“Am I my genes?” she asked me. In her late thirties, she had breast cancer and had just learned that she had a mutation associated with the disease. She looked out the window in my office at the gray sky. We were both silent for a moment, each pondering her question. She seemed to be asking the question to herself, more than expecting a definitive answer from me.
Still, I wasn’t sure what to say. I wondered to what degree exactly genes make us who we are, and how we understand what that means.
“I always knew I shouldn’t have lived on Long Island for those years,” another woman with breast cancer told me a few days later.
“Why is that?” I asked.
“Because that’s why I got the cancer.”
“But you have the mutation,” I reminded her.
“Yes, but it was living on Long Island with all of those high tension wires, and the stress of staying in a crappy relationship that triggered the illness.”
“I don’t believe my cancer is from God or genetics,” a poor African American woman from the Bronx who also had a breast cancer mutation told me a few weeks later. “It’s from the environment. They dump garbage in the water and the landfills in the Bronx that they would never dump in Manhattan.”
“The biggest question I face,” a young, attractive blonde woman at risk for Huntington’s disease said to me a few days afterward, “is whether I should just try to have kids with my husband, or adopt, abort, or have no children.” She stared down into her lap, silent and sad, unsure of how to resolve her dilemma. Again, though a physician, I was uncertain what to say.
These women’s statements all surprised me but, I soon saw, were hardly unique. In interviewing people confronting several diseases for which genetic tests exist, I was continually struck by the wide range of dilemmas they faced. Genetic information forced these individuals to embark on journeys for which they had to navigate many obstacles.
They responded to genetic information about themselves as a kind of Rorschach test—interpreting this information in a wide range of ways, based on their prior views and stories about themselves and other cultural and personal experiences.
The issues they confront differ in several key ways from those of patients with other disorders for which no good genetic tests exist. The men and women I met struggle to make sense of diseases that threaten not only themselves, as single individuals, but others in their families—deceased, living, and not yet born. They feel guilty about the possibility that their mutations could harm their yet unborn children and grandchildren.
Genetic testing (also referred to below as genetic assay, assessment, and analysis) is rapidly spreading. Every year, dozens of new genetic tests are developed and offered. Over time, the amount of testing will soar. In industrialized countries such as the United States, physicians are increasingly offering genetic assays to patients and, it seems probable, will eventually sequence the entire genome of every patient. As outlined more fully below, our DNA consists of chains of four large molecules, known as nucleosides—adenine, cytosine, thymine, and guanine (abbreviated A, C, T, G)—that serve as an “alphabet.” Every person has a unique set of three billion of these “letters” that constitute the genetic program that makes us. We share 99.9% of our DNA with each other, differing from one another only by about 0.1%.
Until now, many direct-to-consumer (DTC) tests have assayed at most only one out of every 10,000 letters, and only the most common of human genetic variants. Imagine comparing War and Peace with the Bible by reading only one letter per page—a single “A” or “C.” The reader would understand nothing of either book, and could not meaningfully differentiate them.
Still, direct-to-consumer marketing companies such as 23andme.com—started and funded by the wife of one of the founders of Google—have begun providing inexpensive online testing, relying on such single letter variations. Individuals simply swipe the inside of their mouth with a swab and mail the sample. Soon after, they receive detailed information about their purported risks of numerous diseases.
Much of the information now provided by these DTC companies has little practical value, and laboratories interpret the same results in different ways. The FDA is now investigating these companies, and pursuing the possibility of regulating them.1 But DTC testing appears likely to continue in some form. At the same time, however, the costs of mapping a person’s entire genome have been plummeting—from billions of dollars to sequence the first person’s complete DNA, to $100 million, to $1 million, to $100,000, to $10,000, and will shortly be even cheaper—$1,000, and eventually even less.
Thus, we are now beginning to read the equivalent of entire books, allowing us to identify and make sense of them. Soon, we will be able to sequence each of our entire genomes, permitting previously unimaginable discoveries.
Eventually, physicians will probably order complete genome sequencing tests on all of us. The Mayo Clinic and Vanderbilt University now already collect and permanently store DNA samples of all patients who walk in the door. Many other academic medical centers are trying to arrange to do the same. Ultimately, DTC testing may well include whole genome sequencing as well.
In upcoming years and decades, the usefulness of many tests, though not all, may thus increase. We will soon have more biological information about ourselves than ever before. But are we ready for it? What will it mean? What will we do with it, and how will we understand it?
In the mere 50 years since DNA was discovered, researchers have made extraordinary advances. Genetics appears likely to enhance our comprehension and treatment of many diseases, and our understandings of important aspects of ourselves. In 1986, investigators identified the first mutation for a human disease—Huntington’s disease (HD). Since then, they have found mutations associated with thousands of other disorders, including cystic fibrosis, sickle cell anemia, and breast cancer. In 2003, an entire human genome was sequenced. Currently, doctors routinely check infants and pregnant mothers for genes associated with dozens of disorders, some of which are readily remediable. Scientists are also reprogramming stem cells, which have not yet specialized to become parts of particular organs (whether liver, muscle, or brain). Researchers are working to turn these protean cells into whatever types of tissue an injured body may need (e.g., heart, pancreas, or spinal cord), hoping to develop new treatments for many disorders. In 2009, researchers found that altering merely four genes can transform a skin cell into many other types of cells in the body. In May of 2010, the scientist Craig Venter announced he had “synthesized life,” using a computer and four chemicals bought off a shelf to replicate the DNA of a bacteria that then took over another cell2.
