As with the examination of any child, the order and extent of the physical examination of infants and children with potential cardiac problems should be individualized. The more innocuous procedures, such as inspection, should be done first, and the more frightening or uncomfortable parts should be delayed until later in the examination.
Supine is the preferred position for examining patients in any age group. However, if older infants and young children between 1 and 3 years of age refuse to lie down, they can be examined initially while sitting on their mothers’ laps.
Growth Pattern
Growth impairment is frequently observed in infants with congenital heart diseases (CHDs). The growth chart should reflect height and weight in terms of absolute values and also in percentiles. Accurate plotting and following of the growth curve are essential parts of the initial and follow-up evaluations of a child with significant heart problems. In overweight children, acanthosis nigricans should be checked in the neck and abdomen.
Different patterns of growth impairment are seen in different types of CHD.
1. Cyanotic patients have disturbances in both height and weight.
2. Acyanotic patients, particularly those with large left-to-right shunts, tend to have more problems with weight gain than with linear growth. The degree of growth impairment is proportional to the size of the shunt.
3. Acyanotic patients with pressure overload lesions without intracardiac shunt grow normally.
Poor growth in a child with mild cardiac anomaly or failure of catch-up weight gain after repair of the defect may be due to failure to recognize certain syndromes, inadequate calorie intake, or the underlying genetic predisposition.
Inspection
Much information can be gained by simple inspection without disturbing a sleeping infant or frightening a child with a stethoscope. Inspection should include the following: general appearance and nutritional state; any obvious syndrome or chromosomal abnormalities; color (i.e., cyanosis, pallor, jaundice); clubbing; respiratory rate, dyspnea, and retraction; sweat on the forehead; and chest inspection.
General Appearance and Nutritional State
The physician should note whether the child is in distress, well nourished or undernourished, and happy or cranky. Obesity should also be noted; besides being associated with other cardiovascular risk factors such as dyslipidemia, hypertension, and hyperinsulinemia, obesity is also an independent risk factor for coronary artery disease.
Chromosomal Syndromes
Obvious chromosomal abnormalities known to be associated with certain congenital heart defects should be noted by the physician. For example, about 40% to 50% of children with Down syndrome have a congenital heart defect; the two most common defects are endocardial cushion defect (ECD) and ventricular septal defect (VSD). A newborn with trisomy 18 syndrome usually has a congenital heart defect. Table 2-1 shows cardiac defects associated with selected chromosomal abnormalities along with other hereditary and nonhereditary syndromes.
Hereditary and Nonhereditary Syndromes and Other Systems Malformations
Congenital cardiovascular anomalies are associated with a number of hereditary or nonhereditary syndromes and malformations of other systems. For example, a child with a missing thumb or deformities of a forearm may have an atrial septal defect (ASD) or VSD (e.g., Holt-Oram syndrome [cardio-limb syndrome]). Newborns with CHARGE (coloboma, heart defects, choanal atresia, growth or mental retardation, genitourinary anomalies, ear anomalies) association show a high prevalence of conotruncal abnormalities (e.g., tetralogy of Fallot [TOF], double-outlet right ventricle (RV), persistent truncus arteriosus). A list of cardiac anomalies in selected hereditary and nonhereditary syndromes is given in Table 2-1. Certain congenital malformations of other organ systems are associated with an increased prevalence of congenital heart defects (Table 2-2).
Color
The physician should note whether the child is cyanotic, pale, or jaundiced. In cases of cyanosis, the degree and distribution should be noted (e.g., throughout the body, only on the lower or upper half of the body). Mild cyanosis is difficult to detect. The arterial saturation is usually 85% or lower before cyanosis is detectable in patients with normal hemoglobin levels (see Chapter 11). Cyanosis is more noticeable in natural light than in artificial light. Cyanosis of the lips may be misleading, particularly in children who have deep pigmentation. The physician should also check the tongue, nail beds, and conjunctiva. When in doubt, the use of pulse oximetry is confirmatory. Children with cyanosis do not always have cyanotic congenital heart defects. Cyanosis may result from respiratory diseases or central nervous system disorders. Cyanosis that is associated with arterial desaturation is called central cyanosis. Cyanosis associated with normal arterial saturation is called peripheral cyanosis. Even mild cyanosis in a newborn requires thorough investigation (see Chapter 14).
Peripheral cyanosis may be noticeable in newborns who are exposed to cold and those with congestive heart failure (CHF) because, in both conditions, peripheral blood flow is sluggish, losing more oxygen to peripheral tissues. Cyanosis is also seen in polycythemic patients with normal O2 saturation (see Chapter 11 for the relationship between cyanosis and hemoglobin levels). Circumoral cyanosis, cyanosis around the mouth, is found in normal children with fair skin. Isolated circumoral cyanosis is not significant. Acrocyanosis is a bluish or red discoloration of the fingers and toes of normal newborns in the presence of normal arterial oxygen saturation.
Pallor may be seen in infants with vasoconstriction from CHF or circulatory shock or in severely anemic infants. Newborns with severe CHF and those with congenital hypothyroidism may have prolonged physiologic jaundice. Patent ductus arteriosus (PDA) and pulmonary stenosis (PS) are common in newborns with congenital hypothyroidism. Hepatic disease with jaundice may cause arterial desaturation because of the development of pulmonary arteriovenous fistula (e.g., arteriohepatic dysplasia).
Clubbing
Long-standing arterial desaturation (usually longer than 6 months’ duration), even if too mild to be detected by an inexperienced person, results in clubbing of the fingernails and toenails. When fully developed, clubbing is characterized by a widening and thickening of the ends of the fingers and toes, as well as by convex fingernails and loss of the angle between the nail and nail bed (Fig. 2-1). Reddening and shininess of the terminal phalanges are seen in the early stages of clubbing. Clubbing appears earliest and most noticeably in the thumb. Clubbing may also be associated with lung disease (e.g., abscess), cirrhosis of the liver, and subacute bacterial endocarditis. Occasionally, clubbing occurs in normal people, such as in familial clubbing.
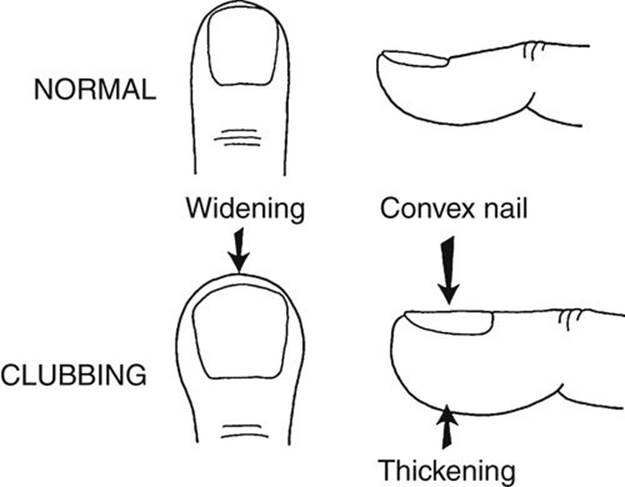
FIGURE 2-1 Diagram of normal and clubbed fingers.
TABLE 2-1
MAJOR SYNDROMES ASSOCIATED WITH CARDIOVASCULAR ABNORMALITIES



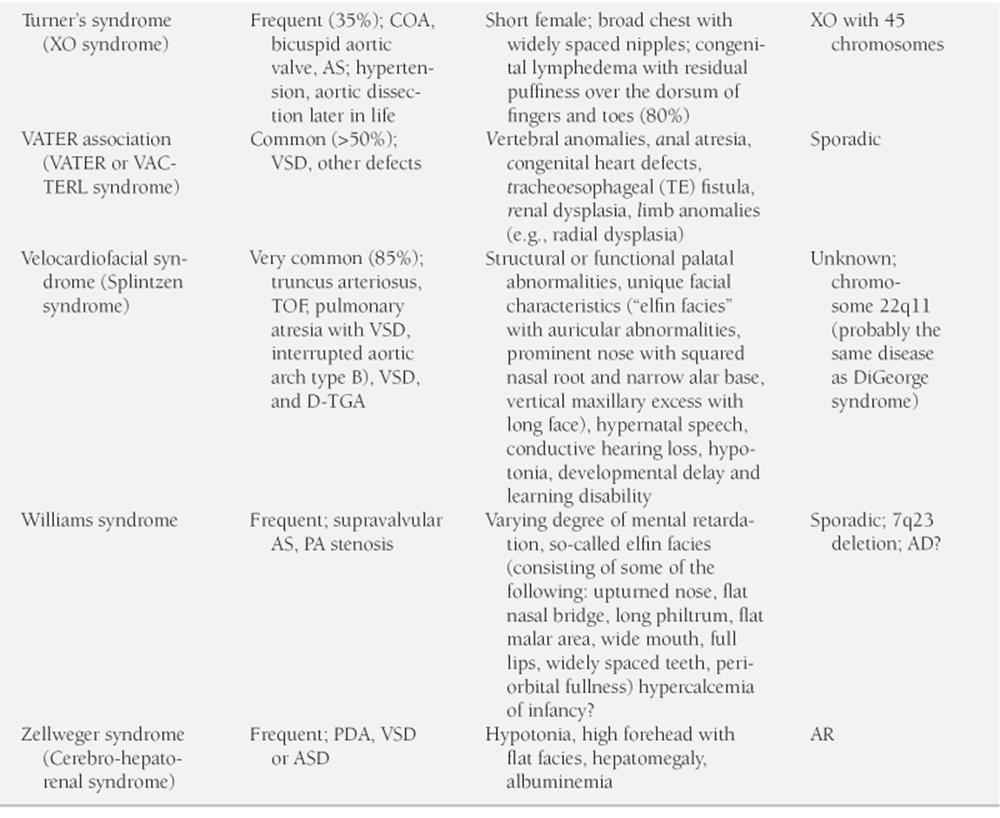
AD, autosomal dominant; AR, autosomal recessive; AS, aortic stenosis; ASD, atrial septal defect; CHD, congenital heart disease; Chrom, chromosome; COA, coarctation of the aorta; CV, cardiovascular; ECD, endocardial cushion defect; ECG, electrocardiogram; FBS, fasting blood sugar; GTT, glucose tolerance test; HOCM, hypertrophic obstructive cardiomyopathy; LVH, left ventricular hypertrophy; MR, mitral regurgitation; MVP mitral valve prolapse; PA, pulmonary artery; PDA, patent ductus arteriosus; PFC, persistent fetal circulation; PPHN, persistent pulmonary hypertension of newborn; PS, pulmonary stenosis; TGA, transposition of the great arteries; TOF, tetralogy of Fallot; VSD, ventricular septal defect; XR, sex-linked recessive; ±, may or may not be present.
TABLE 2-2
PREVALENCE OF ASSOCIATED CONGENITAL HEART DEFECTS IN PATIENTS WITH OTHER SYSTEM MALFORMATION
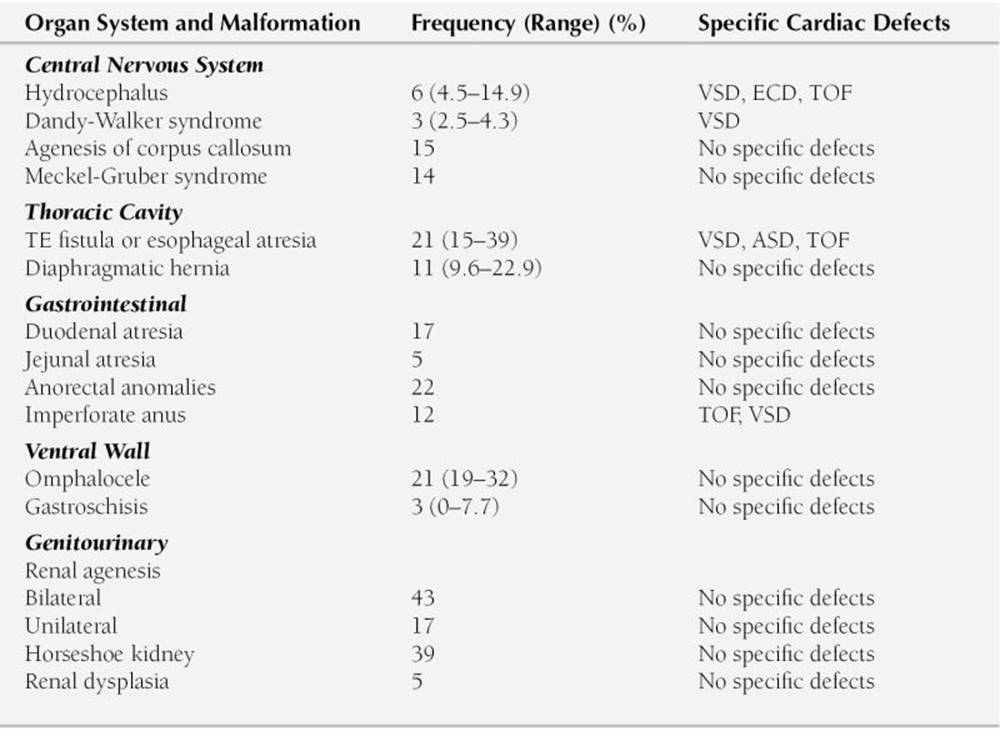
ASD, atrial septal defect; ECD, endocardial cushion defect; TE, trachea-esophageal; TOF, tetralogy of Fallot; VSD, ventricular septal defect.
