ESTIMATED GESTATIONAL AGE USING MENSTRUAL DATES
INCIDENCE
PERINATAL MORTALITY
PATHOPHYSIOLOGY
COMPLICATIONS
MANAGEMENT
INTRAPARTUM MANAGEMENT
The adjectives postterm, prolonged, postdates, and postmature are often loosely used interchangeably to describe pregnancies that have exceeded a duration considered to be the upper limit of normal. We do not recommend use of the term postdates because the real issue in many postterm pregnancies is “post-what dates?” Postmature is reserved for the relatively uncommon specific clinical fetal syndrome in which the infant has recognizable clinical features indicating a pathologically prolonged pregnancy. Therefore, postterm or prolonged pregnancy is our preferred expression for an extended pregnancy.
The international definition of prolonged pregnancy, endorsed by the American College of Obstetricians and Gynecologists (2013a), is 42 completed weeks—294 days—or more from the first day of the last menstrual period. It is important to emphasize the phrase “42 completed weeks.” Pregnancies between 41 weeks 1 day and 41 weeks 6 days, although in the 42nd week, do not complete 42 weeks until the seventh day has elapsed. Thus, technically speaking, prolonged pregnancy could begin either on day 294 or on day 295 following the onset of the last menses. Which is it? Day 294 or 295? We cannot resolve this question, and emphasize this dilemma only to ensure that litigators and others understand that some imprecision is inevitable when there is biological variation such as with prolonged pregnancy. Amersi and Grimes (1998) have cautioned against use of ordinal numbers such as “42nd week” because of imprecision. For example, “42nd week” refers to 41 weeks and 1 through 6 days, whereas the cardinal number “42 weeks” refers to precisely 42 completed weeks.
ESTIMATED GESTATIONAL AGE USING MENSTRUAL DATES
The definition of postterm pregnancy as one that persists for 42 weeks or more from the onset of a menstrual period assumes that the last menses was followed by ovulation 2 weeks later. That said, some pregnancies may not actually be postterm, but rather are the result of an error in gestational age estimation because of faulty menstrual date recall or delayed ovulation. Thus, there are two categories of pregnancies that reach 42 completed weeks: (1) those truly 40 weeks past conception, and (2) those of less-advanced gestation but with inaccurately estimated gestational age. Even with precisely recalled menstrual dates, there is still not precision. Specifically, Munster and associates (1992) reported that large variations in menstrual cycle lengths are common in normal women. Boyce and coworkers (1976) studied 317 French women with periconceptional basal body temperature profiles. Almost 70 percent who completed 42 postmenstrual weeks had a less-advanced gestation based on ovulation dates. These variations in the menstrual cycle may partially explain why a relatively small proportion of fetuses delivered postterm have evidence of postmaturity syndrome. Even so, because there is no accurate method to identify the truly prolonged pregnancy, all those judged to have reached 42 completed weeks should be managed as if abnormally prolonged. Sonographic evaluation of gestational age during pregnancy has been used to add precision. Blondel and colleagues (2002) studied 44,623 women delivered at the Royal Victoria Hospital in Montreal. Postterm pregnancy rates were analyzed according to six algorithms for gestational age based on the last menstrual period, sonographic evaluation at 16 to 18 weeks, or both. The proportion of births at 42 weeks or longer was 6.4 percent when based on the last menstrual period alone, but was 1.9 percent when based on sonographic measurements alone. Similarly, sonographic pregnancy dating at ≤ 12 weeks resulted in a 2.7-percent incidence of postterm gestation compared with 3.7 percent in a group assessed at 13 to 24 weeks (Caughey, 2008). These findings suggest that menstrual dates are frequently inaccurate in predicting postterm pregnancy. Other clinical studies confirmed these observations (Bennett, 2004; Joseph, 2007; Wingate, 2007).
INCIDENCE
From a review, the incidence of postterm pregnancy ranged from 4 to 19 percent (Divon, 2008). Using criteria that likely overestimate the incidence, approximately 6 percent of 4 million infants born in the United States during 2009 were estimated to have been delivered at 42 weeks or more (Martin, 2011). The trend toward fewer births at 42 weeks suggests earlier intervention. Specifically, in 2000, 7.2 percent of births in this country were 42 weeks or beyond compared with 5.5 percent in 2009. Zhang and colleagues (2010) studied live births in this country from 1992 through 2003 and reported dramatic increases in labor induction rates at 41 and 42 weeks’ gestation (Fig. 43-1). This significantly increased use of labor induction was posited to contribute to a downward shift of the overall mean birthweight for these infants born in the United States.
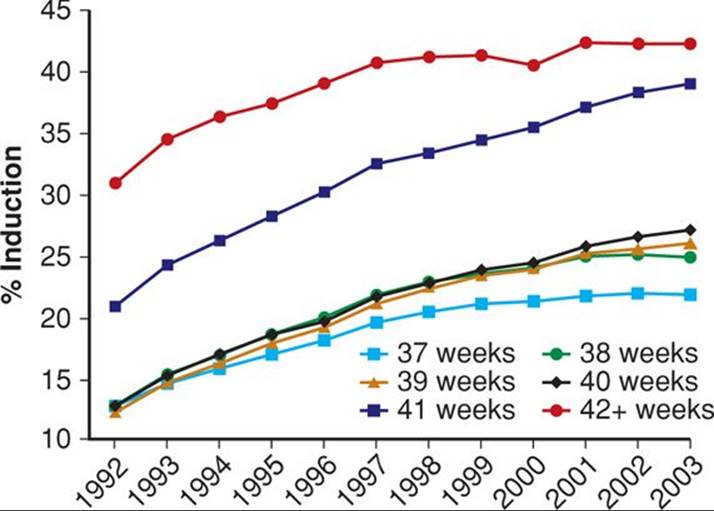
FIGURE 43-1 Rates of induction by gestational age among non-Hispanic white singleton livebirths ≥ 37 weeks from 1992 through 2003. (Redrawn from Zhang, 2010, with permission.)
There are contradictory findings concerning maternal demographic factors such as parity, prior postterm birth, socioeconomic class, and age as risks for postterm pregnancy. Olesen and associates (2006) analyzed various risk factors in 3392 participants in the 1998 to 2001 Danish Birth Cohort. Only prepregnancy body mass index (BMI) ≥ 25 and nulliparity were significantly associated with prolonged pregnancy. Caughey (2009) and Arrowsmith (2011) and their coworkers also reported similar associations.
The tendency for some mothers to have repeated postterm births suggests that some prolonged pregnancies are biologically determined. And Mogren and colleagues (1999) reported that prolonged pregnancy recurred across generations in Swedish women. When mother and daughter had a prolonged pregnancy, the risk for the daughter to have a subsequent postterm pregnancy was increased two- to threefold. In another Swedish study, Laursen and associates (2004) found that maternal, but not paternal, genes influenced prolonged pregnancy. As noted in Chapter 21 (p. 426), rare fetal–placental factors that have been reported as predisposing to postterm pregnancy include anencephaly, adrenal hypoplasia, and X-linked placental sulfatase deficiency (MacDonald, 1965).
PERINATAL MORTALITY
Perinatal mortality rates increase after the expected due date has passed. This is best seen when perinatal mortality rates are analyzed from times before widespread intervention for postterm pregnancies. In two large Swedish studies shown in Figure 43-2, after reaching a nadir at 39 to 40 weeks, the perinatal mortality rate increased as pregnancy exceeded 41 weeks. This trend has also been reported for the United States (MacDorman, 2009). Another study in this country of more than 2.5 million births has similar findings (Cheng, 2008). The major causes of death in these pregnancies included gestational hypertension, prolonged labor with cephalopelvic disproportion, “unexplained anoxia,” and malformations. Similar outcomes were reported by Olesen and colleagues (2003) in 78,022 women with postterm pregnancies delivered before routine labor induction was adopted in Denmark. Moster and associates (2010) found increased rates of cerebral palsy in postterm births, and Yang and coworkers (2010) reported lower intelligence quotient (IQ) scores at age 6.5 years in children born ≥ 42 weeks’ gestation. Conversely, autism was not associated with postterm birth (Gardener, 2011).
