Masashi Ueda • Takashi Temma • Hideo Saji
INTRODUCTION
Integrins are transmembrane receptors that mediate the attachment between a cell and its surroundings. They are members of a glycoprotein family of compounds that form heterodimeric receptors for extracellular matrix molecules. They consist of α- and β-subunits and are involved in the interaction between cells, extracellular matrices (e.g., collagen, laminin, fibronectin, and vitronectin), and secretory molecules (e.g., growth factors). To date, 18 α-subunits and eight β-subunits, forming 24 different integrins, have been defined.1 Of these, αvβ3-integrin is the most widely researched. αvβ3-integrin is highly expressed on the cell membranes of activated endothelial cells in new blood vessels. It is thus a promising molecular target for angiogenic imaging.2 Furthermore, integrins are expressed on the cell membranes of several types of tumors. αvβ6-integrin is interesting because its expression is low or undetectable in most tissues in adults, yet its expression is upregulated dramatically in many different carcinomas.3
For integrin imaging, various imaging modalities, including positron emission tomography (PET), single photon emission computed tomography (SPECT), magnetic resonance imaging (MRI), ultrasonography, and optical imaging have been tested at the preclinical stage, with varying degrees of success.4 Among these, because of their characteristic high sensitivity, nuclear medicine methods using radionuclides (PET and SPECT) have been the only ones to advance beyond animal experiments to succeed in clinical-level translational research. Using PET, which is more sensitive than SPECT, radioactive probes have been successfully used in vivo at the picomole level.5 Use of the minimum possible amount of radioactive tracers can minimize the possibility of producing a pharmacologic effect in the organism, as well as the possibility of monopolizing the integrin target molecules. This is one of the advantageous characteristics of radioactive probes because the molecules that serve as imaging targets are normally present in low expression levels in vivo.
All the radioactive probes that target integrin are based on the amino acid sequence arginine–glycine–aspartic acid (amino acid 1-letter symbol: RGD). The RGD sequence is found in many proteins in extracellular matrices that show connectivity with integrin, and the RGD sequence shows connectivity with the integrin αv-subunit.6 Integrin-specific probes are RGD compounds that have been labeled with radionuclides. Scintigraphy and SPECT use technetium-99m (99mTc) and other γ-ray–emitting nuclides, whereas PET uses positron-emitting nuclides such as fluorine-18 (18F), copper-64 (64Cu), and gallium-68 (68Ga). This review describes the radioactive probes targeting αvβ3- and αvβ6-integrins. These probes contain RGD-based structures and have already been used in clinical trials or have received attention for future clinical use.
αvβ3-Integrin
αvβ3-Integrin is highly expressed on the cell membranes of activated endothelial cells in new blood vessels and is most widely investigated for its involvement in angiogenesis.7 Angiogenesis, the formation of new blood vessels, is a normal biologic process that occurs in physiologic as well as pathologic conditions. Examples of physiologic processes accompanied by angiogenesis are regeneration, growth, and wound repair. Benign pathologic conditions accompanied by angiogenesis include arthritis, psoriasis, and retinopathy.8 Angiogenesis is also an important process in the growth of solid, including malignant, tumors.9 When the tumor size increases to over 1 mm3, oxygen and nutrients supplied by pre-existing blood vessels are insufficient; therefore, the tumor can only continue to grow if additional blood vessels are formed.10 Because this “angiogenic switch” is an event common to many malignant tumors, angiogenesis is an important phenomenon, not only in imaging but also as a therapeutic target. In fact, a therapeutic drug (cilengitide) has been developed that specifically inhibits αvβ3- and αvβ5-integrins.11
Probes for αvβ3-Integrin
18F-Labeled Probes
[18F]gluco-RGD and [18F]galacto-RGD
