14.1 Plain Films Radiography
Description
Images of radiopaque tissues obtained by exposure of special photographic plates to ionizing radiation.
Uses in Ophthalmology
Plain films are of limited use in ophthalmology. They may be used to identify or exclude radiopaque intraorbital or intraocular foreign bodies. However, computed tomography (CT) is the study of choice to evaluate for foreign bodies, as CT has greater contrast sensitivity over conventional radiography. Plain films remain a valid screening modality before magnetic resonance imaging (MRI) if an occult metallic foreign body is suspected. Plain films should never be used in the setting of trauma or for the diagnosis of orbital fractures, as this needlessly exposes the patient to radiation without providing enough detail of orbital bone and soft tissue.
14.2 Computed Tomography
Description
CT uses ionizing radiation and computer-assisted formatting to produce multiple cross-sectional planar images. With the use of multidetector technology, CT now provides direct axial, coronal, and parasagittal images without the need for patient repositioning or data reformatting. Bone and soft tissue windows should always be reviewed in axial, coronal, and parasagittal orientations (see Figures 14.2.1 to 14.2.3). Orbital studies should be obtained using the lowest radiation dose necessary for diagnosis (as low as reasonably acceptable [ALARA]—see below). This is especially important in children; for accreditation by the American College of Radiology, health care facilities must maintain specific pediatric protocols for CT. Radiopaque iodinated contrast allows for more extensive evaluation of vascular structures and areas where there is a breakdown of the normal capillary endothelial barrier (as in inflammation).

FIGURE 14.2.1 Axial soft tissue window of inferior orbit shows abnormality, which is difficult to assess using this window.

FIGURE 14.2.2 Coronal soft tissue window shows a large blowout fracture of the orbital floor. This finding was missed on the axial study in Figure 14.2.1, demonstrating the importance of reviewing both axial and coronal images.

FIGURE 14.2.3 Coronal bone window shows the fracture again. In bone windows, the soft tissue detail fades, but bone detail is enhanced, allowing for better examination of bony anatomy.
Uses in Ophthalmology
1. Excellent for defining bone abnormalities such as fractures (orbital wall or optic canal), calcification, or bony involvement of a soft tissue mass.
2. Locating suspected intraorbital or intraocular metallic foreign bodies. Glass, wood, and plastic are less radiopaque and therefore more difficult to isolate on CT.
3. Soft tissue windows are good for determining some pathologic features, including orbital cellulitis/abscess, inflammation, and tumors. May be useful in determining posterior scleral rupture when clinical examination is inconclusive, but B-scan ultrasonography is likely more sensitive. CT should never be used to definitively rule out globe rupture; clinical examination and/or surgical exploration are more sensitive modalities.
4. Excellent for imaging paranasal sinus anatomy and disease.
5. Head CT is very sensitive for locating parenchymal, subarachnoid, subdural, and epidural hemorrhage in either acute or subacute settings. Although CT may show orbital hemorrhage as a nonspecific opacification within radiolucent orbital fat, it is significantly less sensitive for orbital hemorrhage than central nervous system hemorrhage.
6. Imaging modality of choice for most cases of thyroid eye disease. See 7.2.1, Thyroid Eye Disease.
7. Loss of consciousness or unwitnessed head trauma with poor history requires CT of the brain unless otherwise recommended by a neurosurgical consultant.
8. Note that CT of the brain does not provide adequate detail of the orbital anatomy, and orbital CT does not image the entire brain. Each site has its own specific, separate CT protocol.
Guidelines for Ordering an Orbital Study
1. Always order a dedicated orbital study if ocular or orbital pathology is suspected. Always include views of paranasal sinuses and cavernous sinuses.
2. Order both direct axial and direct coronal views if older scanners are used. Newer, multidetector CT scanners provide excellent views in coronal and parasagittal planes with very short scan times and no patient repositioning. Multidetector CT has supplanted older technology in the vast majority of hospitals and imaging centers.
3. When evaluating traumatic optic neuropathy, request 1-mm cuts of the orbital apex and optic canal to rule out bony impingement of the optic nerve. See 3.11, Traumatic Optic Neuropathy.
4. When attempting to localize ocular or orbital foreign bodies, order 1-mm cuts.
5. Intravenous contrast may be necessary for suspected infections or inflammatory conditions. Contrast is helpful in distinguishing orbital cellulitis from abscess. However, it is not mandatory to rule out orbital inflammation or postseptal involvement. Relative contraindications for contrast include renal failure, diabetes, congestive heart failure, multiple myeloma or other proteinemias, sickle cell disease, multiple severe allergies, and asthma. Check renal function and discuss the options with the radiologist for patients in whom renal insufficiency is suspected. Shellfish allergy is not a contraindication for CT contrast.
