John D. Hummel, MD
CASE PRESENTATION
A 67-year-old man presented with a 3-year history of atrial fibrillation (AF) that was symptomatic with dyspnea on exertion. He underwent echocardiography as well as treadmill nuclear stress testing revealing a left ventricular ejection fraction of 40% without evidence of significant valve or coronary artery disease. The left ventricle was diffusely hypokinetic. Holter monitoring revealed rate-controlled atrial fibrillation. He was initially treated with sotalol and spontaneously converted to and maintained NSR for approximately 14 months with resolution of his cardiomyopathy. His symptomatic AF then recurred, and the patient required two cardioversions with early recurrence of AF, which remained persistent. He presented with dyspnea on moderate exertion and complaints of mild swelling in his lower extremities. His left ventricular ejection fraction was globally depressed with an ejection fraction of 40%. The patient was referred for ablation of his AF and was enrolled in the TTOP-AF trial (a randomized, prospective trial of antiarrhythmic medical therapy versus phased-RF ablation therapy).1 He was randomized to curative ablation, utilizing three specially designed catheters (Figure 38-1). The patient underwent percutaneous catheter placement in the left atrium with pulmonary vein isolation using the PVAC catheter (Figures 38-2 and 38-3). Pulmonary vein isolation was followed by septal ablation with the MASC catheter (Figure 38-4). Subsequent ablation of complex fractionated atrial electrograms using the MAAC catheter completed the procedure (Figure 38-5).
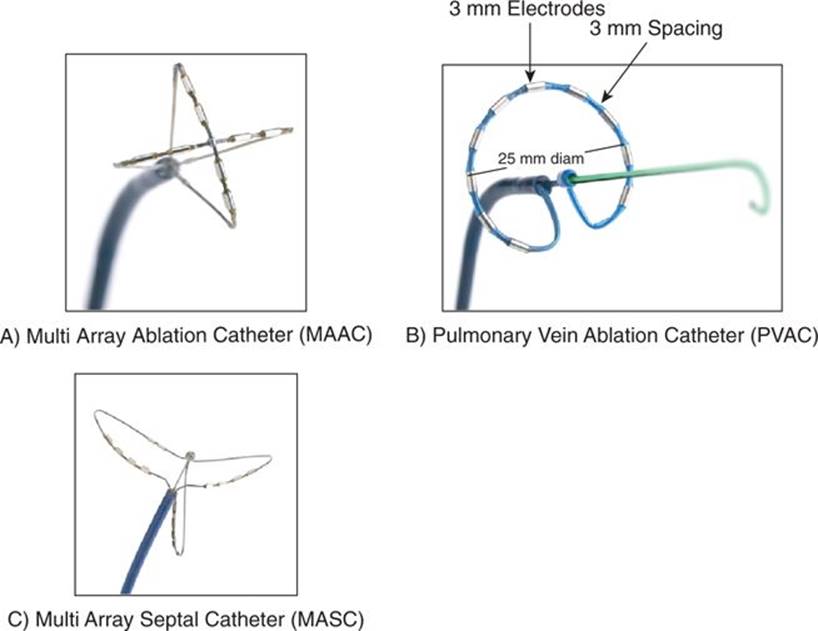
FIGURE 38-1 The three primary catheters for phased radiofrequency energy delivery are (A) the MAAC used for targeting regions of complex fractionated electrograms or creating areas of ablation in the body of the left atrium such as the LA roof and mitral isthmus; (B) the PVAC placed in the pulmonary veins over a guidewire and rotated between RF applications to allow electrical isolation of the pulmonary veins; (C) the MASC delivered through the transseptal sheath, pulled back against the septum and rotated between RF applications to eliminate septal electrical activity.
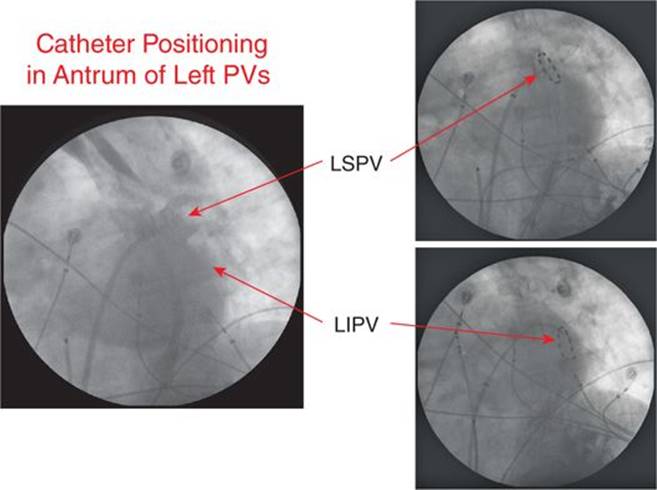
FIGURE 38-2 Fluoroscopic image of the PVAC deployed in the left superior and inferior veins to allow energy delivery.
