David M. Cline
EPIDEMIOLOGY
![]() Ischemic heart disease is the number one killer of adults in the United States, with 204.3 deaths per 100,000 population.
Ischemic heart disease is the number one killer of adults in the United States, with 204.3 deaths per 100,000 population.
![]() Acute coronary syndrome (ACS) is a spectrum of disease ranging from acute episodes of intermittent angina to acute myocardial infarction (AMI).
Acute coronary syndrome (ACS) is a spectrum of disease ranging from acute episodes of intermittent angina to acute myocardial infarction (AMI).
PATHOPHYSIOLOGY
![]() Coronary plaque forms on coronary vessel walls after repetitive injury. With plaque rupture, thrombogenic substances are exposed to platelets.
Coronary plaque forms on coronary vessel walls after repetitive injury. With plaque rupture, thrombogenic substances are exposed to platelets.
![]() Platelet response involves adhesion, activation, and aggregation. Platelet adhesion molecules are strongly thrombogenic and bind to von Willebrand’s factor (VWF). Thrombin, collagen, shearing forces, adenosine diphosphate (ADP), thromboxane A2, and serotonin are potent platelet activators. Platelet glyco-protein IIb/IIIa receptors cross-link fibrinogen or VWF as the common pathway of aggregation.
Platelet response involves adhesion, activation, and aggregation. Platelet adhesion molecules are strongly thrombogenic and bind to von Willebrand’s factor (VWF). Thrombin, collagen, shearing forces, adenosine diphosphate (ADP), thromboxane A2, and serotonin are potent platelet activators. Platelet glyco-protein IIb/IIIa receptors cross-link fibrinogen or VWF as the common pathway of aggregation.
![]() The severity of ACS depends upon the extent of O2 deprivation by thrombus, with complete occlusion resulting in cell death.
The severity of ACS depends upon the extent of O2 deprivation by thrombus, with complete occlusion resulting in cell death.
![]() AMI results in injury to both the conduction system, leading to ectopy and dysrhythmia, and left ventricle (LV) pump function, leading to increased filling pressures.
AMI results in injury to both the conduction system, leading to ectopy and dysrhythmia, and left ventricle (LV) pump function, leading to increased filling pressures.
![]() Cocaine is directly toxic to the myocardium and chronic use accelerates atherosclerosis; acute use may prompt AMI.
Cocaine is directly toxic to the myocardium and chronic use accelerates atherosclerosis; acute use may prompt AMI.
CLINICAL FEATURES
![]() Cardiac risk factors are modestly predictive of CAD in asymptomatic patients, but poor emergency department (ED) predictors for AMI or other ACS.
Cardiac risk factors are modestly predictive of CAD in asymptomatic patients, but poor emergency department (ED) predictors for AMI or other ACS.
![]() The physical examination of a patient with an ACS can range from normal to profound illness.
The physical examination of a patient with an ACS can range from normal to profound illness.
![]() Silent (painless)/atypical presentations of ischemia are common. Women and the elderly are more likely to present in this way and may complain of easy fatigability and/or shortness of breath.
Silent (painless)/atypical presentations of ischemia are common. Women and the elderly are more likely to present in this way and may complain of easy fatigability and/or shortness of breath.
![]() Ischemic/anginal pain is similar to AMI pain. AMI pain usually resolves only with aggressive intervention, whereas anginal pain can resolve with rest or nitroglycerin (NTG).
Ischemic/anginal pain is similar to AMI pain. AMI pain usually resolves only with aggressive intervention, whereas anginal pain can resolve with rest or nitroglycerin (NTG).
![]() Extent and location of myocardial loss determines prognosis and predicts complications. A 25% loss of the LV leads to congestive heart failure (CHF). A 40% LV loss leads to shock. Right ventricle (RV) infarct leads to hypotension.
Extent and location of myocardial loss determines prognosis and predicts complications. A 25% loss of the LV leads to congestive heart failure (CHF). A 40% LV loss leads to shock. Right ventricle (RV) infarct leads to hypotension.
![]() Anterior injury leads to tachydysrhythmia. Inferior injury leads to increased vagal tone and first-degree and Mobitz I blocks.
Anterior injury leads to tachydysrhythmia. Inferior injury leads to increased vagal tone and first-degree and Mobitz I blocks.
![]() Mobitz II block is usually associated with anterior AMI and may lead to complete heart block. Anterior and/or inferior injury may lead to complete heart block.
Mobitz II block is usually associated with anterior AMI and may lead to complete heart block. Anterior and/or inferior injury may lead to complete heart block.
![]() Fifteen to twenty percent of AMI patients have some degree of CHF.
Fifteen to twenty percent of AMI patients have some degree of CHF.
![]() Free wall myocardial rupture accounts for 10% of AMI fatalities and occurs 1 to 5 days post-AMI.
Free wall myocardial rupture accounts for 10% of AMI fatalities and occurs 1 to 5 days post-AMI.
![]() Interventricular wall rupture is signified by pain, shortness of breath (SOB), and a holosystolic murmur.
Interventricular wall rupture is signified by pain, shortness of breath (SOB), and a holosystolic murmur.
![]() Papillary muscle rupture occurs in 1% of all MIs, typically a day into the event and up to 14 days later.
Papillary muscle rupture occurs in 1% of all MIs, typically a day into the event and up to 14 days later.
![]() Pericarditis is seen in up to 20% of all MIs 2 to 4 days after the event. Dressler’s syndrome occurs 2 to 10 weeks post-AMI.
Pericarditis is seen in up to 20% of all MIs 2 to 4 days after the event. Dressler’s syndrome occurs 2 to 10 weeks post-AMI.
![]() Thirty percent of inferior AMIs involve the right ventricle, and are associated with increased mortality and complications.
Thirty percent of inferior AMIs involve the right ventricle, and are associated with increased mortality and complications.
DIAGNOSIS AND DIFFERENTIAL
![]() The diagnosis of ST-segment elevation myocardial infarction (STEMI) is made when criteria on the ECG are met in the setting of symptoms suggestive of MI: ≥ than 1 mm ST elevation in two or more contiguous leads.
The diagnosis of ST-segment elevation myocardial infarction (STEMI) is made when criteria on the ECG are met in the setting of symptoms suggestive of MI: ≥ than 1 mm ST elevation in two or more contiguous leads.
![]() The diagnosis of non-ST-segment elevation myocardial infarction (NSTEMI) depends on abnormal elevation of biologic markers but may include ECG changes not meeting criteria for STEMI.
The diagnosis of non-ST-segment elevation myocardial infarction (NSTEMI) depends on abnormal elevation of biologic markers but may include ECG changes not meeting criteria for STEMI.
![]() The diagnosis of unstable angina is clinical but may include more subtle changes in the ECG and non-diagnostic changes in biomarker levels.
The diagnosis of unstable angina is clinical but may include more subtle changes in the ECG and non-diagnostic changes in biomarker levels.