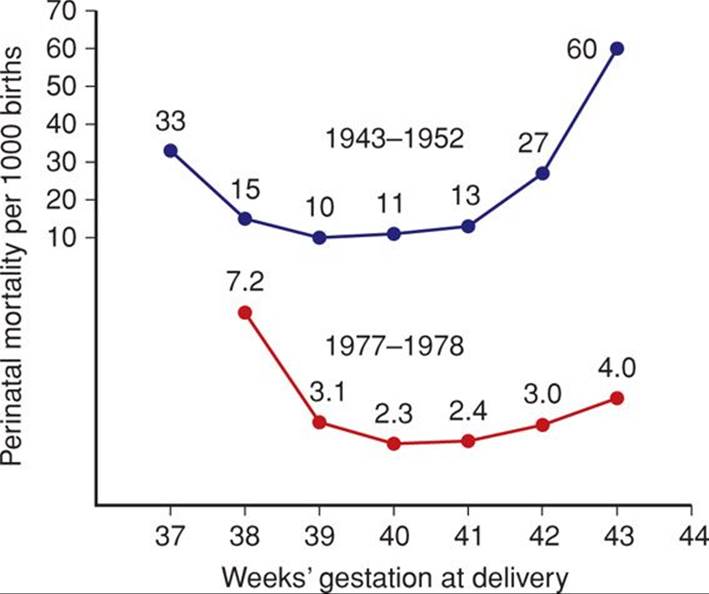
FIGURE 43-2 Perinatal mortality rates in late pregnancy according to gestational age in Sweden of all births during 1943–1952 compared with those during 1977–1978. The partially compressed scale is used for convenience in depiction. (Adapted from Bakketeig, 1991, and Lindell, 1956.)
Alexander and colleagues (2000a) reviewed 56,317 consecutive singleton pregnancies delivered at ≥ 40 weeks between 1988 and 1998 at Parkland Hospital. As shown in Table 43-1, labor was induced in 35 percent of pregnancies completing 42 weeks. The rate of cesarean delivery for dystocia and fetal distress was significantly increased at 42 weeks compared with earlier deliveries. More infants of postterm pregnancies were admitted to intensive care units. Importantly, the incidence of neonatal seizures and deaths doubled at 42 weeks. Smith (2001) has challenged analyses such as these because the population at risk for perinatal mortality in a given week consists of all ongoing pregnancies rather than just the births in a given week. Figure 43-3 shows perinatal mortality rates calculated using only births in a given week of gestation from 37 to 43 completed weeks compared with the cumulative probability—the perinatal index—of death when all ongoing pregnancies are included in the denominator. As shown, delivery at 38 weeks had the lowest risk index for perinatal death.
TABLE 43-1. Pregnancy Outcomes in 56,317 Consecutive Singleton Pregnancies Delivered at or Beyond 40 Weeks at Parkland Hospital from 1988 through 1998
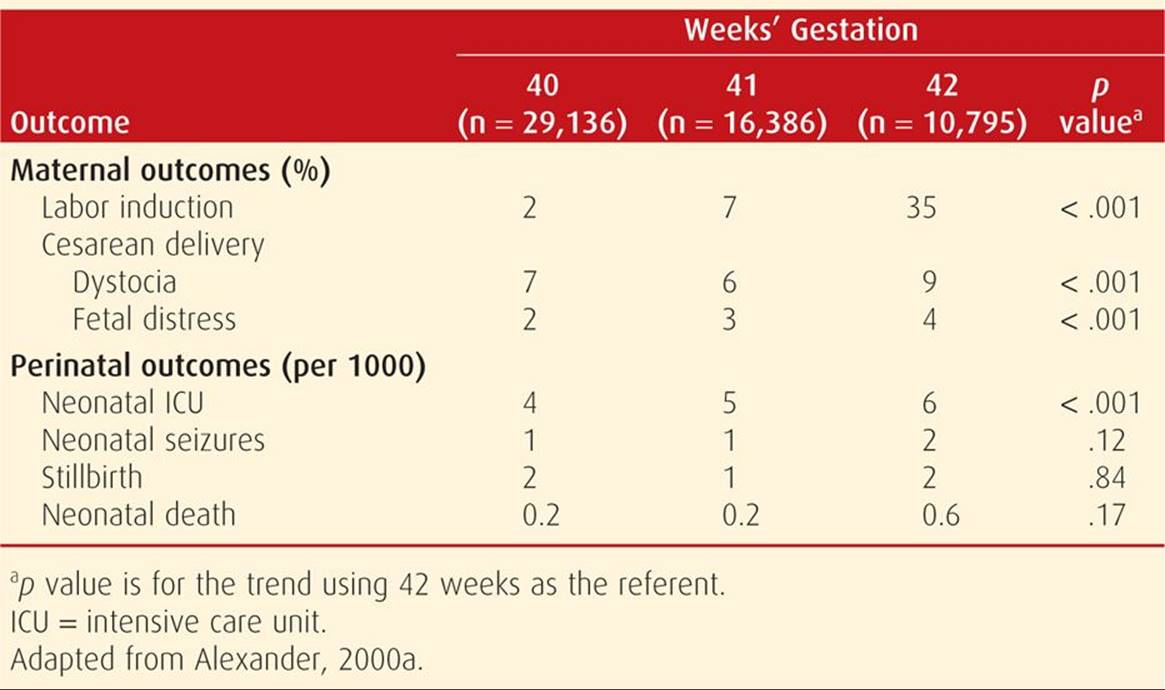
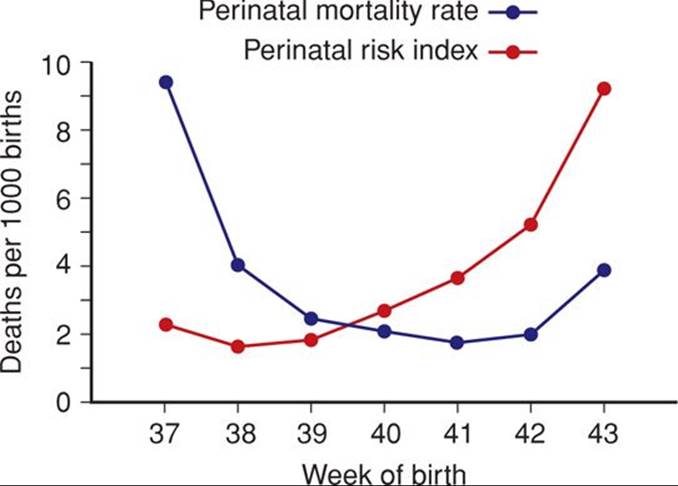
FIGURE 43-3 Perinatal mortality rate and perinatal risk index for births between 37 and 43 weeks in Scotland from 1985 through 1996. The perinatal mortality rate is the number of perinatal deaths with delivery in a given gestational week divided by the total number of births in that week multiplied by 1000. The perinatal risk index is the cumulative probability of perinatal death multiplied by 1000. (Redrawn from Smith, 2001, with permission.)
PATHOPHYSIOLOGY
![]() Postmaturity Syndrome
Postmaturity Syndrome
The postmature infant presents a unique appearance such as shown in Figure 43-4. Features include wrinkled, patchy, peeling skin; a long, thin body suggesting wasting; and advanced maturity in that the infant is open-eyed, unusually alert, and appears old and worried. Skin wrinkling can be particularly prominent on the palms and soles. The nails are typically long. Most postmature infants are not technically growth restricted because their birthweight seldom falls below the 10th percentile for gestational age. On the other hand, severe growth restriction—which logically must have preceded completion of 42 weeks—may be present.
