VIRAL HEPATITIS
Acute viral hepatitis is a systemic infection predominantly affecting the liver. Clinically characterized by malaise, nausea, vomiting, diarrhea, and low-grade fever followed by dark urine, jaundice, and tender hepatomegaly; may be subclinical and detected on basis of elevated aspartate and alanine aminotransferase (AST and ALT) levels. Hepatitis B may be associated with immune-complex phenomena, including arthritis, serum sickness-like illness, glomerulonephritis, and a polyarteritis nodosa–like vasculitis. Hepatitis-like illnesses may be caused not only by hepatotropic viruses (A, B, C, D, E) but also by other viruses (Epstein-Barr, CMV, coxsackievirus, etc.), alcohol, drugs, hypotension and ischemia, and biliary tract disease (Table 163-1).
TABLE 163-1 THE HEPATITIS VIRUSES


HEPATITIS A (HAV)
27-nm picornavirus (hepatovirus) with single-stranded RNA genome.
Clinical Course See Fig. 163-1.

FIGURE 163-1 Scheme of typical clinical and laboratory features of HAV.
Outcome
Recovery within 6–12 months, usually with no clinical sequelae; a small proportion will have one or two apparent clinical and serologic relapses; in some cases, pronounced cholestasis suggesting biliary obstruction may occur; rare fatalities (fulminant hepatitis), no chronic carrier state.
Diagnosis
IgM anti-HAV in acute or early convalescent serum sample.
Epidemiology
Fecal-oral transmission; endemic in underdeveloped countries; food-borne and waterborne epidemics; outbreaks in day-care centers, residential institutions.
Prevention
After exposure: immune globulin 0.02 mL/kg IM within 2 weeks to household and institutional contacts (not casual contacts at work). Before exposure: inactivated HAV vaccine 1 mL IM (unit dose depends on formulation); half dose to children; repeat at 6–12 months; target travelers, military recruits, animal handlers, day-care personnel, laboratory workers, and pts with chronic liver disease (especially hepatitis C).
HEPATITIS B (HBV)
42-nm hepadnavirus with outer surface coat (HBsAg), inner nucleocapsid core (HBcAg), DNA polymerase, and partially double-stranded DNA genome of 3200 nucleotides. Circulating form of HBcAg is HBeAg, a marker of viral replication and infectivity. Multiple serotypes and genetic heterogeneity.
Clinical Course See Fig. 163-2.

FIGURE 163-2 Scheme of typical clinical and laboratory features of HBV.
Outcome
Recovery >90%, fulminant hepatitis (<1%), chronic hepatitis or carrier state (only 1–2% of immunocompetent adults; higher in neonates, elderly, immunocompromised), cirrhosis, and hepatocellular carcinoma (especially following chronic infection beginning in infancy or early childhood) (Chap. 165). Reactivation of HBV has been observed with immunosuppression, particularly with rituximab.
Diagnosis
HBsAg in serum (acute or chronic infection); IgM anti-HBc (early anti-HBc indicative of acute or recent infection). Most sensitive test is detection of HBV DNA in serum; not generally required for routine diagnosis.
Epidemiology
Percutaneous (needle stick), sexual, or perinatal transmission. Endemic in sub-Saharan Africa and Southeast Asia, where up to 20% of population acquire infection, usually early in life.
Prevention
After exposure in unvaccinated persons: hepatitis B immune globulin (HBIg) 0.06 mL/kg IM immediately after needle stick to within 14 days of sexual exposure in combination with vaccine series. For perinatal exposure (HbsAg+ mother) HBIg 0.05 mL in the thigh immediately after birth with the vaccine series started within the first 12 h of life. Before exposure: recombinant hepatitis B vaccine IM (dose depends on formulation as well as adult or pediatric and hemodialysis); at 0, 1, and 6 months; deltoid, not gluteal injection. Has been targeted to high-risk groups (e.g., health workers, persons with multiple sexual partners, IV drug users, hemodialysis pts, hemophiliacs, household and sexual contacts of HBsAg carriers, persons traveling in endemic areas, unvaccinated children <18). Universal vaccination of all children is now recommended in the United States.
