Steven Go
EPIDEMIOLOGY
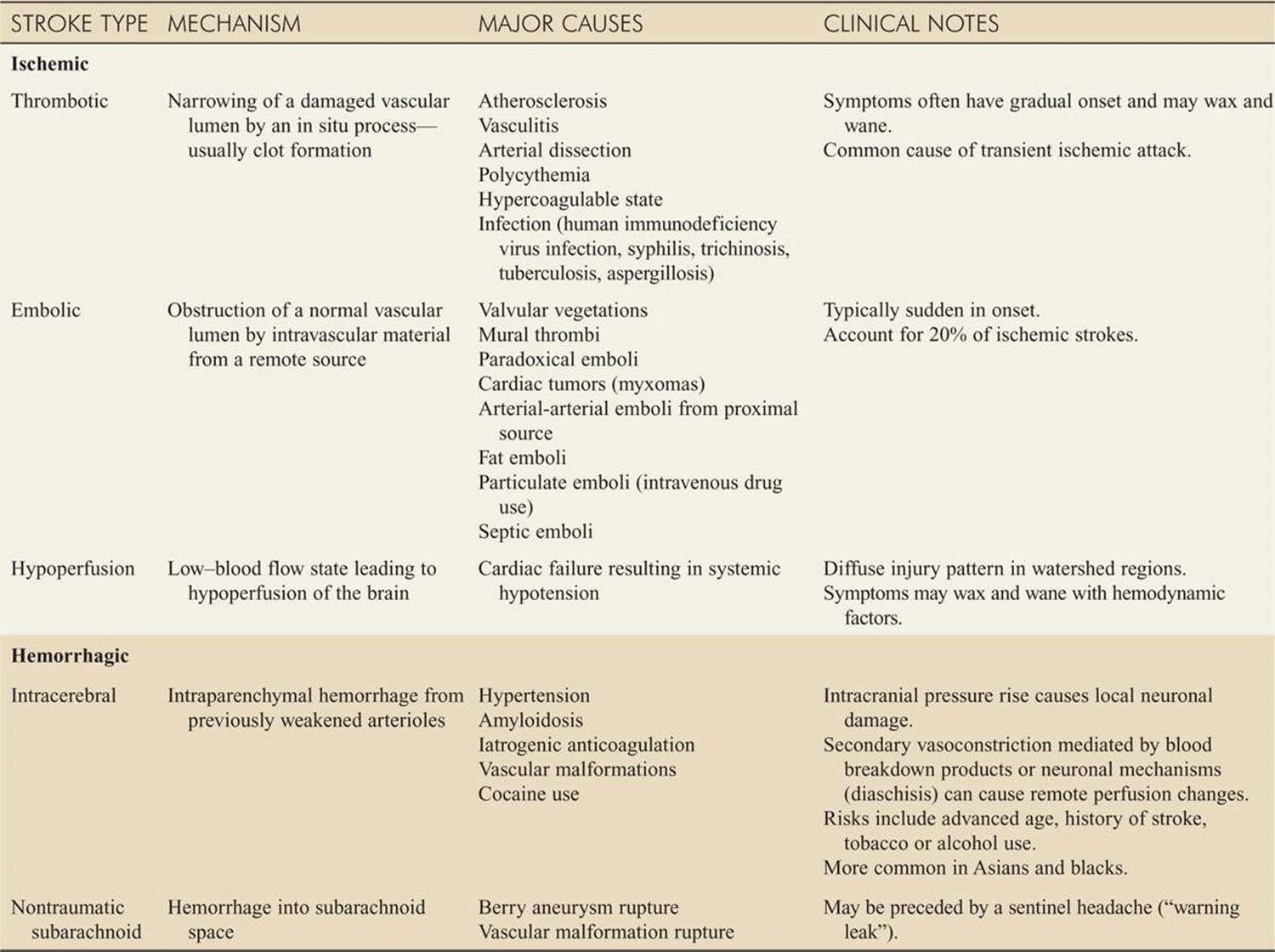
![]() Ischemic strokes (87%) are more common than hemorrhagic strokes (intracerebral [10%] and nontraumatic subarachnoid hemorrhage [3%]). See Table 143-1.
Ischemic strokes (87%) are more common than hemorrhagic strokes (intracerebral [10%] and nontraumatic subarachnoid hemorrhage [3%]). See Table 143-1.
TABLE 143-1 Stroke Classification
![]() Cervical artery dissection (CAD) causes 10% to 25% of strokes in young and middle-aged patients.
Cervical artery dissection (CAD) causes 10% to 25% of strokes in young and middle-aged patients.
PATHOPHYSIOLOGY
![]() Stroke may be defined as any disease process that interrupts blood flow to the brain. Ischemic strokes result from thrombotic, embolic, or low flow states. Hemorrhagic strokes are secondary to intracerebral hemorrhage (ICH) or subarachnoid hemorrhage (SAH).
Stroke may be defined as any disease process that interrupts blood flow to the brain. Ischemic strokes result from thrombotic, embolic, or low flow states. Hemorrhagic strokes are secondary to intracerebral hemorrhage (ICH) or subarachnoid hemorrhage (SAH).
![]() A transient ischemic attack (TIA) is a transient neurologic deficit that typically lasts less than 1 to 2 hours, but duration can no longer be used to discriminate between TIA and stroke; they are best thought of as similar disease processes on a continuum.
A transient ischemic attack (TIA) is a transient neurologic deficit that typically lasts less than 1 to 2 hours, but duration can no longer be used to discriminate between TIA and stroke; they are best thought of as similar disease processes on a continuum.
![]() CAD can be associated with neck trauma, major or trivial (chiropractic manipulation), family history of arterial disease, genetic factors, recent upper respiratory illness, connective tissue disease, and history of migraine.
CAD can be associated with neck trauma, major or trivial (chiropractic manipulation), family history of arterial disease, genetic factors, recent upper respiratory illness, connective tissue disease, and history of migraine.
![]() The vast majority of SAH are caused by a ruptured cerebral aneurysm.
The vast majority of SAH are caused by a ruptured cerebral aneurysm.
