcription of most cytokines and chemokines leads to reduced synthesis of these mediators and decreased activation of leukocytes. ↓ transcription of phospholipase A2 and cyclooxygenase-2 (COX-2) results in decreased formation of prostaglandins and thromboxanes (see page 347). ↓ expression of adhesion molecules necessary for leukocyte chemotaxis ↑ expression of antiinflammatory molecules, such as neutral endopeptidase and lipocortins |
|
Fig. 16.9 ![]() Glucocorticoids: principal and adverse effects.
Glucocorticoids: principal and adverse effects.
Therapeutic levels of cortisol suppress the inflammatory response. This is useful in treating conditions such as allergy and autoimmune disease and to prevent transplant rejection, where the inflammatory response is unwanted. However, cortisol also produces a number of adverse effects related to its antiinflammatory, mineralocorticoid, and glucocorticoid actions. The relative mineralocorticoid potencies (blue) and glucocorticoid potencies (brown) of some corticosteroids are included.
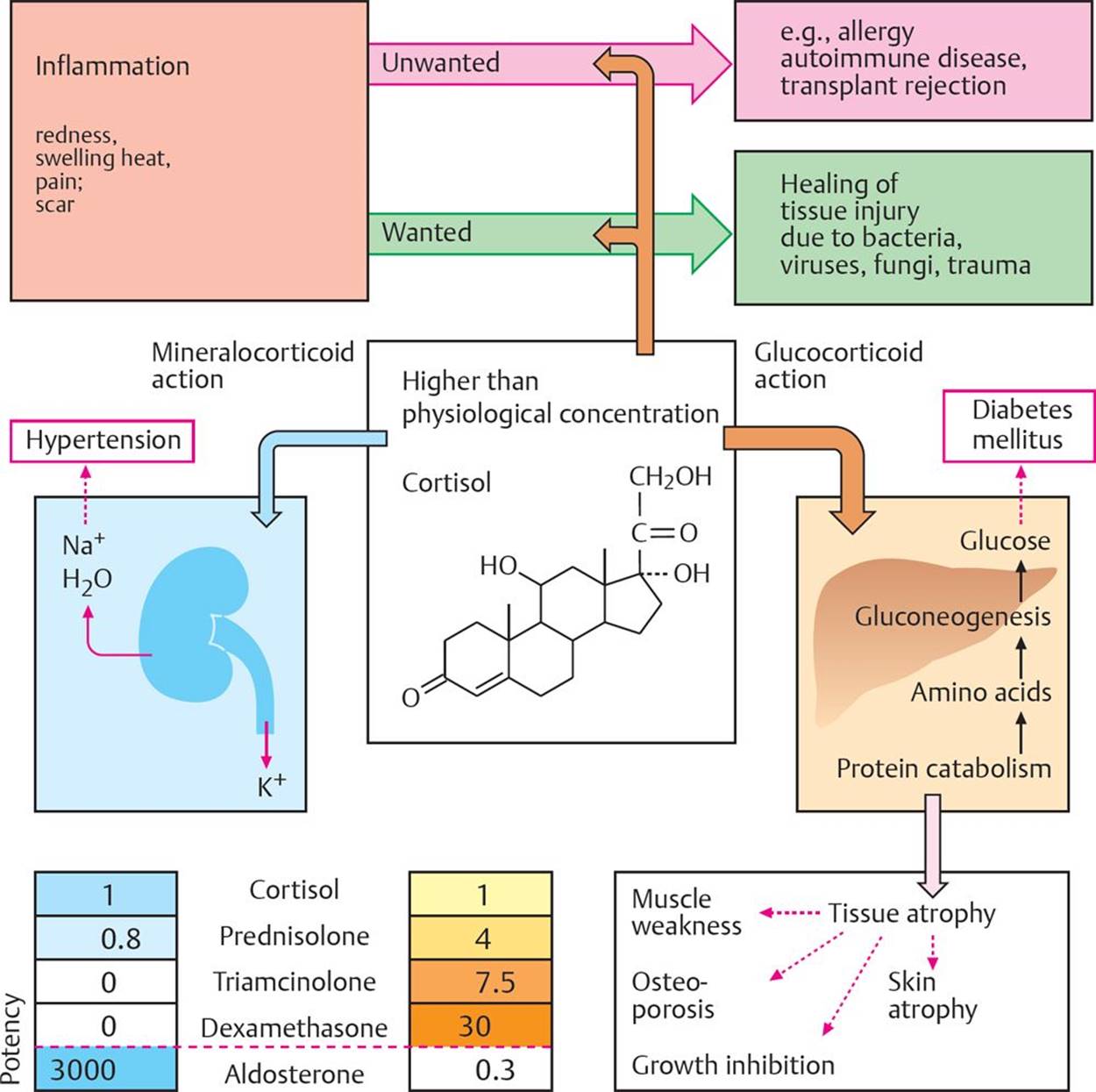
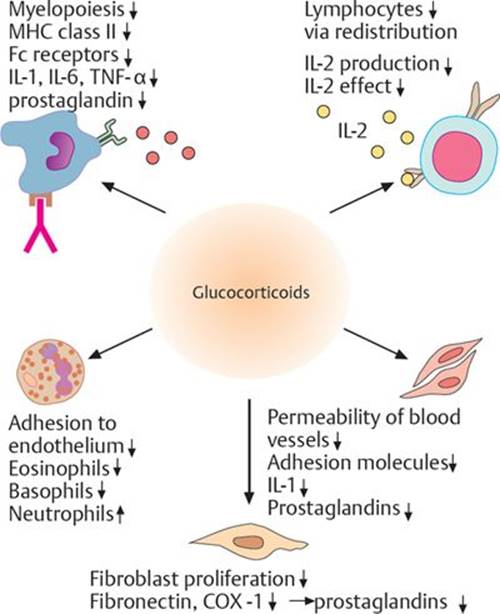
Fig. 16.10 ![]() Effects of glucocorticoids on the immune system.
Effects of glucocorticoids on the immune system.
Glucocorticoids act on a wide range of cells, including blood cells, endothelial cells, and fibroblasts, as well as receptor proteins and proinflammatory mediators, to reduce inflammation and its spread. (Fc, fragment, crystallizable; IL, interleukin; MHC, major histocompatibility complex; TNF, tumor necrosis factor.)
