Introduction
It is easy to assume that all patients referred to a neurologist for the evaluation of limb pain have neurological problems such as radiculopathies, compression neuropathies, or plexopathies, and that more common musculoskeletal problems have been excluded prior to referral. In practice, this is far from true, and a neurologist, therefore, must have a thorough grasp of both the neurological and nonneurological causes of pain and the modalities available for their treatment. This chapter attempts to address these issues and is organized into three sections:
• Common neurological sources of limb pain and the musculoskeletal conditions that most commonly mimic them (Table 16.1).
• Treatment of nociceptive pain.
• Controversial localized pain syndromes.
Shoulder and proximal arm pain
C5 radiculopathy
C5 radiculopathy is characterized by neck pain and paresthesias that radiate into the shoulder and upper arm. On occasion, arm pain may be present without any neck pain. Weakness of the deltoid, biceps, and infraspinatus is usually less prominent than sensory dysfunction.
Brachial plexopathy
Only a small percentage of patients referred to a neurologist for the question of brachial plexopathy will actually have a plexus lesion. The three most important etiologies of brachial plexopathy are trauma, cancer, and idiopathic brachial neuritis, each of which has a distinctive presentation.
Trauma
Trauma may affect the brachial plexus at any level. The best-known traumatic plexopathy is Erb’s palsy, caused by damage to the upper trunk of the plexus, usually from downward pressure on the shoulder with forceful separation of the shoulder from the head. In addition to pain, there is weakness of the deltoid, biceps, supraspinatus, and infraspinatus. Sensation is decreased over the lateral upper arm and sometimes the lateral forearm. Electromyography (EMG) should be performed at least 3 weeks after suspected trauma to provide the most accurate information concerning localization and prognosis. In many cases, it is necessary to image the brachial plexus and cervical spine to differentiate traumatic brachial plexopathy from cervical polyradiculopathy.
Cancer
Neoplastic infiltration of the brachial plexus results in exquisite pain in the suprascapular and supraclavicular regions that radiates into the shoulder and arm, and persists or is worse at night. Weakness and wasting of the arm and hand muscles follows the pain by several weeks. The most common cancers that infiltrate the brachial plexus are breast and lung carcinomas. Unfortunately, patients with infiltrative brachial plexopathies have a poor prognosis, and usually do not survive long enough to make a neurological recovery. Local radiation and pain relief with narcotics and agents for neuropathic pain are used to palliate symptoms.
Radiation-induced brachial plexopathy develops months to years after local radiotherapy for breast or lung cancer. The challenge in diagnosing radiation-induced plexopathy is to differentiate it from direct infiltration by tumor recurrence. Clinically, radiation-induced plexopathy is less likely to be painful or to be associated with Horner’s syndrome. Electrophysiologically, radiation-induced plexopathy can be diagnosed by finding myokymia. MRI of the brachial plexus helps to differentiate between the two conditions: nodular enhancement and mass lesions are more frequent in patients with neoplastic infiltration.1Because radiation-induced plexopathy is secondary to
Table 16.1 Common neurological and musculoskeletal causes of limb pain

ischemic damage and fibrosis, it is frequently refractory to all treatments.
Idiopathic brachial neuritis
Idiopathic brachial neuritis (also known as brachial plexitis or Parsonage–Turner syndrome) has a fairly characteristic presentation, which includes intense local neck or shoulder pain followed several days to weeks later by arm weakness. Most patients report a viral prodrome or vaccination several weeks before the plexopathy develops. The mainstay of treatment of idiopathic brachial neuritis is pain control with agents for neuropathic symptoms. In some cases, the pain is so severe that narcotics are required. Although idiopathic brachial neuritis is a self-limited condition, it usually improves only after several months from onset.
Rotator cuff tendonitis
Inflammation of the tendons of the rotator cuff causes pain localized to the lateral shoulder, which is reproduced by repetitive overhead reaching or by lying on the affected side. On examination, patients have point tenderness over the lateral acromion or supraspinatus or infraspinatus tendons, and pain with passive abduction at the shoulder. The weakness perceived by the patient and often found on examination is due to guarding, tendon instability, and disuse atrophy, rather than to true neurological dysfunction. The cornerstones of treatment include ice, nonsteroidal anti-inflammatory drugs (NSAIDs), and avoidance of activities that exacerbate the pain. For patients who do not respond to conservative measures, local steroid injections are helpful, with surgery reserved for patients with refractory pain or rotator cuff tears.