![]() The history is usually suggestive, but the electrocardiogram (ECG) is the best single test available in the ED.
The history is usually suggestive, but the electrocardiogram (ECG) is the best single test available in the ED.
![]() Patients with more significant ECG abnormalities are more likely to have AMI, unstable angina, and serious cardiovascular complications.
Patients with more significant ECG abnormalities are more likely to have AMI, unstable angina, and serious cardiovascular complications.
![]() Patients with normal or nonspecific ECGs still have a 1% to 5% incidence of AMI and a 4% to 23% incidence of unstable angina. Patients with non-diagnostic ECGs or evidence of ischemia that is age-indeterminate have a 4% to 7% incidence of AMI and a 21% to 48%o incidence of unstable angina.
Patients with normal or nonspecific ECGs still have a 1% to 5% incidence of AMI and a 4% to 23% incidence of unstable angina. Patients with non-diagnostic ECGs or evidence of ischemia that is age-indeterminate have a 4% to 7% incidence of AMI and a 21% to 48%o incidence of unstable angina.
![]() Demonstration of new ischemia in ECG increases the risk of AMI from 25% to 73% and the unstable angina risk from 14% to 43%.
Demonstration of new ischemia in ECG increases the risk of AMI from 25% to 73% and the unstable angina risk from 14% to 43%.
![]() Findings on electrocardiogram predict culprit artery. Figure 20–1 is an ECG that shows an inferiorlateral MI from occlusion of the left circumflex artery.
Findings on electrocardiogram predict culprit artery. Figure 20–1 is an ECG that shows an inferiorlateral MI from occlusion of the left circumflex artery.
![]() Figure 20–2 is an ECG that shows an inferior MI from right coronary artery occlusion.
Figure 20–2 is an ECG that shows an inferior MI from right coronary artery occlusion.
![]() Inferior wall AMIs should have a right-sided lead V4 (V4R) obtained, because ST-segment elevation in V4R is highly suggestive of right ventricular (RV) infarction.
Inferior wall AMIs should have a right-sided lead V4 (V4R) obtained, because ST-segment elevation in V4R is highly suggestive of right ventricular (RV) infarction.
![]() In Fig. 20-3, a right ventricular infarction is shown; Fig. 20-3A shows the standard leads with ST elevation in the lead Vr Right ventricular leads are placed on the same patient, and leads V3R, V4R, V5R, V6R show ST elevation compatible with right-sided ventricular infarction.
In Fig. 20-3, a right ventricular infarction is shown; Fig. 20-3A shows the standard leads with ST elevation in the lead Vr Right ventricular leads are placed on the same patient, and leads V3R, V4R, V5R, V6R show ST elevation compatible with right-sided ventricular infarction.
![]() Figure 20–4 ECG shows anterior myocardial infarction from distal left anterior descending coronary artery occlusion.
Figure 20–4 ECG shows anterior myocardial infarction from distal left anterior descending coronary artery occlusion.
![]() Figure 20–5 shows an ECG demonstrating anterior myocardial infarction due to occlusion of the left anterior descending coronary artery.
Figure 20–5 shows an ECG demonstrating anterior myocardial infarction due to occlusion of the left anterior descending coronary artery.
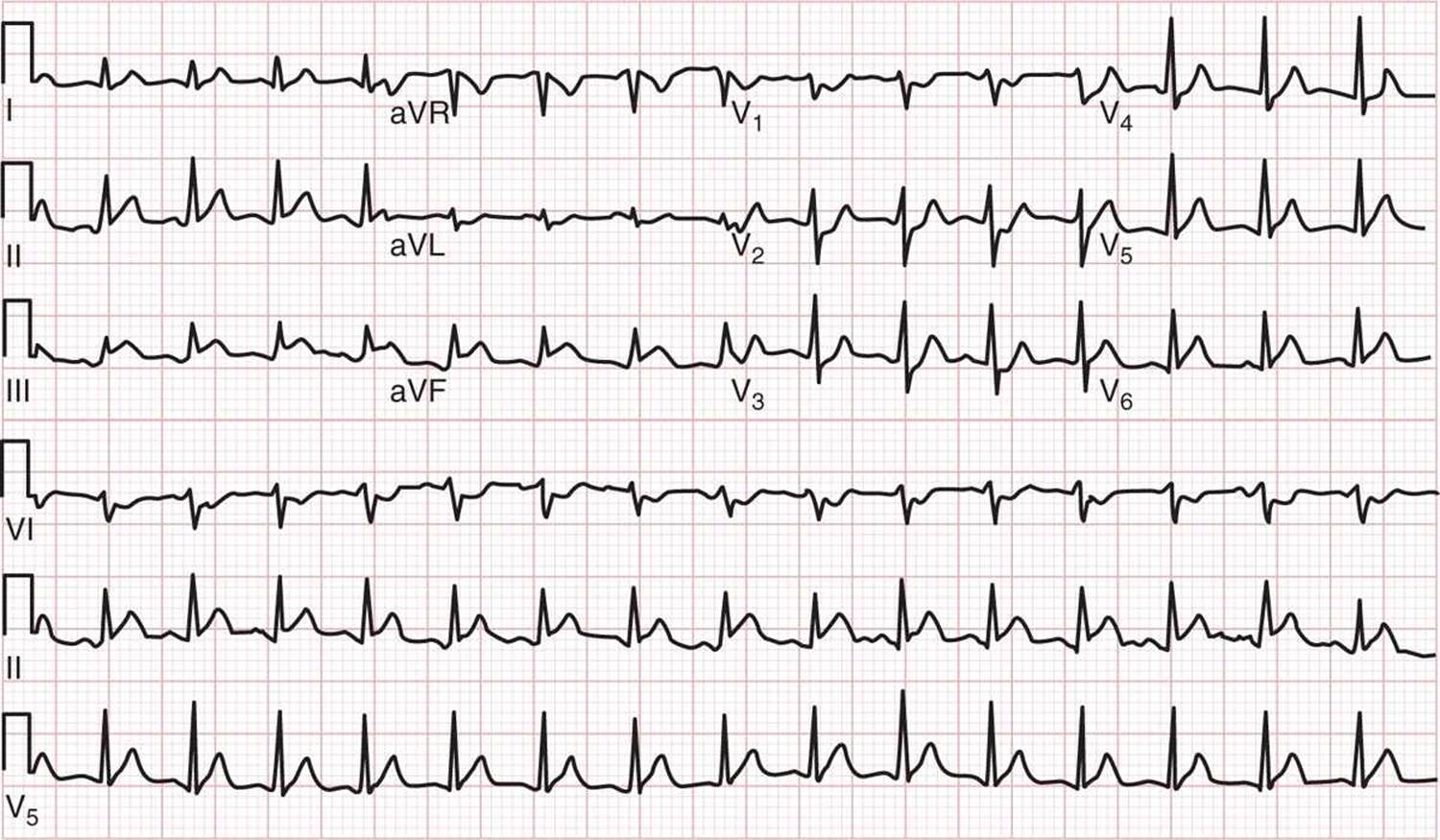
FIG. 20–1. ECG showing inferiorlateral myocardial infarction from left circumflex coronary artery occlusion. (ECG from a 42-year-old man presenting with chest pain. ECG shows ST-segment elevation in limb leads II, III [inferior], and aVF, as well as lead V6 [lateral]. ST-segment depression is evident in leads Vp V2, and V3, reflecting reciprocal changes in the anterior leads. The patient was found to have 100% occlusion of the left circumflex coronary artery at cardiac catheterization. (Courtesy of David M. Cline, MD, Wake Forest Baptist Health.)
