Dusadee Sarangarm and Darren Braude
Peripheral arterial disease (PAD) is usually secondary to atherosclerosis and occurs most frequently in the lower extremities. PAD is analogous in many ways to coronary artery disease (CAD). Like CAD, PAD is often asymptomatic in patients with known cardiovascular disease or risk factors. Stable angina corresponds to noncritical ischemia and presents as predictable claudication. Unstable angina corresponds to the progression of chronic PAD to critical limb ischemia and presents as rest pain, ulceration, or gangrene. Myocardial infarction corresponds to acute, critical limb ischemia caused by thrombus or embolism. Irreversible damage to nerves and muscle may develop within 6 hours and up to 20% of patients may die (1).
CLINICAL PRESENTATION
PAD may be categorized as acute or chronic, critical or noncritical, and limb-threatening or not. Acute ischemia most often occurs secondary to in situ expansion of an atherosclerotic thrombus or less commonly due to an embolus from a more proximal source. Chronic, noncritical ischemia can present as intermittent claudication (IC) and asymptomatic arterial insufficiency. Chronic, critical ischemia can present as rest pain and/or skin ulcerations, infections, or necrosis. Limb-threatening ischemia can be caused by both acute ischemia of an extremity and chronic, critical ischemia (Table 95.1).
TABLE 95.1
Classification of Peripheral Arterial Disease

Because most PAD in developed countries is related to underlying atherosclerosis, the typical patient has known atherosclerotic disease or risk factors including diabetes, obesity, smoking history, hypertension, hyperlipidemia, renal insufficiency, and hyperhomocysteinemia; other risk factors include advanced age, male sex, and non-White ethnicity (1,2,3).
Acute Ischemia
Patients with acute ischemia usually present to the ED because of severe pain in a lower extremity. Pain is usually not well defined and in most cases extends above the ankle, depending on the location of the obstruction and degree of collateral flow. Patients with coexisting diabetic neuropathy, however, may not have typical pain. Patients may also complain of paresthesias, weakness, numbness, and/or a cold, white, or discolored extremity.
Acute ischemia is usually caused by an arterial embolism or thrombus of a native artery or bypass graft. Thrombosis is much more common than embolism. Patients with an arterial thrombosis become symptomatic from thrombus extension and subsequent arterial occlusion. Most patients will have prior chronic arterial insufficiency with claudication, rest pain, typical trophic skin changes, and/or prior bypass grafting. Because long-standing disease has led to the development of collateral circulation, symptoms and physical findings in thrombosis are typically not as dramatic as with embolism.
Arterial embolism arises from the heart in most cases, often from ventricular thrombus such as in postmyocardial infarction ventricular aneurysm or from atrial thrombus in the setting of mitral valve disease or atrial fibrillation. Unless the patient has significant coexisting lower extremity atherosclerosis, there typically is no established collateral circulation to deliver blood distally. Thus, arterial embolism usually presents in a more dramatic fashion than thrombosis. The hallmarks of acute ischemia are sudden onset of symptoms without preceding symptoms of chronic PAD, an identifiable embolic source, and a normal circulatory examination of the unaffected leg.
Chronic Critical Ischemia
Patients with chronic, critical ischemia usually present to the ED due to new or worsening rest pain or skin changes. As circulation is affected by gravity, rest pain typically occurs as the patient is sleeping or when the patient is supine. Pain occurs distally in the forefoot or toes but not usually in the calves. Because the pain is exacerbated by leg elevation and improved with dependency, many patients will sleep in a chair or prefer to hang the affected leg over the side of the bed. Symptoms worsen in the cold and the pain is often severe. Associated ischemic peripheral neuropathy may cause sharp shooting pains and sensations of heat and cold.
Chronic Noncritical Ischemia
Patients with chronic, noncritical ischemia usually present to the ED because of new or worsening claudication, which is essentially angina of the lower extremities. Patients classically report calf cramping or aching after a predictable amount of activity. This prompts the patient to stop activity, thereby relieving the pain. Depending on the site of the occlusion, claudication may occur in the calf, hip, buttock, or thigh. In general, foot claudication is rare with atherosclerosis.