Lateral arm pain
C6 radiculopathy
C6 radiculopathy is characterized by pain and paresthesias that radiate from the neck into the lateral arm and hand, including the thumb. Weakness may involve the biceps, triceps, and wrist flexors.
Lateral epicondylitis
Lateral epicondylitis (tennis elbow) is produced by inflammation of the extensor carpi radialis longus and brevis tendons. Pain secondary to lateral epicondylitis is elicited by palpating the painful area while the patient resists wrist extension. Treatment includes rest, ice, and NSAIDs, with corticosteroid injections reserved for patients with refractory symptoms.
Medial hand and arm pain
Ulnar neuropathy
The most common symptoms of ulnar neuropathy are paresthesias and numbness in the palmar surface of the fifth and medial half of the fourth digits. Ulnar

Figure 16.1 The sensory innervation of the hand.
neuropathy is most often the result of repetitive microtrauma to the nerve as it passes around the elbow. Less common sites of ulnar neuropathy include the forearm and hand. Precipitants include resting the elbows on a table or lying flat on the back while the arms are supinated. On examination, weakness involves finger abduction and flexion of the distal interphalangeal joints of digits four and five. The thenar eminence and first dorsal interosseous may become atrophic. Sensory loss is found over both the palmar and dorsal surfaces of the fifth and medial half of the fourth digits and palm (Figure 16.1). Severe ulnar neuropathy produces a claw hand in which hyperextension at the fourth and fifth metacarpophalangeal joints is accompanied by flexion of the interphalangeal joints. To elicit Tinel’s sign for ulnar neuropathy, percuss the nerve at the elbow as it runs between the olecranon process and medial epicondyle, and ask the patient if paresthesias develop in digits four and five. Ulnar neuropathy may be confirmed electrophysiologically with nerve conduction studies. Conservative treatment of ulnar neuropathy includes avoiding precipitating activities and wearing a padded elbow sleeve to prevent further microtrauma to the nerve. Consider surgical decompression and transposition of the nerve should conservative measures fail.
C8 radiculopathy
Expected complaints of C8 radiculopathy are neck pain and paresthesias radiating into the medial hand and forearm. Numbness in these distributions may accompany weakness of the intrinsic muscles of the hand. Because structural radiculopathies at the C8 levels are relatively uncommon, carefully consider more common possibilities such as ulnar neuropathy at the elbow before making the diagnosis.
Lower trunk brachial plexopathy
The lower trunk of the brachial plexus is comprised of nerve fibers derived from the C8–T1 roots, and damage at this site may cause pain and other sensory complaints in the medial hand, forearm, and arm. The lower trunk of the plexus, like the upper trunk, may be damaged by trauma, cancer (especially in patients with infiltrative apical lung cancer), or idiopathic inflammatory conditions. Because the lower trunk lies in proximity to the oculosympathetic chain, it may be associated with Horner’s syndrome (Chapter 7). One cause of lower trunk brachial plexopathy deserves special mention because of its rarity: thoracic outlet syndrome. This diagnosis is usually incorrect, and often is given to patients with arm pain of unclear etiology. True neurogenic thoracic outlet syndrome, however, is a legitimate diagnosis caused by compression of the lower trunk of the brachial plexus by a cervical rib or accessory band of connective tissue.2 It requires rigorous investigation including EMG and thoracic imaging to confirm the presence of a responsible structural abnormality.
Medial epicondylitis
Medial epicondylitis (golf elbow) is produced by inflammation of the flexor carpi radialis tendon, usually resulting from repetitive gripping movements and wrist flexion. Pain is localized to the medial elbow and, because of the well-known course of the ulnar nerve around the elbow, patients with medial epicondylitis are often referred to neurologists for evaluation of ulnar neuropathy. Pain secondary to medial epicondylitis is elicited by palpating the painful area while the patient resists wrist flexion. Imaging studies should be obtained only to exclude alternative diagnoses. Treatment for medial epicondylitis involves rest, ice, and NSAIDs. Corticosteroid injections and gradual introduction of grip-strengthening exercises may also help.