CLINICAL FEATURES
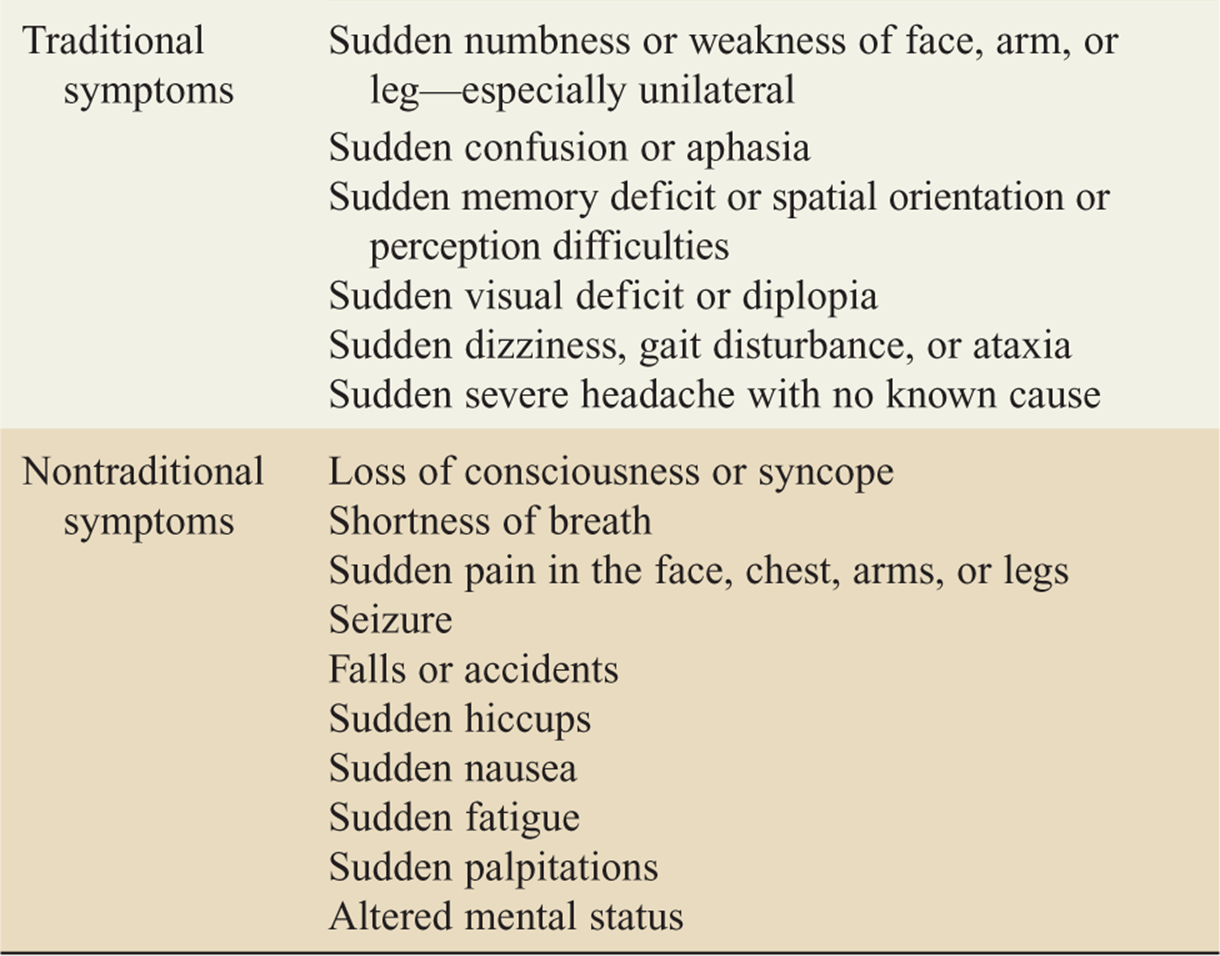
![]() Specific findings in stroke patients depend on the regions of the brain that are compromised and how severe the insult is (Table 143-2). Stroke presentation can vary considerably from classically described syndromes.
Specific findings in stroke patients depend on the regions of the brain that are compromised and how severe the insult is (Table 143-2). Stroke presentation can vary considerably from classically described syndromes.
TABLE 143-2 Symptoms of Stroke
![]() Anterior cerebral artery strokes typically cause contralateral leg weakness and sensory changes.
Anterior cerebral artery strokes typically cause contralateral leg weakness and sensory changes.
![]() Middle cerebral artery strokes classically present with hemiparesis (arm > leg) and sensory loss. Weakness in the lower half of the face (variable) and ipsilat-eral gaze preference may occur. Aphasia (receptive and/or expressive) is often present if the dominant hemisphere (usually left) is affected, while contralateral hemineglect suggests nondominant hemisphere involvement.
Middle cerebral artery strokes classically present with hemiparesis (arm > leg) and sensory loss. Weakness in the lower half of the face (variable) and ipsilat-eral gaze preference may occur. Aphasia (receptive and/or expressive) is often present if the dominant hemisphere (usually left) is affected, while contralateral hemineglect suggests nondominant hemisphere involvement.
![]() Posterior circulation strokes can present very subtly with findings such as unilateral headache, visual field defects, dizziness, vertigo, diplopia, dysphagia, ataxia, cranial nerve deficits, or bilateral limb weakness. Signs and symptoms can occur alone or in various combinations.
Posterior circulation strokes can present very subtly with findings such as unilateral headache, visual field defects, dizziness, vertigo, diplopia, dysphagia, ataxia, cranial nerve deficits, or bilateral limb weakness. Signs and symptoms can occur alone or in various combinations.
![]() Occlusion of the basilar artery causes severe quadriplegia, coma, and the locked-in syndrome.
Occlusion of the basilar artery causes severe quadriplegia, coma, and the locked-in syndrome.
![]() Cerebellar strokes present similarly to other posterior stroke syndromes, but can deteriorate quickly if a hematoma or edema is present.
Cerebellar strokes present similarly to other posterior stroke syndromes, but can deteriorate quickly if a hematoma or edema is present.
![]() CAD can involve both the anterior and posterior arterial systems. The most common symptoms with an internal carotid dissection are unilateral head pain (50-67%), neck pain (25%), or face pain (10%). Vertebral artery dissections commonly present with headache (69%) and posterior neck pain (46%), which can be unilateral or bilateral. Signs and symptoms of CAD can be transient as well as persistent.
CAD can involve both the anterior and posterior arterial systems. The most common symptoms with an internal carotid dissection are unilateral head pain (50-67%), neck pain (25%), or face pain (10%). Vertebral artery dissections commonly present with headache (69%) and posterior neck pain (46%), which can be unilateral or bilateral. Signs and symptoms of CAD can be transient as well as persistent.
![]() Intracranial hemorrhages may present with any of the anatomic syndromes discussed above.
Intracranial hemorrhages may present with any of the anatomic syndromes discussed above.
![]() SAH classically presents with sudden onset of a “thunderclap” headache at its most severe. It occurs 20% of the time during activity associated with elevated blood pressures such as sexual intercourse, weight lifting, defecation, or coughing. Vomiting, photophobia, nuchal irritation, low-grade fever, and altered mental status all may occur. Focal findings can occur depending on the location of the aneurysm. A recent history suggestive of a warning leak may be obtained in many patients.
SAH classically presents with sudden onset of a “thunderclap” headache at its most severe. It occurs 20% of the time during activity associated with elevated blood pressures such as sexual intercourse, weight lifting, defecation, or coughing. Vomiting, photophobia, nuchal irritation, low-grade fever, and altered mental status all may occur. Focal findings can occur depending on the location of the aneurysm. A recent history suggestive of a warning leak may be obtained in many patients.
![]() Symptoms may resolve spontaneously as blood diffuses in the SAH space; therefore, the clinician should not be misled by improving clinical condition when the history is strongly suggestive of SAH.
Symptoms may resolve spontaneously as blood diffuses in the SAH space; therefore, the clinician should not be misled by improving clinical condition when the history is strongly suggestive of SAH.