Aumailley et al. developed a cyclic RGD peptide [cyclo(-Arg-Gly-Asp-D-Phe-Val-), c(RGDf V)] by circularizing a straight chain RGD peptide and introducing a nonnatural D amino acid.12 Because c(RGDf V) has high affinity and selectivity, as well as in vivo stability, nearly all the subsequent αvβ3-integrin imaging probes have been designed with this cyclic pentapeptide, or a slight modification, as their basic structure. In the first radiolabeled probe, c(RGDyV) was labeled with iodine-125 (125I), but the high liposolubility led to considerable nonspecific accumulation in the liver, bile, and digestive tract, which was associated with reduced image quality.13 Accordingly, attempts were made to increase the hydrophilicity. Probes with added glucose or galactose were developed, that is, the gluco-RGD probe, which has c(RGDyK) as the basic structure,14 and the galacto-RGD probe, which has c(RGDfK) as the basic structure. The galacto-RGD probe was developed as an 18F-labeled probe ([18F]galacto-RGD). It exhibits high affinity and selectivity for αvβ3-integrin.15 Further, although this probe shows high accumulation in transplant-tumor tissue that expresses αvβ3-integrin in vivo, the increased water solubility indicates that rapid clearance from nonspecific tissue such as blood and muscle is possible through renal excretion. Further, this probe is stable in vivo; the percentage of unaltered substances in the blood was found to be 86% at 120 minutes after administration in mice; the percentage in tumor tissue was 87%. The expression level of αvβ3-integrin in transplanted tumors was found to be correlated with the accumulation of this probe in the tumors.16
[18F]galacto-RGD was first tested as an integrin probe for human application in 2005,17,18 after which its usefulness has been clarified in a variety of clinical studies. [18F]galacto-RGD can be used for many solid cancers. The accumulation of this probe is correlated with the expression level of αvβ3-integrin. This correlation is not simply a consequence of tumor size.19 This probe is suitable for detection of the primary lesion but not metastatic lesions. However, because [18F]galacto-RGD also accumulates at sites of benign chronic inflammations such as villonodular synovitis,18 it is not suited for differentiating benign from malignant lesions, similar to 2-deoxy-2-[18F]fluoro-D-glucose (18F-FDG). It causes the same level of radiation exposure as other 18F-labeled tracers.
In summary, [18F]galacto-RGD is an excellent probe for αvβ3-integrin imaging, except for the organs showing physiologic accumulation (liver, spleen, and organs of the urinary system), and is the probe that has been the subject of the most research. Because of the complexity of synthetic methods, automatic probe synthesis remains an unresolved issue. The probe is, therefore, not yet ready for routine use.
[18F]fluciclatide (AH111585)
[18F]fluciclatide (AH111585) is another integrin probe that has been made available for clinical use (and has been developed by GE Healthcare). The molecular structure of [18F]fluciclatide contains 1 RGD motif but unlike galacto-RGD, it shows the highest affinity for αvβ5-integrin, followed by αvβ3-integrin. This probe exhibits high stability in vivo because of the polyethylene glycol (PEG) spacer, the disulfide bridge, and cyclization. Imaging has been successfully performed with [18F]fluciclatide PET for all breast cancer lesions detected by CT.20 The radiation exposure with [18F]fluciclatide is roughly equal to that with [18F]galacto-RGD.21 This probe has the advantage of having simpler radiochemical features than [18F]galacto-RGD, and therefore, there is optimism that it will become clinically useful. Research on this probe is currently advancing to a phase II trial.22
[18F]RGD-K5
[18F]RGD-K5 is an RGD probe synthesized through click chemistry and developed by the molecular imaging department at Siemens.23 Its affinity for αvβ3-integrin is high (Kd = 7.9 nM), a selectivity that is approximately two-fold greater than that for other integrin subtypes. It is a promising probe because of in vivo stability achieved through reduced renal excretion. It provides nearly the same results as other integrin probes in examinations of breast cancer patients. However, histologic analyses have shown that [18F]RGD-K5 uptake is not correlated with histologic angiogenic parameters. Therefore, either this probe is not specific for αvβ3-integrin or it may bind to αvβ3-integrin on the surface of tumor cells, in addition to new blood vessels.22
64Cu- and 68Ga-Labeled Probes
64Cu-DOTA-RGD
64Cu-labeled integrin probes have been developed to label integrins by introducing 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid [DOTA] into c(RGDyK) (Fig. 40.1).24 However, the probe has reduced affinity because of the bulky nature of DOTA. 64Cu-DOTA-PEG-RGD introduced with PEG at the linker location connecting RGD and DOTA has been shown to improve pharmacokinetics, decrease accumulation in the liver, and increase clearance via the blood and kidneys. High tumor-tissue ratios have been observed in micro-PET studies on glioblastoma-transplanted mice, showing the usefulness of this probe.25
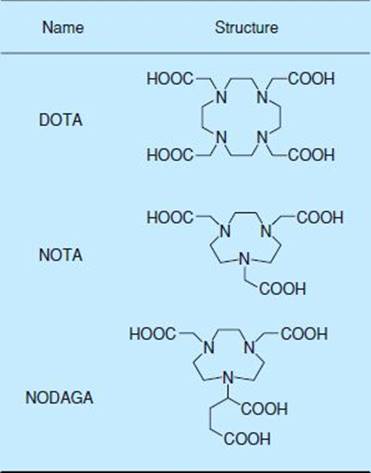
FIGURE 40.1. Chemical structure of three macrocyclic chelators.