Increasingly, in the United States and elsewhere, the field is also affecting human reproduction. Assisted reproductive technologies or ARTs, once criticized for producing “test tube babies,” now account for 7% of all births in some European countries, and 1% in the United States. These numbers are soon expected to reach 10%—equal to the proportion of infertile couples. Preimplantation genetic diagnosis (PGD) enables women to select which embryos to implant into their womb, based on the presence or absence of over 5,000 different mutations. Thus, couples are now eliminating certain lethal mutations from their future descendants, and in some cases, deciding the gender of their future children. Many men and women now buy and sell eggs and sperm on the Internet to assist other couples and individuals in having children, though critics argue that such transactions “commodify” these essential and sacred components of human beings—turning them, and thus us, into mere marketplace goods.
Pharmaceutical companies seek to develop “personalized medicine”—tailoring drugs for specific individuals based on the individual’s genetics. Through this field of pharmacogenomics, genes are being identified that predict responsiveness to particular medications, rather than diseases. Our individual DNA may thus help determine which medication for an ailment will work best for each of us. Genetics may then be used not only to assist in foreseeing and diagnosing certain diseases, but in treating them, too. Knowledge is swiftly advancing about not just genes but the mechanisms through which they make and assemble proteins and regulate themselves, launching new, larger fields of “genomics” and “proteinomics.” Just as antibiotics shaped the last century, eliminating certain epidemics of infectious disease, many researchers argue that genetics is beginning to alter several aspects of the twenty-first century.
Yet while researchers are discovering that single mutations are responsible for thousands of rare diseases, far more common ailments such as diabetes, cancer, heart disease, and depression appear to result not from single isolated mutations but rather from complex interactions, not yet well-understood, between multiple genes and other, environmental inputs. DNA mixes and sorts, and combinations of genes and gene-environment interactions, rather than single point mutations, appear to be responsible for most common diseases. Debates thus rage about the current and future extent of genetic knowledge—whether genetics is overly hyped, and if so, to what degree. Some critics have become wary of the ability of genetics to provide much new, useful information.
Clearly, at times, much hype and “genetic reductionism” occur, but the study of DNA is also still in its infancy. Our comprehension of genetics and its potential effects is still young. Now, at the start of a new millennium, it is impossible to predict what the future, or even just the next 50, 100, or 200 years will bring. To profound and unique extents, our DNA clearly helps shape us—for example, key aspects of how we look and whether we may be predisposed to develop certain diseases. To what degree and how exactly it does so, however, remain unknown.
Countless uncertainties persist. For instance, when scientists first cracked the human genome many were astonished that it contained so few genes compared to other mammals, and that we shared 99% of this DNA with primates, and 85% with mice. How humans thus look and act so differently from other species is not wholly clear. Scientists still think that half of our DNA is “junk,” its function still murky. They do not yet fully know what information and instructions it may hide that they might soon begin to decipher. In upcoming years the functions of countless additional genetic sequences will undoubtedly be discovered.
As the effects of combinations of sequences are explored, understandings of genetics will most likely increase—most likely not to the full extent that some hope, but far more than at present. Use of genetics is becoming ever more common in clinical medicine, confronting patients and their families and physicians with tests associated with medication responses, modestly increased risks for common diseases in conjunction with other, nongenetic risk factors, and genetic modifiers and suppressors that may contribute to illness, but are not primary causes.
This burgeoning genetic knowledge will continue to pose ever-new dilemmas. It is uncertain how people do or will see or fathom such information—whether they want it, and what, if anything, they do with it. Fears, myths, and misunderstandings about genes persist among patients, policy makers, scientists, and the public at large. Many critical aspects of the roles that genetics plays in our lives, and how we in fact view these, remain uncharted.
In numerous ways, genes are counterintuitive. In The Selfish Gene, the British biologist Richard Dawkins asserts that all species are essentially machines that genes have developed for reproducing themselves. Yet people frequently think of themselves as having free will and a transcendent spirit—not as resulting from an egg and a sperm that formed a cell that has kept dividing to produce us. Science suggests that we consist of mere molecules (mostly of carbon, hydrogen, and oxygen) and wouldn’t exist otherwise, and that DNA contains the instructions for building the underlying machine or hardware that constitutes much, if not most, of us. Clearly, environmental factors also shape key aspects of us, though which aspects and to what extent remain uncertain. But in addition to these environmental influences, many people believe that genetics still insufficiently accounts for human beings—feeling, for instance, that we also have an incorporeal soul that results from nonchemical elements of our being. Crucial questions therefore arise of how people understand and make sense of these paradoxes, conceptualize themselves in relation to their genes, and make decisions as a result.