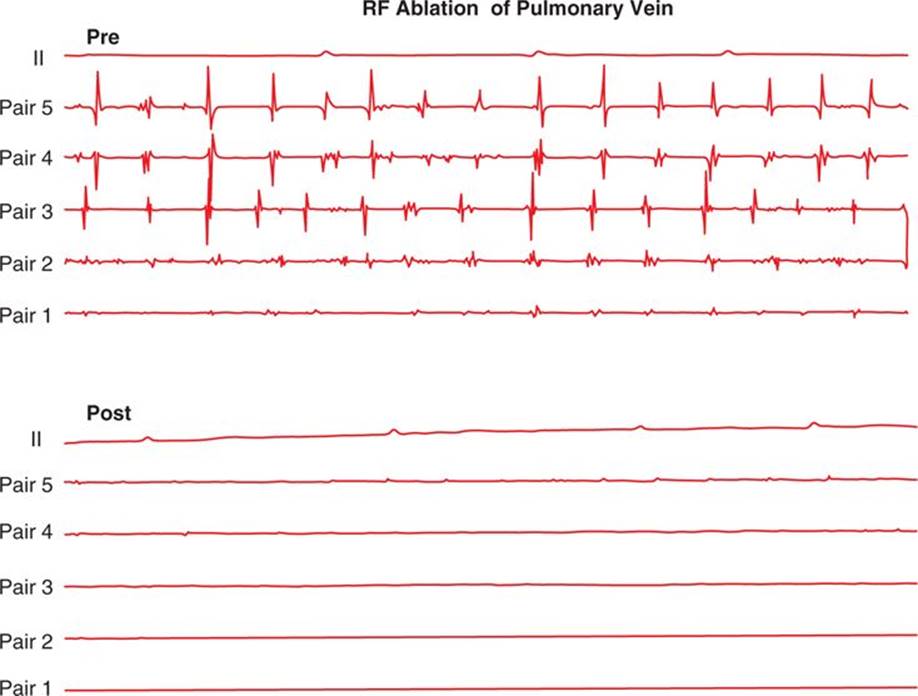
FIGURE 38-3 Elimination of PV activity in the left inferior pulmonary vein after RF delivery.
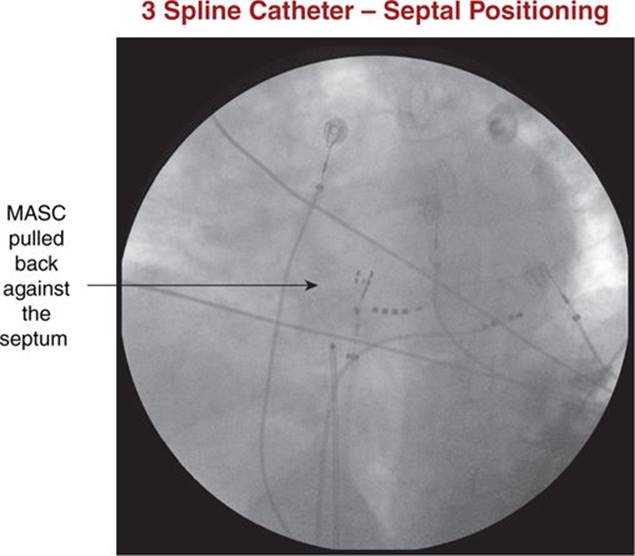
FIGURE 38-4 Position of the MASC against the septum on fluoroscopy.
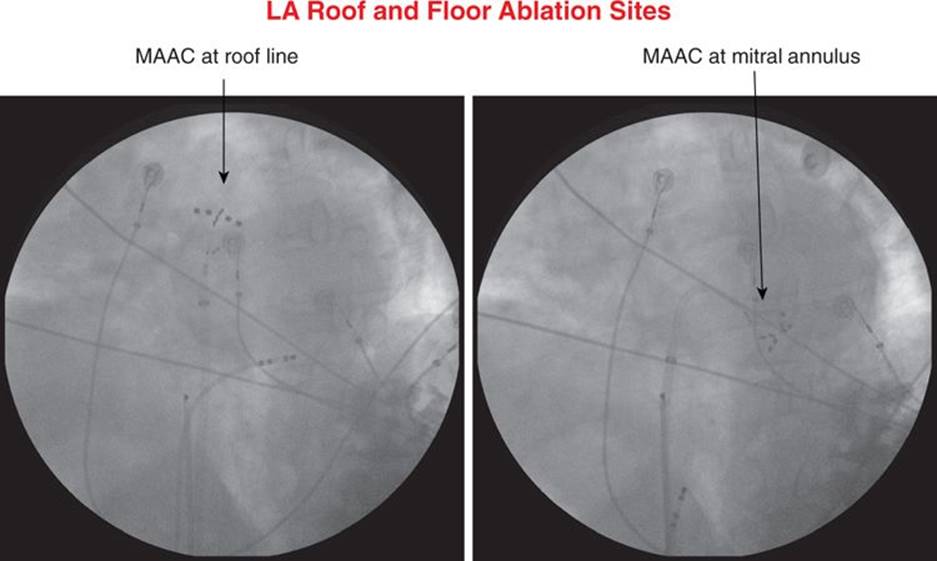
FIGURE 38-5 Position of the MAAC catheter on flouroscopy along the left atrial roof line and between the left inferior pulmonary vein and the mitral isthmus.