68Ga-NOTA-RGD
A stable radioactive metal complex compound for 68Ga can be created using metal chelators such as DOTA, 1,4,7-triazacyclononane-1,4,7-triacetic acid [NOTA] and 1,4,7-triazacyclononane, 1-glutaric acid-4,7-acetic acid [NODAGA] (Fig. 40.1). 68Ga is an ideal positron-emitting nuclide for labeling low–molecular-weight peptide tracers that exhibit rapid pharmacokinetics in vivo because of its ability to be prepared for use with an on-site from a 68Ge/68Ga generator with its short half-life of 68 minutes.26
68Ga-NOTA-RGD is a tracer obtained by joining a lysine side chain of c(RGDyK) to isothiocyanato-benzyl-NOTA.27 68Ga labeling is completed with 10 minutes of incubation at room temperature. 68Ga-NOTA-RGD maintains high connectivity with αvβ3-integrin in in vivo evaluations, and NOTA modification is thought to have little effect on reducing the connectivity of c(RGDyK). In mouse studies, 68Ga-NOTA-RGD exhibited the highest accumulation in the kidneys because of reduced renal excretion and showed extremely low accumulation in the liver and intestines. Studies in tumor-bearing mice have shown high tumor–blood and tumor–muscle ratios, indicating that 68Ga-NOTA-RGD possesses excellent characteristics for micro-PET imaging. Similar findings have also been reported regarding 68Ga-NODAGA-RGD.28
Possible clinical applications of 68Ga-labeling RGD peptides are highly anticipated. In the first clinical study, 18F-FDG and 68Ga-NOTA-RGD were used in PET examinations of colon cancer patients with liver metastases. In three of the six subjects, 68Ga-NOTA-RGD was observed to have relatively high accumulation in metastatic liver lesions but no accumulation was observed in the other three subjects. Upon administration of antiangiogenic therapy, a partial response was achieved in the patients who exhibited 68Ga-NOTA-RGD uptake, whereas the nonaccumulation group presented with stable or progressive disease.22
99mTc-Labeled Probes
99mTc labeling can be performed on site at the time of use with a commercially available generator system in contrast to 18F that requires a cyclotron or a commercial PET radiopharmacy. 99mTc-labeled probes are also being developed because γ-cameras for planar and SPECT imaging are more widely available than PET. Compared to PET, SPECT has the disadvantage of lower spatial resolution and sensitivity, and quantitative analysis can be difficult. In addition, because of the long time required for imaging, routine use of full-body SPECT imaging in clinical settings can be difficult.
99mTc-αP2 is a peptide composed of 10 amino acids with 2 RGD motifs and has been prepared with a cysteine-mediated 99mTc chelator. This tracer was initially reported in 1988.29 In clinical studies on patients with malignant melanomas, this probe successfully detected metastases in the neck, axilla, abdomen, and soft tissue. The sensitivity in the chest was low, because of the activity in the cardiac blood pool. Imaging should be performed 1 to 2 hours after administration to obtain the best tumor–background ratio. However, issues persist regarding matters such as the specificity of integrin binding and the relationship between in vivo accumulation levels and integrin expression levels.
99mTc-NC100692 (developed by GE Healthcare) is a peptide probe that contains RGD inside the array, and exhibits high affinity for αvβ3-integrin. This probe could detect malignant lesions sized 1 cm and larger in clinical studies involving breast cancer patients, detecting 19 of 22 lesions.30 Although this probe exhibited excellent sensitivity for metastatic lesions to the lung and brain in other clinical studies on breast and lung cancer patients, it exhibited low sensitivity for the detection of metastases to the liver and bones.31

FIGURE 40.2. SPECT axial images of pancreatic tumor (A) and purulent inflammatory lesion (B), and an anatomic image of excised pancreas (C) in a hamster pancreatic carcinogenesis model. The tumor in pancreatic head is indicated by an arrow and the inflammatory lesion is indicated by a dotted circle. In, intestine; Ki, kidney; Li, liver; Pg, pancreatic gastric lobe; Ps, pancreatic splenic lobe; Sp, spleen; St, stomach. (Image courtesy of Dr. Yoshimoto, National Cancer Research Center, Tokyo, Japan.)