Fears also exist that eugenics may overly shape the use of this scientific knowledge. At the extreme, some caution that use of genetic information can easily slip down the slope toward the horrors of Nazi Germany. They caution, too, about the dangers of tampering with Nature, invoking images of Aldous Huxley’s Brave New World, Mary Shelley’s Frankenstein, and the 1997 film Gattaca, which portrayed a future society that tested newborns to permanently classify them as genetic “desirables” or “undesirables.’”
Many anthropologists and postmodern critics fear genetic determinism and reductionism, arguing instead that disease is “socially constructed.” Barbara Katz Rothman and other scholars have criticized overemphasis on genetics as “the cause” of many disorders3. “Geneticization” in a variety of social realms claims too much for the potency of DNA. News headlines regularly announce the discovery of “the fat gene,” “the gay gene,” or the “alcoholism gene.” Yet each of these traits in fact appears to involve highly complex interactions of genes with other biological, social, and psychological factors. Patients, too, may inappropriately apply the notion that genes predict certain diseases to other, multi-causal behaviors in which genes may in fact play relatively small roles.
Still, regardless of how much genetics may be oversold, we and future generations will have to live with this ever-expanding field. Every day in the United States, countless people wrestle with whether to get tested, and many proceed to do so. We need to prepare as best we can for these challenges, and the possible meanings of genetic information for us and our descendants—to understand how people perceive the current and potential meanings and implications of this information.
Genetics also sparks other, broader social challenges related, for instance, to privacy, stigma, and discrimination. Many government officials seek to establish genetic databases for law enforcement and public health. Though the U.S. Health Insurance Portability and Accountability Act (HIPAA) of 1996 aims to protect medical privacy, apprehensions persist. The burgeoning electronic storage of personal information heightens these worries. Identity theft has occurred, and hackers have broken into secure computer systems, stealing sensitive data. In 2008, Congress passed the Genetic Information Nondiscrimination Act (GINA), and most states have enacted their own genetic privacy laws. Yet these acts vary widely, and numerous challenges remain.
Genetics can pose larger psychological and metaphysical questions, too, about fate. From oracles in Ancient Greece to crystal balls in medieval tales, and psychics today, people have yearned to predict future events. In the case of HD and various enzyme deficiencies for which mutations with relatively high penetrance have been found, DNA can potentially inform many people about aspects of their future. Genetics may provide some information about the relative likelihood of certain future diseases, though surely not to the degree that some media hype may suggest. After all, genetics is still a relatively new field, and current scientific knowledge remains limited. The future can never be wholly known, and much genetic information will be in terms of relative probabilities, rather than absolutes.
Just as the ancient Greek Oracles famously offered ambiguous messages that recipients misinterpreted, so, too, genetic information will surely present us with complex puzzles and uncertainties. The information will often no doubt involve relatively small augmentations in risk, shaped by many other, still unknown factors. Nevertheless, depending on the specific details, some people may seek these relatively small increases in the chance of a disease, even if the overall predictiveness is still not very high (e.g., learning that one has a 10% or 20% chance, rather than a 1% or 2% likelihood, of getting a particular disease). Such information could potentially help motivate preventive behavior. Yet direct-to-consumer genetic testing companies are taking advantage of widespread popular desires to prophesy the future, aggressively marketing these tests as fortune-tellers, alluring many people. Every day, millions of people read their horoscopes, and many are now beginning to seek genetic tests as well.
As one of my teachers, the late anthropologist Clifford Geertz, has written, to understand complex social phenomena, and avoid external observers imposing their own preconceived notions onto a situation, it is crucial to understand these phenomena from the views of individuals within them.4
Hence, I decided to explore in-depth how individuals in fact now confront and understand some of these complex, multifaceted issues in their daily lives by interviewing a wide range of individuals who have, or are at risk for, several genetic diseases, in order to obtain a “think description.” I interviewed 64 individuals who faced HD, breast and ovarian cancer, or Alpha-1 antitrypsin deficiency. I decided to concentrate on these three diseases, each with different characteristics, to try to grasp in a more fully and nuanced manner several ways in which genetics can affect people, and experiences individuals are now facing.
The degree to which genetic risks affected the whole span of their lives, from birth to death, surprised me. I found how these participants each wrestled with genetics, which has vast implications for their lives and those of their families. These men and women all struggled to make sense of their predicament and its causes, to discern how genetics may influence their lives, at times against their will. In the face of scientific uncertainties, they tried to comprehend these tests and probabilities; avoid fatalism, anxiety, despair, stigma, and discrimination; and find hope and meaning, and a sense of wholeness. They confront a series of quandaries, each explored in a different chapter here—whether to test; whether to disclose their genetic risks to parents, siblings, spouses, offspring, distant relatives, friends, doctors, insurers, employers, and schools; how to view and understand themselves and their genetics; what treatments, if any, to pursue; whether to have children, adopt, screen embryos, or abort; and whether to participate in genetic communities, and if so, how. These decisions are among the most difficult many have ever faced. Forced to wander through a wilderness of shifting sands, they chart paths that many others may eventually follow. They are pioneers, encountering quandaries that many of us, too, will soon face.