Modified from Copel JA, Kleinman CS: Congenital heart disease and extracardiac anomalies: Association and indications for fetal echocardiography. Am J Obstet Gynecol 154:1121, 1986.
Respiratory Rate, Dyspnea, and Retraction
The physician should note the respiratory rate of every infant and child. If the infant breathes irregularly, the physician should count for a whole minute. The respiratory rate is faster in children who are crying, upset, eating, or feverish. The most reliable respiratory rate is that taken during sleep. After finishing a bottle of formula, an infant may breathe faster than normal for 5 to 10 minutes. A resting respiratory rate more than 40 breaths/min is unusual, and that more than 60 breaths/min is abnormal at any age. Tachypnea, along with tachycardia, is the earliest sign of left-sided heart failure. If the child has dyspnea or retraction, this may be a sign of a more severe degree of left-sided heart failure or a significant lung pathology.
Sweat on the Forehead
Infants with CHF often have a cold sweat on their foreheads. This is an expression of heightened sympathetic activity as a compensatory mechanism for decreased cardiac output.
Acanthosis Nigricans
Acanthosis nigricans is a dark pigmentation of skin creases most commonly seen on the neck in the majority of obese children and those with type 2 diabetes. It is also found in the axillae, groins, and inner thighs and on the belt line of the abdomen. Rarely, acanthosis occurs in patients with Addison disease, Cushing syndrome, polycystic ovary syndrome (Stein-Leventhal syndrome), hypothyroidism, and hyperthyroidism. This condition is associated with insulin resistance and a higher risk of developing type 2 diabetes.
Inspection of the Chest
Precordial bulge, with or without actively visible cardiac activity, suggests chronic cardiac enlargement. Acute dilatation of the heart does not cause precordial bulge. Pigeon chest (pectus carinatum), in which the sternum protrudes on the midline, is usually not a result of cardiomegaly.
Pectus excavatum (undue depression of the sternum) rarely, if ever, causes significant cardiac embarrassment. Rather, it may be a cause of a pulmonary systolic murmur or a large cardiac silhouette on a posteroanterior view of a chest roentgenogram, which compensates for the diminished anteroposterior diameter of the chest. As a group, children with a significant pectus excavatum have a shorter endurance time than normal children.
Harrison’s groove, a line of depression in the bottom of the rib cage along the attachment of the diaphragm, indicates poor lung compliance of long duration, such as that seen in large left-to-right shunt lesions.
Palpation
Palpation should include the peripheral pulses (their presence or absence, the pulse rate, the volume of the pulses) and the precordium (the presence of a thrill, the point of maximal impulse [PMI], precordial hyperactivity). Although ordinarily palpation follows inspection, auscultation may be more fruitful on a sleeping infant who might wake up and become uncooperative.
Peripheral Pulses
1. The physician should count the pulse rate and note any irregularities in the rate and volume. The normal pulse rate varies with the patient’s age and status. The younger the patient, the faster the pulse rate. Increased pulse rate may indicate excitement, fever, CHF, or arrhythmia. Bradycardia may mean heart block, effects of drugs, and so on. Irregularity of the pulse suggests arrhythmias, but sinus arrhythmia (an acceleration with inspiration) is normal.
2. The right and left arm and an arm and a leg should be compared for the volume of the pulse. Every patient should have palpable pedal pulses, of the dorsalis pedis, tibialis posterior, or both. It is often easier to feel pedal pulses than femoral pulses. Attempts at palpating a femoral pulse often wake up a sleeping infant or upset a toddler. If a good pedal pulse is felt, coarctation of the aorta (COA) is effectively ruled out, especially if the blood pressure (BP) in the arm is normal.
Weak leg pulses and strong arm pulses suggest COA. If the right brachial pulse is stronger than the left brachial pulse, the cause may be COA occurring near the origin of the left subclavian artery or supravalvular aortic stenosis (AS). A weaker right brachial pulse than the left suggests aberrant right subclavian artery arising distal to the coarctation.
3. Bounding pulses are found in aortic run-off lesions such as PDA, aortic regurgitation (AR), large systemic arteriovenous fistula, or persistent truncus arteriosus (rarely). Pulses are bounding in premature infants because of the lack of subcutaneous tissue and because many have PDA.
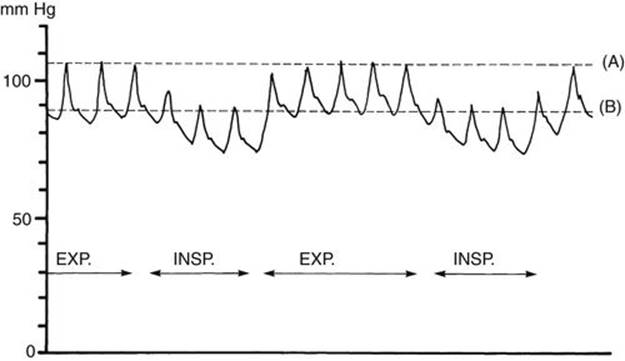
FIGURE 2-2 Diagram of pulsus paradoxus. Note the reduction in systolic pressure of more than 10 mm Hg during inspiration. EXP, expiration; INSP, inspiration.
4. Weak, thready pulses are found in cardiac failure or circulatory shock or in the leg of a patient with COA. A systemic-to-pulmonary artery (PA) shunt (either classic Blalock-Taussig shunt or modified Gore-Tex shunt) or subclavian flap angioplasty for repair of COA may result in an absent or weak pulse in the arm affected by surgery. Arterial injuries resulting from previous cardiac catheterization may cause a weak pulse in the affected limb.
5. Pulsus paradoxus (paradoxical pulse) is suspected when there is marked variation in the volume of arterial pulses with the respiratory cycle. The term pulsus paradoxus does not indicate a phase reversal; rather, it is an exaggeration of normal reduction of systolic pressure during inspiration. When arterial BP is being monitored through an indwelling arterial catheter, the presence of pulsus paradoxus is easily detected by a wide swing (>10 mm Hg) in arterial pressure. In a child without arterial pressure monitoring, accurate evaluation requires sphygmomanometry (Fig. 2-2). Pulsus paradoxus may be associated with cardiac tamponade secondary to pericardial effusion or constrictive pericarditis or to severe respiratory difficulties seen with asthma or pneumonia. It is also seen in patients who are on ventilators with high pressure settings, but in these cases, the BP increases with inflation.
The presence of pulsus paradoxus is confirmed by the use of a sphygmomanometer as described below.
a. The cuff pressure is raised about 20 mm Hg above the systolic pressure.
b. The pressure is lowered slowly until Korotkoff sound 1 is heard for some but not all cardiac cycles, and the reading is noted (line A on Fig. 2-2).
c. The pressure is lowered further until systolic sounds are heard for all cardiac cycles, and the reading is noted (line B on Fig. 2-2).
d. If the difference between readings A and B is greater than 10 mm Hg, pulsus paradoxus is present.
Chest
One should palpate the following on the chest: apical impulse, point of maximal impulse (PMI), hyperactivity of the precordium, and palpable thrill.
Apical Impulse
Palpation of the apical impulse is usually superior to percussion in the detection of cardiomegaly. Its location and diffuseness should be noted. Percussion in infants and children is inaccurate and adds little. The apical impulse is normally at the fifth intercostal space in the midclavicular line after age 7 years. Before this age, the apical impulse is in the fourth intercostal space just to the left of the midclavicular line. An apical impulse displaced laterally or downward suggests cardiac enlargement.
Point of Maximal Impulse
The PMI is helpful in determining whether the RV or left ventricle (LV) is dominant. With RV dominance, the impulse is maximal at the lower left sternal border or over the xiphoid process; with LV dominance, the impulse is maximal at the apex. Normal newborns and infants have RV dominance and therefore more RV impulse than older children. If the impulse is more diffuse and slow rising, it is called a heave. If it is well localized and sharp rising, it is called a tap.Heaves are often associated with volume overload. Taps are associated with pressure overload.
Hyperactive Precordium
The presence of a hyperactive precordium characterizes heart disease with volume overload, such as that seen in defects with large left-to-right shunts (e.g., PDA, VSD) or heart disease with severe valvular regurgitation (e.g., AR, mitral regurgitation [MR]).
Thrills
Thrills are vibratory sensations that represent palpable manifestations of loud, harsh murmurs. Palpation for thrills is often of diagnostic value. A thrill on the chest is felt better with the palm of the hand than with the tips of the fingers. However, the fingers are used to feel a thrill in the suprasternal notch and over the carotid arteries.
1. Thrills in the upper left sternal border originate from the pulmonary valve or PA and therefore are present in PS, PA stenosis, or PDA (rarely).
2. Thrills in the upper right sternal border are usually of aortic origin and are seen in AS.
3. Thrills in the lower left sternal border are characteristic of a VSD.
4. Thrills in the suprasternal notch suggest AS but may be found in PS, PDA, or COA.
5. The presence of a thrill over the carotid artery or arteries accompanied by a thrill in the suprasternal notch suggests diseases of the aorta or aortic valve (e.g., COA, AS). An isolated thrill in one of the carotid arteries without a thrill in the suprasternal notch may be a carotid bruit.
6. Thrills in the intercostal spaces are found in older children with severe COA and extensive intercostal collaterals.
Blood Pressure Measurement
Whenever possible, every child should have his or her BP measured as part of the physical examination. The status of the child at the time of BP measurement, such as moving, crying, or fighting, should be considered in the interpretation of obtained BP values before making any decision about the normalcy of the measurement. When BP is measured in a reasonably quiet situation, an average value of 2 or more BP values is compared with a set of normative BP standards to see if obtained BP values are normal or abnormal. Unfortunately, there have been problems regarding the proper method of measuring BP as well as the normative BP values for children.
Scientifically unsound methods of BP measurement recommended by two previous National Institutes of Health (NIH) Task Forces (1977 and 1987) have dominated the field, and they have been the source of confusion for nearly half a century. At this time, both the methodology and standards recommended by the NIH Task Forces have been abandoned. However, the most recent BP standards recommended by the Working Group of the National High Blood Pressure Education Program (NHBPEP) are still problematic for several reasons.
In this subsection, the following important issues in children’s BP measurement are discussed for a quick overview:
a. What is the currently recommended BP measurement method?
b. How good are normal BP standards recommended by the Working Group of the NHBPEP?
c. Which normal BP standards should be used and why?
d. How accurate are oscillometric BP measurements?
e. Are BP levels obtained by oscillometric devices interchangeable with those obtained by the auscultatory method?
f. How should arm and leg BP values be interpreted in children?
g. What are normative BP levels in neonates and small children?
h. How important is the concept of peripheral amplification of systolic pressure?