111In-Labeled Probe
Recently, a very interesting animal experiment using 111In-DOTA-c(RGDfK) was performed. Yoshimoto et al.32 investigated the usefulness of SPECT imaging with 111In-DOTA-c(RGDfK) for early and accurate detection of pancreatic cancer in a hamster model of pancreatic carcinoma. As a result, 111In-DOTA-c(RGDfK) was clearly visible in chemically induced pancreatic cancers as small as 3 mm in diameter. In contrast, no probe uptake was observed in inflammatory lesions in animals (Fig. 40.2). The uptake of 111In-DOTA-c(RGDfK) strongly correlated with αvβ3-integrin expression. The investigators also compared the uptake of 18F-FDG and 111In-DOTA-c(RGDfK) in a mouse inflammatory model created by turpentine oil injection. Although 18F-FDG was actively taken up in the inflamed regions in all cases, 111In-DOTA-c(RGDfK) was not (Fig. 40.3). The inflammation-to-muscle ratio for 18F-FDG was four-fold higher than that for 111In-DOTA-c(RGDfK). These results imply that 111In-DOTA-c(RGDfK) has a lower rate of false-positive tumor detection than 18F-FDG.

FIGURE 40.3. Autoradiograms (ARGs) of 18F-FDG and 111In-DOTA-c(RGDfK), and images of immunohistochemical (IHC) staining against αvβ3-integrin in a mouse model for inflammation. Acute inflammation was characterized by focal neutrophil infiltration and the expression of αvβ3-integrin was observed outside the inflammatory regions only. (Image courtesy to Dr. Yoshimoto, National Cancer Research Center, Tokyo, Japan.)
Multivalent Probes
As described above, a variety of αvβ3-integrin–targeting probes have been developed, but there is still a lack of probes that can detect lesions in sites with high physiologic accumulation (background) such as the liver. Thus, by focusing on in vivo integrin cluster formation, and the interactions of other integrin molecules and functional molecules in the extracellular matrix, attempts are being made to increase specific accumulation to tumors by creating multivalent probes.33,34
Specifically, a variety of investigations have been carried out on dimers, tetramers, octamers, and other entities with circular pentapeptides as the basic component, in which the distance between components is adjusted through substances such as PEG and glycine linkers. 64Cu,35 68Ga,36 and 18F37 have been used as label nuclides, and higher tumor clustering than with monomers has been observed through the multivalent effect. However, multivalency does not necessarily mean that the tumor–background ratio will increase; therefore, caution is essential. Recent developments in clinical research on 18F-labeled dimers have shown that they have no side effects and have also shown good pharmacokinetics in healthy volunteers.38 Further results from clinical research on patients are eagerly awaited.
αvβ6-INTEGRIN
αvβ6-Integrin is unique in that it is expressed exclusively in epithelial cells. The β6-subunit partners only with the αv-subunit, forming a single heterodimer. The expression of αvβ6-integrin is dramatically increased during wound healing. Elevated αvβ6-integrin expression is observed at the wound edge within a few days of wounding.39 This expression is maintained until wound closure is complete. Moreover, αvβ6-integrin expression is upregulated in many cancers and is often associated with a poor prognosis.40 Because αvβ6-integrin is not expressed in normal adult tissue, it is considered to be a very good molecular target for cancer therapy.41 Although drugs targeting αvβ6-integrin–expressing cells have not been developed yet, imaging probes have allowed researchers to visualize αvβ6-integrin–positive tumors in a mouse xenograft model. These probes will be useful not only for the detection of αvβ6-integrin–positive tumors but also for the evaluation of efficacy of αvβ6-integrin–targeted therapy.
PROBES FOR αvβ6-INTEGRIN
18F-Labeled Probes
Recently, Hausner et al.42 identified a potent inhibitor of αvβ6-integrin. This inhibitor is a 20-amino acid peptide, NAVPNLRGDLQVLAQKVART (A20FMDV2), derived from foot-and-mouth disease virus. The peptide contains the RGDLXXL motif, where X indicates any amino acid. Although the highly conserved RGD motif is recognized by eight different integrins,43 the key moiety responsible for αvβ6-integrin specificity is the DLXXL sequence. This sequence showed only minimal interactions with αvβ3-, αvβ5-, and αIIbβ3-integrins.44 Hausner et al. conjugated 4-[18F]fluorobenzoic acid (18F-FBA) to the amino terminal of the peptide to produce 18F-FBA-A20FMDV2. This probe showed high affinity for αvβ6-integrin, with an IC50 of 3 nM. The selectivity of this probe was 1,000-fold higher for αvβ6-integrin than that for other RGD-directed integrins (αvβ3 and αvβ5), as expected. Although the tumor uptake was relatively low (less than 1% injected dose per gram tissue [%ID/g] 1 hour after injection), the tumor was visualized using PET. The %ID/g ratio of αvβ6-integrin–positive tumor/negative tumor was approximately 3, suggesting specific accumulation of this probe in vivo.