These men and women try to fathom their predicament, and are often both allured and frightened by the possibility of knowing key elements of their future, wanting to avoid stigma and rejection, and maintain a sense of hope and control in their lives. Yet these goals conflict. Complex social contexts—relationships with immediate and extended families, friends, health care providers, insurance companies, coworkers, and patient communities—all shape these choices. Genetics forces people to balance competing external and internal pressures. These men and women attempt to mediate and negotiate with, or work around, these social contexts. This book illuminates how these issues affect these individuals’ lives.
Genetics compel these individuals to venture on long journeys, encountering many challenges. First, genetics can potentially provide certain information about the future—though generally as inexact probabilities, not absolutes. Secondly, genetic information relates to not just one individual, but to his or her family, too. Tensions can result, however, since genetic and social bonds can differ, posing questions about responsibilities toward various family members. An individual must decide, for instance, whether to disclose a mutation to certain relatives, who may then become distraught about their own future. Thirdly, diseases for which definitive genetic tests exist tend to be rare, and thus poorly understood by most people. Fourthly, new reproductive technologies permit screening of embryos and elimination of genes from descendants, posing additional moral and psychological dilemmas. Fifthly, physicians and direct-to-consumer companies offer, but at times oversell, genetic tests. And, finally, fears linger that genetic information can prompt stigma and discrimination in insurance and employment. With increasing electronization of medical records, and a fractured, yet rapidly evolving health care system, genetic information is also becoming available in a dramatically changing landscape. Other organizations—including schools and police departments—would often also like individuals’ genetic data. In part in response, many individuals choose to enter genetic communities, but then face dilemmas—for example, whether to participate in research that will aid others, but not themselves. These phenomena combine, affecting each other.
In facing these moral tensions, individuals often follow implicit “gut feelings” more than explicit ethical or logical principles per se. Superstitions and beliefs about fate prove potent—not necessarily related to established religions, but rather mirroring underlying desires for coherence in one’s life over time.
The bulk of this book examines the series of experiences through which these individuals pass. Heretofore, the medical literature has generally discussed each of these domains separately, with distinct sets of studies examining testing, or disclosure, or understandings of disease, or reproduction. Instead, I have sought to bring these facets together to explore how they fit into the whole of people’s lives. From these patients’ own perspectives, I probed how these issues interact. Such an effort may seem ambitious, but, I think, is vital to advance our knowledge of these realms.
This book groups these experiences into four broad domains concerning, respectively, genes in “the family,” “the mind,” “the clinic,” and “the wider world.” As a brief overview, the first of these parts, “Genes in the family,” explores, in chapter 2, how individuals learn of their genetic risk (usually from other family members), how they decide whether to get tested or not, and how they interact with providers about testing issues, and view and interpret the probabilities and uncertainties involved. Chapter 3 probes how individuals decide whether and what to communicate about genetic test results to other family members. Individuals who get tested must balance ethical mandates to disclose to kin (and perhaps encourage other family members to undergo testing) against the psychological difficulties of doing so, due to shame, fear, denial, or estrangement.
In the second part of the book, “Genes in the mind,” chapter 4 examines how individuals weigh possible causes (i.e., genetics and/or environment) and larger cosmological questions of “why me?” Their understandings of causality and fate powerfully shape their decisions about testing, treatment, reproduction, and disease prevention. Chapter 5 explores how individuals make sense of their “genetic identity”—how they understand information about their genes and integrate it with their past and ongoing notions of themselves, deciding how and to what degree to see their genes as part of their identity. Chapter 6 examines myths and misunderstandings of genetics as individuals struggle to make sense of complex probabilities—how they comprehend their genetic risks, and grasp murky probabilities.
In part IV, “Genes in the clinic,” chapter 7 explores how individuals confront treatment decisions. Chapter 8 probes how these individuals decide whether to have children, and if so, whether to screen embryos, undergo amniocentesis and abortion of affected fetuses, or adopt. Chapter 9 looks into experiences with insurance companies, loss of insurance and privacy, and dilemmas about whether to use insurance for testing and treatment, and how to maintain confidentiality in a world with increasingly fluid electronic medical records.
Part V, “Genes in the wider world,” investigates, in chapter 10, the implications of these issues for disclosures to coworkers and bosses, friends, dates, neighbors, and schools. Patients face potential discrimination, stigma, and decisions about “going public” to varying degrees. Chapter 11 probes the structures and functions of genetic communities—both in-person and, increasingly, online—that affect how individuals process and convey information. These communities can provide benefits, but also expose members to much sicker patients, generating fear and creating questions of whether, and to what degree, to join disease organizations. Chapter 12 explores the broader implications of these experiences for public health policy, widespread clinical testing and screening, professional and public education, law enforcement, and society. The concluding chapter synthesizes many of these themes.
I have arranged these discussions to reflect broadly the sequences of social space and time through which individuals tend to encounter these issues. But these domains are closely interwoven and do not fully follow a fixed progression. Rather, understandings and decisions evolve over time. Myths about genetics affect disclosure decisions, and vice versa. People divulge their genetic risks to ever-widening circles—from immediate to extended family members, friends, physicians, and employers—but not always in this same exact order.