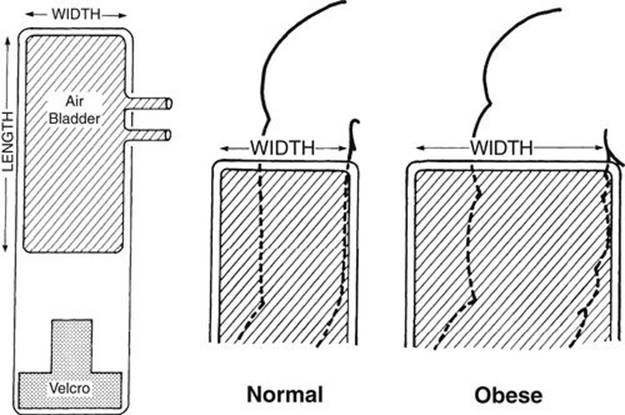
FIGURE 2-3 Diagram showing a method of selecting an appropriate-sized blood pressure cuff. The selection is based on the thickness rather than the length of the arm. The end of the cuff is at the top, and the cuff width is compared with the diameter or circumference of the arm. The width of the inflatable part of the cuff (bladder, cross-hatched areas) should be 125% to 155% of the diameter or 40% to 50% of the circumference of the arm.
1. What is the currently recommended BP measuring method?
In the recent past, two Task Forces of the NIH, 1977 and 1987, recommended BP cuff selection based on the length of the arm, initially recommending the cuff width to be two thirds of the arm length and later changing it to three fourths of the length of the arm. These Task Forces have provided normal BP standards based on these unscientific methods. The BP cuff selection based on the length of the arm is scientifically unsound and violates the physical principles underlying indirect BP measurement, which have been established a century ago. For adults, the Special Task Force of the American Heart Association (AHA) has recommended the correct cuff selection method based on the thickness (or circumference) of the arm, and it has been in use since 1950. The correct width of the BP cuff is 40% to 50% of the circumference of the limb on which the BP is being measured (Fig. 2-3). In 1988, the AHA’s Special Task Force extended the same cuff selection method to children as well, but the 1987 NIH Task Force did not. In 2004, the NHBPEP has accepted the correct BP cuff selection method of the AHA.
The following summarizes current views on BP measurement techniques recommended by the AHA as well as the NHBPEP:
a. The BP cuff width should be 40% to 50% of the circumference (equivalent to 125% to 155% of the diameter) of the extremity with the cuff long enough to completely or nearly completely encircle the extremities (endorsed by both groups).
b. The NHBPEP recommends Korotkoff phase 5 (K5) as the diastolic pressure, but this recommendation is debatable based on a number of earlier reports. Earlier studies indicate that K4 agrees better with direct intraarterial diastolic pressure for children 12 years of age and younger (endorsed by the AHA, NIH Task Forces, and the Bogalusa Heart Study [Hammond et al, 1995]).
c. Both groups recommend averaging two or more readings (because the averaged values are closer to the basal BP level and are more reproducible).
d. Both the AHA and the NHBPEP recommend the sitting position with the arm at the heart level.
2. How good are BP standards recommended by the NHBPEP?
Normative BP standards recommended by the NHBPEP are not as good as it was made to believe for several reasons. Readers should be aware of a few major flaws in the NHBPEP’s normative values.
a. BP data presented in the NHBPEP standards are not obtained by using the same methodology as the program recommends, nor are they from a nationally representative population. They are obtained through an arm’s-length–based cuff selection method, which is currently abandoned because of its unscientific nature. These values are also from single measurement rather than the averages of multiple readings, as currently recommended. In other words, the original source of the elaborate BP standards of the NHBEP is one that has been abandoned by the program itself, yet the program’s recommended BP standards are from these abandoned studies.
b. Expressing children’s BP levels by age and height percentiles is statistically unsound and unjustified on highly variable office BP readings. Height has no statistically important role in children’s BP levels. Partial correlation analysis in the San Antonio Children’s Blood Pressure Study (SACBPS) shows that when auscultatory BP levels were adjusted for age and weight, the correlation coefficient of systolic BP with height was very small (r = 0.068 for boys; r = 0.072 for girls), whereas when adjusted for age and height, the correlation of systolic pressure with weight remained high (r = 0.343 for boys; r = 0.294 for girls). These findings indicate that the contribution of height to BP levels is negligible. The apparent correlation of height to BP levels may all be secondary to a close correlation that exists between height and weight (r = 0.86). A similar conclusion was reached with oscillometric BP levels in the same study. Although weight is very important contributor to BP, weight cannot be used as a second variable because this would interfere with the detection of high BP in obese children. Thus, we found no rationale to use anything other than age and gender to express children’s normative BP standards.
c. Recommending additional computations requiring the use of scientifically unsound complex BP tables on such highly variable office BP readings is unreasonable and unproductive relative to what is to be gained by such efforts. Analyzing unscientifically obtained data by additional computations does not improve their value.
d. The NHBPEP does not point out that the auscultatory and oscillometric BP readings are not interchangeable. SACBPS, in which both auscultatory and oscillometric methods were used, found that oscillometric systolic pressures are significantly higher than auscultatory BP readings (see later discussion for further details). This finding is important in view of the popular use of oscillometric devices in BP measurements in pediatric practice.
e. As a national guideline, the NHBPEP does not emphasize the important contribution of the “white-coat phenomenon” in office BP readings. The white-coat phenomenon refers to the finding that BP readings obtained in a health care facility are often significantly higher than those obtained outside the facility. The white-coat phenomenon may be the most common cause of high BP readings, not true hypertension, in pediatric practice.
3. Which normal BP standards should be used and why?
a. Because the NIH Task Forces’ BP standards (of 1987) were obtained by using the unscientific, arm’s-length–based, cuff selection method, these BP standards are no longer acceptable.
b. The BP standards of the NHBPEP (2004) are riddled with major flaws as discussed earlier; they are not the best standards. Health care providers who choose to use these standards should be aware of the flaws. Although not acceptable as reliable pediatric BP standards, the NHBPEP’s normative values are presented in Appendix B for the sake of completeness (Tables B-1 and B-2).
c. Normative BP percentile values from SACBPS are recommended as a better alternative to BP standards than the NHBPEP’s standards until nationwide data using the currently recommended methods become available. These are the only available BP standards that have been obtained according to the currently recommended method. In the SACBPS, BP levels were obtained in more than 7000 schoolchildren of three ethnic groups (African American, Mexican American, and non-Hispanic white) enrolled in kindergarten through the 12th grade in the San Antonio, Texas, area. Both the auscultatory and oscillometric (model Dinamap 8100) methods were used in the study, and the data were the averages of three readings. No consistent ethnic difference was found among the three ethnic groups, but there were important gender differences. Auscultatory BP data were expressed according to age and gender (Park, 2001). These BP standards are normally distributed from the mean value, and thus the effect of obesity is not a problem in using the standards (Figs. 2-4 and 2-5). Percentile BP values for these figures are presented in Appendix B (Tables B-3 and B-4).
d. When BP is measured using an oscillometric device, one should use device-specific normative BP standards. SACBPS found that the readings by the auscultatory method and those by Dinamap 8100 are significantly different and thus are not interchangeable (Park et al, 2005) as will be described further. This study does not show superiority of any one of the devices but simply indicates that the indirect BP readings are different according to the sensitivity of the detection device used. It may imply that the detection of oscillation by the machine occurs earlier (at a higher level of systolic pressure) than what human ears can hear. The gold standard remains an intraarterial BP reading. Percentile BP values by an oscillometric method (Dinamap 8100) are presented in Tables B-6 and B-7).
4. How accurate are oscillometric BP measurements?
The accuracy of indirect BP measurement by an oscillometric method (Dinamap Model 1846) has been demonstrated. In fact, oscillometric BP levels correlated better with intraarterial pressures than the auscultatory method (Park et al, 1987). Averaging three BP readings makes the BP readings more reproducible. The arm circumference–based cuff selection method is also appropriate for the Dinamap method. The oscillometric method also provides some advantages over auscultation: it eliminates observer-related variations, and it can be successfully used in infants and small children. Auscultatory BP measurement in small infants is not only difficult to obtain but also has not been shown to be accurate. The percentile values of normative oscillometric BPs in neonates and children up to 5 years of age are presented in Appendix B (Table B-5). Aside from the issue of accuracy, the oscillometric method is widely used in large pediatric and pediatric cardiology practices and in the setting of emergency departments. One caution is that not all oscillometric devices in clinical use have been validated for their accuracy.
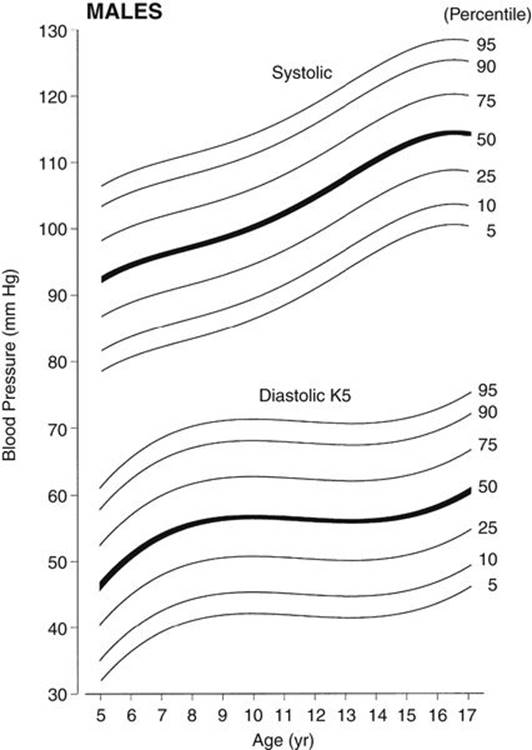
FIGURE 2-4 Age-specific percentile curves of auscultatory systolic and diastolic (K5) pressures in boys 5 to 17 years of age. Blood pressure values are the average of three readings. The width of the blood pressure cuff was 40% to 50% of the circumference of the arm. Percentile values for the figure are shown in Table B-3, Appendix B. (From Park MK, Menard SW, Yuan C: Comparison of blood pressure in children from three ethnic groups. Am J Cardiol 87:1305-1308, 2001.)
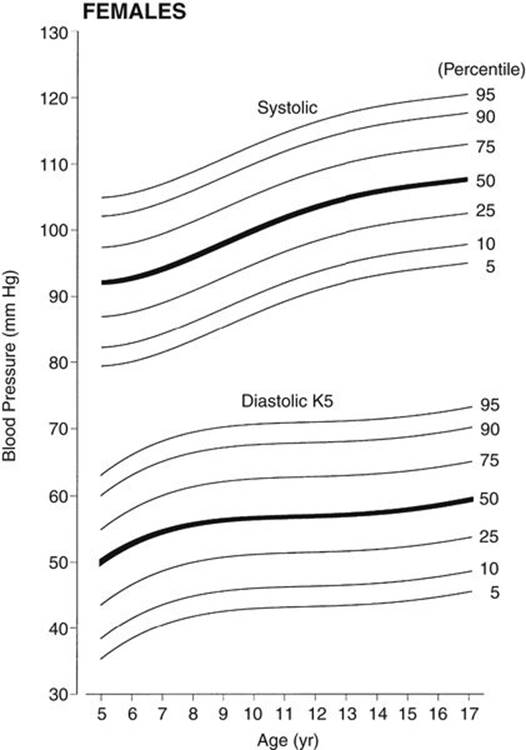
FIGURE 2-5 Age-specific percentile curves of auscultatory systolic and diastolic (K5) pressures in girls 5 to 17 years of age. Blood pressure values are the average of three readings. The width of the blood pressure cuff was 40% to 50% of the circumference of the arm. Percentile values for the figure are shown in Table B-4, Appendix B. (From Park MK, Menard SW, Yuan C: Comparison of blood pressure in children from three ethnic groups. Am J Cardiol 87:1305-1308, 2001.)
5. Are BP levels obtained by oscillometric devices interchangeable with those obtained by the auscultatory method?
We found that BP levels obtained by the Dinamap (Model 8100) were on the average 10 mm Hg higher than the auscultatory method for the systolic pressure and 5 mm Hg higher for the diastolic pressure (Park et al, 2001). Therefore, the auscultatory and the Dinamap BPs are not interchangeable. This necessitates oscillometric-specific normative BP standards so that one does not use normal auscultatory BP standards when the oscillometric method is used (Park et al, 2005). Dinamap 8100–specific BP standards as presented in Appendix B (see Tables B-6 and B-7).