DIAGNOSIS AND DIFFERENTIAL
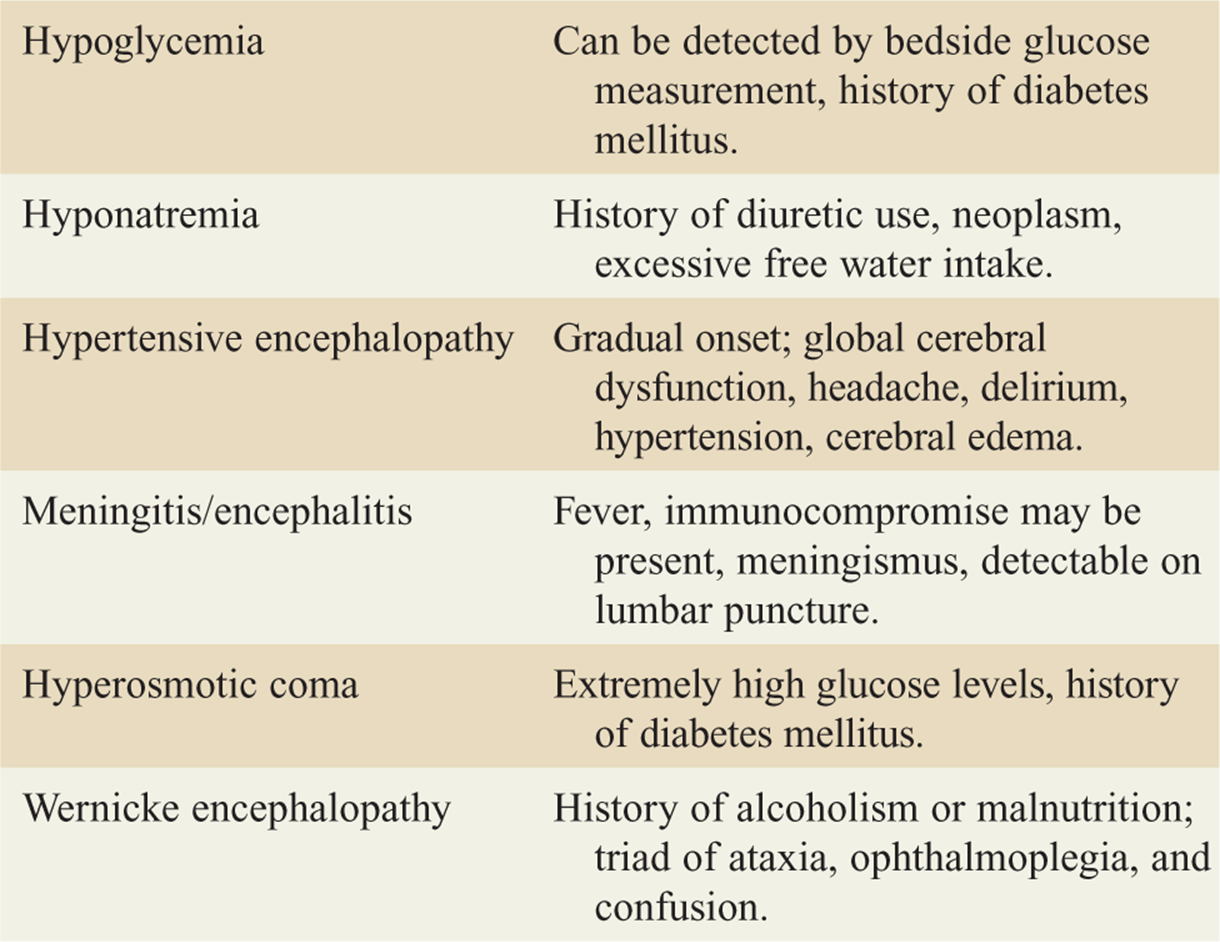
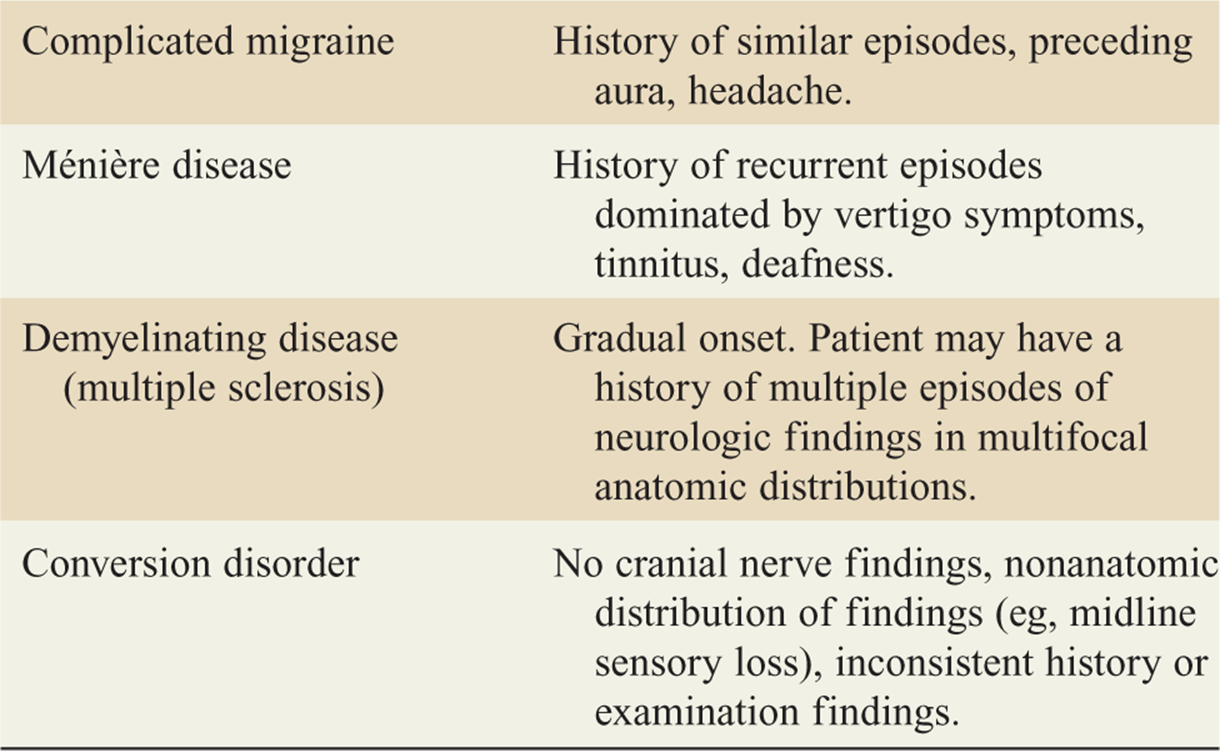
![]() The differential for stroke-like symptoms is found in Table 143-3.
The differential for stroke-like symptoms is found in Table 143-3.
TABLE 143-3 Differential Diagnoses of Consequence for Acute Stroke Symptoms


![]() It is critical that an accurate determination of the time the patient was last known to be at their neurological baseline be made.
It is critical that an accurate determination of the time the patient was last known to be at their neurological baseline be made.
![]() Eligibility for thrombolytic therapy should be determined if stroke is the primary working diagnosis (Tables 143-4 and 143-5).
Eligibility for thrombolytic therapy should be determined if stroke is the primary working diagnosis (Tables 143-4 and 143-5).
TABLE 143-4 American Heart Association/American Stroke Association 2007 Criteria for IV Recombinant Tissue Plasminogen Activator (rtPA) in Acute Ischemic Stroke


TABLE 143-5 Additional Exclusion Criteria for IV Recombinant Tissue Plasminogen Activator (rtPA) in Acute Ischemic Stroke When Given 3 to 4.5 Hours After Symptom Onset
|
Age >80 y |
|
Severe stroke as assessed clinically (NIHSS score >25) |
|
Combination of previous stroke and diabetes mellitus |
|
Blood glucose <50 milligrams/dL or >400 milligrams/dL |
|
Oral anticoagulant treatment |
Abbreviation: NIHSS = National Institutes of Health Stroke Scale.