Lateral hand pain
Carpal tunnel syndrome
Compression of the median nerve at the wrist results in carpal tunnel syndrome (CTS). In the early stages of the disorder, patients describe pain and paresthesias in the hand, mostly in the first three digits and the radial half of the fourth digit. It is also common for patients to report symptoms in the entire hand or extending proximally into the forearm and upper arm, even though these areas are outside the sensory distribution of the median nerve. Hand overuse, particularly typing, mouse use, and assembly work, precipitate or exacerbate the symptoms. Nocturnal symptoms are common, and patients may shake out their hands to relieve the paresthesias. Motor problems are not prominent early in CTS, but as the disorder progresses, the hand and fingers may become weak and grip strength may be reduced. On examination, look for weakness of thumb abduction and flattening of the thenar eminence. The sensory loss in CTS affects the palmar surfaces of the first three digits and the lateral half of the fourth digit (Figure 16.1). Because the palmar sensory branch does not pass through the carpal tunnel, sensation over the palm is spared. Tinel’s and Phalen’s signs support the diagnosis. To elicit Tinel’s sign (Figure 16.2), tap the volar aspect of the wrist briskly with a reflex hammer and inquire about paresthesias in the first three fingers. To elicit Phalen’s sign (Figure 16.3), instruct the patient to appose the dorsal surfaces of the hands to each other while the wrists are flexed for 1 minute: in patients with CTS, paresthesias should develop in the palmar surfaces of the first three fingers. Physical examination abnormalities may be limited in early CTS, and nerve conduction studies may be necessary to make the diagnosis. Patients with mild disease may improve with wrist braces and activity restriction. Local corticosteroid injections are a temporizing measure that may reduce sensory symptoms. Carpal tunnel release surgery is the definitive treatment for patients with more severe CTS.
De Quervain’s tenosynovitis
This condition, caused by inflammation of the tendons of the extensor pollicis brevis and abductor

Figure 16.3 Phalen’s sign. Instruct the patient to appose the dorsal surfaces of the hands for 1 minute. Patients with carpal tunnel syndrome will report paresthesias in the palmar surfaces of digits one to three.
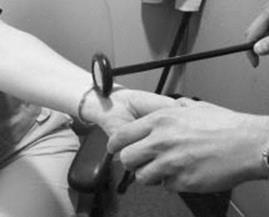
Figure 16.2 Tinel’s sign. Tap the distal volar aspect of the wrist briskly with a reflex hammer. The sign is positive when the patient reports paresthesias in the palmar surfaces of digits one to three.
pollicis longus, may lead to referral for CTS or superficial radial neuropathy, a rare compression neuropathy. Patients with de Quervain’s tenosynovitis describe pain in the thumb and radial styloid, especially with pinching maneuvers. On examination, the radial styloid is tender to palpation. The Finkelstein test is a provocative maneuver for de Quervain’s tenosynovitis in which pain is reproduced by enclosing the thumb inside the fist and deviating the wrist in the ulnar direction (Figure 16.4). Treat de Quervain’s tenosynovitis with rest, NSAIDS, and immobilization of the thumb using

Figure 16.4 The Finkelstein maneuver. Instruct the patient to enclose their thumb inside their fingers (A) and deviate the hand in the ulnar direction (B). Patients with de Quervain’s tenosynovitis will note pain in the lateral aspect of the thumb.
a thumb spica splint. Refractory cases may require corticosteroid injection and possibly surgical referral.
Carpometacarpal joint arthritis
Osteoarthritis of the carpometacarpal (CMC) joint is present in the majority of people over 65 and is characterized by pain at the base of the thumb and radial aspect of the wrist, and sometimes by grip weakness. This condition often leads to neurological consultation for evaluation of CTS. Carpometacarpal joint arthritis is diagnosed on physical examination by demonstrating tenderness to compression at the CMC joint (Figure 16.5). Plain radiographs, although usually not necessary, confirm the diagnosis. Treatment involves immobilization, NSAIDs, and heat application.
Thigh and hip pain
L2 and L3 radiculopathy
While intervertebral disc herniations usually spare the upper lumbar nerve roots, spinal stenosis is a common cause of upper lumbar radiculopathy in older patients (see Chapter 17). Pain and paresthesias may radiate from the back into the lateral (L2) or medial (L3) thigh. Weakness of hip flexion, hip adduction, and knee extension may accompany an upper lumbar radiculopathy. The patellar reflex may be reduced or absent. Because it may be challenging to differentiate among upper lumbar radiculopathy, lumbosacral plexopathy, and femoral neuropathy by history and physical examination, nerve conduction studies and EMG are often required.
Meralgia paresthetica
Entrapment of the lateral femoral cutaneous nerve, usually at the inguinal ligament, leads to meralgia paresthetica. Pain and paresthesias, often quite severe, involve the lateral thigh and sometimes radiate into the knee.
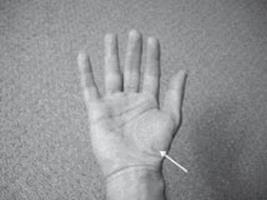
Figure 16.5 The carpometacarpal joint.