Although targeted imaging was achieved using the first-generation probe 18F-FBA-A20FMDV2, low uptake and poor retention in the target tissue limited its utility. To improve pharmacokinetics of the probe, small, monodisperse PEG polymers [PEG28 or (PEG28)2] were introduced to A20FMDV2.45 The modified probes maintained high affinity and selectivity for αvβ6-integrin. In vivo studies revealed that the modified probes showed significantly improved tumor retention (more than 1.5%ID/g for both PEGylated probes 4 hour after injection). Radioactivity accumulation was more than 20-fold greater than for 18F-FBA-A20FMDV2. Thus, 18F-FBA-PEG28-A20FMDV2 and 18F-FBA-(PEG28)2-A20FMDV2 are promising in αvβ6-integrin–specific tumor imaging.
64Cu-Labeled Probes
The A20FMDV2 peptide was also labeled with 64Cu.46 Two different chelators, DOTA and 4,11-bis(carboxymethyl)-1,4,8,11-tetraazabicyclo[6.6.2] hexadecane (CB-TE2A) were used to radiolabel. In vitro, both DOTA-PEG-A20FMDV2 and CB-TE2A-PEG-A20FMDV2 showed excellent αvβ6-integrin–specific binding. In vivo, both probes showed higher accumulation in αvβ6-integrin-positive tumors than in negative tumors. However, unexpectedly high and persistent levels of radioactivity accumulated in the kidneys. Despite identical targeting peptide moieties and similar in vitro performance, both 64Cu-labeled probes displayed inferior pharmacokinetics, making them less suitable candidates than the 18F-labeled probes for in vivo imaging of αvβ6-integrin.
123/125I-Labeled Probe
Several peptides containing RGDLXXL motifs (A20FMDV2,42 TP H2009.1,47 Bpep,48 and Peptide 2949) showed specific binding to αvβ6-integrin in phage display screening (Table 40.1). However, with exception of A20FMDV2, the binding affinities of the peptides were not quantitatively evaluated. A competitive binding assay performed in our laboratory revealed that the A20FMDV2 has the highest affinity for αvβ6-integrin–positive tumor cells. Because 18F- or 64Cu-labeling at the N-terminal amino acid of A20FMDV2 did not inhibit binding to αvβ6-integrin, we introduced glycylcysteine at the N-terminus of A20FMDV2 to label site-specifically using N-(m-[123/125I]iodophenyl) maleimide (123/125I-IPM).50 A biodistribution study showed that the tumor uptake of 123/125I-IPM–conjugated A20FMDV2 (123/125I-IFMDV2) 1 hour after administration was 1.3%ID/g, which was greater than 18F-FBA-A20FMDV2. Pretreatment with excess A20FMDV2 induced a 70% decrease in 123/125I-IFMDV2 accumulation in αvβ6-integrin–positive tumors, and did not affect accumulation in αvβ6-integrin–negative tumors (Fig. 40.4). The higher accumulation of 123/125I-IFMDV2 in αvβ6-integrin–positive tumors was confirmed by autoradiographic study (Fig. 40.5). The detailed characteristics of 123/125I-IFMDV2 are being prepared for publication.
TABLE 40.1
αvβ6-INTEGRIN TARGETING PEPTIDES


FIGURE 40.4. In vivo blocking study. AsPC-1 is αvβ6-integrin–positive tumor and MIAPaca-2 is αvβ6-integrin–negative tumor. The radioactivity accumulated in AsPC-1 xenograft was significantly decreased by the preadministration of an excess amount of A20FMDV2, whereas that accumulated in MIAPaca-2 xenograft was not affected by the pretreatment. **p < 0.01 (Mann–Whitney U test).