DIFFERENTIAL DIAGNOSIS
Acute Thrombus or Embolus
If there is involvement of both legs, the clinician should consider a low cardiac output state or an aortic dissection tracking into the bilateral iliac arteries. If only one lower extremity is involved, the differential diagnosis includes conditions that mimic ischemia, such as phlegmasia cerulea dolens and acute compressive neuropathy, as well as nonatherosclerotic causes of true ischemia, such as phlegmasia alba dolens, arterial trauma, aortic dissection, arteritis, hypercoagulable states, and vasospasm.
Intermittent Claudication
Calf claudication in patients with atherosclerotic risk factors may be confused with venous claudication, compressive neuropathy, calf spasms, or symptomatic Baker’s cyst. Venous claudication follows treatment for deep venous thrombosis, resolves slowly when exercise stops, improves with limb elevation, and is usually associated with edema. Compressive spinal neuropathy usually produces distinctly dermatomal sharp pain, is associated with back pain, and does not resolve immediately when activity stops. The discomfort from a Baker’s cyst persists at rest.
Hip, thigh, or buttock claudication may be confused with arthritis of the hip and spinal stenosis. Pain from arthritis is not consistently reproduced with activity, often present at rest, and not relieved quickly with rest. Spinal stenosis symptoms are dermatomal, usually associated with back pain, and more typically relieved by bending forward than by stopping activity.
Rest Pain
Any distal sensory neuropathy may present with pain primarily at night. This pain tends to be symmetric, foot-based, shooting pains that are neither exacerbated by elevation nor relieved with dependency; an exception is in the case of ischemic neuropathy associated with chronic arterial ischemia. Other important causes of neuropathy include diabetes, vitamin deficiency, and alcohol abuse. Additionally, gout may cause extremity pain at rest, although this pain persists despite dependency, is localized to a joint, and is almost always associated with visible findings. Disc herniation of the lumbosacral nerve roots may also produce extremity pain at rest. This pain usually persists with dependency, follows a dermatomal pattern, is associated with back complaints, and presents with a normal vascular examination. Some confusion may arise with the straight-leg raise examination maneuver as it can cause pain in patients with rest pain, as well as in nerve root compression. Reflex sympathetic dystrophy can present with burning pain and skin changes though the pain is not exacerbated by elevation and pulses should be normal. Finally, simple night cramps cause calf rather than foot pain that is relieved with massage, not exacerbated by elevation, and is not associated with pulse deficits.
Ulcers and Gangrene
Ulcers secondary to arterial ischemia must be differentiated from ulcers secondary to other causes, such as venous stasis and diabetes. Ulcers related to venous insufficiency are the most common, tend to occur more proximally, produce less pain, are relieved with elevation, coexist with edema and stasis dermatitis, and have a wet base with copious granulation tissue. The ulcers of diabetes may occur in the same distal pressure point locations as PAD-associated ulcers but tend to be painless, deep, and surrounded by significant callus and/or inflammation. The primary differential of dry gangrene is frostbite. Wet gangrene may originate in other skin ulcers or wounds.
ED EVALUATION
Acute, critical PAD is primarily a clinical diagnosis based upon history and physical examination, including the ankle-brachial pressure index (ABI). The 2011 American College of Cardiology Foundation and American Heart Association guideline update defines patients with suspected lower extremity PAD as individuals with one or more of the following signs or symptoms: exertional leg symptoms, nonhealing wounds, age 70 and older, or 50 years and older with a history of smoking or diabetes (10). In patients with clinical evidence of acute ischemia, vascular surgery consultation should precede diagnostic studies and may require immediate transfer in some settings. In cases of diagnostic uncertainty, or when an alternative emergent diagnosis such as aortic dissection is highly likely, the ED physician may order imaging studies prior to consultation in the patient with previously undiagnosed PAD and no evidence of critical ischemia. However, diagnostic tests should not delay consultation or transfer if acute, critical PAD is suspected. Definitive diagnostic testing should ideally be at the discretion of the vascular surgery consultant given individual practitioner preference, institutional resources, associated delays, and potential adverse effects.
Examination of the lower extremities should include bilateral inspection for sores, edema, dermatitis, gangrene, and signs of infection; palpation of skin temperature; assessment of capillary refill time; palpation of the femoral, popliteal, dorsalis pedis, and posterior tibial pulses; auscultation of the femoral artery; and motor and sensory examinations. The abdomen should be examined for masses or bruits suggestive of abdominal aortic aneurysm. Capillary refill has not been found to be useful in assessing chronic peripheral vascular disease while the most useful findings to confirm PAD are pulse deficit, bruit, and cool skin (4,5).