As we will see, several key medical, social, and psychological factors also shaped these domains, including symptoms (whether patients displayed physical evidence of a disease or not), genetic testing (whether they had been tested or not, and if so, whether they had a mutation), family history (whether family members had the disease, and if so, who), socioeconomic status, age, gender, ethnicity, religion, education, other personal experiences (e.g., past trauma), and psychological traits (appearing more or less comfortable with, or able to tolerate, ambiguity). Usually, for each person, one or more of these phenomena proved more potent than others, yet no one characteristic wholly determined an individual’s views.
A major overarching theme of this book concerns the degrees to which experiences and responses concerning genetics are in fact social. Specifically, these men and women confront these challenges in interpersonal contexts that profoundly shape the outcomes. DNA involves not simply a single patient at a time, but rather a complex social milieu involving family members (past, present, and future), physicians, pharmaceutical and biotechnology companies, policymakers, and others.
While controversies rage between “socially-constructed” versus biological or “essentialist” approaches in understanding human beings, these men and women highlight intricate negotiations and interactions between these two broad perspectives, highlighting how social and biological factors can in fact interplay.
Of note, genetic reductionism or determinism does not easily sway these interviewees. Rather, these individuals hold much more complex and nuanced perspectives. For instance, in seeking to make sense of their genetic risks, these men and women seek, too, to avoid fatalism and despair. They desire a modicum of control, and frequently a scapegoat for their problems. Genetic determinism competes with beliefs in “being able to control your fate.” In confronting the fact that genetics may lie beyond their control, they struggle with quandaries of what, if anything, they can do about it. They have to balance others’ rights to this information against their own fears of stigma and rejection.
THE DISEASES AND THEIR SYMPTOMS
The three disorders I focus on here vary in several ways, including prevalence, treatability, and their predictability.
Since genetics involves several complex concepts, with which some readers may have little familiarity, Appendix A provides a brief “Genetics Testing 101,” offering additional background in the field.
Huntington’s Disease
The first human disease for which a mutation was discovered, HD, is a fatal autosomal dominant disorder that starts in adulthood—usually in a patient’s forties or fifties. Each of a patient’s children has a 50% chance of having the mutation as well. A person who possesses this mutation will eventually die of it (unless succumbing to another fatal condition first), usually at about the same age as did the affected parent. The disease causes neurological and psychiatric symptoms—including discoordination, psychosis, and loss of memory and cognition—and has no proven treatment. One of the most frightening aspects of the disease is that the patient, in losing his or her mind, seems to become a different person. The mutation actually consists of a sequence of DNA that gets repeated too many times. The severity of the illness increases with the number of abnormal repeats. The mutation is more common among people of Western European descent; worldwide, it occurs in 1 in 20,000 people.5, 6Patients’ adult offspring face quandaries of whether to be tested. After Woody Guthrie died of the mutation, his son, Arlo Guthrie, publicly decided not to undergo testing. Evolutionarily, the mutation has spread because it occurs late in a patient’s life—ordinarily after one had already had children and passed it on to them.
Alpha
Alpha-1 antitrypsin deficiency (AAT or “Alpha”) is the only disease for which the U.S. Equal Employment Opportunity Commission has ruled that genetic discrimination has occurred.7,8 Resulting from defective production of the enzyme Alpha-1 antitrypsin, the disease was discovered in 1963,9 and affects the liver and lungs. If left untreated, it can cause early death. In severe cases, organ transplant can extend life. Symptoms can occur in homozygotes (with two copies of the mutation, one from each parent) and to a lesser extent, heterozygotes (with the mutation from one parent, and a normal gene from the other parent). Early genetic testing can prompt avoidance of environmental factors that can exacerbate illness (e.g., smoking and certain pollutants), enzyme replacement therapy, organ transplants, and reproductive planning to avoid transmitting the disease to offspring.10
In the United States, approximately 60,000 people (1 in 5,000) are homozygotes,11 yet only 5% have been diagnosed.12 Worldwide, about 10,000 patients now receive treatment.13 Of Americans who would benefit from treatment, over 80% do not receive it. Many physicians remain unaware of the illness.14 Diagnosis itself can cause depression and anxiety.15 The World Health Organization,16 the American Thoracic Society,17 and the European Respiratory Society have recommended that in Europe and North America, given the relatively high prevalence of the disease, physicians should test certain patients, including those with emphysema, chronic obstructive pulmonary disease, asthma with air flow obstruction, and unexplained liver disease, and relatives of patients with Alpha.10 To what extent doctors follow these recommendations is unknown, however. In fact, despite the increased attention given to this disorder since its discovery, between 1968 and 2003 diagnoses by physicians did not increase, and the length of time between symptoms and diagnosis increased.12 Patients often report insufficient genetic counseling.14,15
Breast Cancer
Each year in the United States, over 250,000 women are diagnosed, and over 40,000 die of breast cancer, the second leading cause of cancer death among women.18,19 In the early 1990s, the BRCA1 and BRCA2 mutations were discovered, and account for 5 to 10% of all cases.19,20 Of women, 12% in the general population, and 40–60% of those with the BRCA 1/2 mutations will eventually develop breast cancer.21 Treatment can include chemotherapy, radiation, and surgery. The American Cancer Society now recommends that women start screening at age 40, and that those at high risk (a greater than 20% lifetime risk) receive both a mammogram and a MRI yearly.22 The molecular diagnostic company Myriad Genetics has marketed genetic testing for the two mutations, which cost approximately $3,000–$5,000, and are not always covered by insurance. The number of BRCA 1/2 tests performed so far is unknown.