NOTE: The radiologist may recommend premedication with corticosteroids and antihistamines if contrast is required and a prior allergy is suspected. Follow the protocol recommended by your radiology department.
6. Obtain a pregnancy test before obtaining CT scans in females of childbearing age.
7. CT angiography (CTA) is helpful in diagnosing intracranial vascular pathology, including aneurysms. It is available on all multidetector CT scanners and may be more sensitive than magnetic resonance angiography (MRA) in specific clinical situations. However, it requires the use of intravenous iodinated contrast. Also note that CTA requires significantly more radiation than CT. CTA should be avoided if at all possible in children; MRA is the preferred modality in the pediatric age group.
8. CT scans may be obtained in children with careful consideration of the risk of radiation exposure versus benefits of performing the scan. According to the ALARA paradigm, it is best to avoid CT if possible in children unless there is no reasonable substitute. Check with your radiologist about using a pediatric protocol to limit radiation exposure. Each CT scan exposes children to a cumulative radiation dose that may increase the risk of malignancy over their lifetime. Radiation exposure from CT scans in children is of particular concern when serial imaging is required. In these cases, MRI/MRA is often a better choice for children, although intravenous sedation or general anesthesia may be needed because of the significantly longer scanning time.
NOTE: CT is an extremely valuable tool and is an acceptable imaging modality in children when necessary. Because of its easy availability, rapid scan time, excellent bone resolution, and ability to diagnose acute intracranial hemorrhage, CT is the modality of choice in head and orbital trauma in all age groups, including children.
14.3 Magnetic Resonance Imaging
Description
TABLE 14.3.1
Summary of Magnetic Resonance Imaging (MRI) Sequences9

aNote: High-flow areas create a dark area (“flow void”), which is helpful in identifying arterial structures (e.g., carotid siphon within the cavernous sinus).
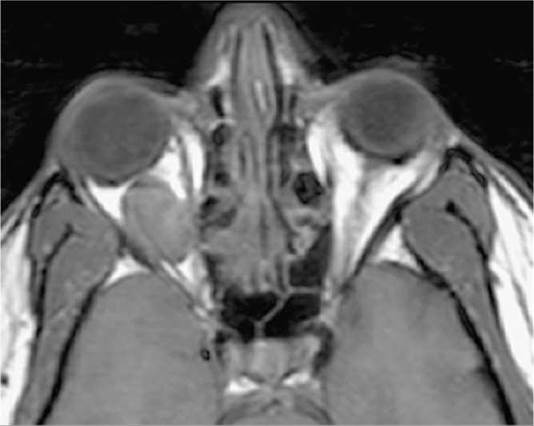
FIGURE 14.3.1 Axial Tl-weighted image without fat suppression or gadolinium. The vitreous is dark (hypointense) relative to the bright signal from fat. A well-circumscribed mass is clearly visible in the right orbit, also hypointense. Most orbital lesions are dark in T1 prior to gadolinium injection. The notable exceptions are listed in Table 14.3.2.
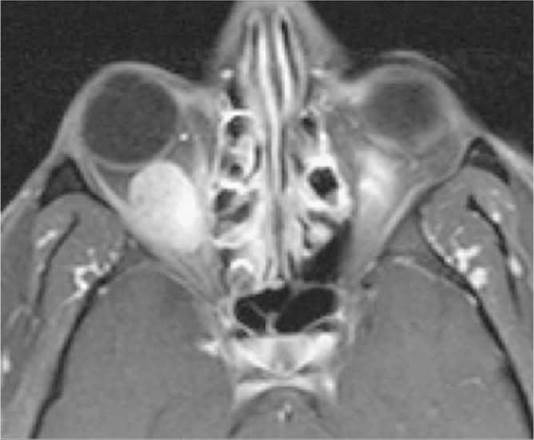
FIGURE 14.3.2 Axial T1 image with fat suppression and gadolinium. Note how both the vitreous and fat are dark, but the extraocular muscles become bright. The orbital mass is now clearly visible. This technique should be performed in all orbital magnetic resonance imagings.

FIGURE 14.3.3 Axial T2-weighted image. The vitreous is hyperintense (bright) relative to the orbital fat. The lesion is also bright but in some cases may be isointense with the surrounding fat.
1. MRI uses a large magnetic field to excite protons of water molecules. The energy given off as the protons re-equilibrate to their normal state is detected by specialized receivers (coils), and that information is reconstructed into a computer image.