The signs and symptoms in acute arterial occlusion resulting from an embolus can be dramatic and reflect the “six Ps” of ischemia: pain, pallor, pulselessness, poikilothermia, paralysis, and paresthesia.
If acute ischemia is embolic in nature, without pre-existing PAD, the unaffected extremity tends to be normal on examination. The affected extremity is white or cyanotic, cool to the touch, and pulseless without appreciable new swelling. There will usually be some component of neuropathy with weakness and sensory deficits. Sensory deficits begin with diminished light touch, two-point discrimination, and proprioception. Loss of deep-pain, pressure sensation, and motor deficits reflect advanced ischemia and possibly irreversible ischemia. The intrinsic muscles of the feet are the most sensitive to ischemia and are tested by having the patient spread their toes.
In arterial thrombosis, findings are usually more subtle than with embolus owing to collateral blood flow and/or slower onset, although the major pulses distal to the obstruction will typically be absent. Unfortunately, there is variability in the presence of peripheral pulses and their detection. Comparison with the unaffected extremity and use of a Doppler are very helpful. The presence of pulses detected on Doppler indicates blood flow but does not necessarily exclude ischemia. Both the affected and the unaffected extremity should show findings of chronic PAD including atrophy of calf muscles, loss of hair over the dorsum of the foot, thickened toenails, shiny skin, diminished pulses, and possibly skin ulcers. Edema may be present as patients prefer to keep their extremities dependent.
Patients with IC will usually have a normal examination at rest, although they may have some subtle findings of chronic ischemia. Further in the disease process, patients with rest pain will show many of the changes noted previously for chronic PAD. These patients do not have complete occlusion; so capillary refill should be present, although delayed, and pulses will be present, at least by Doppler. Another clinical finding of severe ischemia frequently cited is Buerger’s sign. A positive test is the observation of pallor with foot elevation and exaggerated delayed erythema with subsequent dependency (5). This sign is not well validated (4).
Skin ulcerations from arterial insufficiency tend to occur distally at pressure points, such as at the tips of the toes, between the toes, or on the heel. These ulcers often have an irregular border with a pale base and may show signs of infection. Plain radiography is appropriate to screen for underlying osteomyelitis when the ulcer appears sufficiently deep or infected. Gangrene is rarely missed when the distal extremities are examined. Uninfected gangrene is dry while discharge, weeping, crepitance, lymphangitis, and cellulitis suggest infection.
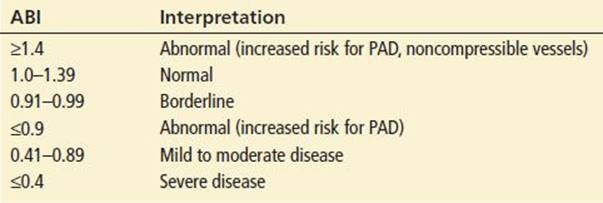
Ankle-Brachial Pressure Index
The ABI is a rapid, objective screening measurement of arterial insufficiency and should be assessed in any patient in whom arterial insufficiency is suspected. If the ABI can be measured, the limb is not immediately threatened, but may still be ischemic. If arterial signals are absent, limb loss is probable without emergent revascularization. Absent venous pulsations indicate severe ischemia. It is helpful to check the ABI in the unaffected leg to ascertain whether the patient has underlying arterial disease or noncompressible vessels. The technique and interpretation of ABI measurement are summarized in Tables 95.2 and 95.3 (6,7). While the typically used ABI cut-off of 0.9 maximizes sensitivity and specificity, any value <1.0 in a symptomatic patient should be considered sufficient evidence of arterial insufficiency (6,7).
TABLE 95.2
Technique for the Measurement of Ankle-Brachial Pressure Index

TABLE 95.3
Interpretation of Ankle-Brachial Pressure Index Results
Imaging Studies
While catheter angiogram remains the gold standard, noninvasive studies are being increasingly used. According to the American College of Radiology 2012 guideline, lower extremity magnetic resonance angiography (MRA) and computed tomography angiography (CTA) have a higher level appropriateness rating than catheter arteriography as the initial radiologic examination for patients with suspected PAD (9). The increased availability of CTA has likely contributed to its increasing use, while MRA has been shown to be more cost-effective and safer than the gold standard of angiography. In addition, these noninvasive diagnostic modalities have been shown to have high sensitivity and specificity (90% to 100%) for the detection of stenosis >50% diameter when compared to catheter angiogram (9). Ultrasound is preferred in patients with moderate to severe renal insufficiency, as both CT contrast and gadolinium are associated with complications in these patients (8).