EPIDEMIOLOGY
AF and heart failure (HF) are two of the most pervasive cardiovascular diseases, costing millions of people loss of quality of life and a decrease in survival. AF affects over 2 million Americans, and the lifetime risk of developing AF for adults aged >40 is 25%, while the AF population carries an elevated risk of developing heart failure in the range of 20%.2-4
ETIOLOGY AND PATHOPHYSIOLOGY
The causes of AF are myriad, and AF and HF represent a deadly intersection with AF promoting HF and HF promoting AF as AF is present in 5% of NYHA class I patients and up to 50% of NYHA class IV patients.5 AF can promote HF through rapid ventricular rates, poor atrial transport, or the irregularity of ventricular contraction. Resolution of AF can result in reverse remodeling and restoration of atrial and ventricular cellular and mechanical function as evidenced by trials comparing AF ablation versus atrioventricular nodal ablation with cardiac resynchronization pacing or medical therapy.6,7
MANAGEMENT
Patients with persistent AF have lower success rates than those with paroxysmal AF, and the use of phased RF in the persistent AF population was examined in the TTOP-AF trial as an initial step in assessing efficacy of a different form of RF ablation in this challenging population. Although no benefit has been demonstrated for attempted medical rhythm control over rate control, several studies of AF and CHF suggest clear benefit with restoration of NSR via AF ablation.7 This may indicate that the deleterious effects of medication offset the beneficial effects of restoration of NSR in previous studies. This patient was enrolled in the TTOP-AF trial. Patients who had failed at least one antiarrhythmic drug were randomized to phased-RF ablation or antiarrhythmic drug. The primary endpoints of TTOP-AF included chronic effectiveness and safety at 6 months and acute safety within 7 days of the ablation.
At 6 months, a greater proportion of ablation patients achieved effectiveness (77/138; 55.8%) compared with medically managed patients (19/72; 26.4%) (p <0.0001). Acutely, 92.8% of procedures were successful, while 12.3% experienced a serious procedure and/or device-related adverse event. The predefined acute safety end point was not met. This trial demonstrated superiority of phased-RF AF ablation over medical therapy but with a higher than expected risk of clinical stroke. Thus, the phased RF system was approved as efficacious by the FDA panel, but rejected for use in the United States due to safety concerns, which are now being addressed after catheter and protocol modifications embodied in the Victory AF trial.
THE TECHNOLOGY
The Phased RF system (Medtronic Ablation Frontiers, Carlsbad, CA, United States) employs three multielectrode catheters to deliver duty-cycled, phased energy via a multichannel RF generator to targeted left atrial regions (Figure 38-1). Phase-shifted energy delivery between adjacent catheter electrodes (bipolar) and return electrodes (unipolar) allows creation of long, contiguous lesions. Modulating unipolar-to-bipolar RF ratios controls lesion depth (Figures 38-6 and 38-7). RF is delivered in temperature-controlled and power-limited fashion (140°F [60°C], 8 or 10 watts) with typical duration of 60 seconds, with the lower power limits being all that is needed due to increased current density on the small electrodes.
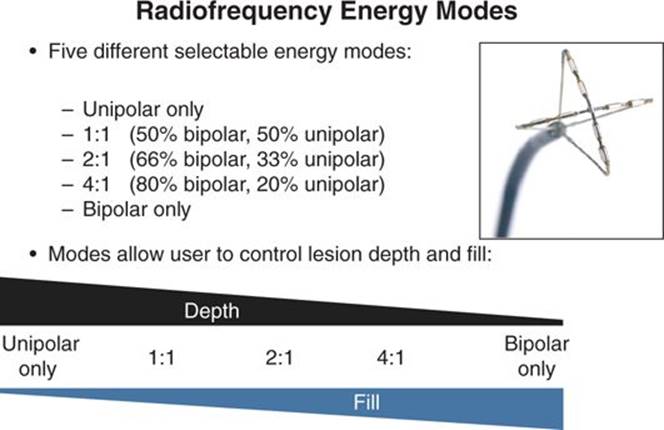
FIGURE 38-6 Schematic diagram of decreasing lesion depth and increasing lesion fill between electrodes as phasing of RF energy transitions from primarily unipolar to primarily bipolar.