2. MRI obtains multiplanar images without loss of resolution.
3. The basic principles of MRI are listed in Table 14.3.1. Figures
14.3.1 to 14.3.3 provide specific examples.
NOTE: In general, conventional “closed” MRI scanners provide better resolution and fat suppression than open scanners, unless the open scanner contains a stronger magnet. As a rule, always try to use conventional “closed” scanners for orbital studies.
4. Contrast studies can be ordered using gadolinium, a well- tolerated, non-iodine-based paramagnetic agent.
5. Diffusion weighted imaging (DWI) is a technique that measures the Brownian motion of water within tissue. Lesions that are tightly packed by cells with a high nuclear to cytoplasmic ratio will minimize Brownian motion and “restrict diffusion.” DWI is often coupled with an apparent diffusion coefficient (ADC) image. The use of DWI/ADC in the orbit is limited because of proximity of the CNS and limitations of resolution, but it can be useful in certain disorders such as lymphoma.
Uses in Ophthalmology
TABLE 14.3.2
Tissues/Lesions That Appear Bright (Hyperintense) Relative to Vitreous on T1 Before Gadolinium Injectiona
|
Tissues/Lesions |
Examples |
|
Fat |
Lipoma, liposarcoma |
|
Mucus/proteinaceous material |
Dermoid cyst, mucocele, dacryocystocele, craniopharyngioma |
|
Melanin |
Melanoma |
|
Subacute blood (3 to 14 days old) |
Lymphangioma with blood cyst, hemorrhagic choroidal detachment, subperiosteal orbital hematoma |
|
Certain fungal infections (iron scavengers) |
Aspergillus |
aNote: Most orbital lesions will become hyperintense after gadolinium infusion.

FIGURE 14.3.4 Magnetic resonance imaging with fluid-attenuated inversion recovery sequence of demyelinating lesions in multiple sclerosis.
1. Excellent for defining the extent of orbital/central nervous system masses. Signal-specific properties of certain pathology may be helpful in diagnosis (see Table 14.3.2).
2. Poor bone definition (e.g., fractures).
3. Excellent for diagnosing intracranial, cavernous sinus, and orbital apex lesions, many of which affect neuro-ophthalmic pathways.
4. Gadolinium is essential in defining lesion extent in suspected neurogenic tumors (meningioma, glioma).
5. All patients with clinical signs or symptoms of optic neuritis from suspected demyelinating disease should undergo brain MRI with gadolinium. Fluid-attenuated inversion recovery images are especially useful. See Figure 14.3.4 and 10.14, Optic Neuritis.
6. For orbital studies, fat suppression (also called fat saturation) should always be used in conjunction with intravenous gadolinium to enhance the visualization of the underlying pathology (e.g., optic neuritis, fat-containing lesions). Note that gadolinium without fat suppression may cause pathology to “disappear” into surrounding orbital fat.
7. DWI can help differentiate the various phases of cerebral infarction (e.g., hyperacute, acute, subacute, and chronic). DWI may also help to distinguish tightly packed, highly cellular tumors (e.g., lymphoma) from inflammation.
Guidelines for Ordering the Study
1. For the vast majority of orbital studies, a head coil is indicated to provide bilateral orbital views extending to the optic chiasm.
2. Intravenous gadolinium is a useful adjunct for evaluating ocular, orbital, and perineural masses. In patients who have kidney failure, sepsis, or recent major surgery, risk of developing nephrogenic systemic fibrosis (NSF) may be a contraindication. NSF is a rare but devastating complication of gadolinium that occurs weeks to months after administration. NSF is characterized by a scleroderma-like fibrosis of the skin, especially over extremities and trunk, which may involve the viscera. There is no known effective therapy or prophylaxis. Evaluate renal function in patients in whom renal insufficiency is suspected and discuss options with your radiology and nephrology departments. Recently, gadolinium deposition with specific areas of the brain has been noted in patients undergoing serial imaging. The clinical significance of this finding is unknown and is under review by the Food and Drug Administration.
3. Contraindications to MRI include severe claustrophobia, marked obesity, some cardiac pacemakers, some cardiac valves, suspected magnetic intraocular/intraorbital foreign bodies, spinal stimulators, vagal nerve stimulators, stapes implants, and specific breast and penile implants. Titanium plates and newer aneurysm clips are MRI safe, as are gold and platinum weights placed in the eyelids. When in doubt, ask the radiologist to look up the specific device in an MRI safety catalog. Any patient with a poorly documented implanted device should NOT be scanned with MRI until the issue is clarified. On occasion, specific types of pacemakers must be turned off and reset for an MRI.