Because this nerve has no motor distribution, there is no muscle weakness: any muscle weakness on examination is due to guarding or to an unrelated condition. Meralgia paresthetica usually develops in obese patients or in those who have recently lost a large amount of weight. Nerve conduction studies are frequently unhelpful in establishing the diagnosis, as a sensory response from the lateral femoral cutaneous nerve is difficult to obtain, even in normal subjects. Meralgia paresthetica may improve with weight loss and avoidance of restrictive clothing such as tight belts. Most patients, however, do not improve with these conservative measures and require local steroid injections at the inguinal ligament.3 Surgery is necessary only on rare occasions.
Lumbosacral plexopathy
Many of the general rules described above for brachial plexopathy also apply to lumbosacral plexopathy. Although etiologies common to both brachial and lumbosacral plexopathies include trauma, cancer, and idiopathic lumbosacral plexitis, there are several distinct pathologies that affect the lumbosacral plexus.
Trauma
Traumatic lumbosacral plexopathy results from severe hip trauma (usually involving a fracture) or pelvic surgery. Depending on the site of trauma, sensorimotor symptoms may involve the entire leg or just a restricted part of it. Electromyography helps to localize and quantify the deficits. Lumbosacral plexopathy related to pregnancy is a specific traumatic etiology that is discussed further in Chapter 11.
Retroperitoneal hematoma
Retroperitoneal hematoma, usually related to anticoagulation, a bleeding diathesis, or a procedure involving femoral catheterization, is an important cause of lumbosacral plexopathy. Rapid identification of the problem and correction of any contributing coagulation abnormalities may prevent life-threatening exsanguination. Plexopathy related to hematoma tends to resolve as the blood is reabsorbed.
Cancer
Neoplasm-related lumbosacral plexopathies, like those that involve the upper extremities, may be divided into infiltrative and radiation-induced plexopathies, and are evaluated in a fashion similar to those that affect the brachial plexus. The most common tumors that infiltrate the lumbosacral plexus are colon and ovarian cancers.
Diabetic amyotrophy
Most patients who develop this disorder of the lumbosacral nerve roots and plexus have mild type 2 diabetes at symptom onset. Weight loss, fevers, and night sweats frequently precede or accompany the lumbosacral plexopathy. Diabetic amyotrophy begins with exquisite pain, usually in the hip and thigh, which often spreads to involve the lower leg and, in many cases, the contralateral leg. Leg weakness, often profound, develops several days later. Patients with diabetic amyotrophy require pain control with agents for neuropathic pain and sometimes with narcotics. Physical therapy may help restore mobility. Immunomodulatory therapy including intravenous methylprednisolone and intravenous immunoglobulin is effective on an anecdotal basis.4 Unfortunately, recovery takes many months to years and is often incomplete.
Idiopathic lumbosacral plexitis
Idiopathic lumbosacral plexitis, although similarly named, should not be considered the lower extremity counterpart of idiopathic brachial neuritis, as it is generally a more severe disorder. Idiopathic lumbosacral plexitis is essentially the same condition as diabetic amyotrophy, except that it occurs in patients without diabetes.5
Trochanteric bursitis
Trochanteric bursitis is caused by the breakdown of the bursa surrounding the greater trochanter of the femur. It often accompanies or is confused with meralgia paresthetica or upper lumbar radiculopathy. Pain is located in the lateral hip, and becomes worse when the patient moves the hip or lies on it. Trochanteric bursitis is diagnosed clinically by finding tenderness to palpation of the trochanteric bursa. Conservative treatment includes a combination of NSAIDs, heat, and physical therapy. Local steroid injections may benefit patients who do not respond to these measures.
Osteoarthritis of the hip and occult hip fracture
Osteoarthritis, almost universal in the elderly population, is a common cause of hip pain. Because pain may radiate into the groin, back, or knee, it may lead to evaluation for lumbosacral radiculopathy. Patients typically report groin pain with attempted internal rotation. Treatment with NSAIDs may help, but patients often require orthopedic referral. Severe anterior or lateral hip pain with difficulty bearing weight suggests the possibility of hip fracture and should prompt plain films of the hip and orthopedic referral.
Knee pain
Isolated knee pain results so rarely from a neurological process that more common orthopedic conditions should be evaluated exhaustively before undertaking any neurological investigation. L3 radiculopathy is a possible source of knee pain, but is almost always accompanied by back pain that radiates into the leg. Gonyalgia paresthetica is an even less common cause of knee pain and results from entrapment of the infrapatellar branch of the saphenous nerve.