KEY TESTING
• Determination of ABI
• Doppler of extremity pulses
• Angiography, MRA, or CTA in consultation with a vascular surgeon
ED MANAGEMENT
Supportive care for patients with PAD includes keeping the affected limb dependent and warm, providing analgesia as needed, and maximizing cardiac output. Aspirin or clopidogrel is indicated in all patients with symptomatic PAD to reduce risk of further vascular injury such as MI and ischemic CVA although the role of these drugs in the acute setting has not been adequately investigated (1,7,10,11).
Acute Ischemia
In the absence of contraindications, patients with acute critical ischemia should receive anticoagulation with unfractionated heparin despite little objective evidence (1,7,11). Supplemental oxygen may be helpful and should be provided when acute ischemia is considered. Options for definitive therapy include surgical or interventional procedures at the discretion of the vascular surgeon. Considerations include degree and duration of ischemia, contraindications, local resources, and results of imaging studies. In patients with severe ischemia, angiography may be performed intraoperatively to minimize delay in care. Surgical options include direct removal of the occlusion (most common with established thrombus), bypass grafting, primary amputation, and Fogarty catheter embolectomy (most common with embolus or suspected new thrombus). Interventional options include direct intra-arterial administration of thrombolytics, mechanical thrombectomy, thromboaspiration, and angioplasty. There is currently no accepted role for intravenous thrombolytics, although this treatment merits discussion with a vascular surgeon if definitive care is expected to be significantly delayed (7,10).
Chronic Critical Ischemia and Rest Pain
Patients with rest pain in the dependent position should be treated as for acute ischemia. Patients experiencing rest pain only when the foot is elevated represent an intermediate group and definitive therapy for this group may involve pharmacotherapy, endovascular procedures, or surgery though these measures may not need to be started emergently. At a minimum, telephone consultation with the patient’s primary care physician and/or vascular surgeon is appropriate. After consultation, these patients may be discharged home on aspirin 75 to 100 mg per day with an adequate follow-up plan and return precautions (11). Should the patient have an allergy to aspirin, clopidogrel 75 mg per day has been found to be a safe and effective alternative (11).
Chronic Ischemia with Ulcerations or Infection
Patients with uninfected ulcerations require wound debridement and dressing, close monitoring for the development of infection, analgesia, and at a minimum, telephone consultation with a vascular surgeon. Admission should be considered but is not mandatory when adequate follow-up is assured.
Patients with infected ulcerations need supportive care, aggressive wound management, systemic antibiotics, admission, and vascular surgery consultation for possible revascularization. Appropriate empiric antibiotic choices should take into account risk factors for gram-negative infections such as diabetes, recent hospitalization, local epidemiology, resistance patterns, and the rising incidence of methicillin-resistant Staphylococcus aureus. A reasonable choice for intravenous therapy is ampicillin- sulbactam (1.5 to 3 g q6 h) plus clindamycin (900 mg q8 h) plus ciprofloxacin (400 mg q12 h) (12). If there is a high suspicion for hospital-acquired methicillin-resistant Staphylococcus aureus, vancomycin (dosing dependent on renal function) may be substituted for clindamycin.
Patients with dry gangrene require supportive care and timely surgery consultation. Patients with evidence of necrotizing infections (gas gangrene, wet gangrene, or sepsis) require intensive care unit admission, immediate surgical consultation, and broad-spectrum antibiotics to cover gram-negative infection, Staphylococcus aureus, Streptococcus sp, and Clostridium sp.
Chronic Noncritical Ischemia and Intermittent Claudication
Although this is a nonemergent condition, these patients require close follow-up with their primary care physician and/or vascular surgeon. In the meantime, they should be reassured that walking, despite causing pain, is not harmful. Pharmacotherapy may be deferred to follow-up, though it is reasonable to start patients without contraindications on 75 to 100 mg per day of aspirin (1,7,10).