METHODS
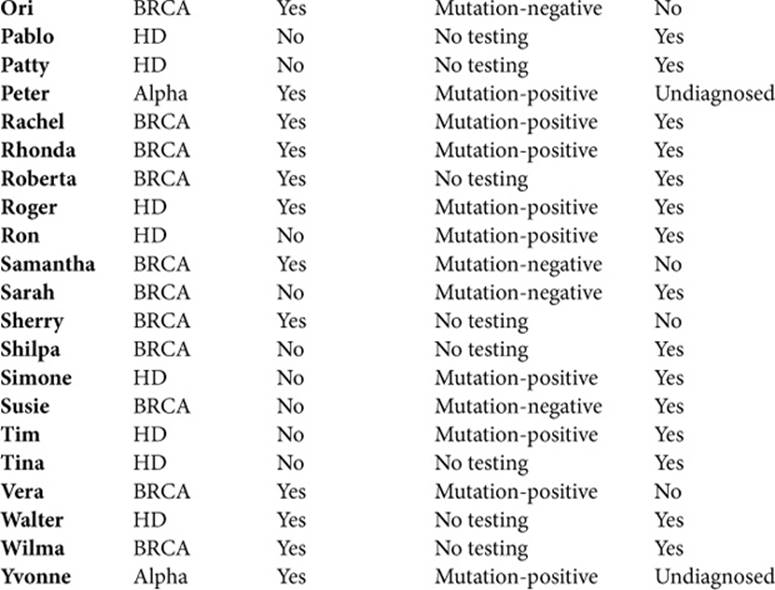
To understand these issues as fully as possible, I interviewed a cross-section of individuals who did and did not have symptoms, and had and had not had genetic testing. Over time, many people had in fact occupied more than one of these categories (going from asymptomatic to symptomatic, and from untested to tested). Table 1.1 summarizes these participants.
Table 1.1. SAMPLE CHARACTERISTICS (N=64)
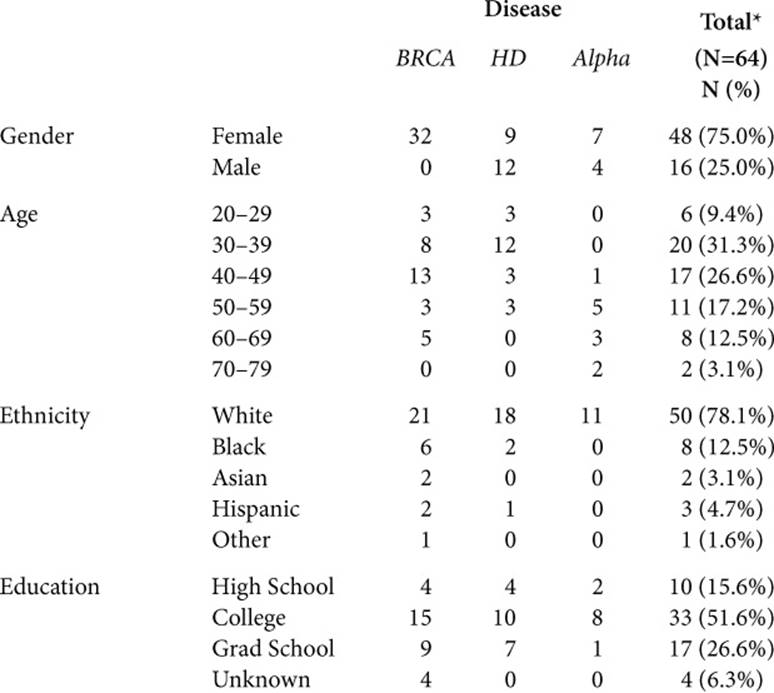
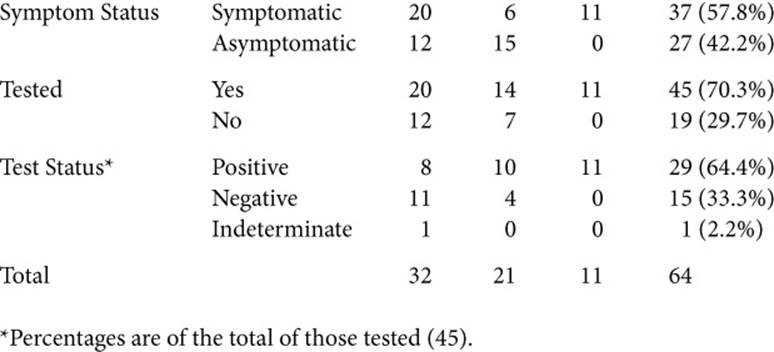
This book aims to illustrate the themes that emerge as richly, but concisely as possible, and thus includes quotations from 62 of these interviewees: 46 women and 16 men. Of these, 30 (all women) confronted breast cancer, 21 faced HD, and 11 Alpha; 49 were Caucasian, 7 were African American, 3 were Hispanic, and 2 were Asian; 44 had had genetic testing, of which 29 had a mutation; 36 had symptoms, and 26 did not.