14.4 Magnetic Resonance Angiography
Description
Special application of MRI technology in which signal from flowing blood is augmented while signal from stationary tissues is suppressed. MRA allows for three-dimensional rotational reconstruction. Importantly, MRA is not performed with gadolinium and is therefore useful in patients with a history of renal disease and other contraindications to gadolinium or CT contrast.
Uses in Ophthalmology
1. Suspected carotid stenosis, occlusion, aneurysm, or dissection.
2. Suspected intracranial and orbital arterial aneurysms (e.g., pupilinvolving third cranial nerve palsy), arteriovenous malformations, and acquired arteriovenous communications.
3. Suspected orbital or intracranial vascular mass. Note that MRA is best for imaging high-flow and large-caliber lesions. Lower-flow lesions (e.g., varix) are not well seen. Both CTA and MRA have limited potential in visualizing cavernous sinus fistulas; color Doppler studies may be more sensitive in making this diagnosis, but conventional arteriography remains the most sensitive and specific modality.
Guidelines for Ordering the Study
Conventional cerebral arteriography remains the gold standard for diagnosis of vascular lesions but carries potential morbidity and mortality in certain populations. Currently, the limit of MRA is an aneurysm larger than about 2 mm. However, the sensitivity is highly dependent on several factors: hardware, software, availability of adequate clinical history, and the experience of the neuroradiologist. Note that in one large study, the two most critical factors in missing a posterior communicating artery aneurysm (PCOM) on MRI/MRA were lack of clinical history by the referring physician and a reading by a radiologist without fellowship training in neuroradiology. Also note that most PCOM aneurysms that manifest as an oculomotor nerve paresis or palsy are at least 4 mm in diameter. Despite these potential limitations, MRA remains a safe and sensitive screening test, especially when coupled with MRI for concomitant soft tissue imaging.
14.5 Magnetic Resonance Venography
Magnetic resonance venography (MRV) is helpful in diagnosing central venous sinus thrombosis. MRI and MRV are essential parts of the workup of any patient presenting with bilateral optic disc swelling. See 10.15, Papilledema.
14.6 Conventional Arteriography
Description
This interventional examination entails intra-arterial injection of radiopaque contrast followed by rapid-sequence x-ray imaging of the region of interest to evaluate the transit of blood through the regional vasculature. Unlike MRA or CTA, catheter arteriography allows the option of simultaneous treatment of lesions by intravascular techniques. Cerebral arteriography is the gold standard for diagnosing intracranial aneurysms, but it is being replaced as the initial imaging modality in many centers by CTA; note that, unlike catheter angiography, CTA is solely a diagnostic modality. Cerebral arteriography carries a stroke risk of 0.3% to 1.8% in patients without transient ischemic attack or previous stroke.
Uses in Ophthalmology
1. Suspected arteriovenous malformations, carotid cavernous fistulas, cavernous sinus fistula, aneurysm, and vascular masses (e.g., varix).
2. Evaluation of ocular ischemic syndrome or amaurosis fugax due to suspected atherosclerotic carotid, aortic arch, or ophthalmic artery occlusive disease. Usually carotid Doppler ultrasound, MRA, or CTA is adequate for diagnosis.
NOTE: Conventional arteriography is generally contraindicated in patients with suspected carotid artery dissection (catheter placement may propagate the dissection). That said, in some centers, arteriography is used to stent areas of carotid dissection.
14.7 Nuclear Medicine
Description
Nuclear medicine imaging uses radioactive contrast (radionuclide) that emits gamma radiation, which is then gathered by a gamma-ray detector. The classic types of radionuclide scanning known to ophthalmologists include bone scanning, liver-spleen scanning, and gallium scanning. Positron emission tomography (PET) is useful in determining metabolic activity within a lesion and is usually coupled with CT for anatomic detail.
Uses in Ophthalmology
1. Scintigraphy (e.g., with technetium-99): Useful for assessing lacrimal drainage physiology in patients with inconsistent irrigation testing.
2. Systemic gallium scan: Useful for detecting extraocular sarcoidosis and Sjogren syndrome. The use of gallium scanning for the diagnosis of sarcoidosis has been largely replaced by other testing, including serum angiotensin-converting enzyme and chest CT. See 12.6, Sarcoidosis.
3. Technetium-99m-tagged red blood cell study: Occasionally used to distinguish cavernous hemangioma from other solid masses in the orbit. This modality has been largely supplanted by specific MRI techniques.