![]() In the setting of AMI, the ECG can range from normal (up to 5%) to distinct ST-segment elevation (Table 20-1).
In the setting of AMI, the ECG can range from normal (up to 5%) to distinct ST-segment elevation (Table 20-1).
![]() Although the cardiac troponins are useful for both diagnosis and risk stratification of patients with chest pain, ACS, and AMI, cardiac marker testing in the ED will not identify most ED patients who subsequently develop adverse events.
Although the cardiac troponins are useful for both diagnosis and risk stratification of patients with chest pain, ACS, and AMI, cardiac marker testing in the ED will not identify most ED patients who subsequently develop adverse events.
![]() Chest radiography may be useful in determining other causes of ischemic-like pain.
Chest radiography may be useful in determining other causes of ischemic-like pain.
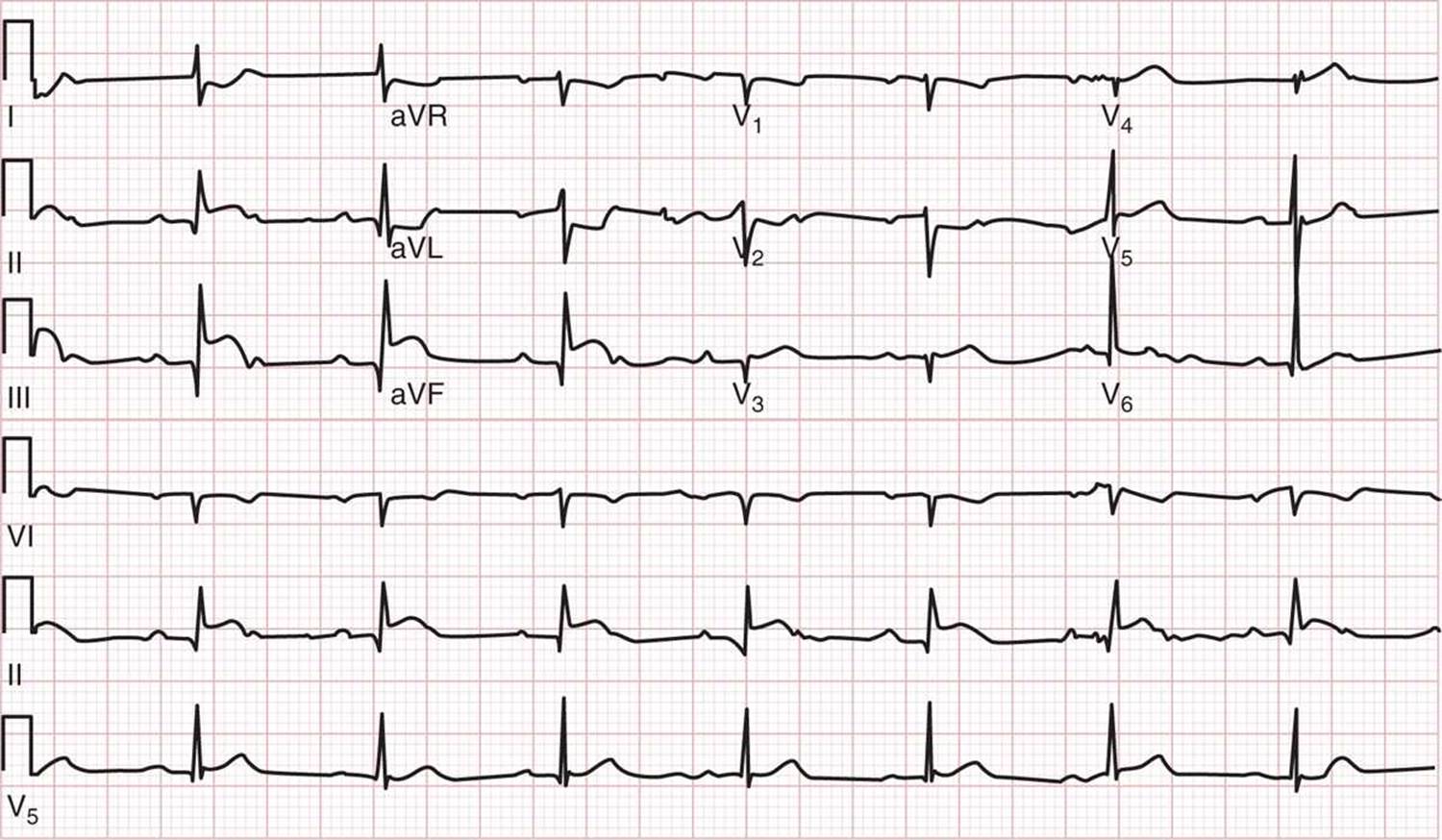
FIG. 20-2. ECG showing inferior myocardial infarction from right coronary artery occlusion. (ECG from an 80-year-old man presenting with acute chest pain. The ECG shows ST-segment elevation in lead III greater than in lead II plus ST-segment depression of >1 mm in lead I and lead aVL. The patient was found to have 100% occlusion of the right coronary artery at cardiac catheterization.) (Courtesy of David M. Cline, MD, Wake Forest Baptist Health.)