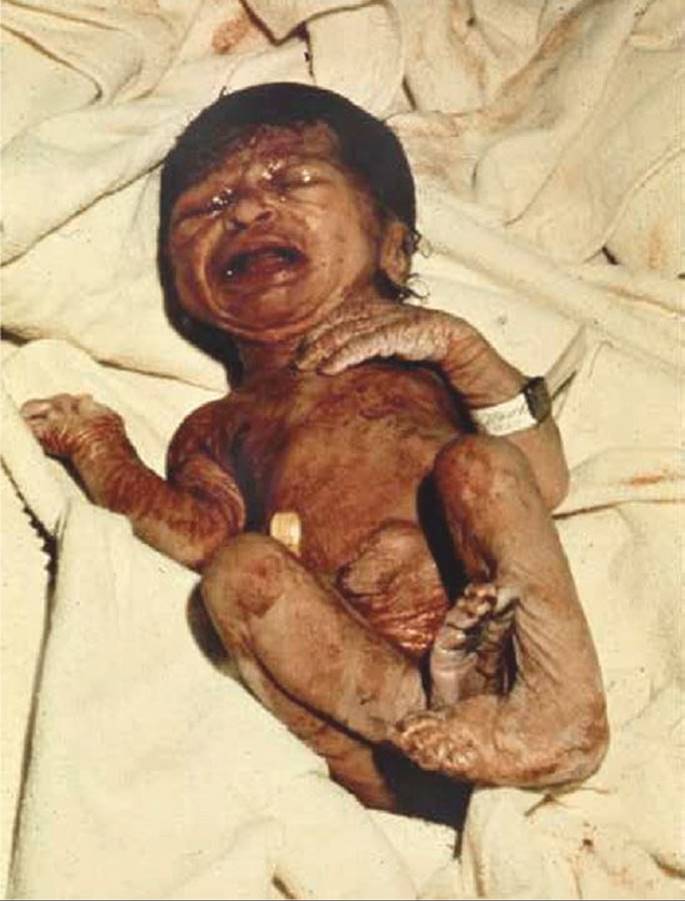
FIGURE 43-4 Postmaturity syndrome. Infant delivered at 43 weeks’ gestation with thick, viscous meconium coating the desquamating skin. Note the long, thin appearance and wrinkling of the hands.
The incidence of postmaturity syndrome in infants at 41, 42, or 43 weeks, respectively, has not been conclusively determined. In one of the rare contemporary reports that chronicle postmaturity, Shime and colleagues (1984) found this syndrome in approximately 10 percent of pregnancies between 41 and 43 weeks. The incidence increased to 33 percent at 44 weeks. Associated oligohydramnios substantially increases the likelihood of postmaturity. Trimmer and associates (1990) reported that 88 percent of infants were postmature if there was oligohydramnios defined by a sonographic maximal vertical amnionic fluid pocket that measured ≤ 1 cm at 42 weeks.
![]() Placental Dysfunction
Placental Dysfunction
Clifford (1954) proposed that the skin changes of postmaturity were due to loss of the protective effects of vernix caseosa. He also attributed the postmaturity syndrome to placental senescence, although he did not find placental degeneration histologically. Still, the concept that postmaturity is due to placental insufficiency has persisted despite an absence of morphological or significant quantitative findings (Larsen, 1995; Rushton, 1991). There are findings that placental apoptosis—programmed cell death—was significantly increased at 41 to 42 completed weeks compared with that at 36 to 39 weeks (Smith, 1999). Several proapoptotic genes such as kisspeptin were shown to be upregulated in postterm placental explants compared with the same genes in term placental explants (Torricelli, 2012). The clinical significance of such apoptosis is currently unclear.
Jazayeri and coworkers (1998) investigated cord blood erythropoietin levels in 124 appropriately grown newborns delivered from 37 to 43 weeks. The only known stimulator of erythropoietin is decreased partial oxygen pressure. Thus, they sought to assess whether fetal oxygenation was compromised due to placental aging in postterm pregnancies. All women had an uncomplicated labor and delivery. These investigators reported that cord blood erythropoietin levels were significantly increased in pregnancies reaching 41 weeks or more. Although Apgar scores and acid-base studies were normal, these researchers concluded that there was decreased fetal oxygenation in some postterm gestations.
Another scenario is that the postterm fetus may continue to gain weight and thus be unusually large at birth. This at least suggests that placental function is not severely compromised. Indeed, continued fetal growth is the norm—albeit at a slower rate beginning at 37 completed weeks (Fig. 43-5). Nahum and colleagues (1995) confirmed that fetal growth continues until at least 42 weeks, however, Link and associates (2007) showed that umbilical blood flow did not increase concomitantly.
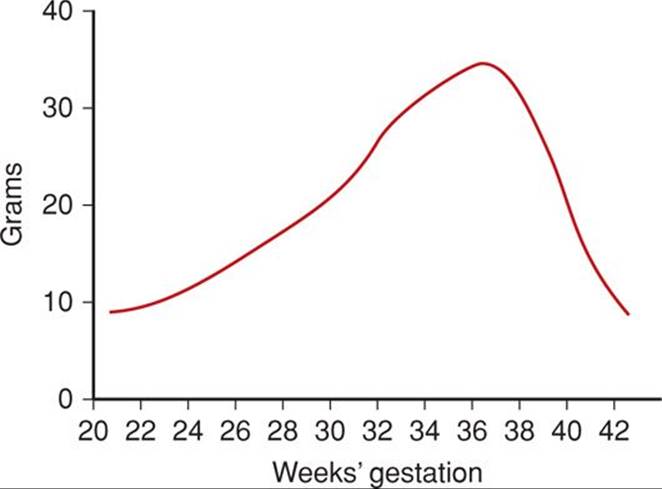
FIGURE 43-5 Mean daily fetal growth during previous week of gestation. (From Hendricks, 1964.)
![]() Fetal Distress and Oligohydramnios
Fetal Distress and Oligohydramnios
The principal reasons for increased risks to postterm fetuses were described by Leveno and associates (1984). Both antepartum fetal jeopardy and intrapartum fetal distress were found to be the consequence of cord compression associated with oligohydramnios. In their analysis of 727 postterm pregnancies, intrapartum fetal distress detected with electronic monitoring was not associated with late decelerations characteristic of uteroplacental insufficiency. Instead, one or more prolonged decelerations such as shown in Figure 43-6 preceded three fourths of emergency cesarean deliveries for nonreassuring fetal heart rate tracings. In all but two cases, there were also variable decelerations (Fig. 43-7). Another common fetal heart rate pattern, although not ominous by itself, was the saltatory baseline shown in Figure 43-8. As described in Chapter 24 (p. 484), these findings are consistent with cord occlusion as the proximate cause of the nonreassuring tracings. Other correlates included oligohydramnios and viscous meconium. Schaffer and colleagues (2005) implicated a nuchal cord in abnormal intrapartum fetal heart rate patterns, meconium, and compromised newborn condition in prolonged pregnancies.
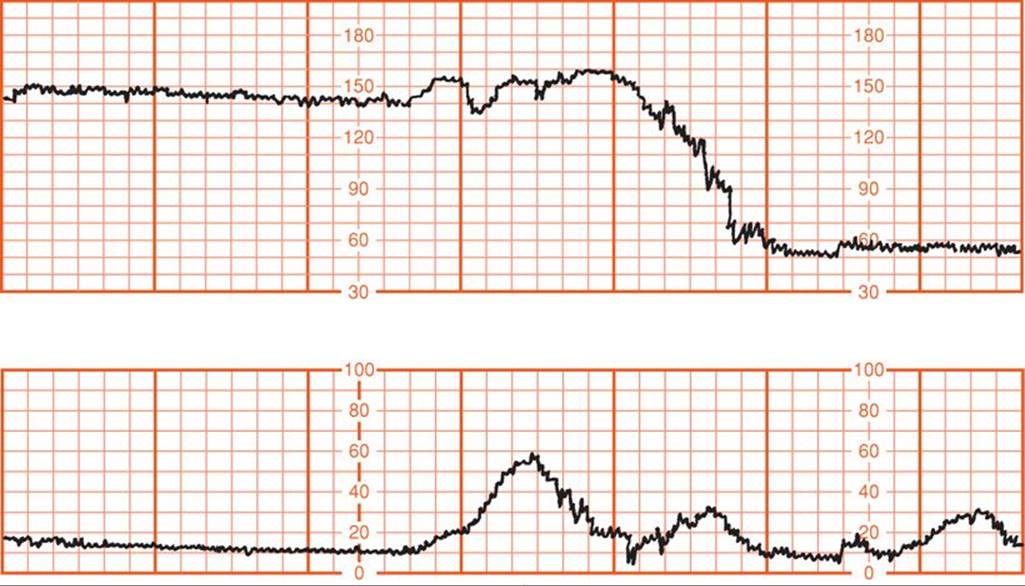
FIGURE 43-6 Prolonged fetal heart rate deceleration before emergency cesarean delivery in a postterm pregnancy with oligohydramnios. (Redrawn from Leveno, 1984, with permission.)