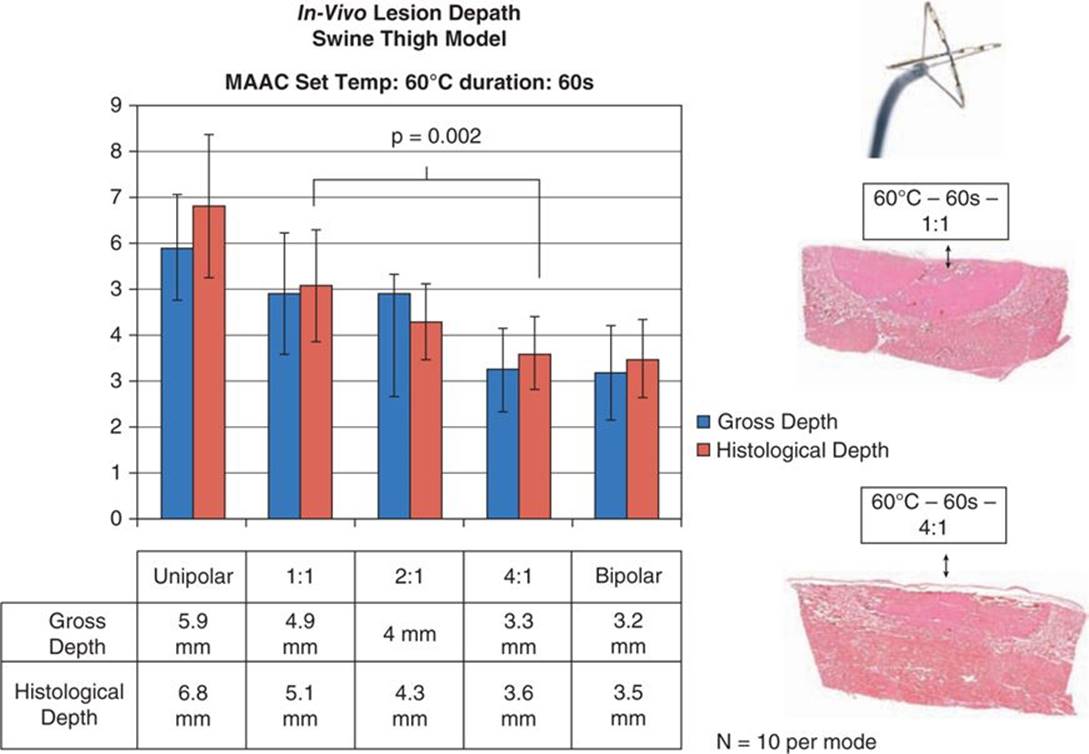
FIGURE 38-7 Preclinical data on differing degrees of lesion depth based on differing ratios of unipolar to bipolar phasing of the RF energy delivery to swine thigh muscle in a perfused tissue bath.
PATIENT OUTCOME
The phased-RF procedure was uncomplicated and successful in maintaining sinus rhythm off antiarrhythmic therapy. Three months postablation the patient’s echocardiogram demonstrated recovery of normal left ventricular function.
REFERENCES
1. Hummel J, Michaud G, Hoyt R, et al. Phased RF ablation of persistent atrial fibrillation. Heart Rhythm. 2014;11(2): 202-209.
2. Lloyd-Jones DM, Larson MG, Leip EP, et al. Lifetime risk for developing congestive heart failure: the Framingham Heart Study. Circulation. 2002;106(24):3068-3072.
3. Roger VL, Go AS, Lloyd-Jones DM, et al. Heart disease and stroke statistics—2011 update: a report from the American Heart Association. Circulation. 2011;123:e18-e209.
4. Lloyd-Jones DM, Wang TJ, Leip EP, et al. Lifetime risk for development of atrial fibrillation: the Framingham Heart Study. Circulation. 2004;110:1042-1046.
5. Maisel WH, Stevenson LW. Atrial fibrillation in heart failure: epidemiology, pathophysiology, and rationale for therapy. Am J Cardiol. 2003;91:2D-8D.
6. Carson PE, Johnson GR, Dunkman WB, Fletcher RD, Farrell L, Cohn JN. The influence of atrial fibrillation on prognosis in mild to moderate heart failure: theV-HeFT Studies. Circulation. 1993;87(6 Suppl):VI102-110.
7. Khan MN, Jaïs P, Cummings J, et al. Pulmonary-vein isolation for atrial fibrillation in patients with heart failure. N Engl J Med. 2008;359:1778-1785.
8. Dagres N, Varounis C, Gaspar T, et al. Catheter ablation for atrial fibrillation in patients with left ventricular systolic dysfunction. A systematic review and meta-analysis. J Card Fail. 2011;17:964-970.