6. How should arm and leg BP values be interpreted in children?
Four-extremity BP measurements are often obtained to rule out COA. The same cuff selection criterion (i.e., 40% to 50% of the circumference) applies for calf or thigh pressure determination, often requiring the use of a larger cuff for the lower extremity. The patient should be in the supine position for BP measurements in the arm and leg. When using the auscultatory method, the thigh pressure is obtained with the stethoscope placed in the popliteal fossa (over the popliteal artery) with the legs bent and in the supine or prone position. Calf BP is difficult to obtain by the auscultatory method.
TABLE 2-3
NORMATIVE BLOOD PRESSURE LEVELS BY DINAMAP MONITOR IN CHILDREN AGE 5 YEARS AND YOUNGER
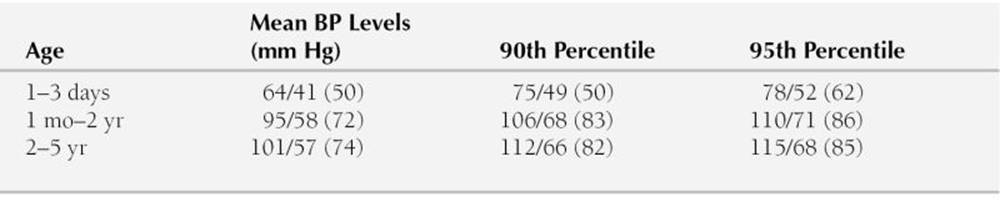
Dinamap Model 1846SX was used.
Blood pressure (BP) levels are systolic/diastolic, with the mean in parentheses.
Modified from Park MK, Menard SM: Normative oscillometric blood pressure values in the first 5 years in an office setting. Am J Dis Child 143:860, 1989.
How do BP levels in the arm and leg compare in normal children? Even when a considerably wider cuff is selected for the thigh, the Dinamap systolic pressure in the thigh or calf is about 5 to 10 mm Hg higher than that in the arm (Park et al, 1993) except in the newborns in whom the arm and calf pressures are the same (see later discuss). This partly reflects the peripheral amplification of systolic pressure (see later discussion). Thus, the systolic pressure in the thigh (or calf) should be higher than or at least equal to that in the arm except in newborns. If the systolic pressure is lower in the leg, COA may be present. Leg BP determinations are mandatory in children with hypertension in the arm to rule out coarctation of he aorta. The presence of a femoral pulse does not rule out a coarctation.
7. What are normative BP levels in neonates and small children?
Blood pressure measurement is important in newborns and small children to diagnose COA, hypertension, or hypotension. In contrast to the recommendations of the NHBEP, the auscultatory method is difficult to apply in newborns and small children because of weak Korotkoff sounds in these age groups, and thus normative standards are not reliable. Therefore, the oscillometric method is frequently used instead. Abbreviated normative Dinamap BP standards for newborns and small children (≤5 years) are presented in Table 2-3. Full percentile values are presented in Appendix B (Table B-5). The same BP cuff selection method as used in older children applies to this age group (i.e., the cuff width approximately 50% of the circumference of the extremity). In newborns, the systolic pressures in the arm and the calf are the same (Park et al, 1989). The absence of a higher systolic pressure in the leg in newborns may be related to the presence of a normally narrow aortic isthmus.
8. How important is the concept of peripheral amplification of systolic pressure?
Many physicians incorrectly assume that peripherally measured BP, such as that measured in the arm, reflects central aortic pressure, which is the perfusing pressure for the brain. This assumption is incorrect in many clinical situations. Some physicians also incorrectly think that the systolic pressure in the central aorta is higher than that in the brachial, radial, and pedal arteries. As shown schematically in Figure 2-6, systolic pressure becomes higher and higher as one moves further peripherally, although the diastolic and mean pressures remain the same or decrease slightly (O’Rourke, 1968). This phenomenon is known as peripheral amplification of systolic pressure. If this is correct, how does blood flow distally? There is a change in the arterial pressure wave form at different levels in an arterial tree, as shown in Figure 2-6, but the area under the curve decreases slightly in the peripheral sites, so that the blood flows to peripheral sites.
It is important for physicians to understand that peripheral systolic pressures obtained, either by direct or indirect methods, do not always reflect the central aortic pressure. Peripherally obtained systolic pressures are usually higher than the central aortic pressure. The magnitude of difference in the systolic pressure between a peripheral artery and the central aorta is not always predictable. There are some clinically important situations in which the peripheral amplification becomes more marked. The following are some key points of the peripheral amplification of systolic pressure:
a. The amplification is limited to systolic pressure only (but not in diastolic and mean pressures).
b. The systolic amplification is greater in children (with more reactive arteries) than in older adults who may have degenerative arterial disease.
c. Pedal artery systolic pressures are higher than the radial artery pressures.
d. The amplification is more marked in vasoconstricted states, and many of them are clinically important.
1) In impending circulatory shock, in which a high level of circulating catecholamines is present, systolic amplification may become more marked. Early diagnosis of an impending circulatory shock can be missed if one pays attention only to systolic pressure; the mean arterial and diastolic pressures should be low in this situation.
2) A child in CHF (in which peripheral vasoconstriction exists) may exhibit an exaggerated systolic amplification. Mean arterial pressure (MAP) may be more meaningful.
3) Arm systolic pressure in subjects running on a treadmill can be markedly higher than the central aortic pressure. A dramatic illustration of this phenomenon is shown in Figure 6-1 (section on exercise stress test) in a young adult running on a treadmill with catheters in the ascending aorta and the radial artery.
4) Subjects receiving catecholamine infusion or other vasoconstrictors in the setting of critical care units may show significant amplification of systolic pressure. MAP may be more meaningful in this situation.
e. A reduced level of peripheral amplification of systolic pressure is noted in vasodilated states.
1) Subjects receiving vasodilators
2) After receiving a contract dye injection (which has vasodilating effects) during cardiac catheterization
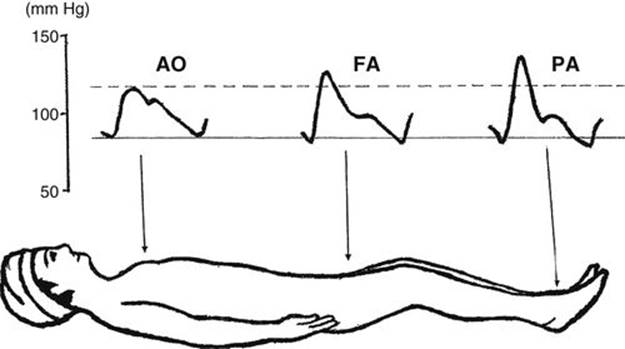
FIGURE 2-6 Schematic diagram of pulse wave changes at different levels of the systemic arteries. AO, aorta; FA, femoral artery; PA, pedal artery. (Modified from Geddes LA: Handbook of Blood Pressure Measurement. Clifton, NJ: Humana Press, 1991.)
Auscultation
Although auscultation of the heart requires more skill, it also provides more valuable information than other methods of physical examination. Whereas the bell-type chest piece is better suited for detecting low-frequency events, the diaphragm selectively picks up high-frequency events. When the bell is firmly pressed against the chest wall, it acts like the diaphragm by filtering out low-frequency sounds or murmurs and picking up high-frequency events. Physicians should ordinarily use both the bell and the diaphragm, although using the bell both lightly and firmly pressed against the chest may be equally effective, especially in sleeping infants. Using only the diaphragm may result in missing some important low-frequency murmurs or sounds, such as mid-diastolic rumble, pulmonary regurgitation (PR) murmur, and faint Still’s innocent heart murmurs. One should not limit examination to the four traditional auscultatory areas. The entire precordium, as well as the sides and back of the chest, should be explored with the stethoscope. Systematic attention should be given to the following aspects:
1. Heart rate and regularity: Heart rate and regularity should be noted on every child. Extremely fast or slow heart rates or irregularity in the rhythm should be evaluated by an electrocardiogram (ECG) and a long rhythm strip.
2. Heart sounds: Intensity and quality of the heart sounds, especially the second heart sound (S2), should be evaluated. Abnormalities of the first heart sound (S1) and the third heart sound (S3) and the presence of a gallop rhythm or the fourth sound (S4) should be noted. Muffled heart sounds should also be noted.
3. Systolic and diastolic sounds: An ejection click in early systole provides a clue to aortic or pulmonary valve stenosis. A midsystolic click provides important clues to the diagnosis of mitral valve prolapse (MVP). An opening snap in diastole (present in mitral stenosis [MS]) should be noted but it is extremely rare in children.
4. Heart murmurs: Heart murmurs should be evaluated in terms of intensity, timing (systolic or diastolic), location, transmission, and quality.
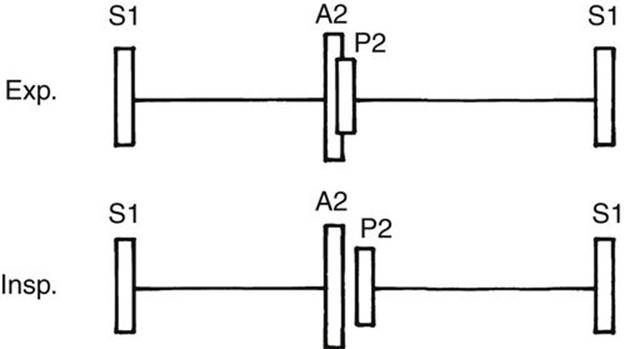
FIGURE 2-7 Diagram showing the relative intensity of A2 and P2 and the respiratory variation in the degree of splitting of the S2 at the upper left sternal border (pulmonary area). Exp, expiration; Insp, inspiration.
Heart Sounds
The heart sound should be identified and analyzed before the analysis of heart murmurs (Fig. 2-7). Muffled and distant heart sounds are present in pericardial effusion and heart failure.
First Heart Sound
The S1 is associated with closure of the mitral and tricuspid valves. It is best heard at the apex or lower left sternal border. Splitting of the S1 may be found in normal children, but it is infrequent. Abnormally wide splitting of S1 may be found in right bundle branch block (RBBB) or Ebstein’s anomaly. Splitting of S1 should be differentiated from ejection click or S4.
1. Ejection click is more easily audible at the upper left sternal border in PS. In bicuspid aortic valve, the click may be louder at the lower left sternal border or apex than at the upper right sternal border.
2. S4 is rare in children.
Second Heart Sound
The S2 in the upper left sternal border (i.e., pulmonary valve area) is of critical importance in pediatric cardiology. The S2 must be evaluated in terms of the degree of splitting and the intensity of the pulmonary closure component of the second heart sound (P2) in relation to the intensity of the aortic closure component of the second heart sound (A2). Although best heard with the diaphragm of a stethoscope, both components are readily audible with the bell. Abnormalities of splitting of the S2 and the intensity of the P2 are summarized in Box 2-1.
BOX 2-1 Summary of Abnormal S2
Abnormal Splitting
Widely Split and Fixed S2
Volume overload (e.g., ASD, PAPVR)
Pressure overload (e.g., PS)
Electrical delay (e.g., RBBB)
Early aortic closure (e.g., MR)
Occasional normal child
Narrowly Split S2
Pulmonary hypertension
AS
Occasional normal child
Single S2
Pulmonary hypertension
One semilunar valve (e.g., pulmonary atresia, aortic atresia, persistent truncus arteriosus)
P2 not audible (e.g., TGA, TOF, severe PS)
Severe AS
Occasional normal child
Paradoxically split S2
Severe AS
LBBB, WPW syndrome (type B)
Abnormal Intensity of P2
Increased P2 (e.g., pulmonary hypertension)
Decreased P2 (e.g., severe PS, TOF, TS)
AS, aortic stenosis; ASD, atrial septal defect; LBBB, left bundle branch block; MR, mitral regurgitation; PAPVR, partial anomalous pulmonary venous return; PS, pulmonary stenosis; RBBB, right bundle branch block; TGA, transposition of the great arteries; TOF, tetralogy of Fallot; TS, tricuspid stenosis; WPW, Wolff-Parkinson-White.