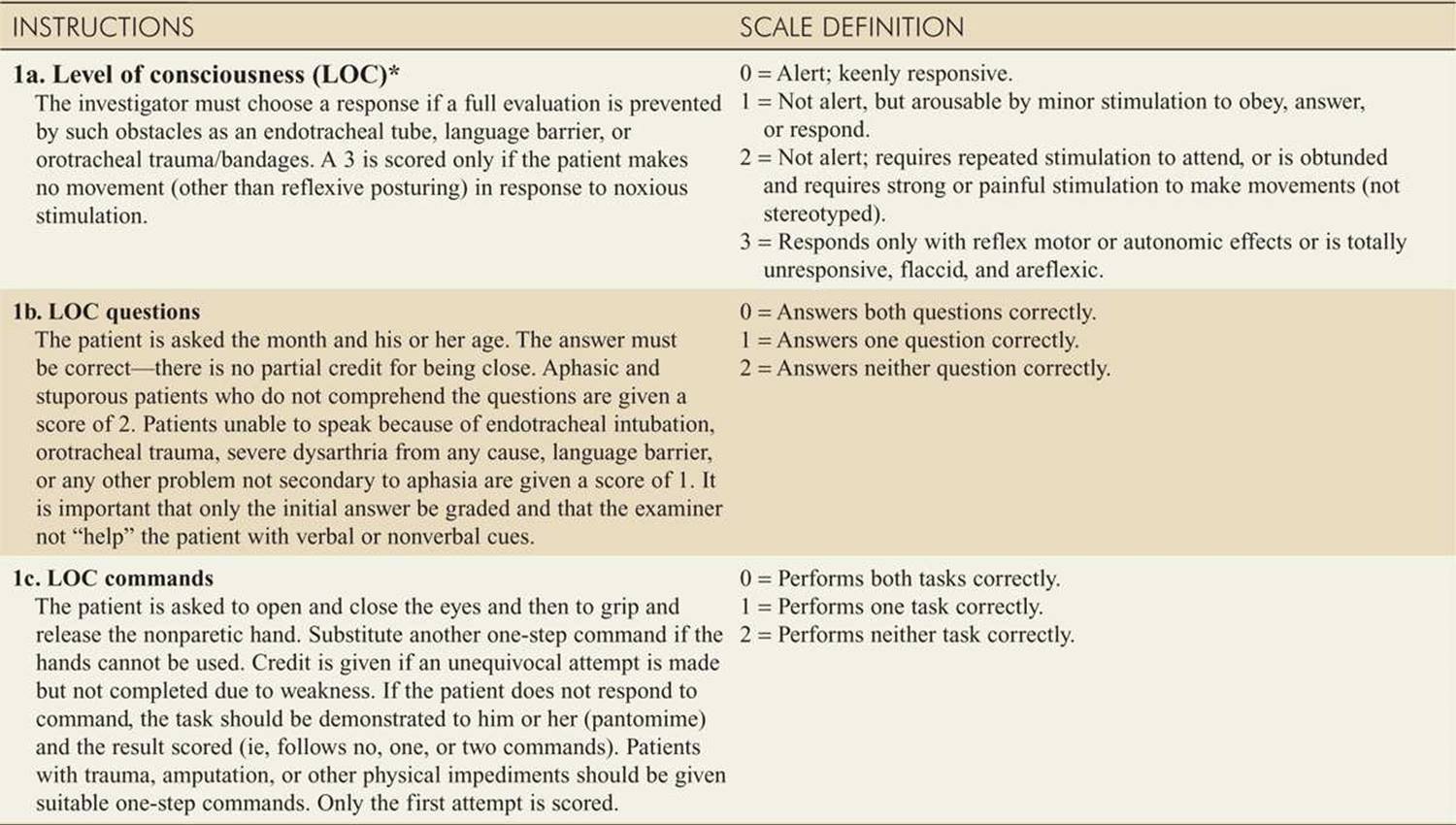
![]() A National Institutes of Health Stroke Scale (NIHSS) score should be quickly calculated upon presentation (Table 143-6).
A National Institutes of Health Stroke Scale (NIHSS) score should be quickly calculated upon presentation (Table 143-6).
TABLE 143-6 National Institutes of Health Stroke Scale (NIHSS)




![]() An emergent noncontrast CT scan (best interpreted by a neuroradiologist) should be rapidly performed within 25 minutes of arrival to determine whether hemorrhage or a stroke mimic is present. Most acute ischemic strokes will not be visualized in the early hours of a stroke.
An emergent noncontrast CT scan (best interpreted by a neuroradiologist) should be rapidly performed within 25 minutes of arrival to determine whether hemorrhage or a stroke mimic is present. Most acute ischemic strokes will not be visualized in the early hours of a stroke.
![]() MRI, MRA, and CT angiography may be of value in detecting particular disease entities (eg, CAD, tumor, circle of Willis aneurysms), but their exact role is currently unclear.
MRI, MRA, and CT angiography may be of value in detecting particular disease entities (eg, CAD, tumor, circle of Willis aneurysms), but their exact role is currently unclear.
![]() Modern CTs are thought to be 98% sensitive to detect SAH within 12 hours of symptom onset; however, some studies have found sensitivity as low as 85.7% when a third-generation CT is performed greater than 6 hours after headache onset.
Modern CTs are thought to be 98% sensitive to detect SAH within 12 hours of symptom onset; however, some studies have found sensitivity as low as 85.7% when a third-generation CT is performed greater than 6 hours after headache onset.
![]() If SAH is suspected and the CT is negative, many authorities agree that a lumbar puncture is indicated.
If SAH is suspected and the CT is negative, many authorities agree that a lumbar puncture is indicated.
![]() Cerebrospinal fluid (CSF) xanthochromia does not develop until 12 hours after symptom onset, and the threshold number of RBCs needed in the CSF to be considered diagnostic of SAH is still unclear. Xanthochromia is best excluded by spectrometric means.
Cerebrospinal fluid (CSF) xanthochromia does not develop until 12 hours after symptom onset, and the threshold number of RBCs needed in the CSF to be considered diagnostic of SAH is still unclear. Xanthochromia is best excluded by spectrometric means.
![]() A normal head CT, no xanthochromia, and zero or few RBCs (<5 × 106 RBCs/L) are generally considered to exclude an SAH.
A normal head CT, no xanthochromia, and zero or few RBCs (<5 × 106 RBCs/L) are generally considered to exclude an SAH.
![]() Other diagnostic tests that may be useful in certain patients to rule out stroke mimics or concurrent conditions include a complete blood count, ECG, pulse oximetry, electrolyte and coagulation studies, cardiac enzyme levels, toxicology screen, blood alcohol level, echocardiogram, and carotid duplex scanning.
Other diagnostic tests that may be useful in certain patients to rule out stroke mimics or concurrent conditions include a complete blood count, ECG, pulse oximetry, electrolyte and coagulation studies, cardiac enzyme levels, toxicology screen, blood alcohol level, echocardiogram, and carotid duplex scanning.
EMERGENCY DEPARTMENT CARE AND DISPOSITION
![]() Rapid assessment and stabilization of any airway, breathing, and circulation abnormalities is paramount.
Rapid assessment and stabilization of any airway, breathing, and circulation abnormalities is paramount.
![]() Routine supplemental oxygen is not indicated, but O2 saturation should be kept ≥94%.
Routine supplemental oxygen is not indicated, but O2 saturation should be kept ≥94%.
![]() IV access should be established while the patient is placed on a cardiac monitor.
IV access should be established while the patient is placed on a cardiac monitor.
![]() A rapid bedside glucose determination should be done and any hypoglycemia normalized.
A rapid bedside glucose determination should be done and any hypoglycemia normalized.
![]() The patient should be kept NPO, on strict bedrest, and aspiration and fall precautions instituted.
The patient should be kept NPO, on strict bedrest, and aspiration and fall precautions instituted.
![]() Once the patient’S condition is stabilized, the patient should be sent for a noncontrast head CT scan.
Once the patient’S condition is stabilized, the patient should be sent for a noncontrast head CT scan.
