A 90-year-old male in perfect condition and no major medical history presents with 3-week onset of dull back pain. The pain is nagging, but not shooting and with no tingling or electrical shock-like sensations. He denies any sensory symptoms or urinary retention. His wife tells us that he had noted some leg weakness and had frequent falls. When specifically asked, she mentions that he has had a 20-pound weight loss. His neurologic examination reveals he is able to walk, but he has proximal leg muscle weakness (MRC 4/5). No sensory level is found. Tendon reflexes are subclonus. There is percussion pain at the thoracic spine level. CT and MRI shows destructive lesions in the left superior pubic ramus and multiple other lesions in the spine. The largest lesion is at T7 involving the posterior body pedicle with epidural extension and spinal cord displacement (Figure 10.1). A PSA level is over 400 ng/ml. He has a fair amount of pain, but believes his leg weakness is not worsening. His wife is not so sure.
What do you do now?
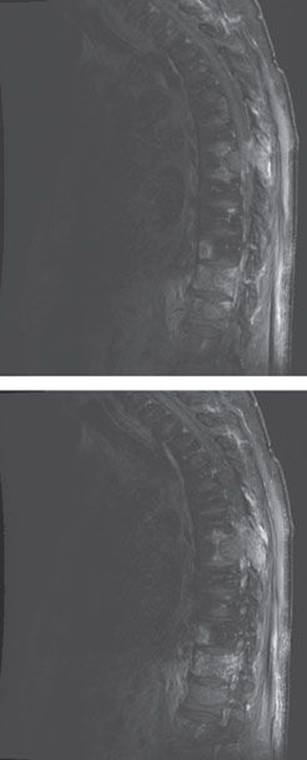
FIGURE 10.1 Large T7 lesion compressing the cord.
Cancer can take a significant and profound turn for the worse with spinal cord compression. Spinal cord compression may be a presenting symptom of metastatic disease, as it is in our patient example. Such a serious presentation requires a careful assessment of treatment options and there are a few.
Decisions to go ahead with surgical decompression are generally guided by the oncological prognosis—this is not as straightforward as it may seem—and the prediction of ambulation in a patient who has pretreatment motor function. What is mostly true is that a patient who can walk at the time of the intervention will continue to walk, and patients who have developed motor deficits slowly may have a better outcome than patients with more rapid onset. Life expectancy prediction in general has been difficult, but scoring systems have been devised. One example is the Tokuhashi score (Table 10.1). There are several elements that take life expectancy into account and in this scoring system, the primary site of the neoplasm and the neurologic deficit are the most important elements to predict survival.
In our patient the first priority is an assessment of the degree of metastatic epidural compression of the spinal cord. The potential instability of the spine is unquestionably severe.
There are several ways to approach this clinical problem medically and surgically (Table 10.2). Corticosteroids are administered immediately in all patients. There is little certainty whether a high-dose dexamethasone (96 mg per day) or moderate dose (16 mg per day) should be used. Most physicians would prefer an aggressive approach in patients who have had rapid progressive motor symptoms and use a high dose.
The next priority is to determine whether the patient is a surgical candidate. In patients with pain only, stable neurologic findings and a radiosensitive tumor, radiotherapy is the first option. Criteria that have been used to favor an operative management include rapid progression of motor deficit, instability of the spine on MRI scan, medically intractable pain, and incomplete neurologic injury. Some algorithms also include complete sensory and motor paraplegia over 24 hours as a reason to proceed with surgery. However, external-beam radiotherapy is used in most patients, and this requires a total of 30 GY in 10 fractions. Radiotherapy is not considered in radioresistant tumors, such as renal cell carcinoma. If the tumor is not radiosensitive, vertebroplasty or kyphoplasty with or without open stabilization surgery is an option.