Splitting of the S2
In every normal child, with the exception of occasional newborns, two components of the S2 should be audible in the upper left sternal border. The first is the A2; the second is the P2. Both components are best heard at the pulmonary valve area (upper left sternal border). The A2 is not the second heart sound at the aortic area; rather, it is the first (or aortic closure) component of the second heart sound at the pulmonary area.
Normal Splitting of the S2
The degree of splitting of the S2 varies with respiration, increasing with inspiration and decreasing or becoming single with expiration (see Fig. 2-7). During inspiration, because of a greater negative pressure in the thoracic cavity, there is an increase in systemic venous return to the right side of the heart. This increased volume of blood in the RV prolongs the duration of RV ejection time, which delays the closure of the pulmonary valve, resulting in a wide splitting of the S2. The absence of splitting (i.e., single S2) or a widely split S2 usually indicates an abnormality.
Abnormal Splitting of the S2
Abnormal splitting may be in the form of wide splitting, narrow splitting, a single S2, or paradoxical splitting of the S2 (rarely).
1. A widely split and fixed S2 is found in conditions that prolong the RV ejection time or that shorten the LV ejection. Therefore, it is found in:
a. ASD or partial anomalous pulmonary venous return (PAPVR) (conditions in which the amount of blood ejected by the RV is increased; volume overload)
b. PS (the valve stenosis prolongs the RV ejection time; pressure overload)
c. RBBB (a delay in electrical activation of the RV) delays the completion of the RV ejection
d. MR (a decreased forward output seen in this condition shortens the LV ejection time, causing aortic closure to occur earlier than normal)
e. An occasional normal child, including “prolonged hangout time” seen in children with dilated PA (a condition called idiopathic dilatation of the PA). In dilated PA, the increased capacity of the artery produces less recoil to close the pulmonary valve, which delays closure.
2. A narrowly split S2 is found in conditions in which the pulmonary valve closes early (e.g., pulmonary hypertension) or the aortic valve closure is delayed (e.g., AS). This is occasionally found in a normal child.
3. A single S2 is found in the following situations:
a. When only one semilunar valve is present (e.g., aortic or pulmonary atresia, persistent truncus arteriosus)
b. When the P2 is not audible (e.g., transposition of the great arteries [TGA], TOF, severe PS)
c. When aortic closure is delayed (e.g., severe AS)
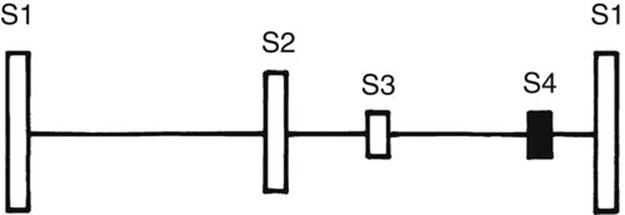
FIGURE 2-8 Diagram showing the relative relationship of the heart sounds. The filled bar shows an abnormal sound.
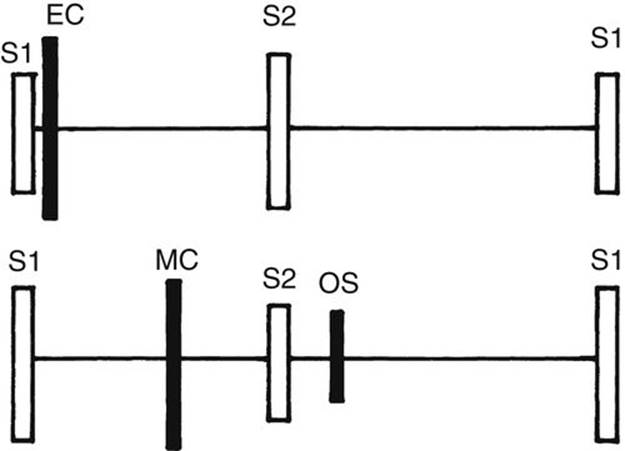
FIGURE 2-9 Diagram showing the relative position of ejection click (EC), midsystolic click (MC), and diastolic opening snap (OS). Filled bars are abnormal sounds.
d. When the P2 occurs early (e.g., severe pulmonary hypertension)
e. In an occasional normal child
4. A paradoxically split S2 is found when the aortic closure (A2) follows the pulmonary closure (P2) and therefore is seen when the LV ejection is greatly delayed (e.g., severe AS, left bundle branch block [LBBB], sometimes Wolff-Parkinson-White [WPW] pre-excitation).
Intensity of the P2
The relative intensity of the P2 compared with the A2 must be assessed in every child. In the pulmonary area, the A2 is usually louder than the P2 (see Fig. 2-7). Judgment as to normal intensity of the P2 is based on experience. There is no substitute for listening to the hearts of many normal children. Abnormal intensity of the P2 may suggest a pathologic condition. Increased intensity of the P2, compared with that of the A2, is found in pulmonary hypertension. Decreased intensity of the P2 is found in conditions with decreased diastolic pressure of the PA (e.g., severe PS, TOF, tricuspid atresia).
Third Heart Sound
The S3 is a somewhat low-frequency sound in early diastole and is related to rapid filling of the ventricle (Fig. 2-8). It is best heard at the apex or lower left sternal border. It is commonly heard in normal children and young adults. A loud S3 is abnormal and is audible in conditions with dilated ventricles and decreased ventricular compliance (e.g., large-shunt VSD or CHF). When tachycardia is present, it forms a “Kentucky” gallop.
Fourth Heart Sound or Atrial Sound
The S4 is a relatively low-frequency sound of late diastole (i.e., presystole) and is rare in infants and children (see Fig. 2-8). When present, it is always pathologic and is seen in conditions with decreased ventricular compliance or CHF. With tachycardia, it forms a “Tennessee” gallop.
Gallop Rhythm
A gallop rhythm is a rapid triple rhythm resulting from the combination of a loud S3, with or without an S4, and tachycardia. It generally implies a pathologic condition and is commonly present in CHF. A summation gallop represents tachycardia and a superimposed S3 and S4.
Systolic and Diastolic Sounds
1. An ejection click (or ejection sound) follows the S1 very closely and occurs at the time of the ventricular ejection’s onset. Therefore, it sounds like a splitting of the S1. However, whereas it is usually audible at the base (either side of the upper sternal border), the split S1 is usually audible at the lower left sternal border (exception with an aortic click, discussed in a later section). If the physician hears what sounds like a split S1 at the upper sternal border, it may be an ejection click (Fig. 2-9).
The pulmonary click is heard at the second and third left intercostal spaces and changes in intensity with respiration, being louder on expiration. The aortic click is best heard at the second right intercostal space but may be louder at the apex or mid-left sternal border. It usually does not change its intensity with respiration. The ejection click is most often associated with:
a. Stenosis of semilunar valves (e.g., PS or AS)
b. Dilated great arteries, which are seen in systemic or pulmonary hypertension, idiopathic dilatation of the PA, TOF (in which the aorta is dilated), and persistent truncus arteriosus
2. Midsystolic click with or without a late systolic murmur is heard at the apex in MVP (see Fig. 2-9 and Chapter 21).
3. Diastolic opening snap is rare in children and is audible at the apex or lower left sternal border. It occurs somewhat earlier than the S3 during diastole and originates from a stenosis of the atrioventricular (AV) valve, such as MS (see Fig. 2-9).
Extracardiac Sounds
1. A pericardial friction rub is a grating, to-and-fro sound produced by friction of the heart against the pericardium. This sounds similar to sandpaper rubbed on wood. Such a sound usually indicates pericarditis. The intensity of the rub varies with the phase of the cardiac cycle rather than the respiratory cycle. It may become louder when the patient leans forward. Large accumulation of fluid (pericardial effusion) may result in disappearance of the rub.
2. A pericardial knock is an adventitious sound associated with chronic (i.e., constrictive) pericarditis. It rarely occurs in children.
Heart Murmurs
Each heart murmur must be analyzed in terms of intensity (grade 1 to 6), timing (systolic or diastolic), location, transmission, and quality (musical, vibratory, blowing, and so on).
Intensity
Intensity of the murmur is customarily graded from 1 to 6:
Grade 1: Barely audible
Grade 2: Soft but easily audible
Grade 3: Moderately loud but not accompanied by a thrill
Grade 4: Louder and associated with a thrill
Grade 5: Audible with the stethoscope barely on the chest
Grade 6: Audible with the stethoscope off the chest
The difference between grades 2 and 3 or grades 5 and 6 may be somewhat subjective. The intensity of the murmur may be influenced by the status of cardiac output. Thus, any factor that increases the cardiac output (e.g., fever, anemia, anxiety, exercise) intensifies any existing murmur or may even produce a murmur that is not audible at basal conditions.
Classification of Heart Murmurs. Based on the timing of the heart murmur in relation to the S1 and S2, the heart murmur is classified into systolic, diastolic, and continuous murmurs.
Systolic Murmurs
Most heart murmurs are systolic in timing in that they occur between the S1 and S2. Systolic murmurs was classified by Aubrey Leatham in 1958 into two subtypes according to the time of onset: (1) ejection type and (2) regurgitant type (Fig. 2-10). Recently, Joseph Perloff classified systolic murmurs according to their time of onset and termination into four subtypes: (1) midsystolic (or ejection), (2) holosystolic, (3) early systolic, or (4) late systolic (Fig. 2-11). The holosystolic and early systolic murmurs of Perloff belong to the regurgitant murmur of Leatham. The types of murmurs (described earlier) and the location of the maximum intensity of the murmur are very important in the assessment of a systolic murmur. Transmission of the murmur to a particular direction and the quality of the murmur also help in deciding the cause of the murmur. These aspects will be discussed in detail below.
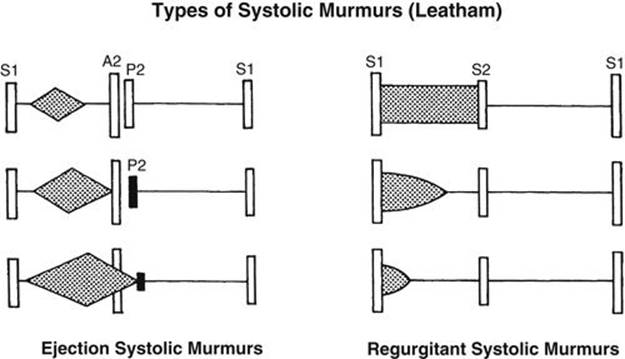
FIGURE 2-10 Diagram of Leatham’s classification of systolic murmurs. This classification is based primarily on the relationship of the S1 to the onset of the murmur. Short ejection-type murmur with the apex of the “diamond” in the early part of systole is found with mild stenosis of semilunar valves (top, left). With increasing severity of stenosis, the murmur becomes louder and longer, and its apex moves toward the S2 (middle, left). In severe pulmonary stenosis, the murmur is louder and may go beyond the A2 (bottom, left). A regurgitant systolic murmur is most often due to ventricular septal defect (VSD) and is usually holosystolic, extending all the way to the S2 (top, right). The regurgitant murmur may end in middle or early systole (not holosystolic) in some children, especially in those with small-shunt VSD and in some neonates with VSD (middle and bottom, right). Regardless of the length or intensity of the murmur, all regurgitant systolic murmurs are pathologic.
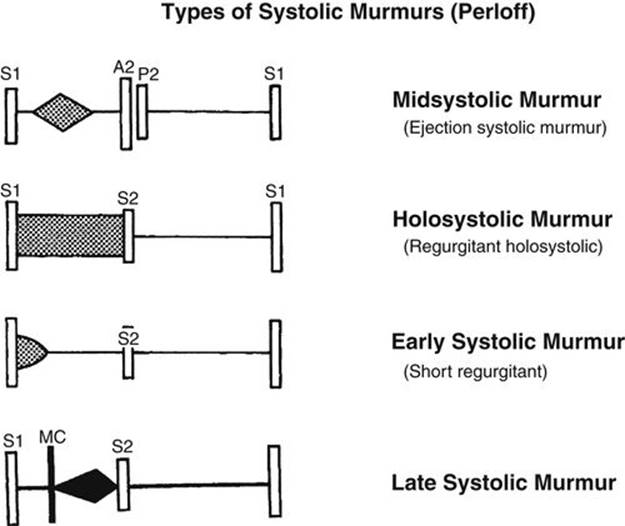
FIGURE 2-11 Diagram of Perloff’s classification of systolic murmurs. Midsystolic murmur is the same as ejection systolic murmur of Leatham. Holosystolic and early systolic murmurs are both regurgitant murmurs of Leatham. Late systolic murmur is typically audible with mitral valve prolapse.