Pharmacokinetics. These agents are effective orally, parenterally, and topically.
Note: Beclomethasone and fluocinonide are for local use only. Table 16.9 lists the duration of action of these agents.
|
Table 16.9 |
||
|
Duration of Action |
Agent (s) |
|
|
Short |
Hydrocortisone (cortisol) |
|
|
Intermediate |
Prednisone, methylprednisolone, triamcinolone |
|
|
Long |
Dexamethasone, betamethasone |
|
Uses
– Endocrine
— Adrenocortical insufficiency (Addison disease)
— Congenital adrenal hyperplasia (to suppress ACTH release)
– Non-endocrine
— Rheumatoid arthritis
— Leukemia
— Lymphoma
— Allergic reactions
— Asthma
— Inflammatory and autoimmune disorders
— Immunosuppression for transplantation
— Collagen disorders
— Cerebral edema
— Bacterial meningitis
Side effects. Large doses of glucocorticoids for 1 week or less do not pose problems, but patients with non-endocrine disorders who receive systemic corticosteroids for longer times develop adverse effects, including inhibition of CRH release from the hypothalamus and ACTH release from the anterior pituitary (Fig. 16.11). Some of the side effects experienced include
– Hyperglycemia
– Increased susceptibility to infection
– Weight gain (cushingoid features)
– Osteoporosis
– Behavioral and personality changes
– Myopathy
– Ocular effects
– Growth retardation in children
When possible, glucocorticoids should be administered locally, e.g., as an aerosol spray in asthma, to minimize adverse effects, or alternate-day therapy should be used.