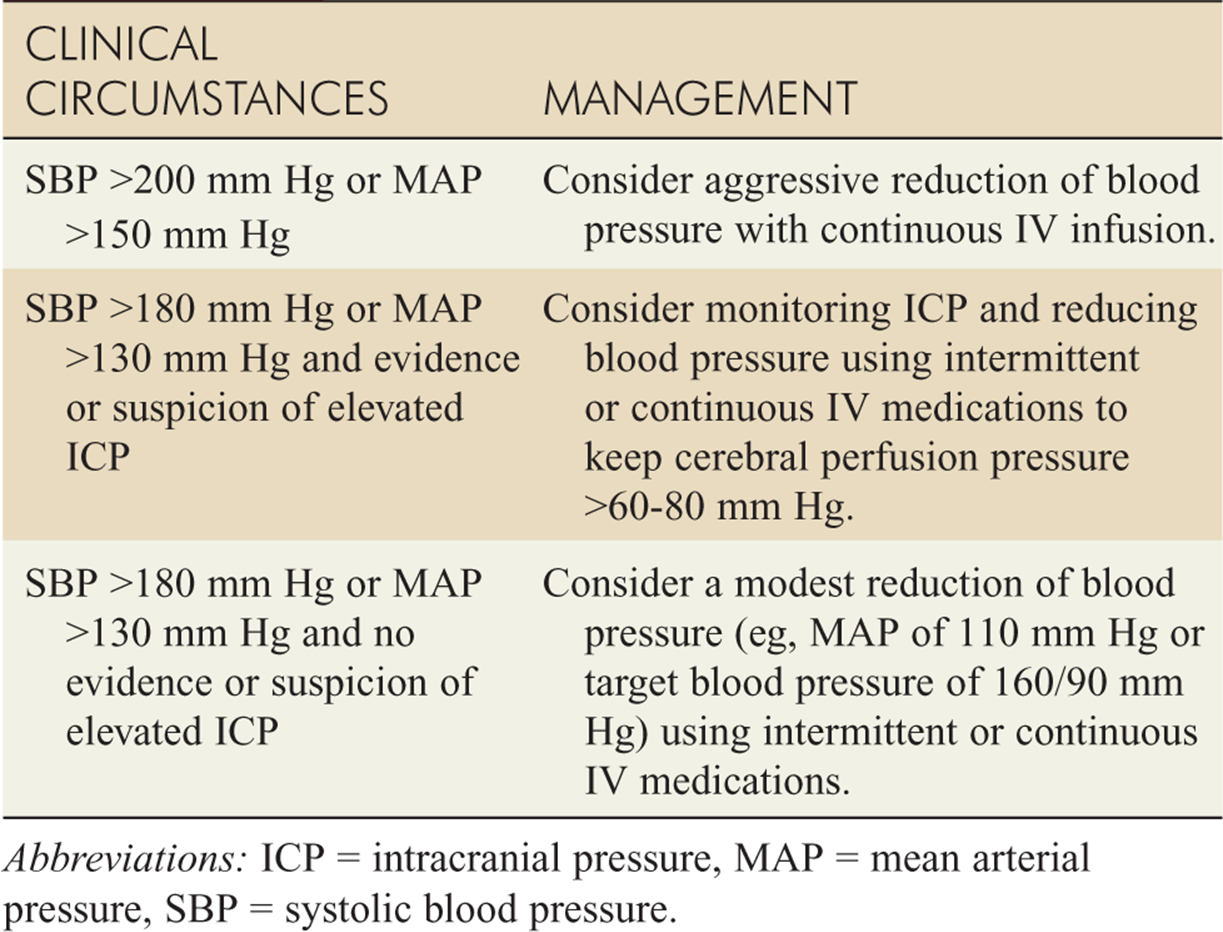
![]() If a patient is not a candidate for thrombolysis, then permissive hypertension is in order (ie, no intervention unless systolic blood pressure [SBP] >220 mm Hg or diastolic blood pressure [DBP] >120 mm Hg). If blood pressure control is needed, a titratable intravenous antihypertensive should be used, such as labetalol (typical starting dose is 10-20 milligrams over 1-2 minutes IVP; continuous infusion dosage generally starts at 2 milligrams/min, titrated to effect) with a target MAP reduction of 10% to 25%. Extreme caution should be taken to avoid overcorrection.
If a patient is not a candidate for thrombolysis, then permissive hypertension is in order (ie, no intervention unless systolic blood pressure [SBP] >220 mm Hg or diastolic blood pressure [DBP] >120 mm Hg). If blood pressure control is needed, a titratable intravenous antihypertensive should be used, such as labetalol (typical starting dose is 10-20 milligrams over 1-2 minutes IVP; continuous infusion dosage generally starts at 2 milligrams/min, titrated to effect) with a target MAP reduction of 10% to 25%. Extreme caution should be taken to avoid overcorrection.
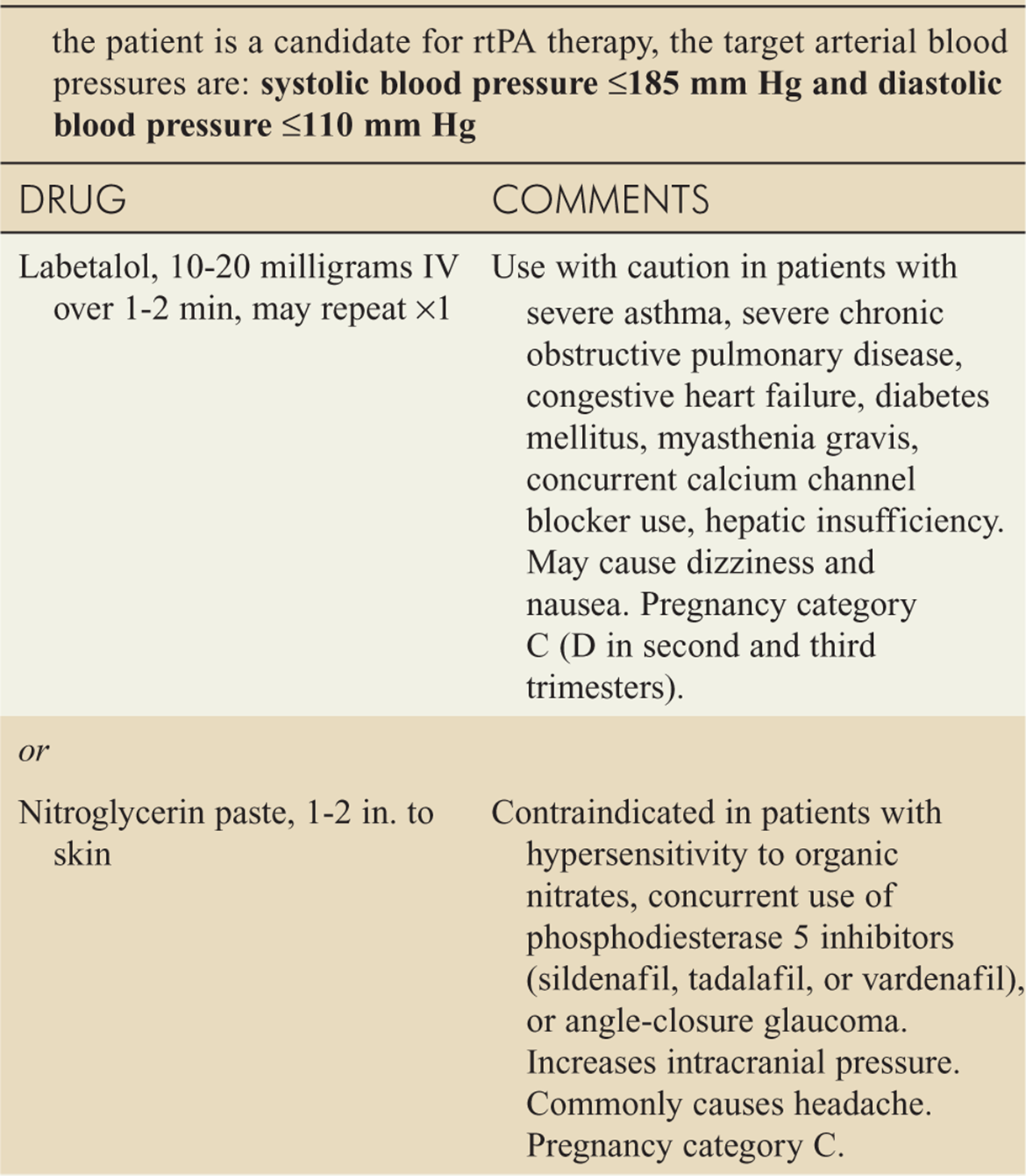
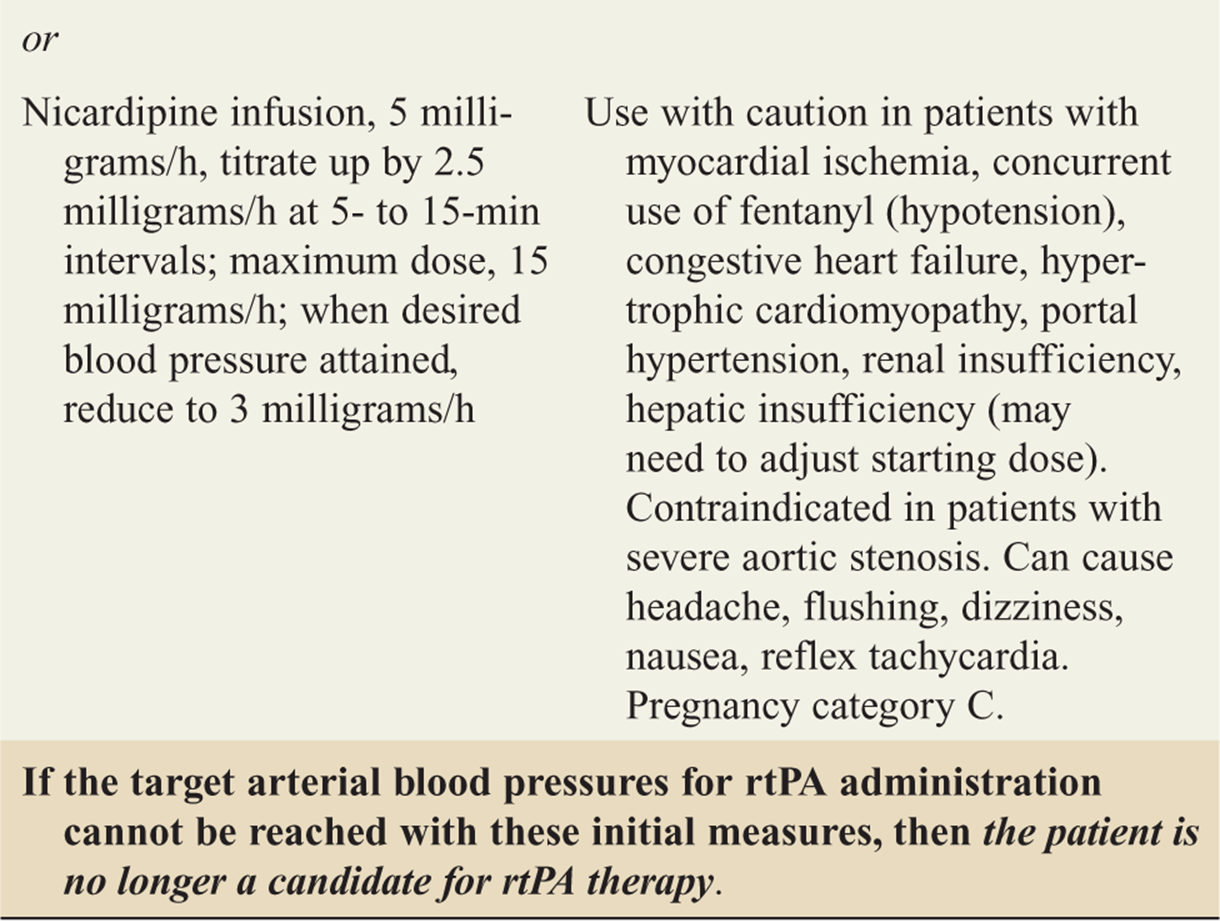
![]() If a patient is a candidate for recombinant tissue plas-minogen activator (rtPA), the target blood pressures are SBP ≤ 185 mm Hg and DBP ≤ 110 mm Hg prior to administration of rtPA (see Table 143-7).
If a patient is a candidate for recombinant tissue plas-minogen activator (rtPA), the target blood pressures are SBP ≤ 185 mm Hg and DBP ≤ 110 mm Hg prior to administration of rtPA (see Table 143-7).
TABLE 143-7 Approach to Management of Arterial Hypertension Before Potential Administration of Recombinant Tissue Plasminogen Activator (rtPA)
![]() IV rtPA has been approved for treatment of acute ischemic stroke ≤3 hours of symptom onset.
IV rtPA has been approved for treatment of acute ischemic stroke ≤3 hours of symptom onset.
![]() The American Heart Association/American Stroke Association recommends expansion of the rtPA treatment window to 4.5 hours, but this indication is still awaiting approval by the U.S. FDA at the time of this writing and is considered “off-label.”
The American Heart Association/American Stroke Association recommends expansion of the rtPA treatment window to 4.5 hours, but this indication is still awaiting approval by the U.S. FDA at the time of this writing and is considered “off-label.”