HEPATITIS C (HCV)
Caused by flavi-like virus with RNA genome of >9000 nucleotides (similar to yellow fever virus, dengue virus); genetic heterogeneity. Incubation period 7–8 weeks.
Clinical Course
Often clinically mild and marked by fluctuating elevations of serum aminotransferase levels; >50% likelihood of chronicity, leading to cirrhosis in >20%.
Diagnosis
Anti-HCV in serum. Current third-generation immunoassay incorporates proteins from the core, NS3, and NS5 regions. The most sensitive indicator of HCV infection is HCV RNA (Fig. 163-3).
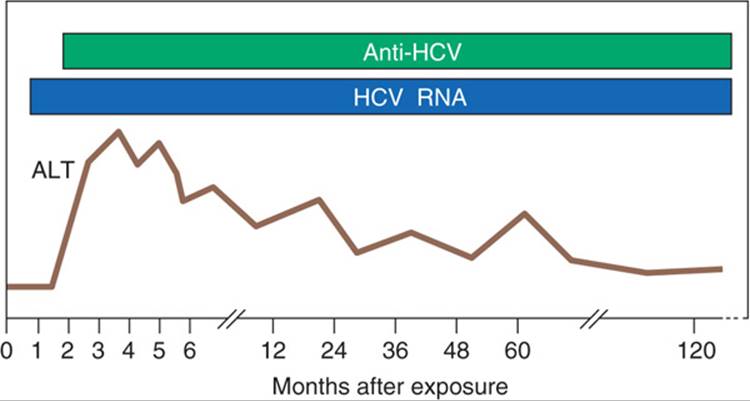
FIGURE 163-3 Scheme of typical laboratory features during acute hepatitis C progressing to chronicity. HCV RNA is the first detectable event, preceding alanine aminotransferase (ALT) elevation and the appearance of anti-HCV.
Epidemiology
HCV accounts for >90% of transfusion-associated hepatitis cases. IV drug use accounts >50% of reported cases of hepatitis C. Little evidence for frequent sexual or perinatal transmission.
Prevention
Exclusion of paid blood donors, testing of donated blood for anti-HCV. Anti-HCV detected by enzyme immunoassay in blood donors with normal ALT is often falsely positive (30%); result should be confirmed by HCV RNA in serum.
HEPATITIS D (HDV, DELTA AGENT)
Defective 37-nm RNA virus that requires HBV for its replication; either co-infects with HBV or superinfects a chronic HBV carrier. Enhances severity of HBV infection (acceleration of chronic hepatitis to cirrhosis, occasionally fulminant acute hepatitis).
Diagnosis
Anti-HDV in serum (acute hepatitis D—often in low titer, is transient; chronic hepatitis D—in higher titer, is sustained).
Epidemiology
Endemic among HBV carriers in Mediterranean Basin, where it is spread predominantly by nonpercutaneous means. In nonendemic areas (e.g., northern Europe, United States) HDV is spread percutaneously among HbsAg+ IV drug users or by transfusion in hemophiliacs and to a lesser extent among HbsAg+ men who have sex with men.
Prevention
Hepatitis B vaccine (noncarriers only).
HEPATITIS E (HEV)
Caused by 29- to 32-nm agent thought to be related to caliciviruses. Enterically transmitted and responsible for waterborne epidemics of hepatitis in India, parts of Asia and Africa, and Central America. Self-limited illness with high (10–20%) mortality rate in pregnant women.
TREATMENT Viral Hepatitis
Activity as tolerated, high-calorie diet (often tolerated best in morning), IV hydration for severe vomiting, cholestyramine up to 4 g PO four times daily for severe pruritus, avoid hepatically metabolized drugs; no role for glucocorticoids. Liver transplantation for fulminant hepatic failure and grades III–IV encephalopathy. In rare instances of severe acute HBV, lamivudine has been used successfully. Most authorities would recommend antiviral therapy for severe acute HBV (Chap. 164). Meta-analyses and small clinical trials suggest that treatment of acute HCV infection with interferon α may be effective at reducing the rate of chronicity. Based on these data, many experts feel that acute HCV infection should be treated with a 24-week course of the best available regimens currently used to treat chronic HCV infection (Chap. 164).