4. PET/CT: The use of PET/CT for the diagnosis and management of orbital disease is still an evolving technique. Limitations in this area include the high background metabolic activity of the adjacent CNS (which may mask orbital abnormalities), the size of the orbital pathology (current PET scanners have a resolution limit of about 7 mm), and the relatively indolent nature of most orbital lymphomas (decreasing the intensity of the signal on PET). PET is extremely useful in the diagnosis and surveillance of systemic pathologies, including metastases and aggressive subtypes of lymphoma. At present, the primary role of PET in the management of orbital disease is for diagnosis, surveillance, and response to therapy of any systemic component of the pathologic process. PET is rarely obtained alone; it is typically coupled with a low-resolution CT to provide anatomic information (PET/CT).
14.8 Ophthalmic Ultrasonography
A-SCAN
Description
A-scan, or amplitude-modulated ultrasonography (US), uses ultrasonic waves (8 to 12 MHz) which travel through different tissues at different velocities in order to generate linear distance versus amplitude of reflectivity curves of the evaluated ocular and orbital tissues. A-scans are one-dimensional and used for measuring and characterizing the composition of tissues on the basis of the reflectivity curves. Not all A-scan instruments are standardized (see Figure 14.8.1).

FIGURE 14.8.1 Normal A-scan ultrasonography.
Uses in Ophthalmology
1. Primary use in ophthalmology is measurement of axial length of the globe. This information is critical for intraocular lens (IOL) power calculations for cataract surgery. Axial length information can also be used to identify certain congenital disorders such as microphthalmia, nanophthalmos, intraocular tumor size, intrinsic tumor vascularity, and congenital glaucoma. A-scan is used to follow patients with congenital glaucoma.
2. A standardized A-scan probe calibrated on an S curve can be used for diagnostic identification of the echogenicity characteristics of masses in the globe or orbit.
3. Specialized A-mode ultrasonography can be used for corneal pachymetry (measurement of corneal thickness).
Guidelines for Ordering the Study
1. When used for IOL power calculations, make sure to check both eyes. The two eyes are usually within 0.3 mm of each other.
2. Spikes along the baseline should be sharply rising at 90 degrees.
3. If needed, keratometry readings should be obtained prior to scan or 30 minutes after the scan for accurate results.
B-SCAN
Description
(See Figure 14.8.2.)

FIGURE 14.8.2 Normal B-scan ultrasonography.
B-scan, or brightness-modulated US, gives real-time, twodimensional (cross-sectional) images of the eye that span from the posterior aspect of the cornea to the posterior aspect of the globe. The B-scan probe may be placed directly on the globe (after anesthetic is applied) or on the closed eyelid, which is preferred in cases of trauma or poorly cooperative patients.
Uses in Ophthalmology
1. Define ocular anatomy in the presence of media opacities (e.g., mature cataract, hyphema, corneal opacity, vitreous hemorrhage, trauma) in order to evaluate retinal and/or choroidal pathology.
2. Diagnosis of scleral rupture posterior to the muscle insertions or when media opacities prevent direct visualization.
3. Identify intraocular foreign bodies especially if made of metal or glass (spherical objects have a specific echo shadow); wood or vegetable matter has variable echogenicity; can also give a more precise location if the foreign body is next to the scleral wall.
4. Evaluation of intraocular tumor/mass consistency and vascularity, retinal detachment, choroidal detachment (serous versus hemorrhagic), and optic disc abnormalities (e.g., optic disc drusen, coloboma). Also used to monitor intraocular tumor/mass over time for response to treatment.
5. Screening of intraocular pathology (especially choroidal melanoma) in blind eyes prior to evisceration.
6. Evaluation of the posterior sclera and anterior orbit for signs of inflammation in cases of suspected posterior scleritis.
Guidelines for Ordering the Study
1. If used in the setting of trauma to determine unknown scleral rupture, the probe is used over closed eyelids with immersion in copious amounts of sterile methylcellulose, such that no pressure is placed on the globe. The gain must be set higher to overcome the sound attenuation of the eyelids. Known ruptured globe is a relative contraindication to B-scan US.
2. When scleral integrity is not in question, B-scan US should be performed dynamically to help differentiate pathologic conditions, such as retinal detachment versus posterior vitreous detachment. To locate the exact location of retinal pathology (e.g., retinal tear, tumor), the scan should be performed directly on the globe.
3. Dense intraocular calcifications (such as those occurring in many eyes with phthisis bulbi) result in poor-quality images.
4. Silicone oil and intraocular gas in the vitreous cause distortion of the scanned image, and therefore the study should be performed in an upright patient to improve image quality.