![]() A careful review of rtPA inclusion and exclusion criteria must be meticulously performed prior to administration of rtPA (Table 143-4).
A careful review of rtPA inclusion and exclusion criteria must be meticulously performed prior to administration of rtPA (Table 143-4).
![]() If the therapeutic window is to be extended to 3 to 4.5 hours, then the additional European Cooperative Acute Stroke Study 3 (ECASS III) exclusion criteria should be used (Table 143-5).
If the therapeutic window is to be extended to 3 to 4.5 hours, then the additional European Cooperative Acute Stroke Study 3 (ECASS III) exclusion criteria should be used (Table 143-5).
![]() Informed consent must be obtained from the patient or their designee prior to thrombolytic therapy.
Informed consent must be obtained from the patient or their designee prior to thrombolytic therapy.
![]() The risk of symptomatic intracerebral hemorrhage (SIH) is 6.4% (45% mortality) when rtPA is given within ≤3 hours of symptom onset and 7.9% (NINDS definition) between 3 and 4.5 hours.
The risk of symptomatic intracerebral hemorrhage (SIH) is 6.4% (45% mortality) when rtPA is given within ≤3 hours of symptom onset and 7.9% (NINDS definition) between 3 and 4.5 hours.
![]() The total dose of rtPA is 0.9 milligrams/kg IV, with a maximum dose of 90 milligrams; 10% of the dose is administered as a bolus, with the remaining amount infused over 60 minutes.
The total dose of rtPA is 0.9 milligrams/kg IV, with a maximum dose of 90 milligrams; 10% of the dose is administered as a bolus, with the remaining amount infused over 60 minutes.
![]() No aspirin or heparin should be administered in the initial 24 hours after treatment with rtPA. Aspirin (325 milligrams orally) is indicated within 24 to 48 hours if the patient is not at risk for aspiration.
No aspirin or heparin should be administered in the initial 24 hours after treatment with rtPA. Aspirin (325 milligrams orally) is indicated within 24 to 48 hours if the patient is not at risk for aspiration.
![]() Intracerebral bleeding should be suspected as the cause of any neurologic worsening.
Intracerebral bleeding should be suspected as the cause of any neurologic worsening.
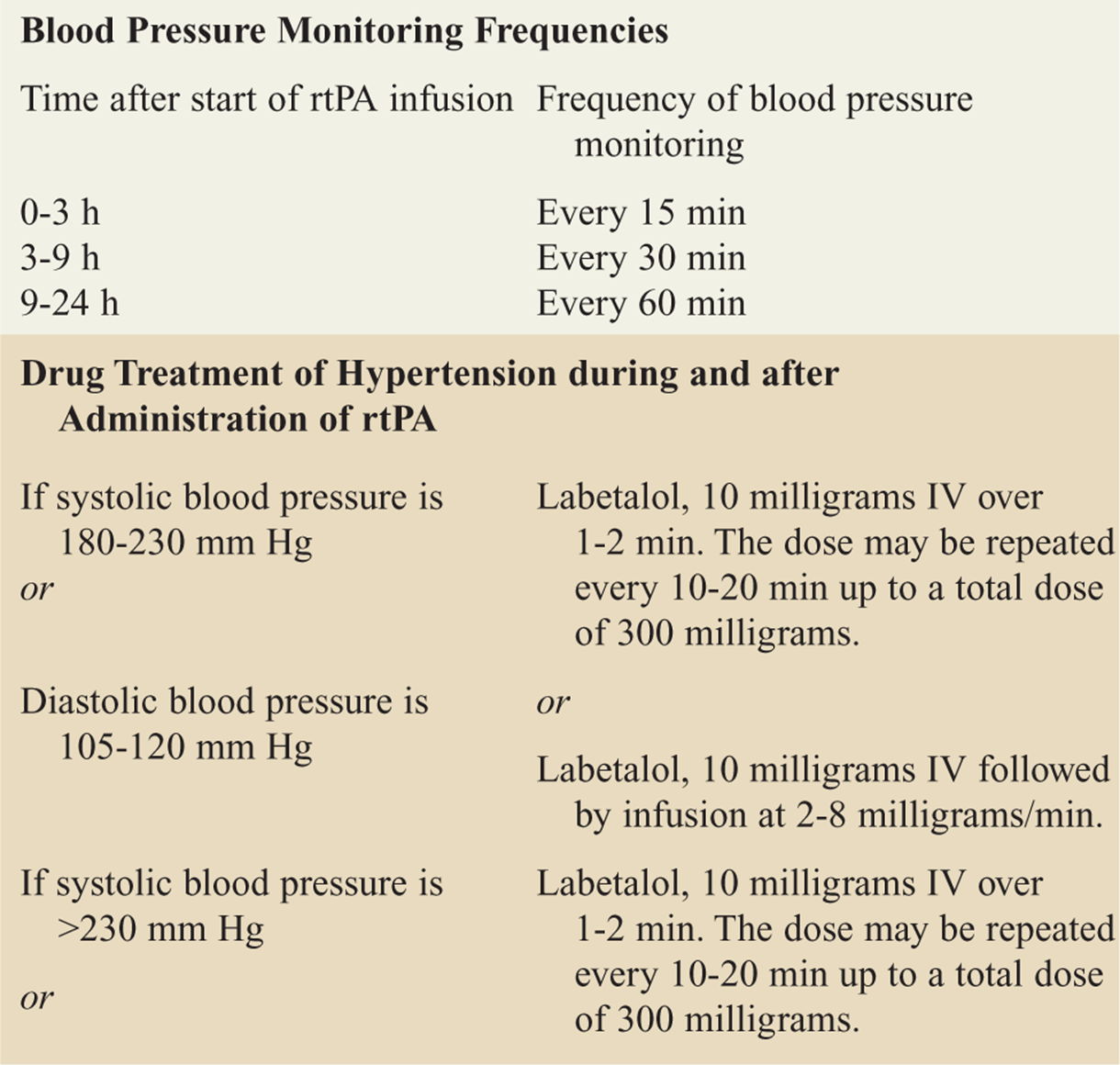
![]() Patients who receive rtPA should have their blood pressure monitored closely and treated if necessary (see Table 143-8).