TOXIC AND DRUG-INDUCED HEPATITIS
DOSE-DEPENDENT (DIRECT HEPATOTOXINS)
Onset is within 48 h, predictable, necrosis around terminal hepatic venule–e.g., carbon tetrachloride, benzene derivatives, mushroom poisoning, acetaminophen, or microvesicular steatosis (e.g., tetracyclines, valproic acid).
IDIOSYNCRATIC
Variable dose and time of onset; small number of exposed persons affected; may be associated with fever, rash, arthralgias, eosinophilia. In many cases, mechanism may actually involve toxic metabolite, possibly determined on genetic basis—e.g., isoniazid, halothane, phenytoin, methyldopa, carbamazepine, diclofenac, oxacillin, sulfonamides.
TREATMENT Toxic and Drug-Induced Hepatitis
Supportive as for viral hepatitis; withdraw suspected agent, and include use of gastric lavage and oral administration of charcoal or cholestyramine. Liver transplantation if necessary. In acetaminophen overdose, more specific therapy is available in the form of sulfhydryl compounds (e.g., N-acetylcysteine). These agents appear to act by providing a reservoir of sulfhydryl groups to bind the toxic metabolites or by stimulating synthesis of hepatic glutathione. Therapy should be begun within 8 h of ingestion but may be effective even if given as late as 24–36 h after overdose.
ACUTE HEPATIC FAILURE
Massive hepatic necrosis with impaired consciousness occurring within 8 weeks of the onset of illness.
CAUSES
Infections [viral, including HAV, HBV, HCV (rarely), HDV, HEV; bacterial, rickettsial, parasitic], drugs and toxins, ischemia (shock), Budd-Chiari syndrome, idiopathic chronic active hepatitis, acute Wilson’s disease, microvesicular fat syndromes (Reye’s syndrome, acute fatty liver of pregnancy).
CLINICAL MANIFESTATIONS
Neuropsychiatric changes—delirium, personality change, stupor, coma; cerebral edema—suggested by profuse sweating, hemodynamic instability, tachyarrhythmias, tachypnea, fever, papilledema, decerebrate rigidity (though all may be absent); deep jaundice, coagulopathy, bleeding, renal failure, acid-base disturbance, hypoglycemia, acute pancreatitis, cardiorespiratory failure, infections (bacterial, fungal).
ADVERSE PROGNOSTIC INDICATORS
Age <10 or >40, certain causes (e.g., halothane, hepatitis C), duration of jaundice >7 d before onset of encephalopathy, serum bilirubin > 300 μmol/L (>18 mg/dL), coma (survival <20%), rapid reduction in liver size, respiratory failure, marked prolongation of PT, factor V level < 20%. In acetaminophen overdose, adverse prognosis is suggested by blood ![]() , serum creatinine >266 μmol/L (>3 mg/dL), markedly prolonged PT.
, serum creatinine >266 μmol/L (>3 mg/dL), markedly prolonged PT.
TREATMENT Acute Hepatic Failure
Endotracheal intubation often required. Monitor serum glucose—IV D10 or D20 as necessary. Prevent GI bleeding with H2 receptor antagonists and antacids (maintain gastric ![]() ). In many centers intracranial pressure is monitored—more sensitive than CT in detecting cerebral edema. Value of dexamethasone for cerebral edema unclear; IV mannitol may be beneficial. Liver transplantation should be considered in pts with grades III–IV encephalopathy and other adverse prognostic indicators.
). In many centers intracranial pressure is monitored—more sensitive than CT in detecting cerebral edema. Value of dexamethasone for cerebral edema unclear; IV mannitol may be beneficial. Liver transplantation should be considered in pts with grades III–IV encephalopathy and other adverse prognostic indicators.

For a more detailed discussion, see Dienstag JL: Acute Viral Hepatitis, Chap. 304, p.2537, and Dienstag JL: Toxic and Drug-Induced Hepatitis, Chap. 305, p. 2558, in HPIM-18.