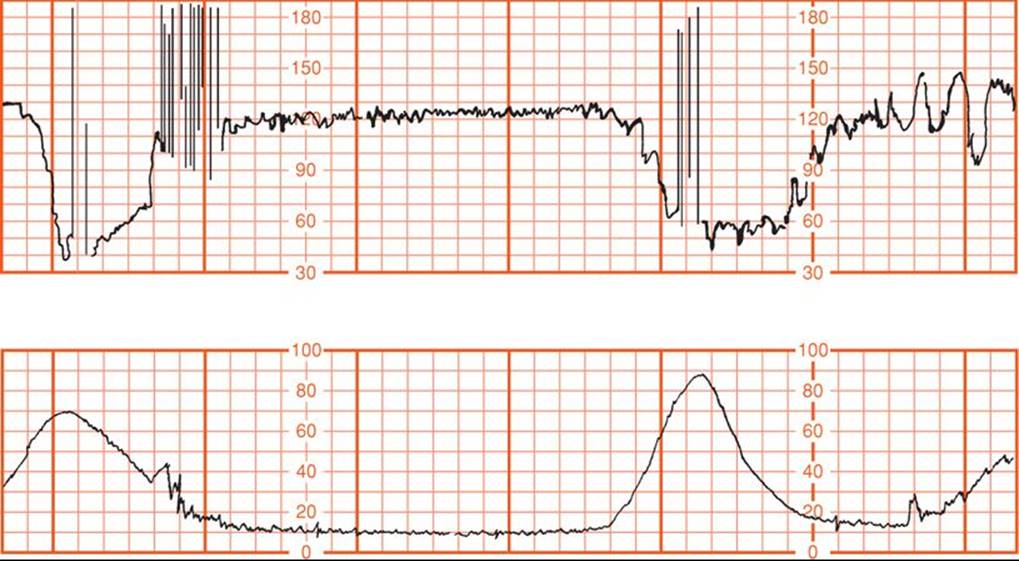
FIGURE 43-7 Severe—less than 70 bpm for 60 seconds or longer—variable decelerations in a postterm pregnancy with oligohydramnios. (Redrawn from Leveno, 1984, with permission.)
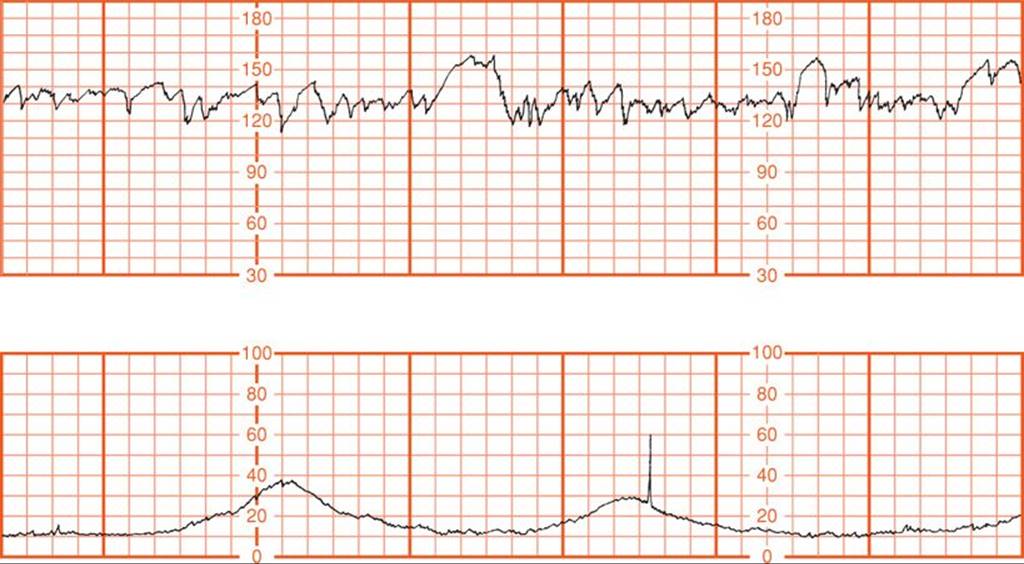
FIGURE 43-8 Saltatory baseline fetal heart rate showing oscillations exceeding 20 bpm and associated with oligohydramnios in a postterm pregnancy. (Redrawn from Leveno, 1984, with permission.)
The volume of amnionic fluid normally continues to decrease after 38 weeks and may become problematic (Fig. 11-1, p. 233). Moreover, meconium release into an already reduced amnionic fluid volume results in thick, viscous meconium that may cause meconium aspiration syndrome (Chap. 33, p. 637).
Trimmer and coworkers (1990) sonographically measured hourly fetal urine production using sequential bladder volume measurements in 38 pregnancies of ≥ 42 weeks. Diminished urine production was found to be associated with oligohydramnios. They hypothesized that decreased fetal urine flow was likely the result of preexisting oligohydramnios that limited fetal swallowing. Oz and associates (2002), using Doppler waveforms, concluded that fetal renal blood flow is reduced in those postterm pregnancies complicated by oligohydramnios. The study mentioned above, by Link and associates (2007), showed that umbilical blood flow did not increase past term.
![]() Fetal-Growth Restriction
Fetal-Growth Restriction
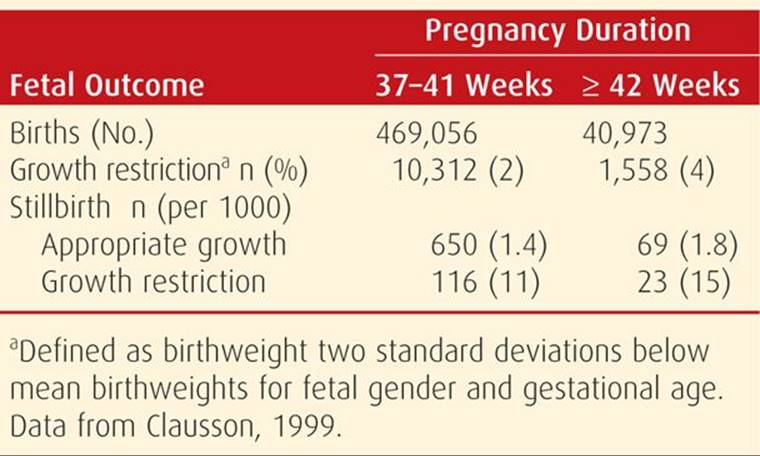
It was not until the late 1990s that the clinical significance of fetal-growth restriction in the otherwise uncomplicated pregnancy became more fully appreciated. Divon (1998) and Clausson (1999) and their coworkers analyzed births between 1991 and 1995 in the National Swedish Medical Birth Registry. As shown in Table 43-2, stillbirths were more common among growth-restricted infants who were delivered after 42 weeks. Indeed, a third of postterm stillborn infants were growth restricted. During this time in Sweden, labor induction and antenatal fetal testing usually commenced at 42 weeks. In a study from Parkland Hospital, Alexander and colleagues (2000d) analyzed outcomes for 355 infants who were ≥ 42 weeks and whose birthweights were < 3rd percentile. They compared these with outcomes of 14,520 similarly aged infants above the 3rd percentile and found that morbidity and mortality rates were significantly increased in the growth-restricted infants. Notably, a fourth of all stillbirths associated with prolonged pregnancy were in this comparatively small number of growth-restricted infants.