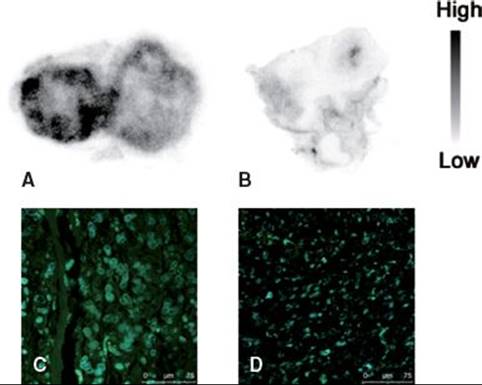
FIGURE 40.5. Autoradiograms of 123I-IFMDV2 in AsPC-1 xenograft (A) and MIAPaca-2 xenograft (B), and images of immunohistochemical staining against αvβ6-integrin in AsPC-1 xenograft (C) and MIAPaca-2 xenograft (D). Strong signal (green) derived from αvβ6-integrin was detected in C, whereas rare signal was detected in D. Nuclei (blue) were observed in both tumors.
CONCLUSIONS
Integrin probes with RGD peptide cores, especially those targeting αvβ3-integrin, represent an example of success in translation from basic to clinical research. Hopefully, integrin imaging will become useful for evaluating patient risk and deciding whether to apply antiangiogenic therapy in clinical settings. Because antiangiogenic therapy usually leads to a reduction of tumor growth rather than to tumor shrinkage, the morphologic imaging methods (e.g., x-ray CT and MRI) used for evaluating tumor size are not entirely appropriate to monitor the effect of treatment. We believe that αvβ3-integrin imaging will be a viable and highly sensitive strategy for the early evaluation of the effects of antiangiogenic therapies. As for αvβ6-integrin imaging, some promising probes have been developed, although no clinical research has yet been performed. Because αvβ6-integrin is not expressed in normal adult tissue but is overexpressed in many cancers, αvβ6-integrin imaging probes may be powerful tools for the specific detection of cancers.
FUTURE CONSIDERATIONS
Recently, αvβ3-integrin was implicated in both the suppression and promotion of angiogenic processes. Therefore, caution should be exercised when interpreting integrin imaging data and the clinical significance of imaging integrin expression is yet to be fully established. Thus, to define the ultimate role of imaging of integrin expression in the clinic, large-scale trials using αvβ3-integrin imaging probes within the context of response assessment or evaluation of patient prognosis are eagerly awaited in the next step.
ACKNOWLEDGMENTS
The authors are grateful to Dr. Yoshimoto (National Cancer Center Research Institute, Tokyo, Japan) for allowing us to present the data shown in Figures 40.2 and 40.3. The development of 123/125I-IFMDV2 was partially supported by Research and Development Project on Molecular Probes for Detection of Biological Features on Cancer from New Energy and Industrial Technology Development Organization (NEDO) of Japan, and a grant from Pancreas Research Foundation of Japan.
REFERENCES
1. van der Flier A, Sonnenberg A. Function and interactions of integrins. Cell Tissue Res. 2001;305:285–298.
2. Rupp PA, Little CD. Integrins in vascular development. Circ Res. 2001;89:566–572.
3. Breuss JM, Gallo J, Delisser HM, et al. Expression of the beta 6 integrin subunit in development, neoplasia and tissue repair suggests a role in epithelial remodeling. J Cell Sci. 1995;108(Pt 6):2241–2251.
4. Cai W, Chen X. Multimodality molecular imaging of tumor angiogenesis. J Nucl Med. 2008;49(suppl 2):113S–128S.
5. Lee CM, Farde L. Using positron emission tomography to facilitate CNS drug development. Trends Pharmacol Sci. 2006;27:310–316.
6. Ruoslahti E, Pierschbacher MD. New perspectives in cell adhesion: RGD and integrins. Science. 1987;238:491–497.
7. Somanath PR, Malinin NL, Byzova TV. Cooperation between integrin alphavbeta3 and VEGFR2 in angiogenesis. Angiogenesis. 2009;12:177–185.
8. Folkman J. Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat Med. 1995;1:27–31.
9. Weis SM, Cheresh DA. Tumor angiogenesis: Molecular pathways and therapeutic targets. Nat Med. 2011;17:1359–1370.
10. Otrock ZK, Hatoum HA, Awada AH, et al. Hypoxia-inducible factor in cancer angiogenesis: Structure, regulation and clinical perspectives. Crit Rev Oncol Hematol. 2009;70:93–102.
11. Eskens FA, Dumez H, Hoekstra R, et al. Phase I and pharmacokinetic study of continuous twice weekly intravenous administration of Cilengitide (EMD 121974), a novel inhibitor of the integrins alphavbeta3 and alphavbeta5 in patients with advanced solid tumours. Eur J Cancer. 2003;39:917–926.
12. Aumailley M, Gurrath M, Müller G, et al. Arg-Gly-Asp constrained within cyclic pentapeptides. Strong and selective inhibitors of cell adhesion to vitronectin and laminin fragment P1. FEBS Lett. 1991;291:50–54.