General principles of nociceptive pain treatment
In an ideal world, neurologists would treat pain that was exclusively of neuropathic origin. The musculoskeletal pain syndromes described in this chapter, however, produce nociceptive rather than neuropathic pain and therefore require a different strategy. The standard approach to nociceptive pain management involves escalating from weaker doses of medications with few side effects, to stronger medications that may have more side effects, to multidisciplinary pain treatment, and finally to alternative therapies:
• Although most nociceptive pain responds to over-the-counter medications such as acetaminophen, aspirin, ibuprofen, and naproxen, patients who respond to one of these treatments usually do not reach neurological attention.
• Prescription NSAIDs are effective for some patients who do not respond to over-the counter analgesics. Common prescription-strength NSAIDs include oxaprozin (600–1200 mg qd), ketorolac (10 mg qid for up to 5 consecutive days), and diclofenac (100 mg bid). Side effects of these medications include gastrointestinal hemorrhage and nephrotoxicity.
• Physical therapy, ice, heat, and massage are effective treatments for some patients with musculoskeletal pain.
• The opioid agonist tramadol (50–100 mg bid) has a lower rate of side effects than the true narcotic medications, and may be used to treat a variety of musculoskeletal complaints refractory to acetaminophen or NSAIDs.
• Narcotic analgesics should be avoided if possible, as these medications have addictive properties, are sedating, and have a thriving black market for resale. Avoid rapid-onset, short-acting narcotics such as hydrocodone, oxycodone, hydromorphone, meperidine, and short-acting morphine, as their brief durations of action lead to frequent spikes in pain. Longer-acting narcotic options include fentanyl patches or methadone, which should still be prescribed sparingly.
• Local anesthetic or steroid injections may be helpful for patients with herniated intervertebral discs, bursitis, and other musculoskeletal conditions that do not respond to oral medications.
• Many pain syndromes are difficult to treat due to the severity of the underlying condition or to superimposed or preexisting psychological factors. Tricyclic antidepressants, selective serotonin reuptake inhibitors, and newer agents such as duloxetine and pregabalin may help patients with refractory chronic pain and comorbid depression.
• Psychological counseling, biofeedback, and alternative treatments such as acupuncture and homeopathic treatments may be effective in treating severe pain.
Controversial localized pain syndromes
Complex regional pain syndrome
Complex regional pain syndrome (CRPS), alternatively known as reflex sympathetic dystrophy or causalgia, is characterized by pain, edema, trophic skin changes, and a decreased range of motion in an extremity. In the early stages, however, pain may be unaccompanied by other signs and symptoms. The condition is divided into CRPS type I in which there is no definable nerve lesion and CRPS type II in which there is a history of a preceding nerve injury. The mechanism by which CRPS develops is unclear, and the diagnosis is, in itself, a controversial one. Many patients with limb pain of uncertain etiology are told that they have CRPS when no clear source for their pain can be identified. However, I have never made this diagnosis or seen a convincing case. There are staunch advocates for6 and against7 the existence of CRPS. While I tend to agree with the latter school, I maintain an open mind.
Myofascial pain syndrome
Myofascial pain syndrome is the name that is often assigned to any focal musculoskeletal pain of unclear etiology despite thorough investigation. Problems such as depression, fatigue, and forgetfulness frequently accompany the pain. Myofascial pain syndrome may represent a restricted form of fibromyalgia, and responds (or fails to respond) to the same treatments as that disorder (Chapter 15).
References
1. Thyagarajan D, Cascino T, Harms G. Magnetic resonance imaging in brachial plexopathy of cancer. Neurology 1995;45:421–427.
2. Cuetter AC, Bartoszek DM. The thoracic outlet syndrome: controversies, overdiagnosis, overtreatment, and recommendations for management. Muscle Nerve 1989;12:410–419.
3. Harney D, Patijn J. Meralgia paresthetica: diagnosis and management strategies. Pain Med 2007;8:669–677.
4. Chan YC, Lo YL, Chan ESY. Immunotherapy for diabetic amyotrophy. Cochrane Database Syst Rev 2009; (3): CD006521.
5. Dyck PJB, Windebank AJ. Diabetic and nondiabetic lumbosacral radiculoplexus neuropathies: new insights into pathophysiology and treatment. Muscle Nerve 2002;25:477–491.
6. Schwartzman RJ, Popescu A. Reflex sympathetic dystrophy. Curr Rheum Rep 2002;4:165–169.
7. Ochoa JL. Truths, errors, and lies around “reflex sympathetic dystrophy” and “complex regional pain syndrome”. J Neurol 1999;246:875–879.