TABLE 10.1 Tokuhashi Revised Scoring System for Preoperative Prognosis of Metastatic Spinal Tumors
|
Parameter |
Score |
|
General Condition |
|
|
Poor |
0 |
|
Moderate |
1 |
|
Good |
2 |
|
No. of extraspinal metastases |
|
|
>3 |
0 |
|
1-2 |
1 |
|
0 |
2 |
|
No. of vertebral body metastases |
|
|
>3 |
0 |
|
2 |
1 |
|
1 |
2 |
|
Metastases to the major internal organs |
|
|
Nonremovable |
0 |
|
Removable |
1 |
|
None |
2 |
|
Primary site of cancer |
|
|
Lung, stomach, bladder, bone |
0 |
|
Esophagus,pancreas, liver, gallbladder, unidentified |
1 |
|
Others |
2 |
|
Kidney, uterus |
3 |
|
Rectum |
4 |
|
Thyroid, breast, prostate, carcinoid |
5 |
|
Palsy or myelopathy |
|
|
Complete |
0 |
|
Incomplete |
1 |
|
None |
2 |
The information in this table is based on Tokuhashi et al. The lower the score, the worse the prognosis. Those patients scoring from 0 to 8 have a prognosis of less than 6 months to live; a score of 9–11, between 6 and 12 months; and a score of 12 to 15, more than a year.
Pain treatment is essential not only before but also during and after radiotherapy. There are many options for adequate pain management. A typical approach is using transdermal patches of fentanyl that can be changed every three to four days, oxycodone 10 to 20 mg every two hours as needed, or hydromorphone 4 mg every four hours. This generally provides good pain palliation. For patients with significant neuropathic pain—that may come later—gabapentin 100 mg twice a day and 300 mg at bedtime is a good starting dose; the medication should be then titrated to between 1,800 and 3,600 mg per day. Bone pain adjuvants are necessary as well, and this includes zoledronic acid 4 mg intravenously every three to four weeks or pamidronate 90 mg intravenously every three to four weeks. Most patients would need bladder catheterization and mostly can learn self-catheterization. Bowel regimen medication is also provided with bisacodyl or glycerin suppository daily, and docusate plus senna.
TABLE 10.2 Initial Treatment Options in Acute Spinal Cord Compression from Cancer
|
Assess surgical options |
|
Assess radiotherapy option |
|
Assess options to minimize effect of tumor |
So what were the decisions in our patient? His Tokuhashi revised score was 10, thus predicting 6 to 12 months of life expectancy. The patient underwent an orchidectomy that resulted in a good response based on his PSA. He was reluctant to undergo surgery and favored radiotherapy first. He was planned to undergo two to five radiation cycles, but unfortunately after three radiation therapies, he fairly rapidly became paraplegic. This resulted in immediate decompressive surgery, but the paraplegia persisted. Despite this motor deficit he remained relatively functional and lived for another 4 years.
This case illustrates well how difficult it is to choose radiotherapy over surgery. In this patient, the decision to postpone surgery seemed justified due to his advanced age and absence of any major neurologic deficit. The sudden appearance of paraplegia may have had a vascular cause, as ischemic myelopathy is not uncommon in these cases. It is uncertain whether this could have been prevented with earlier epidural decompression. Life expectancy prediction after epidural spine compression is difficult and remains a gross estimation. This is particularly the case in patients with metastatic prostate cancer when there is good response to castration, since they may have an extended period of time with reasonable quality of life.
KEY POINTS TO REMEMBER REGARDING OPTIONS IN ACUTE SPINAL CORD COMPRESSION
· Outcome after epidural cord compression is dependent on the type of tumor, radiosensitivity, rapidity of progression of neurologic symptoms, and instability of the spine.
· Aggressive surgical management is not indicated if there is pain only and no major neurologic deficit.
· Aggressive pain management with opioids and corticosteroids may provide adequate palliation.
· Which treatment modality is best for the patient is determined based on severity and acuity of the neurologic deficit, nature of the tumor, instability of the spine, and general performance status and life expectancy of the patient.
Further Reading
Abrahm JL, Banffy MB, Harris MB. Spinal cord compression in patients with advanced metastatic cancer. JAMA 2008; 299:937-946
Cole JS, Patchell RA. Metastatic epidural spinal cord compression. Lancet Neurol 2008; 7:459-466.
Graber JJ, Nolan CP. Myelopathies in patients with cancer. Arch Neurol 2010; 67:298-304.
Prasad D, Schiff D. Malignant spinal-cord compression. Lancet Oncol 2005; 6:15-24.
Quraishi NA, Gokaslan ZL, Boriani S. The surgical management of metastatic epidural compression of the spinal cord. J Bone Surg Br. 2010; 92-B:1054-1060.
Tokuhashi Y, Matsuaki H, Oda H et al. A revised scoring system for preoperative evaluation of metastatic spine tumor prognosis. Spine 2005; 30:2186-2191.