13. Haubner R, Wester HJ, Reuning U, et al. Radiolabeled alpha(v)beta3 integrin antagonists: A new class of tracers for tumor targeting. J Nucl Med. 1999;40: 1061–1071.
14. Haubner R, Wester HJ, Burkhart F, et al. Glycosylated RGD-containing peptides: Tracer for tumor targeting and angiogenesis imaging with improved biokinetics. J Nucl Med. 2001;42:326–336.
15. Haubner R, Wester HJ, Weber WA, et al. Noninvasive imaging of alpha(v)beta3 integrin expression using 18F-labeled RGD-containing glycopeptide and positron emission tomography. Cancer Res. 2001;61:1781–1785.
16. Haubner R, Kuhnast B, Mang C, et al. [18F]Galacto-RGD: Synthesis, radiolabeling, metabolic stability, and radiation dose estimates. Bioconjug Chem. 2004;15: 61–69.
17. Beer AJ, Haubner R, Goebel M, et al. Biodistribution and pharmacokinetics of the alphavbeta3-selective tracer 18F-galacto-RGD in cancer patients. J Nucl Med. 2005;46:1333–1341.
18. Haubner R, Weber WA, Beer AJ, et al. Noninvasive visualization of the activated alphavbeta3 integrin in cancer patients by positron emission tomography and [18F]Galacto-RGD. PLoS Med. 2005;2:e70.
19. Beer AJ, Haubner R, Sarbia M, et al. Positron emission tomography using [18F]Galacto-RGD identifies the level of integrin alpha(v)beta3 expression in man. Clin Cancer Res. 2006;12:3942–3949.
20. Kenny LM, Coombes RC, Oulie I, et al. Phase I trial of the positron-emitting Arg-Gly-Asp (RGD) peptide radioligand 18F-AH111585 in breast cancer patients. J Nucl Med. 2008;49:879–886.
21. Mcparland BJ, Miller MP, Spinks TJ, et al. The biodistribution and radiation dosimetry of the Arg-Gly-Asp peptide 18F-AH111585 in healthy volunteers. J Nucl Med. 2008;49:1664–1667.
22. Gaertner FC, Kessler H, Wester HJ, et al. Radiolabelled RGD peptides for imaging and therapy. Eur J Nucl Med Mol Imaging. 2012;39(suppl 1):S126–S138.
23. Doss M, Kolb HC, Zhang JJ, et al. Biodistribution and radiation dosimetry of the integrin marker 18F-RGD-K5 determined from whole-body PET/CT in monkeys and humans. J Nucl Med. 2012;53:787–795.
24. Chen X, Park R, Tohme M, et al. MicroPET and autoradiographic imaging of breast cancer alpha v-integrin expression using 18F- and 64Cu-labeled RGD peptide. Bioconjug Chem. 2004;15:41–49.
25. Chen X, Hou Y, Tohme M, et al. Pegylated Arg-Gly-Asp peptide: 64Cu labeling and PET imaging of brain tumor alphavbeta3-integrin expression. J Nucl Med. 2004;45:1776–1783.
26. Prata MI. Gallium-68: A new trend in PET radiopharmacy. Curr Radiopharm. 2012;5:142–149.
27. Jeong JM, Hong MK, Chang YS, et al. Preparation of a promising angiogenesis PET imaging agent: 68Ga-labeled c(RGDyK)-isothiocyanatobenzyl-1,4,7-triazacyclononane-1,4,7-triacetic acid and feasibility studies in mice. J Nucl Med. 2008;49:830–836.
28. Knetsch PA, Petrik M, Griessinger CM, et al. [68Ga]NODAGA-RGD for imaging αvβ3 integrin expression. Eur J Nucl Med Mol Imaging. 2011;38:1303–1312.
29. Sivolapenko GB, Skarlos D, Pectasides D, et al. Imaging of metastatic melanoma utilising a technetium-99m labelled RGD-containing synthetic peptide. Eur J Nucl Med. 1998;25:1383–1389.
30. Bach-Gansmo T, Danielsson R, Saracco A, et al. Integrin receptor imaging of breast cancer: A proof-of-concept study to evaluate 99mTc-NC100692. J Nucl Med. 2006;47:1434–1439.
31. Axelsson R, Bach-Gansmo T, Castell-Conesa J, et al. An open-label, multicenter, phase 2a study to assess the feasibility of imaging metastases in late-stage cancer patients with the alpha v beta 3-selective angiogenesis imaging agent 99mTc-NC100692. Acta Radiol.2010;51:40–46.