Patients who receive rtPA should have their blood pressure monitored closely and treated if necessary (see Table 143-8).
TABLE 143-8 Management of Blood Pressure during and after Administration of Recombinant Tissue Plasminogen Activator (rtPa)
![]() For TIA patients, aspirin (325 milligrams orally) plus dipyridamole (400 milligrams orally) is recommended. Antiplatelet therapy is contraindicated for hemorrhagic stroke.
For TIA patients, aspirin (325 milligrams orally) plus dipyridamole (400 milligrams orally) is recommended. Antiplatelet therapy is contraindicated for hemorrhagic stroke.
![]() Heparin or warfarin in the acute treatment of TIA or stroke in the ED is not indicated, even in the presence of atrial fibrillation. A possible exception exists for a weight-based heparin protocol in CAD, but this remains controversial.
Heparin or warfarin in the acute treatment of TIA or stroke in the ED is not indicated, even in the presence of atrial fibrillation. A possible exception exists for a weight-based heparin protocol in CAD, but this remains controversial.
![]() If an ischemic stroke patient presents outside the rtPA therapeutic time window, then aggressive supportive care should be given in the ED (aspiration prevention, normalization of glucose level, fall precautions, and treatment for comorbidities).
If an ischemic stroke patient presents outside the rtPA therapeutic time window, then aggressive supportive care should be given in the ED (aspiration prevention, normalization of glucose level, fall precautions, and treatment for comorbidities).
![]() For patients with evidence of increased intracranial pressure (ICP), head elevation to 30°, analgesia, and sedation are indicated. If more aggressive ICP reduction is indicated, mannitol (0.25-1.0 gram/kg IV), intubation with neuromuscular blockade with mild hyperventilation, and invasive monitoring of ICP may be required.
For patients with evidence of increased intracranial pressure (ICP), head elevation to 30°, analgesia, and sedation are indicated. If more aggressive ICP reduction is indicated, mannitol (0.25-1.0 gram/kg IV), intubation with neuromuscular blockade with mild hyperventilation, and invasive monitoring of ICP may be required.
![]() In SAH, the chances of rebleeding can be reduced by maintaining the patient’S prebleed blood pressure (or MAP <130 mm Hg if baseline blood pressure is unknown). An IV titratable antihypertensive such as labetalol (typical adult starting dose is 10-20 milligrams over 1-2 minutes IVP; continuous infusion dosage generally starts at 2 milligrams/min, titrated to effect) should be used. Nimodipine (60 milligrams orally every 4 hours) may produce modest improvements in outcome by decreasing vasospasm.
In SAH, the chances of rebleeding can be reduced by maintaining the patient’S prebleed blood pressure (or MAP <130 mm Hg if baseline blood pressure is unknown). An IV titratable antihypertensive such as labetalol (typical adult starting dose is 10-20 milligrams over 1-2 minutes IVP; continuous infusion dosage generally starts at 2 milligrams/min, titrated to effect) should be used. Nimodipine (60 milligrams orally every 4 hours) may produce modest improvements in outcome by decreasing vasospasm.
![]() Emergent neurosurgical consultation is indicated in SAH patients. Pain medications and antiemetics should be used as needed.
Emergent neurosurgical consultation is indicated in SAH patients. Pain medications and antiemetics should be used as needed.
![]() Seizure prophylaxis for SAH is controversial and should be discussed with the specialist who will manage the patient after they leave the ED.
Seizure prophylaxis for SAH is controversial and should be discussed with the specialist who will manage the patient after they leave the ED.
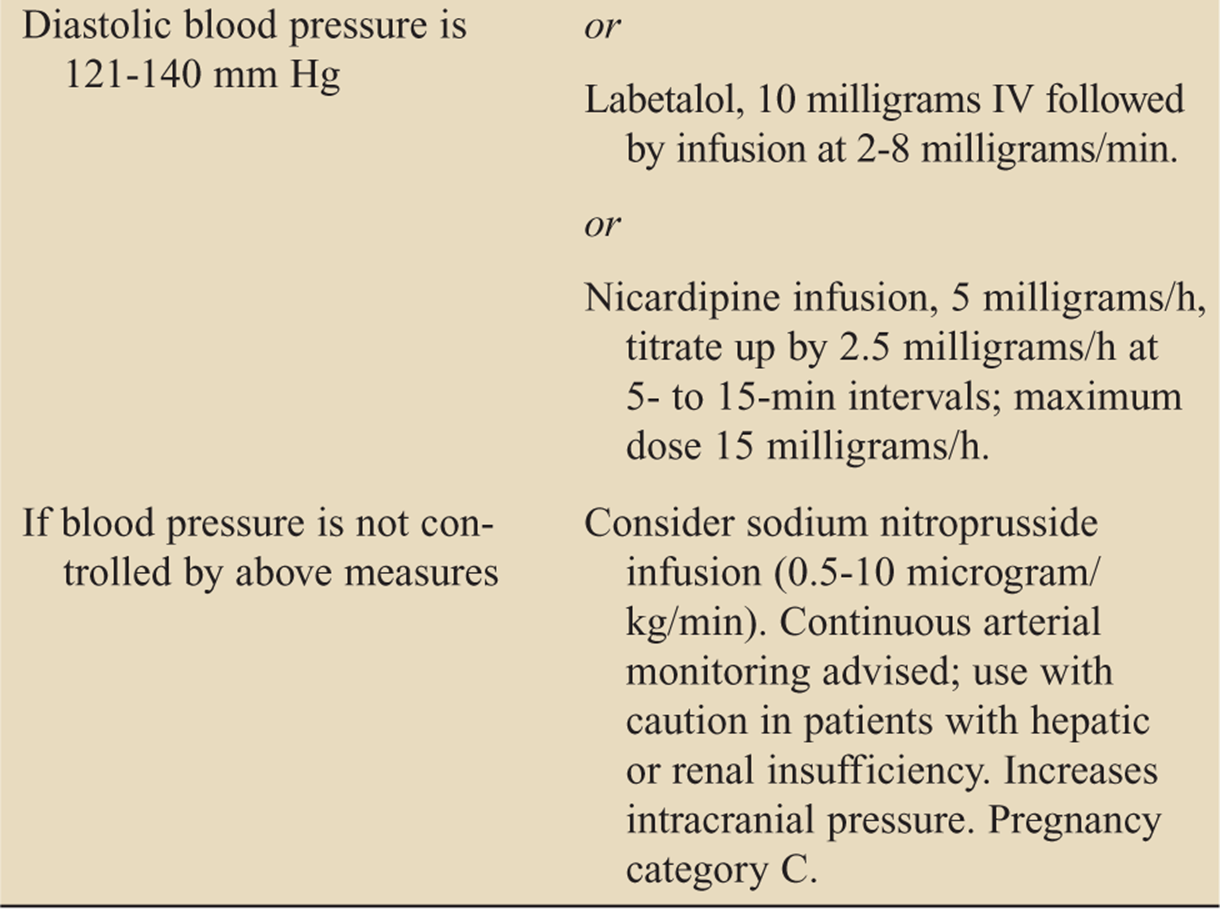
![]() Evidence for the management of blood pressure when spontaneous ICH is present remains controversial. Current guidelines are listed in Table 143-9.