TABLE 43-2. Effects of Fetal-Growth Restriction by Gestational Age on Stillbirth Rates in 537,029 Swedish Women Delivered before and after 42 Weeks
COMPLICATIONS
![]() OLIGOHYDRAMNIOS
OLIGOHYDRAMNIOS
Most clinical studies support the view that diminished amnionic fluid determined by various sonographic methods identifies a postterm fetus with increased risks. Indeed, decreased amnionic fluid in any pregnancy signifies increased fetal risk (Chap. 11, p. 238). Unfortunately, lack of an exact method to define “decreased amnionic fluid” has limited investigators, and many different criteria for sonographic diagnosis have been proposed. Fischer and colleagues (1993) attempted to determine which criteria were most predictive of normal versus abnormal outcomes in postterm pregnancies. As shown in Figure 43-9, the smaller the amnionic fluid pocket, the greater the likelihood that there was clinically significant oligohydramnios. Importantly, normal amnionic fluid volume did not preclude abnormal outcomes. Alfirevic and coworkers (1997) randomly assigned 500 women with postterm pregnancies to assessment using either the amnionic fluid index (AFI) or the deepest vertical pocket (Chap. 11, p. 232). They concluded that the AFI overestimated the number of abnormal outcomes in postterm pregnancies.
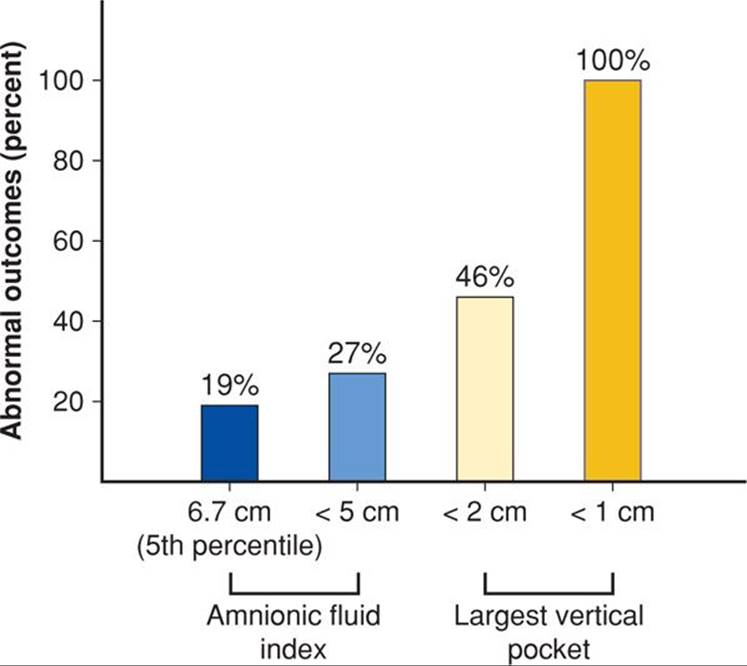
FIGURE 43-9 Comparison of the prognostic value of various sonographic estimates of amnionic fluid volume in prolonged pregnancies. Abnormal outcomes include cesarean or operative vaginal delivery for fetal jeopardy, 5-minute Apgar score ≤ 6, umbilical arterial blood pH < 7.1, or admission to the neonatal intensive care unit. (Adapted from Fischer, 1993.)
Regardless of the criteria used to diagnose oligohydramnios in postterm pregnancies, most investigators have found an increased incidence of some sort of “fetal distress” during labor. Thus, oligohydramnios by most definitions is a clinically meaningful finding. Conversely, reassurance of continued fetal well-being in the presence of “normal” amnionic fluid volume is tenuous. This may be related to how quickly pathological oligohydramnios develops. Although such cases are unusual, Clement and coworkers (1987) described six postterm pregnancies in which amnionic fluid volume diminished abruptly over 24 hours, and in one of these, the fetus died.
![]() Macrosomia
Macrosomia
The velocity of fetal weight gain peaks at approximately 37 weeks as shown in Figure 43-5. Although growth velocity slows at that time, most fetuses continue to gain weight. For example, the percentage of fetuses born in 2009 whose birthweight exceeded 4000 g was 8.2 percent at 37 to 41 weeks and increased to 11.0 percent at 42 weeks or more (Martin, 2011). However, in some studies, brachial plexus injury was not related to postterm gestation (Walsh, 2011). Intuitively at least, it seems that both maternal and fetal morbidity associated with macrosomia would be mitigated with timely induction to preempt further growth. This does not appear to be the case, however, and the American College of Obstetricians and Gynecologists (2013b) has concluded that current evidence does not support such a practice in women at term with suspected fetal macrosomia. Moreover, the College concluded that in the absence of diabetes, vaginal delivery is not contraindicated for women with an estimated fetal weight up to 5000 g. Cesarean delivery was recommended for estimated fetal weights greater than 4500 g if there is prolonged second-stage labor or arrest of descent. Obvious problems with all such recommendations are substantive variations in fetal weight estimation (Chap. 44, p. 885).
![]() Medical or Obstetrical Complications
Medical or Obstetrical Complications
In the event of a medical or other obstetrical complication, it is generally not recommended that a pregnancy be allowed to continue past 42 weeks. Indeed, in many such instances, earlier delivery is indicated. Common examples include gestational hypertensive disorders, prior cesarean delivery, and diabetes.
MANAGEMENT
Although some form of intervention is considered to be indicated for prolonged pregnancies, the types and timing of interventions are not unanimous (Divon, 2008). The decision centers on whether labor induction is warranted or if expectant management with fetal surveillance is best. In a survey done 10 years ago, Cleary-Goldman and associates (2006) reported that 73 percent of members of the American College of Obstetricians and Gynecologists routinely induced women at 41 weeks. Most of the remainder performed twice weekly fetal testing until 42 weeks.
![]() Prognostic Factors for Successful Induction
Prognostic Factors for Successful Induction
Unfavorable Cervix
Although all obstetricians know what an “unfavorable cervix” is, the term unfortunately defies precise objective definition. Thus, various investigators have used different criteria for studies of prolonged pregnancies. For example, Harris and coworkers (1983) defined an unfavorable cervix by a Bishop score < 7 and reported this in 92 percent of women at 42 weeks. Hannah and colleagues (1992) found that only 40 percent of 3407 women with a 41-week pregnancy had an “undilated cervix.” In a study of 800 women undergoing induction for postterm pregnancy at Parkland Hospital, Alexander and associates (2000b) reported that women in whom there was no cervical dilatation had a twofold increased cesarean delivery rate for “dystocia.” Yang and coworkers (2004) found that cervical length ≤ 3 cm measured with transvaginal sonography was predictive of successful induction. In a similar study, Vankayalapati and associates (2008) found that cervical length ≤ 25 mm was predictive of spontaneous labor or successful induction.
Cervical Ripening. A number of investigators have evaluated prostaglandin E2 (PGE2) for induction in women with an unfavorable cervix and prolonged pregnancies. The study by the National Institute of Child Health and Human Development Network of Maternal–Fetal Medicine Units (1994) found that PGE2 gel was not more effective than placebo. Alexander and associates (2000c) treated 393 women with a postterm pregnancy with PGE2, regardless of cervical “favorability,” and reported that almost half of the 84 women with cervical dilatation of 2 to 4 cm entered labor with prostaglandin E2 use alone. The American College of Obstetricians and Gynecologists (2011) previously concluded that PGE2 gel could be used safely in postterm pregnancies. In another study, mifepristone was reported to increase uterine activity without uterotonic agents in women beyond 41 weeks (Fasset, 2008). Prostaglandins and other agents used for cervical ripening are discussed in Chapter 26 (p. 525).