ULTRASONOGRAPHIC BIOMICROSCOPY
Description
(See Figure 14.8.3.)

FIGURE 14.8.3 Normal anterior ultrasonographic biomicroscopy.
Ultrasonographic biomicroscopy uses ultrahigh frequency (50 to 100 MHz) B-mode US of the anterior one-fifth of the globe to give cross sections at near-microscopic resolution. Uses a water-bath eyelid speculum with viscous liquid in the bowl of the speculum.
Uses in Ophthalmology
1. Excellent for defining the following anatomic sites: Corneoscleral or limbal region, anterior chamber angle, iris, ciliary body, and sulcus. Useful for anterior segment detail and identification of various pathologic conditions (e.g., small anterior ocular foreign bodies, ciliary body mass/cyst, plateau iris, IOL location).
2. Unexplained unilateral angle narrowing or closure.
3. Suspected cyclodialysis.
Guidelines for Ordering the Study
Known ruptured globe is a contraindication to the study.
ORBITAL ULTRASONOGRAPHY/DOPPLER
Description
Uses B-mode US coupled with Doppler technology to visualize flow in the vessels in the orbit.
Uses in Ophthalmology
1. Superior ophthalmic vein pathology: Cavernous sinus fistula, superior ophthalmic vein thrombosis.
2. Orbital varix.
3. Arteriovenous malformations.
4. Vascular disease including central retinal artery occlusion, central retinal vein occlusion, ocular ischemic syndrome, and giant cell arteritis.
14.9 Photographic Studies
Description
Various methods of imaging the eye(s) or selected regions of the eye(s), using white light or various spectral wavelengths of light.
Types of Ophthalmic Photographic Imaging Studies

FIGURE 14.9.1 Normal fundus photograph.

FIGURE 14.9.2 Normal fundus autofluorescence.
1. Documentary photography: Color pictures of the face, external eye, cornea, anterior segment, and fundus (white light or red-free lighting). (See Figure 14.9.1.)
2. Fundus autofluorescence (FAF): Imaging modality that takes advantage of the naturally and pathologically occurring fluorophores in the fundus. Provides sensitive information regarding the health of the retinal pigment epithelium (RPE) and allows early detection and monitoring of a variety of conditions such as age-related macular degeneration (AMD), macular dystrophies, and medication toxicity. Additionally, FAF is useful in the evaluation of certain ocular tumors, specifically, choroidal nevi and melanomas. (See Figure 14.9.2.)
3. Specular microscopy: Contact and noncontact photographic techniques used to image the corneal endothelium. The images can then be used to evaluate the quality and quantity of the endothelial cells.
4. Adaptive optics: A super high-resolution means of imaging the retina, allowing identification of rod and cone morphology, useful in delineating retinal dystrophies.
14.10 Intravenous Fluorescein Angiography
Description
(See Figure 14.10.1.)

FIGURE 14.10.1 Normal intravenous fluorescein angiography.
Intravenous fluorescein angiography is a type of angiographic photography that does not rely on ionizing radiation or iodine-based contrast. After intravenous injection of fluorescein solution (usually in a hand or arm vein), rapid-sequence photography is performed by using a camera with spectral excitation and barrier filters. Fluorescein sodium absorbs blue light, with peak absorption and excitation occurring at wavelengths of 465 to 490 nm. Fluorescence then occurs at the yellow-green wavelengths of 520 to 530 nm. The fluorescein molecule is 80% protein bound and does not pass through the tight junctions of a healthy blood-retinal barrier (RPE and retinal capillaries are impermeable, whereas Bruch membrane and choriocapillaris lack tight junctions and are freely permeable).
Phases of the Intravenous Fluorescein Angiography
1. Choroidal filling (background fluorescence): Begins 8 to 15 seconds after injection. Choroid is normally completely filled within 5 seconds after dye appearance within the tissue.
2. Arterial phase: Starts 1 to 2 seconds after choroidal filling.
3. Arteriovenous phase (laminar flow).
4. Venous phase: Arteriovenous transit time is the time from the first appearance of dye within the retinal arteries of the temporal arcade until the corresponding veins are completely filled, normally <11 seconds.
5. Recirculation phase: Occurs 45 to 60 seconds after arterial phase.
6. Late phase: Occurs 10 to 30 minutes post injection.
Foveal dark spot can result from xanthophyll pigment in the outer plexiform layer or tall RPE cells with increased melanin or lipofuscin. The foveal avascular zone is the central area which has no retinal capillaries (300 to 500 microns in diameter).