In brief, as described more fully in Appendix B, I first conducted pilot interviews, and then further developed and refined the interview guide (see Appendix C) to obtain detailed descriptions of these issues. I recruited participants through staff and advertisements in medical clinics, patient organizations, a breast cancer registry, and through word of mouth. Individuals contacted me if they were interested. I interviewed each participant in-depth for two hours concerning experiences of being at risk for a genetic disease, undergoing genetic testing, and/or learning their gene status. I conducted the interviews at participants’ offices or homes or in my office—whichever was more convenient for them.
These men and women spoke not only of their own experiences, but those of siblings, parents, other family members, and friends as well. I interviewed relatively fewer patients with Alpha because their experiences were, overall, more uniform. Known patients with this disorder have essentially all undergone testing because of symptoms. With HD and breast cancer, patients included those who had tested but not had symptoms, had symptoms but no testing, and had neither symptoms nor testing. Given these additional categories, I conducted more interviews regarding these two disorders.
As we shall see, overall, between these three diseases, the similarities in themes far outweigh the differences. Hence, I have presented the themes for these diseases together. Yet “genetic disease” is not monolithic, and when differences arose, I have presented these as well. I use the term “genetic disease” to refer to these three diseases, since strong genetic components have been identified for each.
Many of the issues here do not relate primarily to the fact that the diagnosis is associated with a genetic marker, but to other aspects of these disorders as well. To completely disentangle genetics from others features of these diseases is impossible. Stresses concerning genetics occur in addition to other challenges a diagnosis presents. Nonetheless, I have tried to tease out, and focus on, these genetic realms.
I have set out to convey how these individuals see and experience the predicaments they face—how they frame concepts to make sense of these psychological, social, and moral dilemmas, and what words they use. I was continually struck by their eloquence as they conveyed their struggles, physical and metaphysical concerns, and quests for meaning. For this reason I have presented their views in their own voices, as they chart what they learned and how they changed.
I have woven together these individuals’ stories, presenting here a tapestry that portrays a series of journeys. In doing so, I beg the readers’ patience and indulgence: I could have instead focused on only one of these diseases, or picked merely a handful of these people to present, and just ignored the rest. But that would have limited the range of insights that could be presented about genetics as a whole. Each of these men and women underwent a somewhat different journey, and offers unique perspectives. They varied in whether they had genetic testing, symptoms, or affected family members; were single or married; and wanted children or not. Each brought their individual background and past experiences to the quandaries they now faced.
Consequently, to illuminate the wide and varied realms of life that genetics affects, and the array of issues and experiences they encountered, I have presented a group portrait. Given that genetics continues to evolve, to understand as full a range as possible of these challenges is vital. Though this approach may make it harder for readers to grasp the full details of any one person’s life, at various points I also draw on particular people more than others, providing more of a sense of a person’s perspectives and experiences. Their names (all pseudonyms) are listed below, (see Table 1.2) here along with brief descriptions. In addition, each interviewee also appears in the index, along with a list of the pages on which he or she is quoted, allowing readers to refer back to other quotations by that person. To protect confidentiality, I have changed a few identifying details.
A critic might aver that these interviewees’ experiences, confronting mutations that are relatively predictive of particular disorders, have little, if any, relevance to the issues that future, additional types of genetic testing may pose. In upcoming decades, more widespread testing may identify genetic markers that predict responses to medications, and only modestly increased risks of disease.
Yet though genetic discoveries advance, our comprehension of their multiple ethical, legal, social, psychological, cultural, and medical implications lags woefully behind. Hence, understanding experiences in the present can best help us anticipate challenges that may emerge in the future. Moreover, though scientists will most likely find that the most common diseases do not result from a single mutation, researchers may discover that several of these markers together, at times also in conjunction with environmental factors, may prove somewhat predictive. Unfortunately, we do not yet fully know what these future kinds of genetic tests will be (e.g., how much they will predict medication responses or small increased chances of disease) or how individuals will respond to all of this information. Certainly, however, future generations will encounter growing and bewildering amounts and types of genetic data.