Sweeping or stripping of the membranes to induce labor and thereby prevent postterm pregnancy was studied in 15 randomized trials during the 1990s. Boulvain and coworkers (1999) performed a metaanalysis of these and found that membrane stripping at 38 to 40 weeks decreased the frequency of postterm pregnancy. Moreover, maternal and neonatal infection rates were not increased by cervical manipulation. However, this practice did not modify the cesarean delivery risk. Since then, randomized trials by Wong (2002), Kashanian (2006), Hill (2008), and their coworkers found that sweeping membranes did not reduce the need to induce labor. Drawbacks of membrane stripping included pain, vaginal bleeding, and irregular contractions without labor.
Station of Vertex
The station of the fetal head within the pelvis is another important predictor of successful postterm pregnancy induction. Shin and colleagues (2004) studied 484 nulliparas who underwent induction after 41 weeks. The cesarean delivery rate was directly related to station. It was 6 percent if the vertex before induction was at −1 station; 20 percent at −2; 43 percent at −3; and 77 percent at −4.
![]() Induction versus Fetal Testing
Induction versus Fetal Testing
Because of marginal benefits for induction with an unfavorable cervix as discussed above, some clinicians prefer to use the alternative strategy of fetal testing beginning at 41 completed weeks. There now are several quality studies designed to resolve these important questions.
In a Canadian study, 3407 women were randomly assigned at 41 or more weeks to induction or to fetal testing (Hannah, 1992). In the surveillance group, evaluation included: (1) counting fetal movements during a 2-hour period each day, (2) nonstress testing three times weekly, and (3) amnionic fluid volume assessment two to three times weekly with pockets < 3 cm considered abnormal. Labor induction resulted in a small but significant reduction in cesarean delivery rate compared with fetal testing—21 versus 24 percent, respectively. This difference was due to fewer procedures for fetal distress. Importantly, the only two stillbirths were in the fetal testing group.
The Maternal–Fetal Medicine Network performed a randomized trial of induction versus fetal testing beginning at 41 weeks (Gardner, 1996). Fetal surveillance included nonstress testing and sonographic estimation of amnionic fluid volume performed twice weekly in 175 women. Perinatal outcomes were compared with those of 265 women also at 41 weeks randomly assigned to induction with or without cervical ripening. There were no perinatal deaths, and the cesarean delivery rate was not different between management groups. The results of this study could be used to support the validity of either of these management schemes. Similar results were subsequently reported from a Norwegian randomized trial of 508 women (Heimstad, 2007).
In an analysis of 19 trials in the Cochrane Pregnancy and Childbirth Trials Registry, Gulmezoglu and colleagues (2006) found that induction after 41 weeks was associated with fewer perinatal deaths without significantly increasing the cesarean delivery rate. In a review of two metaanalyses and a recent randomized controlled study, similar conclusions were reached (Mozurkewich, 2009).
In an attempt to “lower” the number of postterm pregnancies, Harrington and associates (2006) randomly assigned 463 women to pregnancy dating with sonography between 8 and 12 weeks versus no first-trimester sonographic evaluation. Their primary end point was the labor induction rate for prolonged pregnancy, and they found no advantages to early sonographic pregnancy dating.
At 42 weeks, labor induction has a higher cesarean delivery rate compared with spontaneous labor. From Parkland Hospital, Alexander and coworkers (2001) evaluated pregnancy outcomes in 638 such women in whom labor was induced and compared them with outcomes of 687 women with postterm pregnancies who had spontaneous labor. Cesarean delivery rates were significantly increased—19 versus 14 percent—in the induced group because of failure to progress. When these investigators corrected for risk factors, however, they concluded that intrinsic maternal factors, rather than the induction itself, led to the higher rate. These factors included nulliparity, an unfavorable cervix, and epidural analgesia.
Evidence to substantiate intervention—whether induction or fetal testing—commencing at 41 versus 42 weeks is limited. Most evidence used to justify intervention at 41 weeks is from the randomized Canadian and American investigations cited earlier. No randomized studies have specifically assessed intervention at 41 weeks versus an identical intervention used at 42 weeks. But there have been observational studies. In one, Usher and colleagues (1988) analyzed outcomes in 7663 pregnancies in women at 40, 41, or 42 weeks confirmed by early sonographic evaluation. After correction for malformations, perinatal death rates were 1.5, 0.7, and 3.0 per 1000 at 40, 41, and 42 weeks, respectively. These results could be used to challenge the concept of routine intervention at 41 instead of 42 weeks.
![]() Management Recommendations
Management Recommendations
Because of the studies discussed above, the American College of Obstetricians and Gynecologists (2013a) defines postterm pregnancies as having completed 42 weeks. There is insufficient evidence to recommend a management strategy between 40 and 42 completed weeks. Thus, although not considered mandatory, initiation of fetal surveillance at 41 weeks is a reasonable option. After completing 42 weeks, recommendations are for either antenatal testing or labor induction. These are summarized in Figure 43-10.
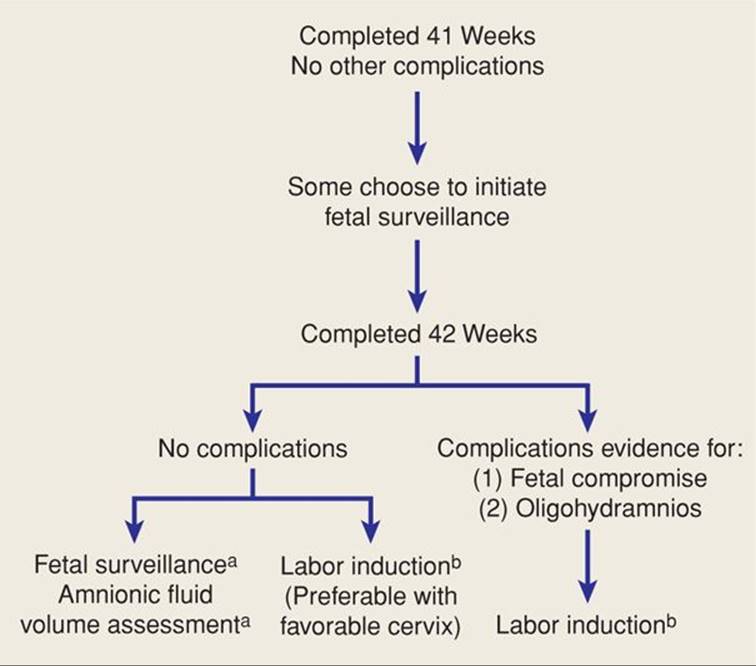
FIGURE 43-10 Management of postterm pregnancy.
aSee text for options
bprostaglandins may be used for cervical ripening or induction.
At Parkland Hospital, based on results discussed above, we consider 41-week pregnancies without other complications to be normal. Thus, no interventions are practiced solely based on fetal age until 42 completed weeks. If there are complications such as hypertension, decreased fetal movement, or oligohydramnios, then labor induction is carried out. It is our view that large, randomized trials should be performed before otherwise uncomplicated 41-week gestations are routinely considered pathologically prolonged. In women in whom a certain gestational age is known, labor is induced at the completion of 42 weeks. Almost 90 percent of such women are induced successfully or enter labor within 2 days of induction. For those who do not deliver with the first induction, a second induction is performed within 3 days. Almost all women are delivered using this management plan, but in the unusual few who are not delivered, management decisions involve a third—or even more—induction versus cesarean delivery. Women classified as having uncertain postterm pregnancies are managed with weekly nonstress fetal testing and assessment of amnionic fluid volume. Women with an AFI ≤ 5 cm or with reports of diminished fetal movement undergo labor induction.