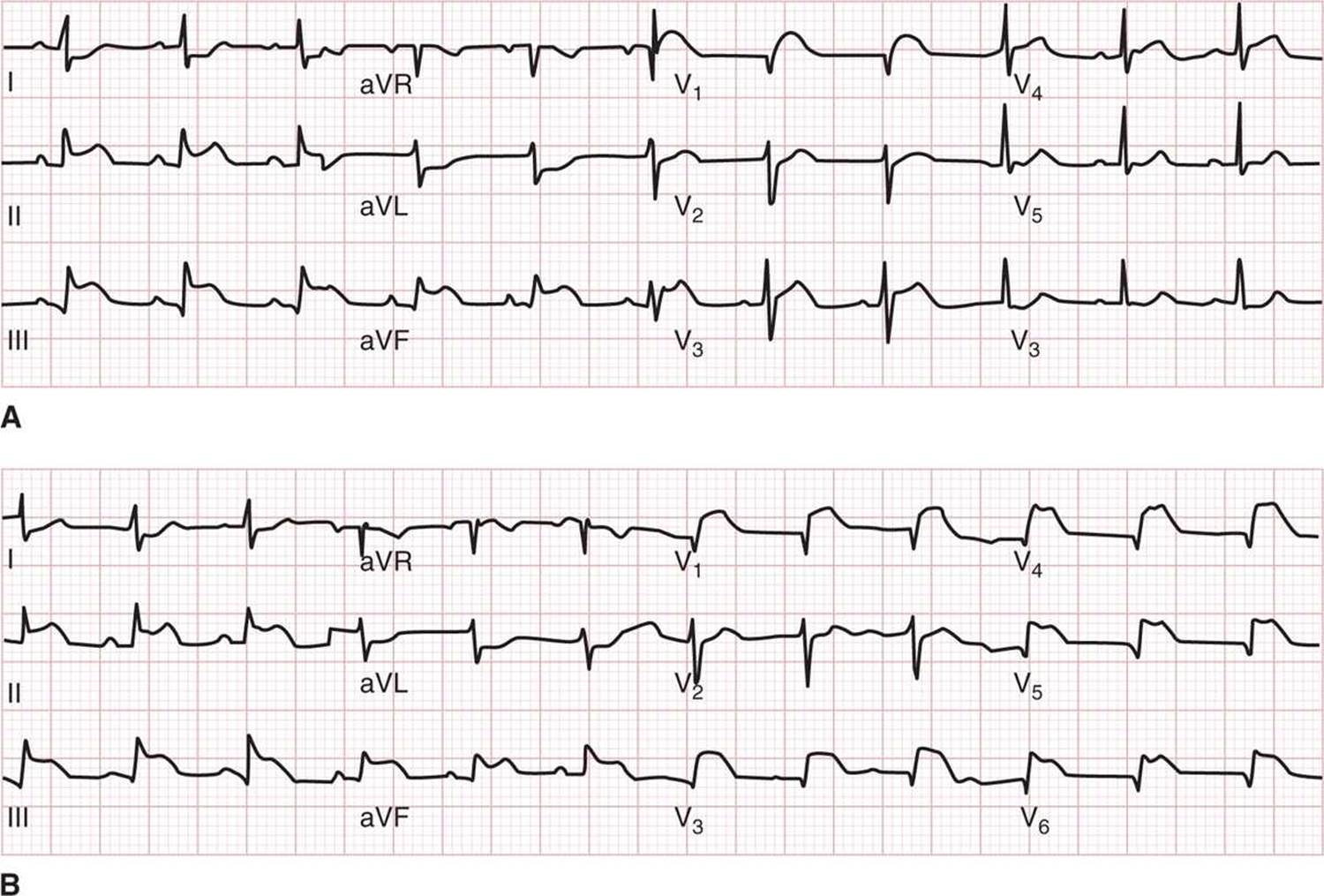
FIG. 20-3. Right ventricular infarction. A. Inferior wall myocardial infarction with ST elevation in lead Vr. ECG showing inferior ST-segment elevation myocardial infarction, also with ST-segment elevation in lead V1 suggestive of right ventricular infarction. B. Inferior wall myocardial infarction with right ventricular leads. Same patient with placement of right ventricular leads, showing ST-segment elevation in V3R, V4R, V5R, and V6R compatible with right ventricular infarction. (Courtesy of J. Stephan Stapczynski, Maricopa Medical Center.)
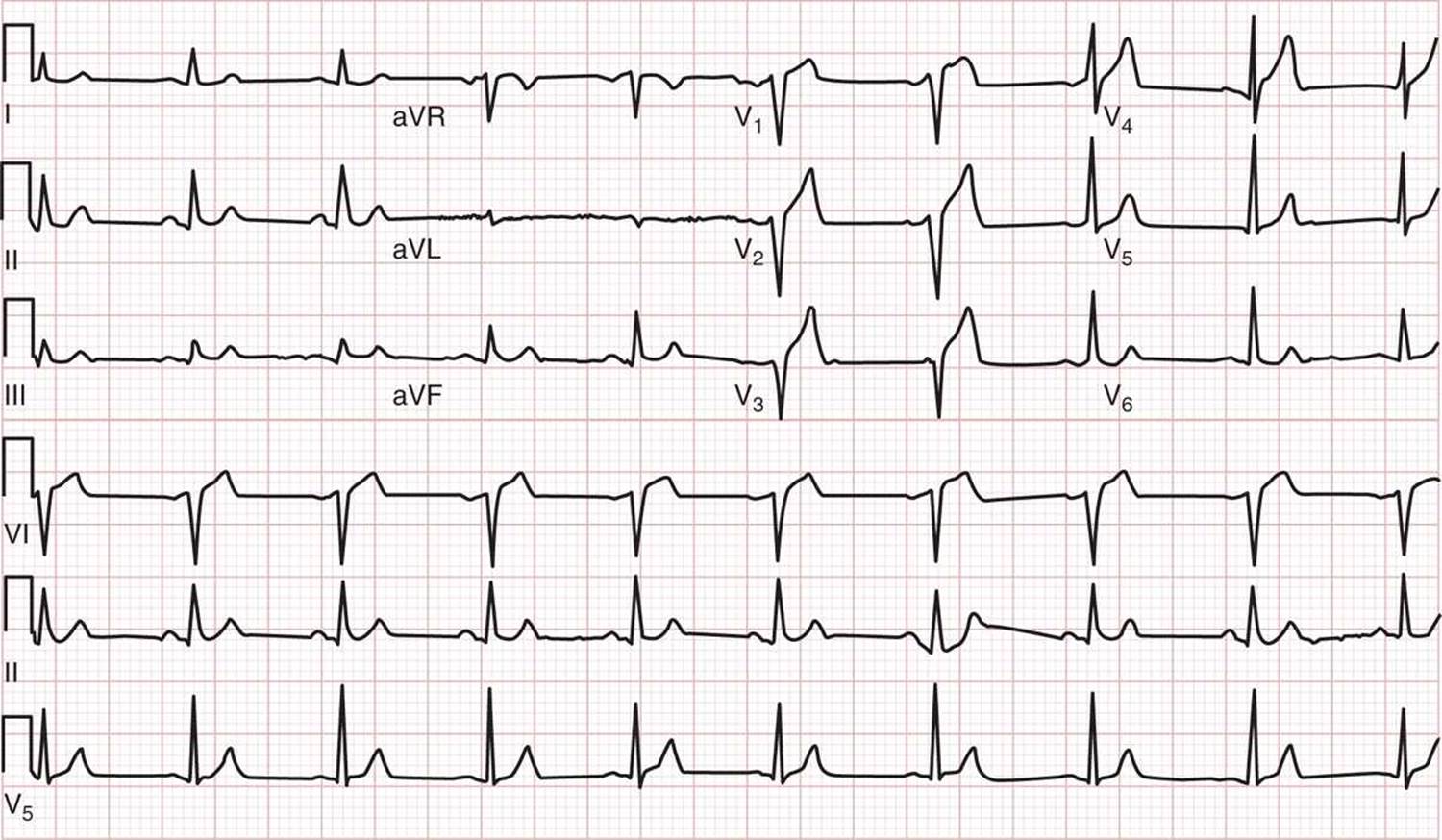
FIG. 20-4. ECG showing anterior myocardial infarction from distal left anterior descending coronary artery occlusion. (ECG from a 52-year-old man presenting with chest pain. ECG shows ST-segment elevation in Vp V2, and V3, with the absence of ST-segment epres-sion in leads II, III, and aVF. The patient was found to have 100% occlusion of the distal left anterior descending coronary artery at cardiac catheterization. (Courtesy of David M. Cline, MD, Wake Forest Baptist Health.)