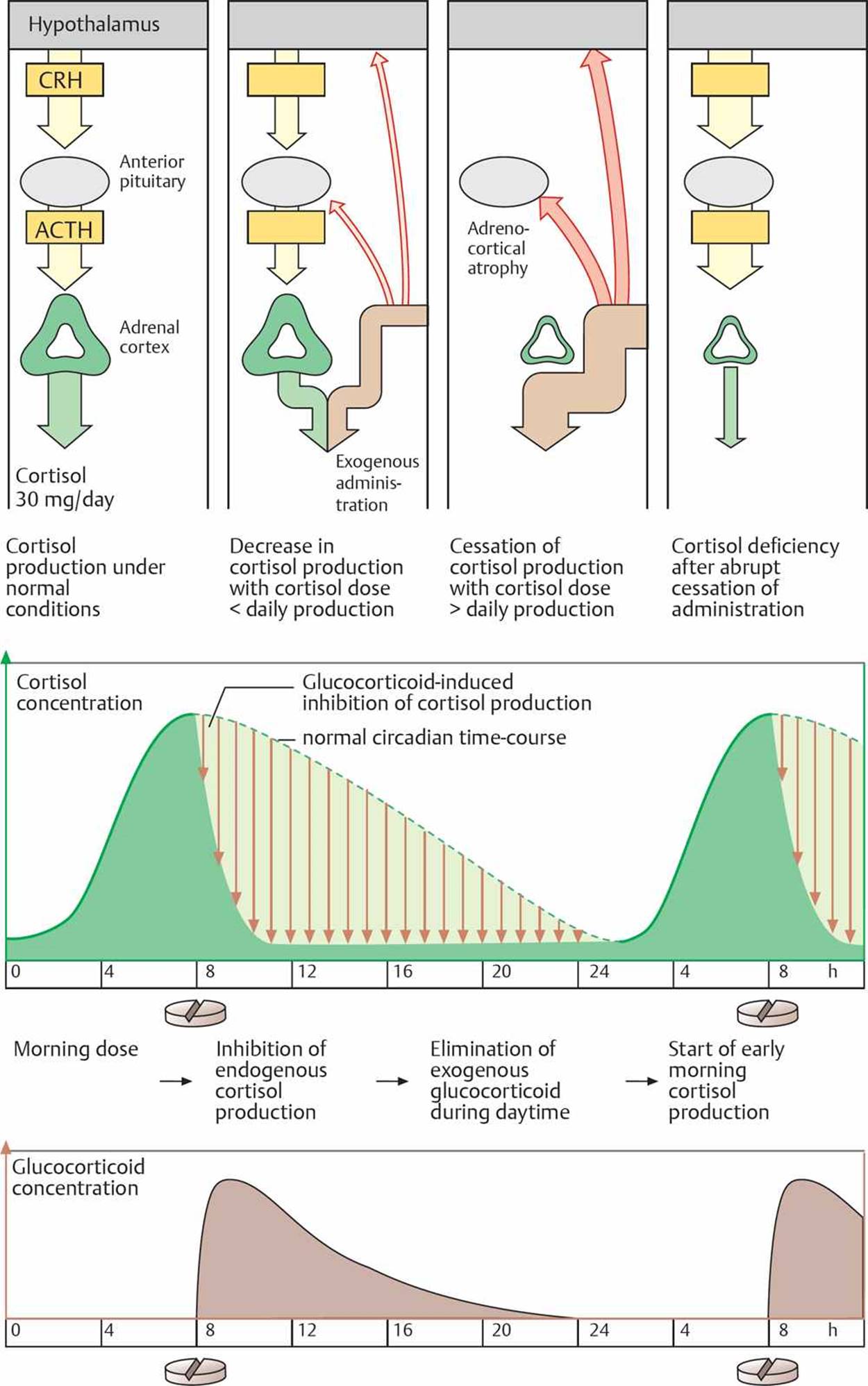
Fig. 16.11 ![]() Cortisol release and its modification by glucocorticoids.
Cortisol release and its modification by glucocorticoids.