INTRAPARTUM MANAGEMENT
Labor is a particularly dangerous time for the postterm fetus. Therefore, women whose pregnancies are known or suspected to be postterm should come to the hospital as soon as they suspect labor. While being evaluated for active labor, we recommend that fetal heart rate and uterine contractions be monitored electronically for variations consistent with fetal compromise.
The decision to perform amniotomy is problematic. Further reduction in fluid volume following amniotomy can certainly enhance the possibility of cord compression. Conversely, amniotomy aids identification of thick meconium, which may be dangerous to the fetus if aspirated. Also, after membrane rupture, a scalp electrode and intrauterine pressure catheter can be placed. These usually provide more precise data concerning fetal heart rate and uterine contractions.
Identification of thick meconium in the amnionic fluid is particularly worrisome. The viscosity probably signifies the lack of liquid and thus oligohydramnios. Aspiration of thick meconium may cause severe pulmonary dysfunction and neonatal death (Chap. 33, p. 637). Because of this, amnioinfusion during labor has been proposed as a way of diluting meconium to decrease the incidence of aspiration syndrome (Wenstrom, 1989). As discussed in Chapter 24 (p. 494), the benefits of amnioinfusion remain controversial. In a large randomized trial by Fraser and colleagues (2005), amnioinfusion did not reduce the risk of meconium aspiration syndrome or perinatal death. According to the American College of Obstetricians and Gynecologists (2012), amnioinfusion does not prevent meconium aspiration, however, it remains a reasonable treatment approach for repetitive variable decelerations.
The likelihood of a successful vaginal delivery is reduced appreciably for the nulliparous woman who is in early labor with thick, meconium-stained amnionic fluid. Therefore, if the woman is remote from delivery, strong consideration should be given to prompt cesarean delivery, especially when cephalopelvic disproportion is suspected or either hypotonic or hypertonic dysfunctional labor is evident. Some practitioners choose to avoid oxytocin use in these cases.
Until recently, it was taught—including at Parkland Hospital—that aspiration of meconium could be minimized but not eliminated by suctioning the pharynx as soon as the head was delivered. According to the American Academy of Pediatrics guidelines, this in no longer recommended (Perlman, 2010). The American College of Obstetricians and Gynecologists (2013c) does not recommend routine intrapartum suctioning. Alternatively, if the depressed newborn has meconium-stained fluid, then intubation is carried out. The American Academy of Pediatrics states that tracheal suctioning is neither supported nor refuted (Perlman, 2010).
REFERENCES
Alexander JM, McIntire DD, Leveno KJ: Forty weeks and beyond: pregnancy outcomes by week of gestation. Obstet Gynecol 96:291, 2000a
Alexander JM, McIntire DD, Leveno KJ: Postterm pregnancy: does induction increase cesarean rates? J Soc Gynecol Invest 7:79A, 2000b
Alexander JM, McIntire DD, Leveno KJ: Postterm pregnancy: is cervical “ripening” being used in the right patients? J Soc Gynecol Invest 7:247A, 2000c
Alexander JM, McIntire DD, Leveno KJ: The effect of fetal growth restriction on neonatal outcome in postterm pregnancy. Abstract No. 463. Am J Obstet Gynecol 182:S148, 2000d
Alexander JM, McIntire DD, Leveno KJ: Prolonged pregnancy: induction of labor and cesarean births. Obstet Gynecol 97:911, 2001
Alfirevic Z, Luckas M, Walkinshaw SA, et al: A randomized comparison between amniotic fluid index and maximum pool depth in the monitoring of postterm pregnancy. Br J Obstet Gynaecol 104:207, 1997
American College of Obstetricians and Gynecologists: Management of postterm pregnancy. Practice Bulletin No. 55, September 2004, Reaffirmed 2011
American College of Obstetricians and Gynecologists: Amnioinfusion does not prevent meconium aspiration syndrome. Committee Opinion No. 346, October 2006, Reaffirmed 2012
American College of Obstetricians and Gynecologists: Definition of term pregnancy. Committee Opinion No. 579, November 2013a
American College of Obstetricians and Gynecologists: Fetal macrosomia. Practice Bulletin No. 22, November 2000, Reaffirmed 2013b
American College of Obstetrics and Gynecologists: Management of delivery of a newborn with meconium-stained amniotic fluid. Committee Opinion No. 379, September 2007, Reaffirmed 2013c
Amersi S, Grimes DA: The case against using ordinal numbers for gestational age. Obstet Gynecol 91:623, 1998
Arrowsmith S, Wray S, Quenby S: Maternal obesity and labour complications following induction of labour in prolonged pregnancy. BJOG 118(5):578, 2011
Bakketeig LS, BergsjØ P: Post-term pregnancy: magnitude of the problem. In Chalmers I, Enkin M, Keirse M (eds): Effective Care in Pregnancy and Childbirth. Oxford, Oxford University Press, 1991, p 765
Bennett KA, Crane JM, O’Shea P, et al: First trimester ultrasound screening is effective in reducing postterm labor induction rates: a randomized controlled trial. Am J Obstet Gynecol 190:1077, 2004
Blondel B, Morin I, Platt RW, et al: Algorithms for combining menstrual and ultrasound estimates of gestational age: consequences for rates of preterm and postterm birth. Br J Obstet Gynaecol 109:718, 2002
Boulvain M, Irion O, Marcoux S, et al: Sweeping of the membranes to prevent post-term pregnancy and to induce labour: a systematic review. Br J Obstet Gynaecol 106:481, 1999
Boyce A, Magaux MJ, Schwartz D: Classical and “true” gestational post maturity. Am J Obstet Gynecol 125:911, 1976
Caughey AB, Nicholson JM, Washington AE: First- vs second-trimester ultrasound: the effect on pregnancy dating and perinatal outcomes. Am J Obstet Gynecol 198(6):703.e1, 2008
Caughey AB, Stotland NE, Washington AE, et al: Who is at risk for prolonged and postterm pregnancy? Am J Obstet Gynecol 200(6):683.e1, 2009
Cheng YW, Nicholson JM, Nakagawa S, et al: Perinatal outcomes in low-risk term pregnancies: do they differ by week of gestation? Am J Obstet Gynecol 199(4):370.e1, 2008
Clausson B, Cnattingus S, Axelsson O: Outcomes of postterm births: the role of fetal growth restriction and malformations. Obstet Gynecol 94:758, 1999
Cleary-Goldman J, Bettes B, Robinon JN, et al: Postterm pregnancy: practice patterns of contemporary obstetricians and gynecologists. Am J Perinatol 23:15, 2006
Clement D, Schifrin BS, Kates RB: Acute oligohydramnios in postdate pregnancy. Am J Obstet Gynecol 157:884, 1987
Clifford SH: Postmaturity with placental dysfunction. Clinical syndromes and pathologic findings. J Pediatr 44:1, 1954
Divon MY, Feldman-Leidner N: Postdates and antenatal testing. Semin Perinatol 32(4):295, 2008
Divon MY, Haglund B, Nisell H, et al: Fetal and neonatal mortality in the postterm pregnancy: the impact of gestational age and fetal growth restriction. Am J Obstet Gynecol 178:726, 1998
Fasset MJ, Wing DA: Uterine activity after oral mifepristone administration in human pregnancies beyond 41 weeks’ gestation. Gynecol Obstet Invest 65(2):112, 2008
Fischer RL, McDonnell M, Bianculli KW, et al: Amniotic fluid volume estimation in the postdate pregnancy: a comparison of techniques. Obstet Gynecol 81:698, 1993