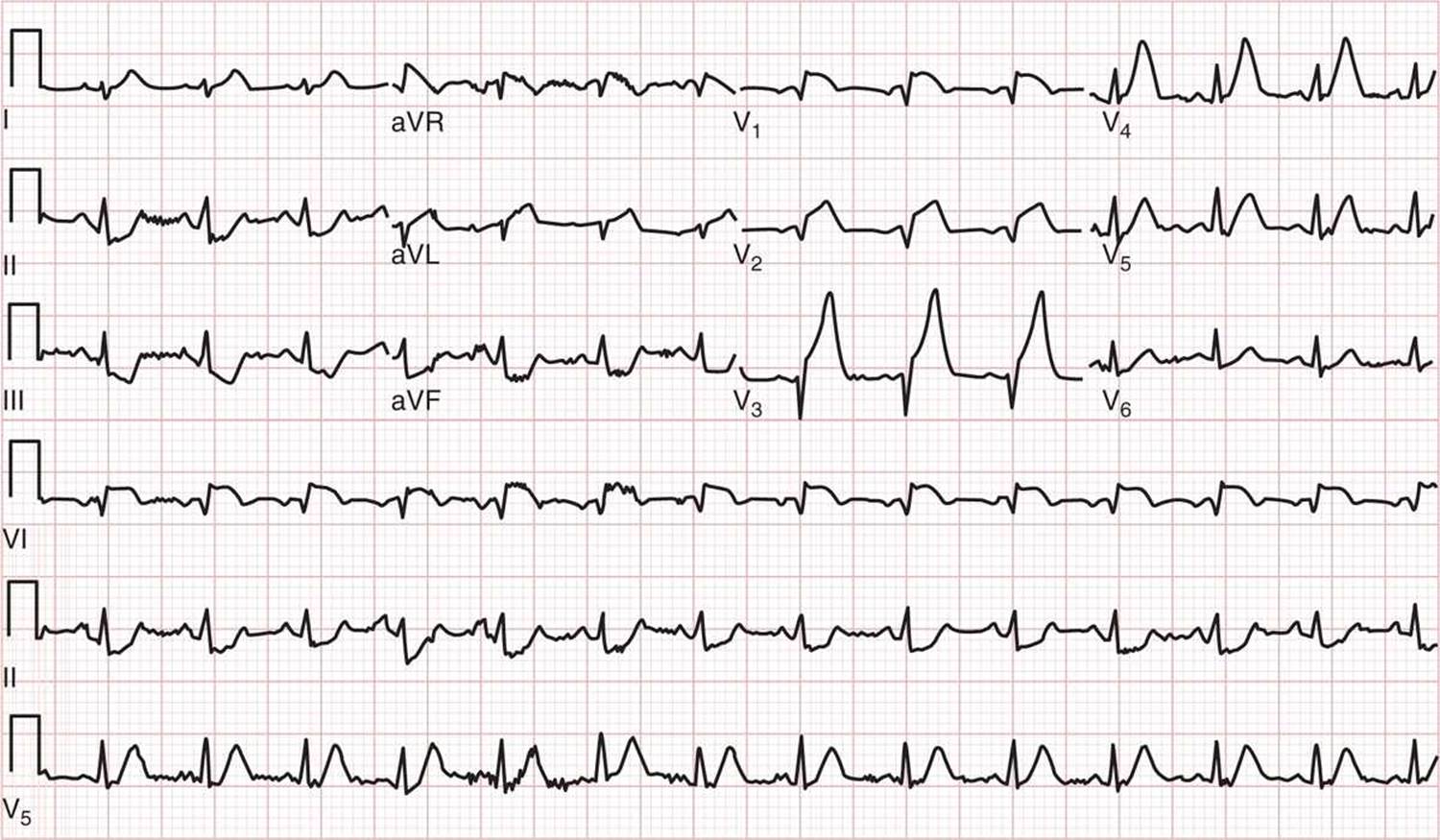
FIG. 20-5. ECG showing anterior myocardial infarction from proximal left anterior descending coronary artery occlusion. (ECG from a 65-year-old man presenting with chest pain. ECG shows ST-segment elevation in V1, V2, and V3, and >1 mm of ST-segment depression in leads II, III, and aVF. The patient was found to have 100% occlusion of the proximal left anterior descending coronary artery at cardiac catheterization. (Courtesy of David M. Cline, MD, Wake Forest Baptist Health.)
![]() The differential diagnosis of cardiac ischemia is particularly broad (see Chapter 19). Entities that should be considered include pericarditis, cardiomyopathies, cardiac valvular disease, pulmonary embolism, pneumonia, pneumothorax, asthma or chronic obstructive pulmonary disease, gastrointestinal disorders (especially esophageal disease), chest trauma, chest wall disorders, hyperventilation, aortic aneurysm, and dissection.
The differential diagnosis of cardiac ischemia is particularly broad (see Chapter 19). Entities that should be considered include pericarditis, cardiomyopathies, cardiac valvular disease, pulmonary embolism, pneumonia, pneumothorax, asthma or chronic obstructive pulmonary disease, gastrointestinal disorders (especially esophageal disease), chest trauma, chest wall disorders, hyperventilation, aortic aneurysm, and dissection.
TABLE 20-1 Electrocardiograph«: Q-Wave-Based Criteria for AMI

EMERGENCY DEPARTMENT CARE AND DISPOSITION
![]() The primary goal of initial treatment is early reperfusion, achieved by the preferred modality, percutaneous coronary intervention (PCI), or fibrinolytics.
The primary goal of initial treatment is early reperfusion, achieved by the preferred modality, percutaneous coronary intervention (PCI), or fibrinolytics.
![]() Institutional goals for reperfusion are PCI within 90 minutes of ED arrival, or fibrinolysis within 30 minutes of ED arrival. Patients ineligible for fibri-nolytic therapy should be considered for transfer to a PCI facility, even if arrival at the cardiac center will be delayed beyond the target time frame.