Types of Systolic Murmurs. Midsystolic (or Ejection Systolic) Murmurs
A midsystolic murmur (or ejection-type murmur) begins after S1 and ends before S2. Midsystolic murmurs coincide with turbulent flow through the semilunar valves and they occur in the following settings: (1) the flow of blood through stenotic or deformed semilunar valves (e.g., AS or PS); (2) accelerated systolic flow through normal semilunar valves, such as seen during pregnancy, fever, anemia, or thyrotoxicosis; and (3) innocent (normal) midsystolic murmurs (see Innocent Heart Murmurs later in this section). There is an interval between the S1 and the onset of the murmur that coincides with the isovolumic contraction period. The intensity of the murmur increases toward the middle and then decreases during systole (crescendo–decrescendo or diamond shaped in contour). The murmur usually ends before the S2 (see Fig. 2-10, left). The murmur may be short or long and is audible at the second left or second right intercostal space.
Holosystolic Murmurs
Holosystolic murmurs begin with S1 and occupy all of systole up to the S2. No gap exists between the S1 and the onset of the murmur. Analysis of the presence or absence of a gap between the S1 and the onset of the systolic murmur is of utmost importance in distinguishing between midsystolic murmurs and holosystolic or early systolic murmurs. Holosystolic and early systolic murmurs of Perloff are regurgitant systolic murmurs of Leatham. The intensity of holosystolic murmurs usually plateaus all the way to the S2. Holosystolic murmurs are caused by the flow of blood from a chamber that is at a higher pressure throughout systole than the receiving chamber, and they usually occur while the semilunar valves are still closed. These murmurs are associated with only the following three conditions: VSD, MR, and tricuspid regurgitation (TR). None of these ordinarily occurs at the base (i.e., second left or right intercostal space).
Early Systolic Murmurs
Early systolic murmurs (or short regurgitant murmurs) begin with the S1, diminish in decrescendo, and end well before the S2, generally at or before midsystole (see Fig. 2-11). Only the three conditions that cause holosystolic murmurs (VSD, MR, and TR) are the causes of an early systolic murmur. An early systolic murmur is a feature of TR with normal RV systolic pressure. When the RV systolic pressure is elevated, a holosystolic murmur results. Early systolic murmurs may occur in a neonate with a large VSD, children or adults with a very small VSD, or with a large VSD and pulmonary hypertension.
Late Systolic Murmurs
The term late systolic applies when a murmur begins in mid- to late systole and proceeds up to the S2 (see Fig. 2-11). The late systolic murmur of MVP is prototypical (see Chapter 21).
Location of Systolic Murmurs
In addition to the type of systolic murmur, the location of maximal intensity of the murmur is important when diagnosing the heart murmur’s origin. The following four locations are important: (1) upper left sternal border (pulmonary valve area), (2) upper right sternal border (aortic valve area), (3) lower left sternal border, and (4) the apex. For example, a holosystolic murmur heard maximally at the lower left sternal border is characteristic of a VSD. A midsystolic murmur maximally audible at the second left intercostal space is usually pulmonary in origin. The location of the heart murmur often helps differentiate between a midsystolic murmur and a holosystolic murmur. For example, a long PS murmur may sound like the holosystolic murmur of a VSD; however, because the maximal intensity is at the upper left sternal border, it is unlikely that a VSD caused the murmur. Although rare, a subarterial infundibular VSD murmur may be maximally heard at the upper left sternal border. The differential diagnosis of systolic murmurs according to the location is discussed in detail in this section (see Tables 2-4 to 2-7; Fig. 2-12).
Transmission of Systolic Murmurs
The transmission of systolic murmurs from the site of maximal intensity may help determine the murmur’s origin. For example, an apical systolic murmur that transmits well to the left axilla and lower back is characteristic of MR, but one that radiates to the upper right sternal border and the neck is more likely to originate in the aortic valve. A systolic ejection murmur at the base that transmits well to the neck is more likely to be aortic in origin; one that transmits well to the back is more likely to be of pulmonary valve or PA origin.
Quality of Systolic Murmurs
The quality of a murmur may help diagnose heart disease. Systolic murmurs of MR or of a VSD have a uniform, high-pitched quality, often described as blowing. Midsystolic murmurs of AS or PS have a rough, grating quality. A common innocent murmur in children, which is best audible between the lower left sternal border and apex, has a characteristic “vibratory” or humming quality.
Differential Diagnosis of Systolic Murmurs at Various Locations
Systolic murmurs that are audible at the four locations are presented in Figure 2-12. More common conditions are listed in larger type and less common conditions in smaller type. For quick reference, characteristic physical, ECG, and radiography findings that are helpful in differential diagnoses are listed in Tables 2-4 through 2-7.
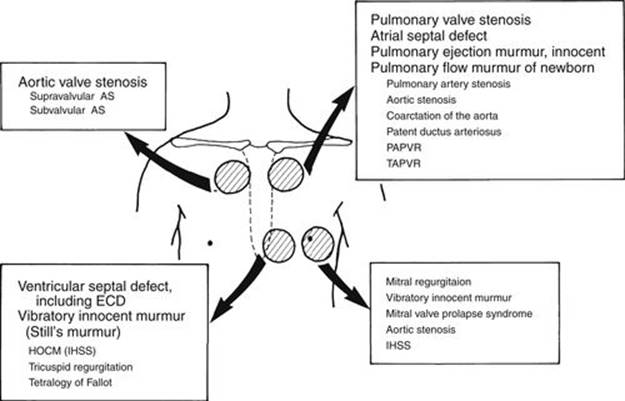
FIGURE 2-12 Diagram showing systolic murmurs audible at various locations. Less common conditions are shown in smaller type (see Tables 2-4 to 2-7). AS, aortic stenosis; ECD, endocardial cushion defect; HOCM, hypertrophic obstructive cardiomyopathy; IHSS, idiopathic hypertrophic subaortic stenosis; PAPVR, partial anomalous pulmonary venous return; TAPVR, total anomalous pulmonary venous return.
TABLE 2-4
DIFFERENTIAL DIAGNOSIS OF SYSTOLIC MURMURS AT THE UPPER LEFT STERNAL BORDER (PULMONARY AREA)
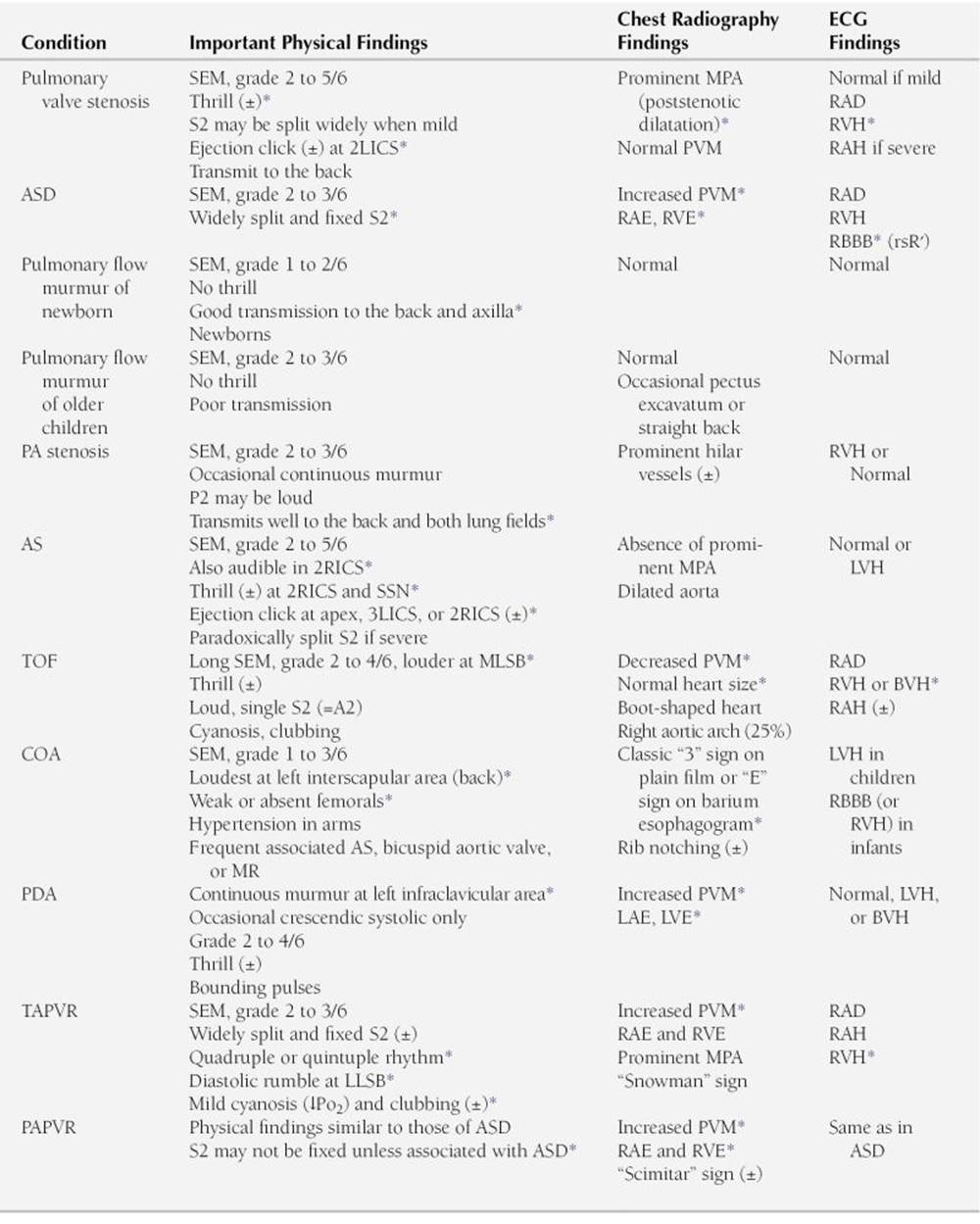
AS, aortic stenosis; ASD, atrial septal defect; BVH, biventricular hypertrophy; COA, coarctation of the aorta; LAE, left atrial enlargement; 2LICS, second left intercostal space; 3LICS, third left intercostal space; LLSB, lower left sternal border; LVE, left ventricular enlargement; LVH, left ventricular hypertrophy; MLSB, mid-left sternal border; MPA, main pulmonary artery; MR, mitral regurgitation; PA, pulmonary artery; PAPVR, partial anomalous pulmonary venous return; PDA, patent ductus arteriosus; PVM, pulmonary vascular markings; RAD, right axis deviation; RAE, right atrial enlargement; RAH, right atrial hypertrophy; RBBB, right bundle branch block; 2RICS, second right intercostal space; RVE, right ventricular enlargement; RVH, right ventricular hypertrophy; SEM, systolic ejection murmur; SSN, suprasternal notch; TAPVR, total anomalous pulmonary venous return; TOF, tetralogy of Fallot; ±, may or may not be present.
∗ Findings that are characteristic of the condition.
TABLE 2-5
DIFFERENTIAL DIAGNOSIS OF SYSTOLIC MURMURS AT THE UPPER RIGHT STERNAL BORDER (AORTIC AREA)
AR, aortic regurgitation; BP, blood pressure; BVH, biventricular hypertrophy; 3LICS, third left intercostal space; LVE, left ventricular enlargement; LVH, left ventricular hypertrophy; PA, pulmonary artery; 2RICS, second right intercostal space; SEM, systolic ejection murmur; SSN, suprasternal notch; URSB, upper right sternal border; ±, may or may not be present.
∗ Findings that are characteristic of the condition.