Describing an Abnormal Study
Hyperfluorescence
1. Leakage: Fluorescein penetrates the blood-retinal barrier and accumulates subretinally, intraretinally, or preretinally. Hyperfluorescence increases in size and brightness as study progresses (e.g., choroidal or retinal neovascularization, central serous chorioretinopathy [CSCR], cystoid macular edema [CME]).
2. Staining: Mild fluorescence appears in the late phase while its borders remain fixed (e.g., scar).
3. Pooling: Accumulation of fluorescein in fluid-filled space in retina or choroid. The margins of the space trapping fluorescein are distinct (e.g., pigment epithelial detachment [PED], CSCR).
4. Window or transmission defect: Focal area of hyperfluorescence without leakage usually due to RPE atrophy that appears early and stays stable in brightness (e.g., geographic atrophy, RPE rip, laser scar).
5. Autofluorescence: Structures that naturally fluoresce can be captured on film prior to intravenous fluorescein injection (e.g., optic nerve drusen and lipofuscin).
Hypofluorescence
1. Blockage: Due to optical density such as blood, pigment, or fibrous tissue interposed between the camera and the choriocapillaris.
2. Nonperfusion: Nonfilling vessel(s) causing relative or absolute hypofluorescence (e.g., central retinal artery occlusion). Applies to both capillaries and larger vessels.
Uses in Ophthalmology
1. Used to image retinal, choroidal, optic disc, iris vasculature, or a combination of these. It is used for diagnosis and therapeutic planning (e.g., retinal lasers).
2. Transit times between injection and appearance of dye in the choroid, retinal arteries, and veins also can be used to evaluate vascular flow. Arm to retina time is less accurate than intraretinal circulation times.
3. Suspected retinal ischemia (capillary nonperfusion) and neovascularization from various conditions (e.g., diabetes).
4. Suspected choroidal neovascularization (CNV) from various diseases (e.g., AMD).
Guidelines for Ordering the Study
1. Side effects of intravenous fluorescein are nausea (10%), vomiting (2%), hives, pruritus, and vasovagal response. True anaphylaxis is rare. Death may occur in 1 out of 220,000 injections. Extravasation into extracellular space at the injection site can produce local necrosis. Treat with cool compresses. Excretion in urine occurs within 24 to 36 hours. Urine will be bright yellow; remember to warn all patients about this.
2. Because it is a photographic method, moderately clear media is required for visualization.
14.11 Indocyanine Green Angiography
Description
Photographic method of ocular angiography similar to intravenous fluorescein angiography utilizing tricarbocyanine dye, an iodine- based dye. Indocyanine green (ICG) angiography differs in that fluorescence occurs in the infrared spectrum (835 nm), allowing for penetration through pigment, fluid, and blood. ICG provides better evaluation of the choroidal vasculature. ICG excitation occurs at 805 nm, with fluorescence at 835 nm. The ICG molecule is approximately 95% protein bound.
Uses in Ophthalmology
1. Suspected occult CNV.
2. Suspected recurrent CNV after prior treatment.
3. Suspected CNV with retinal PED.
4. Suspected polypoidal choroidal vasculopathy. See 11.18, Idiopathic Polypoidal Choroidal Vasculopathy (Posterior Uveal Bleeding Syndrome).
5. Other accepted uses: Identifying feeder vessels in retinal angiomatous proliferation lesions in AMD, chronic CSCR, certain inflammatory conditions (e.g., birdshot choroidopathy), and occasionally helpful in diagnosing certain posterior segment tumors.
Guidelines for Ordering the Study
1. Contraindicated in patients with iodine or shellfish allergies.
2. Most common side effect of ICG dye administration is a vasovagal response.
3. Excreted by hepatic parenchymal cells via bile.
14.12 Optical Coherence Tomography
Description
(See Figure 14.12.1.)
FIGURE 14.12.1 Normal optical coherence tomography.
Optical coherence tomography (OCT) provides noninvasive, noncontact two-dimensional or three-dimensional images by measuring optical reflections of light. In this manner, OCT is similar to US except that OCT is based on the reflection of light, not sound. The OCT scanner sends low-coherence light (~820-nm wavelength for posterior segment imaging and ~ 1310-nm wavelength for anterior segment imaging) emitted by a superluminescent diode to the tissue to be examined and to a reference beam. The time delays of the light reflections from retinal structures are recorded by an interferometer. Using a reference mirror, these light reflections are then translated into an imaged object with a high resolution of up to 3 microns. The most highly reflective structures are the nerve fiber layer and the RPE. Highly reflective lesions include dense pigmentation, scar tissue, subhyaloid hemorrhage, and hard exudates. Low reflectivity in pathologic conditions include intraretinal or subretinal fluid.