Institutional goals for reperfusion are PCI within 90 minutes of ED arrival, or fibrinolysis within 30 minutes of ED arrival. Patients ineligible for fibri-nolytic therapy should be considered for transfer to a PCI facility, even if arrival at the cardiac center will be delayed beyond the target time frame.
![]() Measures given to maintain coronary artery patency through anticoagulation and antiplatelet activity are discussed below. Table 20-2 lists agents and doses.
Measures given to maintain coronary artery patency through anticoagulation and antiplatelet activity are discussed below. Table 20-2 lists agents and doses.
![]() Dysrhythmias should be treated if their effect on heart rate exacerbates oxygen supply-demand imbalance, or if the dysrhythmia seems capable of electrical deterioration (eg, to a nonperfusing rhythm; see Chapter 4). The process of risk stratification using tools such as the Thrombolysis in Myocardial Infarction (TIMI) Risk Score should be rapidly undertaken.
Dysrhythmias should be treated if their effect on heart rate exacerbates oxygen supply-demand imbalance, or if the dysrhythmia seems capable of electrical deterioration (eg, to a nonperfusing rhythm; see Chapter 4). The process of risk stratification using tools such as the Thrombolysis in Myocardial Infarction (TIMI) Risk Score should be rapidly undertaken.
![]() Immediate management includes IV access, O2, cardiac monitoring, and obtaining an ECG.
Immediate management includes IV access, O2, cardiac monitoring, and obtaining an ECG.
![]() Patients meeting criteria for STEMI should receive the following treatments: aspirin, clopidogrel, nitroglycerin, oral beta blocker, an antithrombin, and PCI if available or fibrinolysis if PCI is not available within a reasonable time frame (within 90 minutes is the goal, times in practice are longer on average, especially for transferred patients). Glycoprotein IIb/IIIa inhibitors can be delayed until arrival in the catheterization laboratory.
Patients meeting criteria for STEMI should receive the following treatments: aspirin, clopidogrel, nitroglycerin, oral beta blocker, an antithrombin, and PCI if available or fibrinolysis if PCI is not available within a reasonable time frame (within 90 minutes is the goal, times in practice are longer on average, especially for transferred patients). Glycoprotein IIb/IIIa inhibitors can be delayed until arrival in the catheterization laboratory.
TABLE 20-2 Recommended Doses* of Drugs Used in the Emergency Treatment of Acute Coronary Syndromes

![]() Patients who do not meet criteria for STEMI who have positive markers, or meet otherwise high-risk criteria should be given aspirin, clopidogrel, nitroglycerin, oral beta-blockers, an antithrombin, and should be considered for early intervention with PCI.
Patients who do not meet criteria for STEMI who have positive markers, or meet otherwise high-risk criteria should be given aspirin, clopidogrel, nitroglycerin, oral beta-blockers, an antithrombin, and should be considered for early intervention with PCI.
![]() Aspirin should be administered in a dose of 160 to 325 milligrams (chewed) in patients with suspected ACS, unless contraindicated or already taken by the patient.
Aspirin should be administered in a dose of 160 to 325 milligrams (chewed) in patients with suspected ACS, unless contraindicated or already taken by the patient.
![]() Oral and transdermal nitroglycerin (NTG) are useful in treating angina. A sublingual dose should be repeated twice, for a total of three tablets, administered at 2- to 5-min intervals. If there is no improvement with sublingual NTG, intravenous NTG should be started at 5 to 10 micrograms/min.
Oral and transdermal nitroglycerin (NTG) are useful in treating angina. A sublingual dose should be repeated twice, for a total of three tablets, administered at 2- to 5-min intervals. If there is no improvement with sublingual NTG, intravenous NTG should be started at 5 to 10 micrograms/min.
![]() IV NTG is recommended for MI or recurrent ischemia. The dose should be adjusted by 5 to 10 micrograms/min increments every 3 to 5 minutes, titrated to pain level and blood pressure reduction up to 200 micrograms/min maximum. NTG should be used cautiously in the setting of borderline-low blood pressure, as hypotension may worsen ischemia.
IV NTG is recommended for MI or recurrent ischemia. The dose should be adjusted by 5 to 10 micrograms/min increments every 3 to 5 minutes, titrated to pain level and blood pressure reduction up to 200 micrograms/min maximum. NTG should be used cautiously in the setting of borderline-low blood pressure, as hypotension may worsen ischemia.
![]() NTG is contraindicated in the setting of RV infarction, due to risk of hypotension related to loss of preload. A common side effect of NTG is headache.
NTG is contraindicated in the setting of RV infarction, due to risk of hypotension related to loss of preload. A common side effect of NTG is headache.
![]() Morphine sulfate can be used if there is uncontrolled ischemic chest discomfort despite NTG. Morphine may decrease cardiac output, and should be used with caution in the presence of hypotension, and in patients with inferior MI.
Morphine sulfate can be used if there is uncontrolled ischemic chest discomfort despite NTG. Morphine may decrease cardiac output, and should be used with caution in the presence of hypotension, and in patients with inferior MI.
![]() The thienopyridine clopidogrel, when added to ASA, reduces the composite risk of cardiovascular death, MI, or stoke. Thienopyridines bind to the ADP receptor and inhibit platelet aggregation.
The thienopyridine clopidogrel, when added to ASA, reduces the composite risk of cardiovascular death, MI, or stoke. Thienopyridines bind to the ADP receptor and inhibit platelet aggregation.
![]() Clopidogrel should be considered in addition to standard care (ASA, anticoagulants) for patients with moderate-to high-risk NSTEMI and STEMI, and in patients in whom PCI is planned.
Clopidogrel should be considered in addition to standard care (ASA, anticoagulants) for patients with moderate-to high-risk NSTEMI and STEMI, and in patients in whom PCI is planned.
![]() Clopidogrel is used without aspirin in patients allergic to aspirin. Clopidogrel increases risk of bleeding, and should be withheld at least 5 days before coronary artery bypass grafting (CABG).
Clopidogrel is used without aspirin in patients allergic to aspirin. Clopidogrel increases risk of bleeding, and should be withheld at least 5 days before coronary artery bypass grafting (CABG).
![]() Prasugrel is an oral thienopyridine prodrug and should not be used in patients with prior stroke or age > 74 years.
Prasugrel is an oral thienopyridine prodrug and should not be used in patients with prior stroke or age > 74 years.
![]() Unfractionated heparin (UFH) is used for its anticoagulant properties. UFH has several disadvantages, including: (1) the need for IV administration, (2) the requirement for frequent monitoring of the activated partial thromboplastin time (aPTT) (3) an unpredictable anticoagulant response in individual patients, (4) heparin-induced thrombocytopenia (HIT), and (5) increased risk of bleeding.
Unfractionated heparin (UFH) is used for its anticoagulant properties. UFH has several disadvantages, including: (1) the need for IV administration, (2) the requirement for frequent monitoring of the activated partial thromboplastin time (aPTT) (3) an unpredictable anticoagulant response in individual patients, (4) heparin-induced thrombocytopenia (HIT), and (5) increased risk of bleeding.