Exogenous glucocorticoids cause feedback inhibition of endogenous cortisol production. Depending on the dose, this can cause endogenous cortisol production to decrease or cease completely. If cortisol production is not required by the adrenal cortex, it undergoes atrophy and cannot resume normal cortisol output if exogenous glucocorticoids are abruptly stopped. The dosing regimen of glucocorticoids can minimize adrenocortical atrophy. If they are given when normal cortical secretion is high and feedback inhibition is low (late morning), then the glucocorticoid is eliminated during daytime, and normal cortisol production starts early the following morning.
Acute adrenal crisis (addisonian crisis)
Acute adrenal crisis (addisonian crisis) is due to acute insufficiency of adrenal corticosteroids, mainly cortisol. It usually occurs in people with known Addison disease who undergo some form of stress, such as surgery, trauma, or infection, but it may also occur on abrupt cessation of long-term steroids. The main sign of acute adrenal crisis is shock (hypotension, tachycardia, or oliguria), but there may also be acute abdominal pain, diarrhea, vomiting, hypoglycemia, fever, weakness, and confusion. It may progress to seizures, coma, and death if untreated. If there is a high index of suspicion for acute adrenal crisis, treatment should begin before the results of any laboratory tests are known. Treatment involves giving intravenous (IV) fluids, hydrocortisone, antibiotics, and glucose if necessary. In the longer term, the patient can be switched to oral steroids, and the precipitating factor should be treated.
Perioperative steroid coverage
Patients who have been on long-term steroids or have stopped steroids recently will have some adrenal suppression. Consequently, the perioperative administration of steroids prior to undergoing the stress of surgery is necessary to prevent adrenal crisis, the major effect of which is shock.
Mineralocorticoids
The major mineralocorticoid produced by the adrenal gland is aldosterone. Aldosterone has a very short half-life and is not used therapeutically.
Regulation of secretion. Aldosterone secretion is regulated by the renin-angiotensin- aldosterone system (see pages 181–183).
Renin-angiotensin-aldosterone system
The renin-angiotensin-aldosterone system regulates Na+ balance, fluid volume, and blood pressure. Renin is released by the kidneys in response to reduced perfusion. Renin then stimulates the production of angiotensin II from angiotensin I in the lungs. Angiotensin II causes vasoconstriction and aldosterone secretion from the adrenal glands. Aldosterone causes Na+ (and water) reabsorption, thus increasing fluid volume, blood pressure, and renal perfusion.
Effects. Mineralocorticoids help maintain normal blood volume by promoting Na+ reabsorption by the distal tubules. K+ and H+ ions are excreted in exchange.
Fludrocortisone
Mechanism of action. Fludrocortisone is a synthetic corticosteroid that has much greater mineralocorticoid than glucocorticoid activity.
Pharmacokinetics
– Orally effective
Uses
– Used in salt-losing forms of adrenal insufficiency
Spironolactone
See page 188 for further discussion of this agent.
Mechanism of action. Spironolactone is an aldosterone receptor antagonist.
Uses
– Primary hyperaldosteronism
– Cushing syndrome
– Adrenal adenoma or carcinoma
– Ectopic ACTH-producing tumors
Aminoglutethimide
Mechanism of action. Aminoglutethimide blocks the conversion of cholesterol to pregnenolone, thereby decreasing secretion of all adrenal cortical steroids.
Cholesterol and steroid synthesis
Steroid hormones are produced from cholesterol (C27H46O). All steroid-producing tissues cleave the side chain of cholesterol between carbons 20 and 22 to form pregnenolone (via cholesterol desmolase). This is the rate-limiting step. In most cases, pregnenolone is then converted to progesterone. In the gonads, progesterone is converted to testosterone via cleavage of the remaining side chain. In many tissues, a 5α-reductase converts testosterone to 5α-dihydrotestosterone (DHT), which is the more active form of the hormone. The ovary also makes testosterone but does not release it. Instead, it aromatizes ring A (resulting in the loss of carbon 19) to form 18 carbon steroids (estradiol, estrone). In the adrenal cortex, hydroxylations produce cortisol and aldosterone. Some conversion of pregnenolone to androgens (especially dehydroepiandrosterone [DHEA]) and estrogens may occur.
Uses
– Cushing syndrome
– Adrenal adenoma or carcinoma
– Ectopic ACTH-producing tumors