Spectral domain OCT has largely replaced time domain OCT due to increased image resolution, shorter acquisition time, and fewer artifacts. Enhanced depth imaging OCT is a technique used to improve the detail of the choroid. Newer modalities such as swept- source OCT and multicolor laser imaging are being evaluated. Swept- source OCT provides better resolution of retinal and choroidal structures within the same scan. Multicolor imaging complements OCT and uses red, green, and blue lasers to provide a topographic map of the outer, mid, and inner retina, respectively. OCT angiography (OCT-A) allows for rapid and noninvasive imaging of the retinal microvasculature and may also be applied to the anterior segment. Limitations of OCT-A include a small field of view and need for good patient cooperation.
Uses in Ophthalmology
1. Retinal diseases, including macular edema, macular atrophy, CSCR, AMD, CNV, CME, retinal detachment, PED, retinal tumors, drusen, and hard exudates.
2. Vitreoretinal interface abnormalities, including macular holes, cysts, epiretinal membranes, subhyaloid hemorrhage, and vitreoretinal strands or traction.
3. Suspected glaucoma, including quantification of the nerve fiber layer thickness, macular thickness, and optic nerve cup characteristics.
4. Suspected optic neuritis, other optic neuropathies, optic nerve drusen, disc edema, and multiple sclerosis.
5. Anterior segment pathology, such as Descemet detachment in an edematous cornea.
Guidelines for Ordering the Study
Requires patient’s ability to fixate and relatively clear media.
14.13 Confocal Scanning Laser Ophthalmoscopy
Description
Confocal scanning laser ophthalmoscopy is a noninvasive imaging technique used to obtain high-resolution optical images and evaluate the topography of ocular structures. This confocal optical system provides a contour map of the desired structure in a process known as “optical sectioning.” The system aims to detect reflected light from a very thin optical plane, the focal plane. A series of “focal planes" or images may be recorded and combined to create a three-dimensional image (e.g., Heidelberg retinal tomography).
Uses in Ophthalmology
1. Suspected optic nerve disease, including glaucoma and papilledema.
2. Suspected fundus elevations, including macular edema and choroidal nevi.
Guidelines for Ordering the Study
1. Requires patient’s ability to fixate and relatively clear media.
2. Because the hallmark of the test is to provide comparative data, subsequent tests in the same patient need accurate alignment in the same focal plane to provide useful information.
14.14 Confocal Microscopy
Description
The confocal microscope optically sections the cornea to noninvasively obtain structural information of the different corneal layers.
Uses in Ophthalmology
The high level of detail available may be helpful in the detection of corneal microorganisms such as Acanthamoeba and fungi. It may also permit visualization of noninfectious changes such as those seen in corneal dystrophies, iridocorneal endothelial syndrome, corneal neuropathies, and epithelial downgrowth. It is excellent for imaging the corneal endothelium and obtaining endothelial cell density measurements.
14.15 Corneal Topography and Tomography
Description
Standard keratometry measures the radius of corneal curvature and then converts the radius into dioptric corneal power. Computerized corneal topography is performed using various methods, including Placido disc analysis and rasterstereography. These techniques project an image onto the cornea, most commonly a series of concentric rings, and analyze the reflection to determine corneal curvature. They can provide information on anterior corneal power and regularity. Simulated keratometry readings can be generated and the results can be represented in graphical formats, such as a variety of color maps.
Corneal tomography, the computerized reconstruction of multiple images of cornea, can give detailed information about the anterior and posterior corneal curvatures as well as corneal thickness. These techniques include scanning slit, rotating Scheimpflug photography, and anterior segment OCT-based systems. Scanning slit and rotating Scheimpflug imaging are particularly helpful in imaging posterior corneal elevation. Rotating Scheimpflug photography and anterior segment OCT can image corneal and anterior segment anatomy.
Uses in Ophthalmology
Detecting irregular astigmatism secondary to keratoconus, pellucid marginal degeneration, corneal surgery, corneal trauma, and contact lens warpage; evaluating depth of opacities in inherited corneal dystrophies and corneal scars from inflammatory or infectious etiologies. Has the ability to image normal and abnormal corneal and anterior segment structures. It may be helpful in identifying the cause of decreased vision in patients with no known cause. It is useful for refractive surgical screening and imaging the postkeratorefractive cornea. Serial imaging is critical in the evaluation of progression of corneal ectasias.