TABLE 2-6
DIFFERENTIAL DIAGNOSIS OF SYSTOLIC MURMURS AT THE LOWER LEFT STERNAL BORDER

AV, atrioventricular; BVH, biventricular hypertrophy; ECD, endocardial cushion defect; HOCM, hypertrophic obstructive cardiomyopathy; IHSS, idiopathic hypertrophic subaortic stenosis; LAE, left atrial enlargement; LLSB, lower left sternal border; LVE, left ventricular enlargement; LVH, left ventricular hypertrophy; MR, mitral regurgitation; PVM, pulmonary vascular markings; RAE, right atrial enlargement; RAH, right atrial hypertrophy; RBBB, right bundle branch block; SEM, systolic ejection murmur; TR, tricuspid regurgitation; TOF, tetralogy of Fallot; ULSB, upper left sternal border; VSD, ventricular septal defect; ±, may or may not be present.
∗ Findings that are characteristic of the condition.
TABLE 2-7
DIFFERENTIAL DIAGNOSIS OF SYSTOLIC MURMURS AT THE APEX

HOCM, hypertrophic obstructive cardiomyopathy; IHSS, idiopathic hypertrophic subaortic stenosis; LAE, left atrial enlargement; LAH, left atrial hypertrophy; LVE, left ventricular enlargement; LVH, left ventricular hypertrophy; MR, mitral regurgitation; MVP, mitral valve prolapse; 2RICS, second right intercostal space.
∗ Findings that are characteristic of the condition.
1. Upper left sternal border (or pulmonary area)
In many conditions, both pathologic and physiologic (i.e., innocent murmur), a systolic murmur is most audible at the upper left sternal border. Audible systolic murmurs at this location are usually midsystolic murmurs and may be the result of one of the following:
a. PS
b. ASD
c. Innocent (normal) pulmonary flow murmur of newborns
d. Innocent pulmonary flow murmur of older children
e. PA stenosis
f. AS
g. TOF
h. COA
i. PDA with pulmonary hypertension (a continuous murmur of a PDA is usually loudest in the left infraclavicular area)
j. Total anomalous pulmonary venous return (TAPVR)
k. PAPVR
Conditions a through d are more common than the other listed conditions. Table 2-4 summarizes other clinical findings that are useful in the differential diagnosis of systolic murmurs audible at the upper left sternal border.
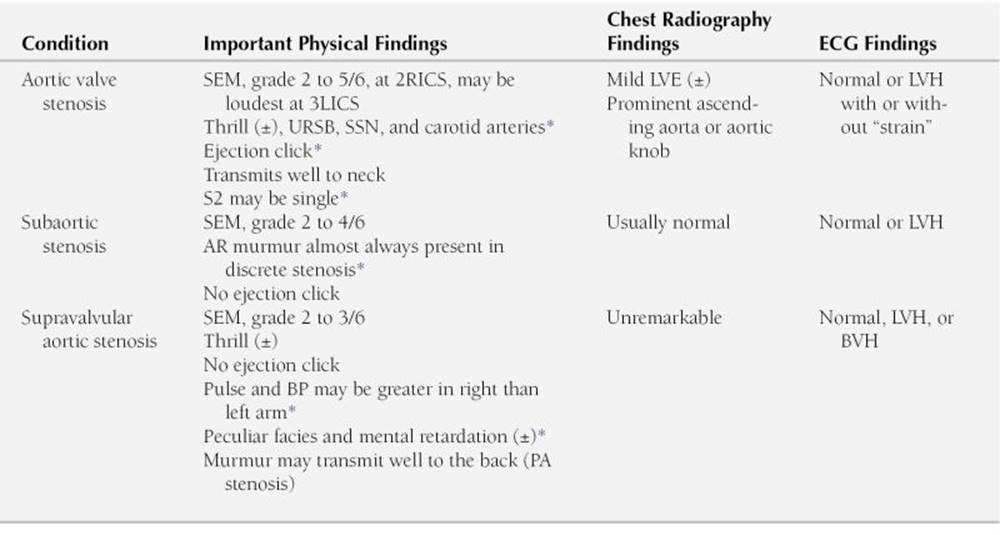
2. Upper right sternal border or aortic area
Systolic murmurs at the upper right sternal border are also midsystolic type. They are caused by narrowing of the aortic valve or its neighboring vascular structures. The murmur transmits well to the neck. Often it transmits with a thrill over the carotid arteries. The midsystolic murmur of AS may be heard with equal clarity at the upper left sternal border (i.e., “pulmonary area”), as well as at the apex. However, the PS murmur does not transmit well to the upper right sternal border and the neck; rather, it transmits well to the back and the sides of the chest. Systolic murmurs in the upper right sternal border are caused by the following:
a. AS
b. Subvalvular AS (subaortic stenosis)
c. Supravalvular AS
Characteristic physical, ECG, and radiography findings that help in the differential diagnosis of these conditions are presented in Table 2-5.
3. Lower left sternal border
Systolic murmurs that are maximally audible at this location may be a holosystolic, early systolic, or midsystolic type and may result from one of the following conditions:
a. VSD murmur is either holosystolic or early systolic in timing (a small muscular VSD murmur may be heard best between the lower left sternal border and the apex)
b. Vibratory or musical innocent murmur (e.g., Still’s murmur); this murmur may be equally loud or even louder toward the apex, and the maximal intensity may be in the midprecordium
c. Hypertrophic obstructive cardiomyopathy (HOCM) (formerly known as idiopathic hypertrophic subaortic stenosis)
d. TR
e. TOF
Characteristic physical, ECG, and radiography findings that help in the differential diagnosis of these conditions are presented in Table 2-6.
4. Apical area
Systolic murmurs that are maximally audible at the apex may be a holosystolic, midsystolic, or late systolic murmur and result from one of the following conditions:
a. MR (holosystolic)
b. MVP (late systolic murmur, usually preceded by a midsystolic click)
c. AS (midsystolic)
d. HOCM (midsystolic)
e. Vibratory innocent murmur (midsystolic)
Characteristic physical, ECG, and radiography findings are summarized in Table 2-7.
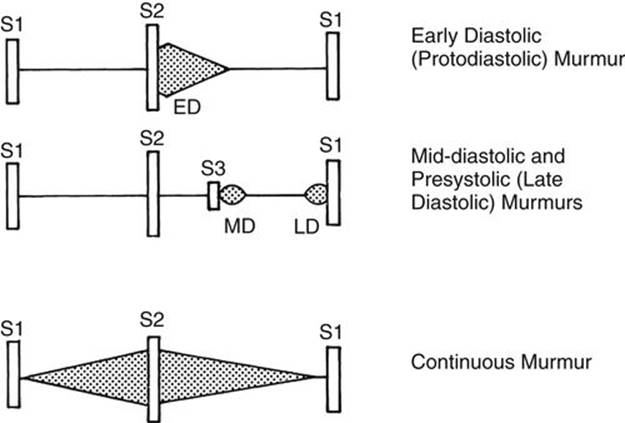
FIGURE 2-13 Diagram of diastolic murmurs and the continuous murmur. ED, early diastolic or protodiastolic murmur; LD, late diastolic or presystolic murmur; MD, mid-diastolic murmur.
Diastolic Murmurs
Diastolic murmurs occur between the S2 and S1. Based on timing and relation to the heart sounds, they are classified into three types: early diastolic (or protodiastolic), mid-diastolic, and late diastolic (or presystolic) (Fig. 2-13).
1. Early diastolic decrescendo murmurs
occur early in diastole, immediately after the S2, and are caused by incompetence of the aortic or pulmonary valve (see Fig. 2-13).
Because the aorta is a high-pressure vessel, AR murmurs are high pitched and best heard with the diaphragm of a stethoscope at the third left intercostal space. The AR murmur radiates well to the apex because the regurgitation is directed toward the apex. Bounding peripheral pulses may be present if the AR is significant. AR murmurs are associated with congenital bicuspid aortic valve, subaortic stenosis, after an intervention for AS (i.e., postvalvotomy or after balloon dilatation), and rheumatic heart disease with AR. Occasionally, a subarterial infundibular VSD with prolapsing aortic cusps may cause an AR murmur.
Pulmonary regurgitation murmurs also occur early in diastole. They are usually medium pitched but may be high pitched if pulmonary hypertension is present. They are best heard at the third left intercostal space and radiate along the left sternal border. These murmurs are associated with postoperative TOF (because of surgically induced PR), pulmonary hypertension, postoperative pulmonary valvotomy or after balloon valvuloplasty for PS, and mild deformity of the pulmonary valve.
2. Mid-diastolic murmurs
start with a loud S3 and are heard in early or mid-diastole but are not temporally midway through diastole (see Fig. 2-13). These murmurs are always low pitched and best heard with the bell of the stethoscope applied lightly to the chest. These murmurs are caused by turbulence caused by the mitral or tricuspid flow secondary to anatomic stenosis or relative stenosis of these valves.
Mitral mid-diastolic murmurs are best heard at the apex and are often referred to as an apical rumble, although frequently they sound more like a hum than a rumble. These murmurs are associated with MS or a large left-to-right shunt VSD or PDA, which produces relative MS secondary to a large flow across the normal-sized mitral valve.
Tricuspid mid-diastolic murmurs are best heard along the lower left sternal border. These murmurs are associated with ASD, PAPVR, TAPVR, and ECD because they all result in relative tricuspid stenosis (TS). Anatomic stenosis of the tricuspid valve is also associated with these murmurs, but such cases are rare.
3. Presystolic (or late diastolic) murmurs
are also caused by flow through the AV valves during ventricular diastole. They result from active atrial contraction that ejects blood into the ventricle rather than a passive pressure difference between the atrium and ventricle. These low-frequency murmurs occur late in diastole or just before the onset of systole (see Fig. 2-13) and are found with anatomic stenosis of the mitral or tricuspid valve.
Continuous Murmurs
Continuous murmurs begin in systole and continue without interruption through the S2 into all or part of diastole (see Fig. 2-13). Continuous murmurs are caused by the following:
1. Aortopulmonary or arteriovenous connection (e.g., PDA, arteriovenous fistula, after systemic-to-PA shunt surgery, persistent truncus arteriosus [rarely])
2. Disturbances of flow patterns in veins (e.g., venous hum)
3. Disturbance of flow pattern in arteries (e.g., COA, PA stenosis)
The murmur of PDA has a machinery-like quality, becoming louder during systole (crescendo), peaking at the S2, and diminishing in diastole (decrescendo). This murmur is maximally heard in the left infraclavicular area or along the upper left sternal border. With pulmonary hypertension, only the systolic portion can be heard, but it is crescendic during systole.
Venous hum is a common innocent murmur that is audible in the upright position, in the infraclavicular region, unilaterally or bilaterally. It is usually heard better on the right side. The murmur’s intensity also changes with the position of the neck. When the child lies supine, the murmur usually disappears. Less common continuous murmurs of severe COA may be heard over the intercostal collaterals. The continuous murmurs of PA stenosis may be heard over the right and left anterior chest, the sides of the chest, and in the back.
The combination of a mid-systolic murmur (e.g., VSD, AS, or PS) and a diastolic murmur (e.g., AR or PR) is referred to as a to-and-fro murmur to distinguish it from a machinery-like continuous murmur.
Innocent Heart Murmurs
Innocent heart murmurs, also called functional murmurs, arise from cardiovascular structures in the absence of anatomic abnormalities. Innocent heart murmurs are common in children. More than 80% of children have innocent murmurs of one type or another sometime during childhood (Table 2-8). All innocent heart murmurs (as well as pathological murmurs) are accentuated or brought out in a high-output state, usually during a febrile illness.
TABLE 2-8
COMMON INNOCENT HEART MURMURS IN CHILDREN
|
Type (Timing) |
Description of Murmur |
Age Group |
|
Classic vibratory murmur (Still’s murmur) (systolic) |
Maximal at MLSB or between LLSB and apex |
3–6 yr |
|
Pulmonary ejection murmur (systolic) |
Maximal at ULSB |
8–14 yr |
|
Pulmonary flow murmur of newborn (systolic) |
Maximal at ULSB |
Prematures and full-term newborns |
|
Venous hum (continuous) |
Maximal at right (or left) supraclavicular and infraclavicular areas |
3–6 yr |
|
Carotid bruit (systolic) |
Right supraclavicular area and over the carotids |
Any age |
LLSB, lower left sternal border; MLSB, mid-left sternal border; ULSB, upper left sternal border.