![]() Anticoagulation due to UFH can be reversed with protamine. The dosage is 1 milligram of protamine per 100 U of UFH infused in the previous 4 hours.
Anticoagulation due to UFH can be reversed with protamine. The dosage is 1 milligram of protamine per 100 U of UFH infused in the previous 4 hours.
![]() As compared to UFH, low molecular weight heparins (LMWH) offer greater bioavailability, lower protein binding, longer half-life, improved safety, and more reliable anticoagulant effect.
As compared to UFH, low molecular weight heparins (LMWH) offer greater bioavailability, lower protein binding, longer half-life, improved safety, and more reliable anticoagulant effect.
![]() LMWH are administered in fixed subcutaneous doses and do not require laboratory monitoring. As compared to UFH, LMWH administration for ACS is associated with decreased ischemia and MI although there is an increase in minor bleeding complications.
LMWH are administered in fixed subcutaneous doses and do not require laboratory monitoring. As compared to UFH, LMWH administration for ACS is associated with decreased ischemia and MI although there is an increase in minor bleeding complications.
![]() For patients with UA/NSTEMI, enoxaparin (a LMWH) or UFH are both reasonable choices for patients undergoing PCI revascularization. Improved outcomes are demonstrated with consistent therapy (use of a single antithrombin from the ED through the catheterization laboratory) and increased bleeding is seen when patients are switched from one antithrombin to another.
For patients with UA/NSTEMI, enoxaparin (a LMWH) or UFH are both reasonable choices for patients undergoing PCI revascularization. Improved outcomes are demonstrated with consistent therapy (use of a single antithrombin from the ED through the catheterization laboratory) and increased bleeding is seen when patients are switched from one antithrombin to another.
![]() In patients in whom CABG is planned, LMWH should be avoided (due to its half-life) in favor of UFH. In patients > 75 years of age, enoxaparin must be used with caution due to an increased risk of ICH. Enoxaparin dosing adjustments are recommended in patients with impaired renal function (creatinine clearance < 30 mL/min).
In patients in whom CABG is planned, LMWH should be avoided (due to its half-life) in favor of UFH. In patients > 75 years of age, enoxaparin must be used with caution due to an increased risk of ICH. Enoxaparin dosing adjustments are recommended in patients with impaired renal function (creatinine clearance < 30 mL/min).
![]() Factor Xa Inhibitors such as fondaparinux, a synthetic pentasaccharide, have similar efficacy to UFH in patients with UA/NSTEMI; bleeding risk is lower than that with enoxaparin.
Factor Xa Inhibitors such as fondaparinux, a synthetic pentasaccharide, have similar efficacy to UFH in patients with UA/NSTEMI; bleeding risk is lower than that with enoxaparin.
![]() Current ACC/AHA guidelines consider fondaparinux an option as an antithrombin. In STEMI patients lacking renal impairment, fondaparinux may be considered for those patients treated with thrombolytics that are not fibrin-specific (ie, streptokinase).
Current ACC/AHA guidelines consider fondaparinux an option as an antithrombin. In STEMI patients lacking renal impairment, fondaparinux may be considered for those patients treated with thrombolytics that are not fibrin-specific (ie, streptokinase).
![]() Direct thrombin inhibitors bind directly to thrombin in clot and are resistant to agents that degrade heparin. Comparison of bivalirudin with UFH found no outcomes benefit in NSTEMI patients, but less bleeding occurred and no dosage adjustment is required in renal impairment. For patients with STEMI, bivalirudin may be considered as an alternative to UFH and GP IIb/IIIa inhibitors.
Direct thrombin inhibitors bind directly to thrombin in clot and are resistant to agents that degrade heparin. Comparison of bivalirudin with UFH found no outcomes benefit in NSTEMI patients, but less bleeding occurred and no dosage adjustment is required in renal impairment. For patients with STEMI, bivalirudin may be considered as an alternative to UFH and GP IIb/IIIa inhibitors.
![]() Percutaneous Coronary Intervention (PCI), coronary angioplasty with or without stent placement, is the treatment of choice for the management of STEMI when PCI can be performed within 90 minutes of initial ED presentation.
Percutaneous Coronary Intervention (PCI), coronary angioplasty with or without stent placement, is the treatment of choice for the management of STEMI when PCI can be performed within 90 minutes of initial ED presentation.
![]() PCI may be offered to patients presenting to a non-PCI facility when prompt transfer can result in acceptable door-to-balloon times. Early invasive therapy (PCI) within 48 hours is recommended in high-risk patients with UA/STEMI, in patients with recurrent angina/ischemia, and in those who have elevated troponin, new or presumably new ST-segment depression, or high-risk findings on stress testing.
PCI may be offered to patients presenting to a non-PCI facility when prompt transfer can result in acceptable door-to-balloon times. Early invasive therapy (PCI) within 48 hours is recommended in high-risk patients with UA/STEMI, in patients with recurrent angina/ischemia, and in those who have elevated troponin, new or presumably new ST-segment depression, or high-risk findings on stress testing.
![]() PCI is also more likely to be beneficial in the setting of depressed LV function, hemodynamic instability, sustained ventricular tachycardia, PCI within the previous 6 months, or prior CABG.
PCI is also more likely to be beneficial in the setting of depressed LV function, hemodynamic instability, sustained ventricular tachycardia, PCI within the previous 6 months, or prior CABG.
![]() In treatment settings without timely access to PCI, fibrinolytics are indicated for patients with STEMI if time to treatment is <6 to 12 hours from symptom onset, and the ECG has at least 1-mm ST-segment elevation in two or more contiguous leads.
In treatment settings without timely access to PCI, fibrinolytics are indicated for patients with STEMI if time to treatment is <6 to 12 hours from symptom onset, and the ECG has at least 1-mm ST-segment elevation in two or more contiguous leads.
![]() The dosages of individual fibrinolytic agents are listed in Table 20-2. STEMI patients who have received fibrinolytics should receive full-dose anticoagulants, started in the ED and maintained for a minimum of 48 hours.
The dosages of individual fibrinolytic agents are listed in Table 20-2. STEMI patients who have received fibrinolytics should receive full-dose anticoagulants, started in the ED and maintained for a minimum of 48 hours.
![]() Similar efficacy and safety profiles have been demonstrated for tissue plasminogen activator (tPA), reteplase (rPA), and tenecteplase (TNK). Contraindications for fibrinolytics are listed in Table 20-3. Before administering thrombolytics, informed consent should be obtained (with particular attention paid to an understanding of the risks). Arterial puncture should be avoided, as should veni-puncture or central line placement in areas which are not readily compressible.