Evidence for the management of blood pressure when spontaneous ICH is present remains controversial. Current guidelines are listed in Table 143-9.
TABLE 143-9 Suggested Guidelines for Treating Elevated Blood Pressure in Spontaneous Intracranial Hemorrhage
![]() Emergent consultation with a neurologist may be helpful in difficult stroke cases where thrombolytics are a consideration; however, therapy should not be unduly delayed while waiting for a response.
Emergent consultation with a neurologist may be helpful in difficult stroke cases where thrombolytics are a consideration; however, therapy should not be unduly delayed while waiting for a response.
![]() Early neurosurgical consultation is indicated for patients with ICH with evidence of increased ICP or in other conditions where surgical intervention may be indicated (eg, cerebellar stroke).
Early neurosurgical consultation is indicated for patients with ICH with evidence of increased ICP or in other conditions where surgical intervention may be indicated (eg, cerebellar stroke).
![]() All patients with acute ischemic stroke or ICH should be admitted, even if they are not candidates for interventional therapy.
All patients with acute ischemic stroke or ICH should be admitted, even if they are not candidates for interventional therapy.
![]() Admission to specialized stroke units is associated with improved outcomes; therefore, transfer to a designated stroke center may be indicated if the patient presents to a non-stroke center.
Admission to specialized stroke units is associated with improved outcomes; therefore, transfer to a designated stroke center may be indicated if the patient presents to a non-stroke center.
![]() The ABCD2 scoring system may be used to predict stroke risk in TIA patients (see Table 143-10).
The ABCD2 scoring system may be used to predict stroke risk in TIA patients (see Table 143-10).
TABLE 143-10 ABCD2 Score to Predict Very Early Stroke Risk after Transient Ischemic Attack
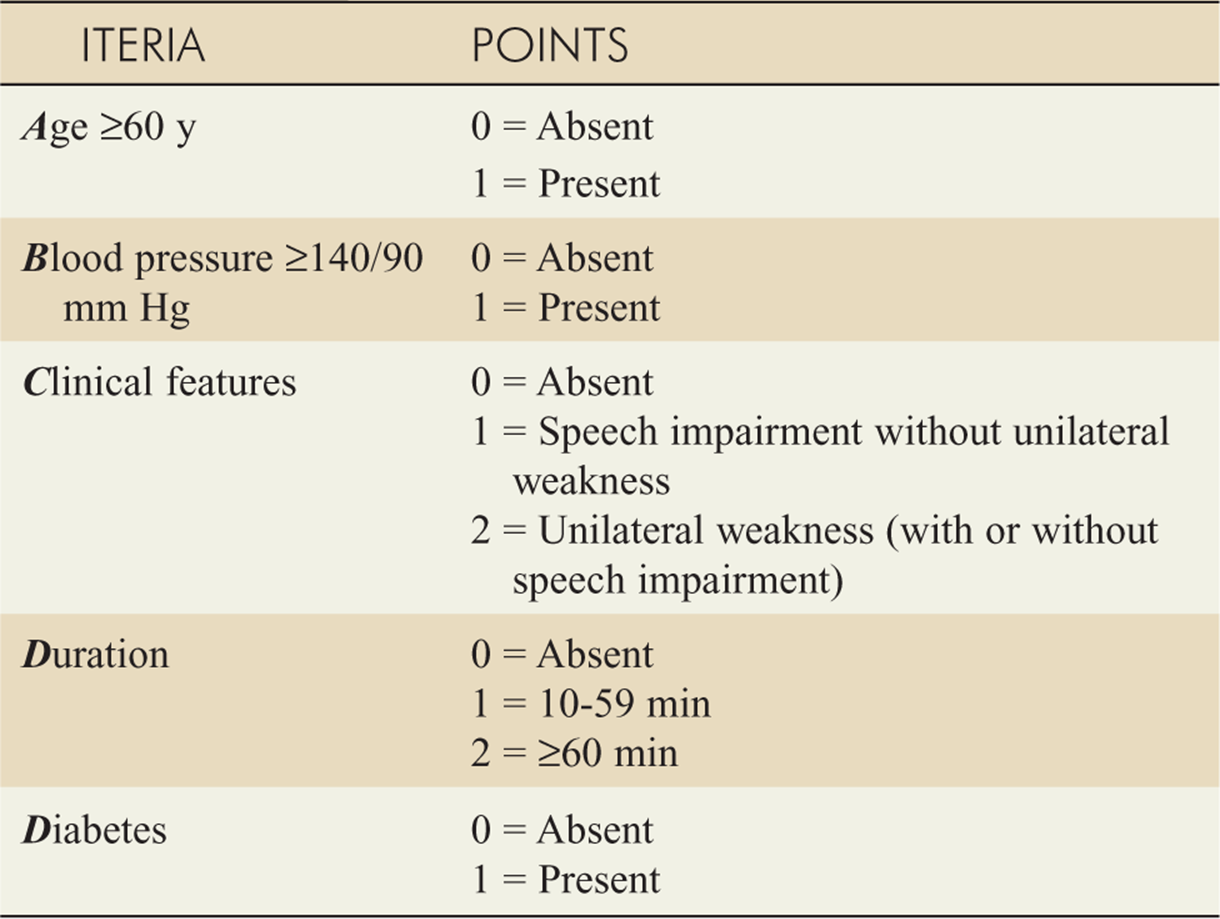
![]() Using the ABCD2, the 2-day risks of subsequent stroke are 1% (ABCD2 score 0-3); 4.1% (4-5); and 8.1% (6-7).
Using the ABCD2, the 2-day risks of subsequent stroke are 1% (ABCD2 score 0-3); 4.1% (4-5); and 8.1% (6-7).
![]() Because of the proven efficacy of early carotid endar-terectomy, many stroke experts recommend admission for most TIA patients for inpatient evaluation and observation. In select low-risk, asymptomatic patients, next-day follow-up and evaluation with a specialist may be appropriate, but responsible adults to observe the patient in a favorable social situation must be available and very explicit return precautions given.
Because of the proven efficacy of early carotid endar-terectomy, many stroke experts recommend admission for most TIA patients for inpatient evaluation and observation. In select low-risk, asymptomatic patients, next-day follow-up and evaluation with a specialist may be appropriate, but responsible adults to observe the patient in a favorable social situation must be available and very explicit return precautions given.
![]() All CAD and SAH patients should be admitted with appropriate specialty consultation.
All CAD and SAH patients should be admitted with appropriate specialty consultation.
For further reading in Tintinalli’s Emergency Medicine: A Comprehensive Study Guide, 7th ed., see Chapter 160, “Spontaneous Subarachnoid and Intracerebral Hemorrhage,” by Jeffrey L. Hackman, Melissa D. Johnson, and O. John Ma, and Chapter 161, “Stroke, Transient Ischemic Attack, and Cervical Artery Dissection,” by Steven Go and Daniel J. Worman.