32. Yoshimoto M, Hayakawa T, Mutoh M, et al. In vivo SPECT imaging with 111In-DOTA-c(RGDfK) to detect early pancreatic cancer in a hamster pancreatic carcinogenesis model. J Nucl Med. 2012;53:765–771.
33. Janssen M, Oyen WJ, Massuger LF, et al. Comparison of a monomeric and dimeric radiolabeled RGD-peptide for tumor targeting. Cancer Biother Radiopharm. 2002; 17:641–646.
34. Dijkgraaf I, Kruijtzer JA, Liu S, et al. Improved targeting of the alpha(v)beta (3) integrin by multimerisation of RGD peptides. Eur J Nucl Med Mol Imaging. 2007;34:267–273.
35. Chen X, Sievers E, Hou Y, et al. Integrin alpha v beta 3-targeted imaging of lung cancer. Neoplasia. 2005;7:271–279.
36. Liu Z, Niu G, Shi J, et al. 68Ga-labeled cyclic RGD dimers with Gly3 and PEG4 linkers: Promising agents for tumor integrin alphavbeta3 PET imaging. Eur J Nucl Med Mol Imaging. 2009;36:947–957.
37. Cai W, Zhang X, Wu Y, et al. A thiol-reactive 18F-labeling agent, N-[2-(4-18F-fluorobenzamido)ethyl]maleimide, and synthesis of RGD peptide-based tracer for PET imaging of alpha v beta 3 integrin expression. J Nucl Med. 2006;47: 1172–1180.
38. Mittra ES, Goris ML, Iagaru AH, et al. Pilot pharmacokinetic and dosimetric studies of 18F-FPPRGD2: A PET radiopharmaceutical agent for imaging α(v)β(3) integrin levels. Radiology. 2011;260:182–191.
39. Haapasalmi K, Zhang K, Tonnesen M, et al. Keratinocytes in human wounds express alpha v beta 6 integrin. J Invest Dermatol. 1996;106:42–48.
40. Bandyopadhyay A, Raghavan S. Defining the role of integrin alphavbeta6 in cancer. Curr Drug Targets. 2009;10:645–652.
41. Bates RC. The alphaVbeta6 integrin as a novel molecular target for colorectal cancer. Future Oncol. 2005;1:821–828.
42. Hausner SH, DiCara D, Marik J, et al. Use of a peptide derived from foot-and-mouth disease virus for the noninvasive imaging of human cancer: Generation and evaluation of 4-[18F]fluorobenzoyl A20FMDV2 for in vivo imaging of integrin alphavbeta6 expression with positron emission tomography. Cancer Res. 2007;67: 7833–7840.
43. Ruoslahti E. RGD and other recognition sequences for integrins. Annu Rev Cell Dev Biol. 1996;12:697–715.
44. Kraft S, Diefenbach B, Mehta R, et al. Definition of an unexpected ligand recognition motif for alphav beta6 integrin. J Biol Chem. 1999;274:1979–1985.
45. Hausner SH, Abbey CK, Bold RJ, et al. Targeted in vivo imaging of integrin alphavbeta6 with an improved radiotracer and its relevance in a pancreatic tumor model. Cancer Res. 2009;69:5843–5850.
46. Hausner SH, Kukis DL, Gagnon MK, et al. Evaluation of [64Cu]Cu-DOTA and [64Cu]Cu-CB-TE2A chelates for targeted positron emission tomography with an alphavbeta6-specific peptide. Mol Imaging. 2009;8:111–121.
47. Elayadi AN, Samli KN, Prudkin L, et al. A peptide selected by biopanning identifies the integrin alphavbeta6 as a prognostic biomarker for nonsmall cell lung cancer. Cancer Res. 2007;67:5889–5895.
48. Pameijer CR, Navanjo A, Meechoovet B, et al. Conversion of a tumor-binding peptide identified by phage display to a functional chimeric T cell antigen receptor. Cancer Gene Ther. 2007;14:91–97.
49. Hsiao JR, Chang Y, Chen YL, et al. Cyclic alphavbeta6-targeting peptide selected from biopanning with clinical potential for head and neck squamous cell carcinoma. Head Neck. 2010;32:160–172.
50. Khawli LA, van den Abbeele AD, Kassis AI. N-(m-[125I]iodophenyl)maleimide: An agent for high yield radiolabeling of antibodies. Int J Rad Appl Instrum B. 1992;19:289–295.