Fraser WD, Hofmeyr J, Lede R, et al: Amnioinfusion for the prevention of the meconium aspiration syndrome. New Engl J Med 353:909, 2005
Gardener H, Spiegelman D, Buka SL: Perinatal and neonatal risk factors for autism: a comprehensive meta-analysis. Pediatrics 128:344, 2011
Gardner M, Rouse D, Goldenberg R, et al: Cost comparison of induction of labor at 41 weeks versus expectant management in the postterm pregnancy. Am J Obstet Gynecol 174:351, 1996
Gulmezoglu AM, Crowther CA, Middleton P: Induction of labour for improving birth outcomes for women at or beyond term. Cochrane Database System Rev 4:CD004945, 2006
Hannah ME, Hannah WJ, Hellman J, et al: Induction of labor as compared with serial antenatal monitoring in post-term pregnancy. N Engl J Med 326:1587, 1992
Harrington DJ, MacKenzie IZ, Thompson K, et al: Does a first trimester data scan using crown rump length measurement reduce the rate of induction of labour for prolonged pregnancy? BJOG 113:171, 2006
Harris BA Jr, Huddleston JF, Sutliff G, et al: The unfavorable cervix in prolonged pregnancy. Obstet Gynecol 62:171, 1983
Heimstad R, Skogvoll E, Mattsson LK, et al: Induction of labor or serial antenatal fetal monitoring in postterm pregnancy: a randomized controlled trial. Obstet Gynecol 109:609, 2007
Hendricks CH: Patterns of fetal and placental growth: the second half of pregnancy. Obstet Gynecol 24:357, 1964
Hill MJ, McWilliams GC, Garcia-Sur, et al: The effect of membrane sweeping on prelabor rupture of membranes: a randomized controlled trial. Obstet Gynecol 111(6):1313, 2008
Jazayeri A, Tsibris JC, Spellacy WN: Elevated umbilical cord plasma erythropoietin levels in prolonged pregnancies. Obstet Gynecol 92:61, 1998
Joseph KS, Huang L, Liu S, et al: Reconciling the high rates of preterm and postterm birth in the United States. Obstet Gynecol 109(4):798, 2007
Kashanian M, Aktarian A, Baradaron H, et al: Effect of membrane sweeping at term pregnancy on duration of pregnancy and labor induction: a randomized trial. Gynecol Obstet Invest 62:41, 2006
Larsen LG, Clausen HV, Andersen B, et al: A stereologic study of postmature placentas fixed by dual perfusion. Am J Obstet Gynecol 172:500, 1995
Laursen M, Bille C, Olesen AW, et al: Genetic influence on prolonged gestation: a population-based Danish twin study. Am J Obstet Gynecol 190:489, 2004
Leveno KJ, Quirk JG, Cunningham FG, et al: Prolonged pregnancy, I. Observations concerning the causes of fetal distress. Am J Obstet Gynecol 150:465, 1984
Lindell A: Prolonged pregnancy. Acta Obstet Gynecol Scand 35:136, 1956
Link G, Clark KE, Lang U: Umbilical blood flow during pregnancy: evidence for decreasing placental perfusion. Am J Obstet Gynecol 196(5)489.e1, 2007
MacDonald PC, Siiteri PK: Origin of estrogen in women pregnant with an anencephalic fetus. J Clin Invest 44:465, 1965
MacDorman MF, Kirmeyer S: Fetal and perinatal mortality, United States, 2005. Natl Vital Stat Rep 57(8):1, 2009
Martin JA, Hamilton BE, Sutton PD, et al: Births: final data for 2009. Natl Vital Stat Rep 60(1):1, 2011
Mogren I, Stenlund H, Högberg U: Recurrence of prolonged pregnancy. Int J Epidemiol 28:253, 1999
Moster D, Wilcox AJ, Vollset SE, et al: Cerebral palsy among term and postterm births. JAMA 304(9):976, 2010
Mozurkewich E, Chilimigras J, Koepke E, et al: Indications for induction of labour: a best-evidence review. BJOG 116(5):626, 2009
Munster K, Schmidt L, Helm P: Length and variation in the menstrual cycle—a cross-sectional study from a Danish county. Br J Obstet Gynaecol 99:422, 1992
Nahum GG, Stanislaw H, Huffaker BJ: Fetal weight gain at term: linear with minimal dependence on maternal obesity. Am J Obstet Gynecol 172:1387, 1995
National Institute of Child Health and Human Development Network of Maternal–Fetal Medicine Units: A clinical trial of induction of labor versus expectant management in postterm pregnancy. Am J Obstet Gynecol 170:716, 1994
Olesen AW, Westergaard JG, Olsen J: Perinatal and maternal complications related to postterm delivery: a national register-based study, 1978–1993. Am J Obstet Gynecol 189:227, 2003
Olesen AW, Westergaard JG, Olsen J: Prenatal risk indicators of a prolonged pregnancy. The Danish Birth Cohort 1998–2001. Acta Obstet Gynecol Scand 85:1338, 2006
Oz AU, Holub B, Mendilcioglu I, et al: Renal artery Doppler investigation of the etiology of oligohydramnios in postterm pregnancy. Obstet Gynecol 100:715, 2002
Perlman JM, Wyllie J, Kattwinkel J, et al: Neonatal resuscitation: 2010 International Consensus on Cardiopulmonary Resuscitation and emergency Cardiovascular Care Science with Treatment Recommendations. Pediatrics 126:e1319, 2010
Rushton DI: Pathology of placenta. In Wigglesworth JS, Singer DB (eds): Textbook of Fetal and Perinatal Pathology. Boston, Blackwell, 1991, p 171
Schaffer L, Burkhardt T, Zimmerman R, et al: Nuchal cords in term and postterm deliveries—do we need to know? Obstet Gynecol 106:23, 2005
Shime J, Gare DJ, Andrews J, et al: Prolonged pregnancy: surveillance of the fetus and the neonate and the course of labor and delivery. Am J Obstet Gynecol 148:547, 1984
Shin KS, Brubaker KL, Ackerson LM: Risk of cesarean delivery in nulliparous women at greater than 41 weeks’ gestational age with an unengaged vertex. Am J Obstet Gynecol 190:129, 2004
Smith GC: Life-table analysis of the risk of perinatal death at term and post term in singleton pregnancies. Am J Obstet Gynecol 184:489, 2001
Smith SC, Baker PN: Placental apoptosis is increased in postterm pregnancies. Br J Obstet Gynaecol 106:861, 1999
Torricelli M, Novembri R, Conti N, et al: Correlation with kisspeptin in postterm pregnancy and apoptosis. Reprod Sci 19(10):1133, 2012
Trimmer KJ, Leveno KJ, Peters MT, et al: Observation on the cause of oligohydramnios in prolonged pregnancy. Am J Obstet Gynecol 163:1900, 1990
Usher RH, Boyd ME, McLean FH, et al: Assessment of fetal risk in postdate pregnancies. Am J Obstet Gynecol 158:259, 1988
Vankayalapati P, Sethna F, Roberts N, et al: Ultrasound assessment of cervical length in prolonged pregnancy: prediction of spontaneous onset of labor and successful vaginal delivery. Ultrasound Obstet Gynecol 31(3):328, 2008
Walsh JM, Kandamany N, Shuibhne NN, et al: Neonatal brachial plexus injury: comparison of incidence and antecedents between 2 decades. Am J Obstet Gynecol 204:324, 2011.
Wenstrom KD, Parsons MT: The prevention of meconium aspiration in labor using amnioinfusion. Obstet Gynecol 73:647, 1989
Wingate MS, Alexander GR, Buekens, et al: Comparison of gestational age classifications: date of last menstrual period vs clinical estimate. Ann Epidemiol 17(6):425, 2007
Wong SF, Hui SK, Choi H, et al: Does sweeping of membranes beyond 40 weeks reduce the need for formal induction of labour? Br J Obstet Gynaecol 109:632, 2002
Yang S, Platt RW, Kramer MS: Variation in child cognitive ability by week of gestation among healthy term births. Am J Epidemiol 171:399, 2010
Yang SH, Roh CR, Kim JH: Transvaginal ultrasonography for cervical assessment before induction of labor. Obstet Gynecol Surv 59:577, 2004
Zhang X, Joseph KS, Kramer MS: Decreased term and postterm birthweight in the United States: impact of labor induction. Am J Obstet Gynecol 203:124.e1, 2010