CRITICAL INTERVENTIONS
• Aspirin and weight-based heparin (80 units/kg bolus followed by 18 units/kg/hr continuous infusion) for acute ischemia and rest pain that persists in the dependent position.
• Emergent vascular surgery consultation, including transfer as necessary, for limb-threatening ischemia or infection.
• Supportive care, keeping affected extremity warm and dependent, maximizing cardiac output, and providing supplemental oxygen.
• Early initiation of antibiotics for patients with signs of infection.
DISPOSITION
All patients with PAD require vascular surgery consultation. The emergency physician must decide according to the acuity of the situation whether that consultation must take place emergently in the ED or on an outpatient basis. In some situations, the patient may be admitted to their primary care provider or hospitalist with inpatient vascular surgery consultation.
Emergent vascular surgery consultation is appropriate for patients with acute ischemia or evidence of serious infection in an ischemic extremity. If vascular surgery consultation is not available, the patient should be transferred expeditiously. Patients with chronic critical ischemia require emergent vascular surgery consultation in most cases. Patients with rest pain that is well controlled with supportive measures, and patients with uninfected ulcers without gangrene, may be candidates for admission with urgent vascular surgery consultation as an inpatient.
Patients with noncritical ischemia may be discharged home with close outpatient follow-up. Consultation with a vascular surgeon before discharge is recommended when possible.
Common Pitfalls
• Delaying consultation or transfer for testing and definitive diagnosis.
• Failing to consider diabetic neuropathy masking the pain of an ischemic extremity.
• Failing to elicit a history of claudication because the patient’s baseline activity is limited.
• Failing to recognize IC can occur in the hip, buttock, or thigh.
REFERENCES
1. Norgren L, Hiatt WR, Dormandy JA, et al. TASC II Working Group. Inter-society consensus for the management of peripheral arterial disease (TASC II). J Vasc Surg. 2007;45(1):S5A–S67A.
2. Jones WS, Patel MR, Dai D, et al. Temporal trends and geographic variation of lower-extremity amputation in patients with peripheral artery disease: Results from U.S. Medicare 2000–2008. J Am Coll Cardiol. 2012 ;60(21):2230–2236.
3. Joosten MM, Pai JK, Bertoia ML, et al. Associations between conventional cardiovascular risk factors and risk of peripheral artery disease in men. JAMA. 2012;308(16):1660–1667.
4. Khan NA. Does the clinical examination predict lower extremity peripheral arterial disease? JAMA. 2006;295:536–546.
5. McGee SR, Boyko EJ. Physical examination and chronic lower-extremity ischemia: A critical review. Arch Intern Med. 1998;158:1357–1364.
6. Aboyans V, Criqui MH, Abraham P, et al. Measurement and interpretation of the ankle-brachial index: A scientific statement from the American Heart Association. Circulation. 2012;126(24):2890–2909. Erratum in Circulation2013; 127(1):e264
7. Rooke TW, Hirsch AT, Misra S, et al. 2011 ACCF/AHA focused update of the guideline for the management of patients with peripheral artery disease (updating the 2005 guideline): A report of the American College of Cardiology Foundation/American Heart Association task force on practice guidelines. J Am Coll Cardiol. 2011;58(19):2020–2045.
8. Von Ziegler F, Costa MA. The role of CT and MRI in the assessment of peripheral vascular disease. Curr Cardiol Rep. 2007;9:412–419.
9. Dill KE, Rybicki FJ, Desjardins B, et al. Expert Panel on Vascular Imaging. ACR Appropriateness Criteria® claudication – suspected vascular etiology [online publication]. Reston, VA: American College of Radiology (ACR); 2012:7. http://www.guideline.gov/content.aspx?id=37943
10. Critical Leg Ischaemia Prevention Study (CLIPS) Group, Catalano M, Born G, Peto R, et al. Prevention of serious vascular events by aspirin amongst patients with peripheral arterial disease: randomized, double-blind trial. J Intern Med. 2007;261:276–284.
11. Alonso-Coello P, Bellmunt S, McGorrian C, et al. Antithrombotic therapy in peripheral artery disease: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e669S–e690S.
12. Stevens DL, Bisno AL, Chambers HR, et al. Practice guidelines for the diagnosis and management of skin and soft-tissue infections (IDSA Guidelines). Clin Infectious Dis. 2005;41(15):1373–1830.