Probably the only way a physician can recognize an innocent heart murmur is to become familiar with the more common forms of these murmurs by auscultating under the supervision of pediatric cardiologists. All innocent heart murmurs are associated with normal ECG and radiography findings. When one or more of the following are present, the murmur is more likely pathologic and requires cardiac consultation:
1. Symptoms
2. Abnormal cardiac size or silhouette or abnormal pulmonary vascularity on chest roentgenograms
3. Abnormal ECG findings
4. Diastolic murmur
5. A systolic murmur that is loud (i.e., grade 3 of 6 or with a thrill), long in duration, and transmits well to other parts of the body
6. Cyanosis
7. Abnormally strong or weak pulses
8. Abnormal heart sounds
Classic Vibratory Murmur
This is the most common innocent murmur in children, first described by Still in 1909. Most vibratory murmurs are detected between 3 and 6 years of age, but the same murmur may be present in neonates, infants, and adolescents. It is maximally audible at the mid-left sternal border or over the midprecordium (between the lower left sternal border and the apex). It is generally of low frequency and best heard with the bell of the stethoscope with the patient in the supine position. The murmur is midsystolic (i.e., not regurgitant) in timing and grade 2 to 3 of 6 in intensity. This murmur is not accompanied by a thrill or ejection click. It has a distinctive quality, described as a “twanging string,” groaning, squeaking, buzzing, or vibratory sound, giving a pleasing musical character to the murmur. The murmur is generally loudest in the supine position and often changes in character, pitch, and intensity with upright positioning. The vibratory quality may disappear, and the murmur may become softer when the bell is pressed harder, thereby proving its low frequency. The intensity of the murmur increases during febrile illness or excitement, after exercise, and in anemic states. The murmur may disappear briefly at a maximum Valsalva maneuver. The ECG and chest radiography findings are normal (see Table 2-8; Fig. 2-14).
An inexperienced examiner may confuse this murmur with the murmur of a VSD. The murmur of a VSD is usually harsh, grade 2 to 3 of 6 in intensity, holosystolic starting with the S1 rather than midsystolic, and often accompanied by a palpable thrill. The ECG and radiography findings are often abnormal.
The origin of the murmur remains obscure. It is believed to be generated by low-frequency vibrations of normal pulmonary leaflets at their attachments during systole or periodic vibrations of a left ventricular false tendon.
Pulmonary Ejection Murmur (Pulmonary Flow Murmur) of Childhood
This is common in children between 8 and 14 years of age but is most frequent in adolescents. The murmur is maximally audible at the upper left sternal border. This murmur represents an exaggeration of normal ejection vibrations within the pulmonary trunk. The murmur is exaggerated by the presence of pectus excavatum, straight back, or kyphoscoliosis. The murmur is midsystolic in timing and slightly grating (rather than vibratory) in quality, with relatively little radiation. The intensity of the murmur is usually a grade 1 to 3 of 6. The S2 is normal, and there is no associated thrill or ejection click (see Table 2-8; Fig. 2-14). The ECG and chest radiography findings are normal.
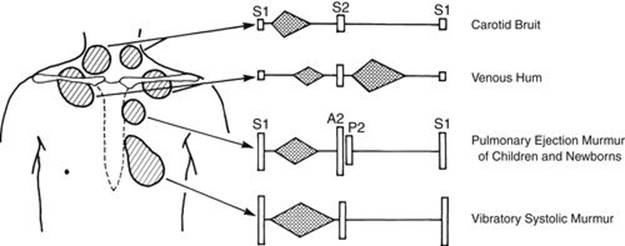
FIGURE 2-14 Diagram of innocent heart murmurs in children.
This murmur may be confused with the murmur of pulmonary valve stenosis or an ASD. In pulmonary valve stenosis, there may be an ejection click, systolic thrill, widely split S2, right ventricular hypertrophy (RVH) on ECG, and poststenotic dilatation of the main PA segment on chest radiography. Important differential points of ASD include a widely split and fixed S2, a mid-diastolic murmur of relative TS audible at the lower left sternal border if the shunt is large, RBBB or mild RVH on ECG manifested by rsR’ in V1, and chest radiography revealing increased pulmonary vascular markings and enlargement of the right atrium, RV, and main PA.
Pulmonary Flow Murmur of Newborns
This murmur is commonly present in newborns, especially those with low birth weight. The murmur usually disappears by 3 to 6 months of age. If it persists beyond this age, a structural narrowing of the pulmonary arterial tree (i.e., PA stenosis) should be suspected. It is best audible at the upper left sternal border. Although the murmur is only a grade 1 to 2 of 6 in intensity, it transmits impressively to the right and left chest, both axillae, and the back. There is no ejection click. The ECG and chest radiography findings are normal (see Table 2-8; Fig. 2-14).
In a fetus, the main PA trunk is large, but the branches of the PA are relatively hypoplastic because they receive a small amount of blood flow during fetal life (only 15% of combined ventricular output goes to these vessels). When the ductus closes after birth, the large dome-shaped main PA trunk gives off two small branch pulmonary arteries. The flow through these small vessels produces turbulence with a faster flow velocity, and the turbulence is transmitted along the smaller branches of the PAs. Therefore, this murmur is heard well around the chest wall. The murmur is louder in small preterm babies than the larger full-term neonates.
The murmur resembles the murmur of organic PA stenosis, which may be seen as a component of rubella syndrome, Williams syndrome, or Alagille syndrome. Characteristic noncardiac findings in children with these syndromes lead physicians to suspect that the PA stenosis murmur has an organic cause. Organic PA stenosis is frequently associated with other cardiac defects (e.g., VSD; pulmonary valve stenosis, or TOF), at the site of a previous Blalock-Taussig shunt, or is seen occasionally as an isolated anomaly. The heart murmur of organic PA stenosis persists beyond infancy, and the ECG may show RVH if the stenosis is severe.
Venous Hum
This murmur is commonly audible in children between the ages of 3 and 6 years. It originates from turbulence in the jugular venous system. This is a continuous murmur in which the diastolic component is louder than the systolic component. The murmur is maximally audible at the right or left infraclavicular and supraclavicular areas (see Table 2-8; Fig. 2-14). The venous hum is heard only in the upright position and disappears in the supine position. It can be obliterated by rotating the head or by gently occluding the neck veins with the fingers.
It is important to differentiate a venous hum from the continuous murmur of a PDA. The murmur of a PDA is loudest at the upper left sternal border or left infraclavicular area and may be associated with bounding peripheral pulses and wide pulse pressure if the shunt is large. The systolic component is louder than the diastolic component. The radiographs show increased pulmonary vascular markings and cardiac enlargement. The ECG may be normal (with a small shunt) or show left ventricular hypertrophy or combined ventricular hypertrophy (with a large shunt).
Carotid Bruit (or Supraclavicular Systolic Murmur)
This is an early systolic ejection murmur that is best heard in the supraclavicular fossa or over the carotid arteries (see Table 2-8; Fig. 2-14). It is produced by turbulence in the brachiocephalic or carotid arteries. The murmur is a grade 2 to 3 of 6 in intensity. Although it rarely occurs, a faint thrill is palpable over a carotid artery. This bruit may be found in normal children of any age.
The murmur of AS often transmits well to the carotid arteries with a palpable thrill, requiring differentiation from carotid bruits. In AS, the murmur is louder at the upper right sternal border, and a systolic thrill is often present in the upper right sternal border and suprasternal notch, as well as over the carotid artery. An ejection click is often present in aortic valve stenosis. The ECG and radiography findings may appear abnormal.
Some Special Features of the Cardiac Examination of Neonates
The following section briefly summarizes some unique aspects of normal and abnormal physical findings in newborns, which are different from those of older infants and children. The difference is caused by the normal RV dominance and elevated pulmonary vascular resistance (PVR) seen in the early neonatal period. Premature infants in general have less RV dominance and a lower PVR than do full-term neonates, adding variability to this generalization.
Normal Physical Findings of Neonates
The following are normal cardiovascular findings in newborn infants:
1. The heart rate generally is faster in newborns than in older children and adults (the newborn rate is usually 100 beats/min, with a normal range of 70 to 180 beats/min).
2. A varying degree of acrocyanosis is the rule rather than the exception.
3. Mild arterial desaturation with arterial Po2 as low as 60 mm Hg is not unusual in an otherwise normal neonate. This may be caused by an intrapulmonary shunt through an as-yet unexpanded portion of the lungs or by a right atrium–to–left atrium shunt through a patent foramen ovale.
4. The RV is relatively hyperactive, with the point of maximal impulse at the lower left sternal border rather than at the apex.
5. The S2 may be single in the first days of life.
6. An ejection click (representing pulmonary hypertension) occasionally is heard in the first hours of life.
7. A newborn may have an innocent heart murmur. Four common innocent murmurs in the newborn period are pulmonary flow murmur of the newborn (see Fig. 2-14), transient systolic murmur of PDA, transient systolic murmur of TR, and vibratory innocent systolic murmur.
a. Pulmonary flow murmur of the newborn is the most common heart murmur in newborn infants (see previous section).
b. Transient systolic murmur of PDA is caused by a closing ductus arteriosus and is audible on the first day of life. It is a grade 1 to 2 of 6 only systolic, at the upper left sternal border and in the left infraclavicular area.
c. Transient systolic murmur of TR is indistinguishable from that of VSD. It is believed that a minimal tricuspid valve abnormality produces regurgitation in the presence of high PVR (and high RV pressure), but the regurgitation disappears as the PVR falls. Therefore, this murmur is more common in infants who had fetal distress or neonatal asphyxia because they tend to maintain high PVR for a longer period.
d. Vibratory innocent murmur is a counterpart of Still’s murmur in older children (see previous section).
8. Peripheral pulses are easily palpable in all extremities, including the feet, in every normal infant. The peripheral pulses normally appear to be bounding in premature babies because of the lack of subcutaneous tissue.
Abnormal Physical Findings in Neonates
The following abnormal physical findings suggest cardiac malformation. Repeated examination is important because physical findings change rapidly in normal infants as well as in infants with cardiac problems.
1. Cyanosis, particularly when it does not improve with the administration of oxygen, suggests a cardiac abnormality.
2. Decreased or absent peripheral pulses in the lower extremities suggest coarctation of the aorta (COA). Weak peripheral pulses throughout suggest hypoplastic left heart syndrome (HLHS) or circulatory shock. Bounding peripheral pulses suggest aortic run-off lesions, such as PDA or persistent truncus arteriosus.
3. Tachypnea of greater than 60 breaths/min with or without retraction suggests a cardiac abnormality.
4. Hepatomegaly may suggest a heart defect. A midline liver suggests asplenia or polysplenia syndrome.
5. A heart murmur may be a presenting sign of a congenital heart defect, although innocent murmurs are much more frequent than pathologic murmurs. Most pathologic murmurs should be audible during the first month of life, with the exception of an ASD. However, the time of appearance of a heart murmur depends on the nature of the defect.
a. Heart murmurs of stenotic lesions (e.g., aortic stenosis, pulmonary stenosis) and those caused by AV valve regurgitation are audible immediately after birth and persist because these murmurs are not affected by the level of PVR.
b. Heart murmurs of large VSD may not be audible until 1 to 2 weeks of age when the PVR becomes sufficiently low to allow shunt to occur.
c. The murmur of an ASD appears after 1 or 2 years when the compliance of the RV improves to allow a significant atrial shunt. A newborn or a small infant with a large ASD may not have a heart murmur.
6. Even in the absence of a heart murmur, a newborn infant may have a serious heart defect that requires immediate attention (e.g., severe cyanotic heart defect such as TGA or pulmonary atresia with a closing PDA). Infants who are in severe CHF may not have a loud murmur until the myocardial function is improved through anticongestive measures.
7. An irregular cardiac rhythm and abnormal heart rate suggest a cardiac abnormality.