Similar efficacy and safety profiles have been demonstrated for tissue plasminogen activator (tPA), reteplase (rPA), and tenecteplase (TNK). Contraindications for fibrinolytics are listed in Table 20-3. Before administering thrombolytics, informed consent should be obtained (with particular attention paid to an understanding of the risks). Arterial puncture should be avoided, as should veni-puncture or central line placement in areas which are not readily compressible.
(a) Tissue plasminogen activator (tPA) is a naturally occurring human protein and is not antigenic. This is fibrin-specific and has a half-life of 5 minutes. When compared with traditional dosing, front-loaded tPA has been shown to have superior 90-minutes patency rates and reocclu-sion rates, with no increase in bleeding risk.
(b) Reteplase (rPA) is a non-fibrin-specific deletion mutant of tPA with a prolonged half-life of 18 minutes (as compared to tPA’s half-life of 3 minutes). Reteplase may have a faster time to perfusion. The main advantage of reteplase is that it is given as a (double) bolus rather than infusion.
TABLE 20-3 Contraindications to Fibrinolytic Therapy in ST-segment Elevation Myocardial Infarction
Absolute
Any prior intracranial hemorrhage
Known structural cerebral vascular lesion (e.g., AVM)
Known malignant intracranial neoplasm (primary or metastatic)
ischemic stroke within 3 months EXCEPT acute stroke within 3 hours
Suspected aortic dissection
Active bleeding or bleeding diathesis (excluding menses)
Significant closed head trauma or facial trauma within 3 months
Relative
History of chronic, severe, poorly controlled hypertension
Severe uncontrolled hypertension on presentation
(SBP >180 mm Hg or DBP >110 mm Hg
History of prior ischemic stroke > 3 months, dementia, or known intracranial pathology not covered in contraindications
Traumatic or prolonged (>10 minutes) CPR or major surgery (< 3 weeks)
Recent (within 2 to 4 weeks) internal bleeding
Non-compressible vascular punctures
Current use of anticoagulants: the higher the INR, the higher the risk of bleeding
Pregnancy
Active peptic ulcer
Streptokinase/anistreplase should not be given a second time
(c) Tenecteplase (TNK) is a fibrin-specific substitution mutant of tPA that is given as a single weight-based bolus.
(d) Streptokinase (SK) activates circulating plas-minogen, is not fibrin-specific, and is capable of generating an allergic reaction (minor: 5–5.7%, anaphylaxis: < 0.2–2.0%). Hypotension occurs in up to 15% of patients and is usually responsive to fluids and slowing of SK infusion. Contraindications to SK include hypotension, prior SK administration (within 6 months), and streptococcal infection within a year. SK’s half-life is 23 minutes, but systemic fibrinolysis persists for 24 hours. Heparin must be given within 4 hours of starting SK.
(e) The most significant complication of thrombolyt-ics is hemorrhage, particularly ICH. Significant bleeding, especially internal, requires cessation of thrombolytics, heparin, and aspirin. Crystalloid and red blood cell infusion may be necessary. Cryoprecipitate (cryo) and fresh frozen plasma (FFP) may be used in an attempt to reverse fibrinolysis due to thrombolytics. Initially, 10 U of cryo are given, and fibrinogen levels are obtained. If the fibrinogen level is < 1 gram/L, the dose of cryo should be repeated. If bleeding continues despite a fibrinogen > 1 gram/L, or if the fibrinogen level is < 1 gram/L after 20 U of cryo, then 2 U of FFP should be administered. If this does not control hemorrhage, then platelets or antifibrinolytic agents (aminocaproic acid or tranexamic acid) are indicated.
![]() Recent evidence shows no particular benefit to the early IV administration of β-blockers on cardiac rhythm, infarct size, reinfarction, or mortality. Oral β-blocker therapy does not need to be initiated in the ED unless there is a specific indication (eg, tachycardia), but β-blockers may be initiated within the first 24 hours of hospitalization for patients lacking contraindications, alternatives include metoprolol and atenolol among others.
Recent evidence shows no particular benefit to the early IV administration of β-blockers on cardiac rhythm, infarct size, reinfarction, or mortality. Oral β-blocker therapy does not need to be initiated in the ED unless there is a specific indication (eg, tachycardia), but β-blockers may be initiated within the first 24 hours of hospitalization for patients lacking contraindications, alternatives include metoprolol and atenolol among others.
![]() Glycoprotein Ilb/IIIa (GP Ilb/IIIa) antagonists bind to platelets and inhibit their aggregation. Abciximab, eptifibatide, and tirofiban are currently available.
Glycoprotein Ilb/IIIa (GP Ilb/IIIa) antagonists bind to platelets and inhibit their aggregation. Abciximab, eptifibatide, and tirofiban are currently available.
![]() There is no current evidence supporting the routine use of GP Ilb/IIIa inhibitor therapy prior to angio-graphy in patients with STEMI, and the use of these agents upstream is uncertain. Use of GP Ilb/IIIa inhibitors should be guided by local interdisciplinary review of ongoing clinical trials, guidelines, and recommendations.
There is no current evidence supporting the routine use of GP Ilb/IIIa inhibitor therapy prior to angio-graphy in patients with STEMI, and the use of these agents upstream is uncertain. Use of GP Ilb/IIIa inhibitors should be guided by local interdisciplinary review of ongoing clinical trials, guidelines, and recommendations.
![]() AMI patients with continued hemodynamic instability and pain or those who have not reperfused after administration of thrombolytics are candidates for rescue angioplasty.
AMI patients with continued hemodynamic instability and pain or those who have not reperfused after administration of thrombolytics are candidates for rescue angioplasty.
![]() Emergent CABG may also be indicated for these patients. Patients in refractory cardiogenic shock should undergo emergent angioplasty. Intraaortic balloon pump or other LV-assisting devices may also be indicated for these patients.
Emergent CABG may also be indicated for these patients. Patients in refractory cardiogenic shock should undergo emergent angioplasty. Intraaortic balloon pump or other LV-assisting devices may also be indicated for these patients.
![]() Patients with AMI or UA who have ongoing chest pain, ECG changes, dysrhythmias, or hemodynamic compromise require cardiac intensive care. Patients with UA and resolved chest pain, normal or nonspecific ECG changes, and no complications should be admitted to a monitored bed. Certain patients, usually those with low risk, may undergo rule-out protocols in chest pain observation units (see Chapter 22).
Patients with AMI or UA who have ongoing chest pain, ECG changes, dysrhythmias, or hemodynamic compromise require cardiac intensive care. Patients with UA and resolved chest pain, normal or nonspecific ECG changes, and no complications should be admitted to a monitored bed. Certain patients, usually those with low risk, may undergo rule-out protocols in chest pain observation units (see Chapter 22).
For further reading in Tintinalli’s Emergency Medicine: A Comprehensive Study Guide, 7th ed., see Chapter 53, “Acute Coronary Syndromes: Unstable Angina, Myocardial Ischemia, and Infarction,” by Judd E. Hollander and Deborah B. Dierck.