Table 1.2. INDIVIDUALS INTERVIEW
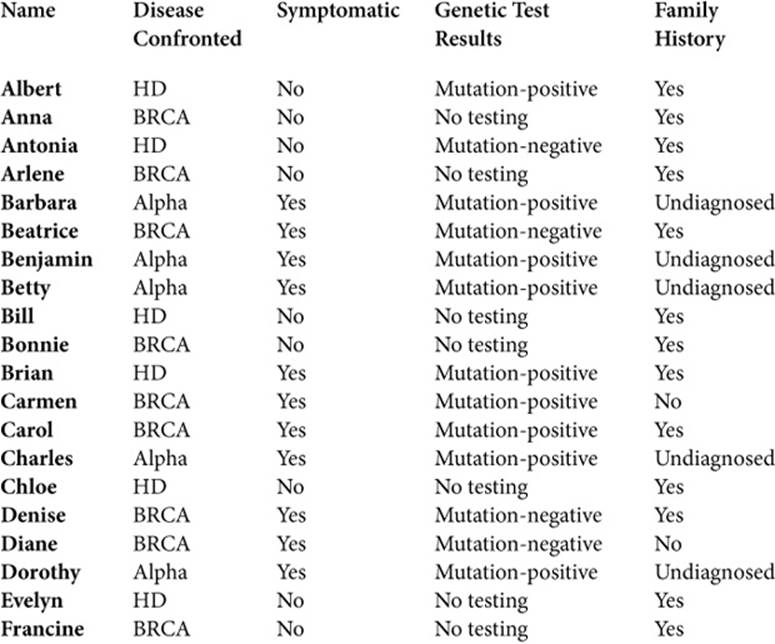
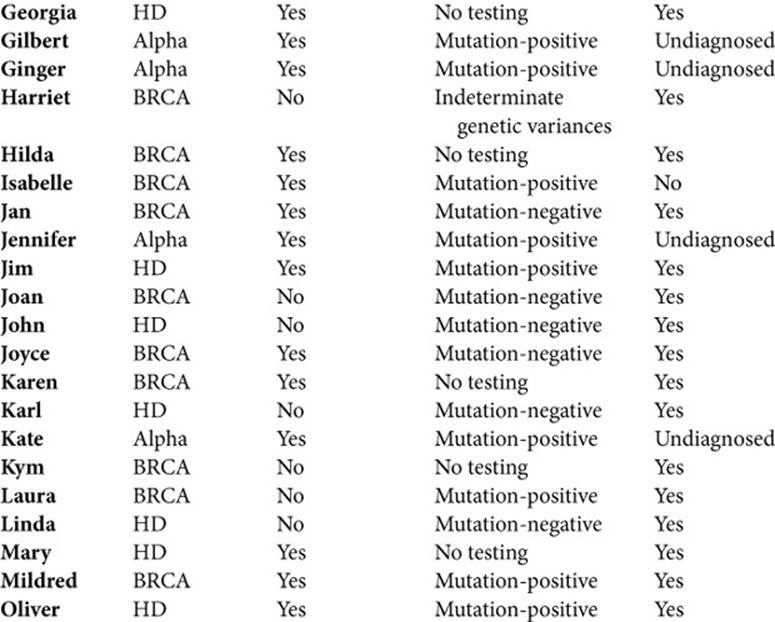
The three disorders here are not the most common diseases, but the insights these men and women offer are thus nonetheless critical. Given that the predictiveness of genetic markers discovered in the future remains unclear, we need to prepare ourselves as best we can in the meantime, based on what we can now know.
It is impossible to gauge definitively whether a larger sample of interviewees, or one recruited from a different culture or with other sociodemographics, would generate the same themes as those here. These data are not necessarily generalizable to other groups, but are valuable in and of themselves as embodiments of fundamental human issues. Indeed, perhaps most importantly, the stories of the men and women here illuminate how people seek to understand, at the most profound levels, themselves, their lives, and their world. Qualitative research can generate key hypotheses and insights, and elucidate the experiences and views of interviewees. But no psychosocial data are generalizable to all people everywhere. Surely, individuals from other cultures will differ in certain ways. But I would suggest that the men and women here highlight crucial underlying tensions—for example, whether to test, whom to tell, what genetic risks mean, how to view fate, identity, and family bonds, and how others will see them as a result—and that these broad challenges will probably have strong underlying similarities to those that arise among other individuals confronting genetic risks in the future. These interviews are noteworthy, too, because no other book has brought together this breadth of experiences concerning the roles of different genetic markers in individuals’ lives. Clearly, further research is also needed to determine how often and in what ways the issues here appear among persons facing different diseases in other cultures.
But importantly, these men and women have much to teach us, suggesting that people confronting future tests will most likely also struggle to make sense of this bewildering information. New assays will also no doubt serve as Rorschachs, with which individuals grapple to construct narratives. The themes here reflect key universal dilemmas related to who we are as human beings, and in what ways, and to what degrees biology defines us. These men and women illustrate how genetics throws into bold relief numerous ancient, timeless conundra. In fact, one of the major findings here—concerning the considerable difficulties people have making sense of these genetic risks, and the subjective ways they wrestle to frame and construct understandings—will most likely be of ever-increasing importance, since future tests of less certain predictiveness will, I suspect, be harder, rather than easier, to comprehend.
Over coming years and decades, we cannot fully foresee what science will bring, but we will, it seems, have more control over our future and certainly that of our children. Genetics will no doubt continue to challenge key aspects of how we see ourselves, but many of us will be unsure how to proceed. Countervailing pushes will continue, based on religion, wariness of science, notions of social construction, and other views and beliefs. Unintended consequences will result. Increasing genetic information is also entering into a world of widening gaps between the haves and have-nots. Corporations will seek to profit as much as they can from genetics. The wealthy will seek to use it to help themselves and their offspring. The poor will have less access to it, given that many still lack basic health care. Unfortunately, among some people, certain discoveries may fuel racist attitudes. Challenges will thus persist in how we understand, seek, use, discuss, and respond to genetic tests.
Hence, the feelings, reactions, and lived experiences of the individuals here, confronting a series of dilemmas posed by current science—even if due to relatively rare disorders—are valuable. Their tales can help us, shedding crucial light on issues that many of us—whether as patients, family members, friends, coworkers, or voters—may one day face.