Definition (Pediatr Rev 2004;25:350; Pediatr Rev 2011;32:240)
• Net gain of H+ or loss of HCO3 and can be divided into AG or NAG
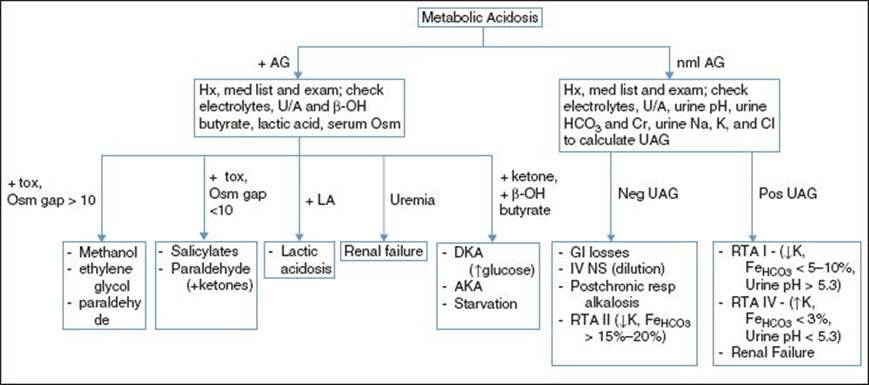
• AG acidosis: Implies presence of unmeasured anions; ↑ organic acids from ingestion, production, inborn errors of metab, or ↓ excretion 2/2 renal failure
• AG = [Na+] − ([Cl−] + [HCO3−]); normal defined as 8–12 w/ normal values believed to be higher in pts <2 yo (some use 16 +/− 4 mEq/L as normal for this group)
• Figge formula corrects AG for albumin: Adjusted AG = observed AG + 2.5 (nl albumin − observed albumin)
• NAG acidosis: 2/2 inability to excrete H+ or loss of HCO3 from GI or urinary tracts
• Severe met acidosis: HCO3 ≤ 8 mEq/L (if nl resp compensation)
Clinical Manifestations (Pediatr Rev 1996;17:395; Pediatr Rev 2011;32:240)
• Nl response to met acidosis is hyperpnea (deep, pauseless respirations) w/ resultant 4–8×’s increase in minute ventilation. As acidosis worsens, resp muscle strength decreases and WOB increases → resp failure
• At pH <7–7.1, cardiac contractility can be impaired and the risk of malignant arrhythmias is increased. Can p/w arteriolar dilation, HoTN, pulmonary edema
• CNS: Cerebral edema, lethargy, coma
• Other: Oxyhemoglobin dissociation curve shifts right, hyperK, insulin resistance, protein denaturing
Etiologies (Rose & Post. Clinical Physiology of Acid-Base & Electrolyte Disorders 2001:535; Pediatr Rev 2011;32:240)
• AG acidosis: Due to exogenous and endogenous H+ excess: MUD PILES
• Methanol: Found in wood alcohol, varnishes, shellac, and sterno, metabolized to formaldehyde, Osm gap >10; p/w sx 12–36 hr after ingestion w/ weakness, N/V, can result in blindness, coma, and death
• Uremia (2/2 renal failure w/ decreased clearance of sulfate, urate, phosphate)
• Diabetic ketoacidosis, starvation ketosis
• Paraldehyde (solvent, anticonvulsant and hypnotic, not available in the United States but still used elsewhere)
• Iron, isoniazid, and ingestions (other): Toluene (glue sniffing), sulfur, etc.
• Lactic acidosis: Most common, 2/2 anaerobic metab, can be 2/2 ↑ production (severe hypoxia, ischemia, shock, CO or cyanide poisoning, szr, inborn errors of metab) or ↓ clearance (hypoperfusion, liver disease)
• Ethylene glycol (in antifreeze, w/ glycolate, oxalate, and Osm gap >10; Ethylene glycol ingestion p/w drunkenness or even coma, then pulmonary edema and then renal failure, consider fomepizole and/or dialysis for rx), Ethanol (alcoholic ketoacidosis)
• Salicylate: Results in combination resp alkalosis and met acidosis, tinnitus
• If concern for ingestions, check Osmolar gap = Measured Osm − Calculated Osm
• Calculated Osm = 2[Na+] + [glucose]/18 + [BUN]/2.8 + [EtOH]/4.6
• If Osm gap >10, presence of unmeasured Osm; methanol, ethylene glycol, etc.
• W/ +AG; to check if >1 cause for low HCO3 can check Δ/Δ = Δ AG/Δ HCO3
• Δ/Δ <1; loss of HCO3 greater than expected (AG acidosis + NAG acidosis)
• Δ/Δ = 1–2; typical AG acidosis as almost 1:1 buffering of H+ w/ HCO3
• Δ/Δ >2; loss of HCO3 less than expected (AG acidosis + metabolic alkalosis)
• NAG acidosis: 2/2 ↑ GU or GI loss of HCO3 or inability to excrete H+: HARD UP
• Hyperalimentation, Hypoaldosteronism, HyperPTH
• Acetazolamide, Ammonium chloride
• RTA (see later), Renal loss 2/2 rapid correction of chronic resp alkalosis
• RTA II (proximal RTA): Dec HCO3 reabsorption, p/w normal or low serum [K+], variable urine pH, fractional excretion of HCO3 (FeHCO3) >15–20%
• Can result in rickets or osteomalacia
• Carbonic anhydrase inhibitors (e.g., acetazolamide) impair HCO3 reabsorption
• Treat w/ 10–15 mEq/kg HCO3 divided multiple times a day
• Renal dysfunction: Some cases of renal failure and RTA
• RTA IV (hypoaldosteronism): W/ normal or inc serum [K+], urine pH < 5.3, FeHCO3 < 3%, check serum aldo; treat w/ HCO3 1–3 mEq/kg qd
• RTA I (distal RTA): Dec H+ secretion w/ urine pH >5.3, low serum [K+], and fractional excretion of HCO3 <3% in adults but can be 5–10% in children
• Can be 2/2 autoimmune process, hyperPTH, amphotericin
• Can result in nephrocalcinosis or nephrolithiasis
• Treat w/ daily HCO3 4–14 mEq/kg qd in children, 1–2 mEq/kg qd in adults
• Diarrhea (most common cause) and other GI losses (ostomy, etc.)
• Ureteral diversion (ureteroenterostomy/ureterosigmoidostomy → intestinal HCO3− loss)
• Parenteral NaCl (common cause, due to rapid dilution of HCO3− by Cl−; similar effect from KCl or MgCl2), Pancreatic fistula
• Urine AG can distinguish between situations w/ intact NH4+ secretion (GI losses RTA II, rapid correction of chronic resp alkalosis) vs. those where secretion of NH4+ is impaired (renal failure, RTA I, and IV)
• UAG is inaccurate w/ high AG acidosis or w/ significant volume depletion
Management (Pediatr Rev 2004;25:350)
• Correct underlying abnormalities as described above
• Sodium-bicarb is the agent of choice for severe metabolic acidosis (especially NAG) but controversial as evidence of improved outcomes is weak
• Amt bicarb to infuse = (weight in kg) × (15 − [measured HCO3]) × (0.5)
• Mix amount calculated in hypotonic solution and infuse over 1 HR
• Provides enough bicarb to correct pH >7.2 or bicarb concentration >15 mEq/L
• Rapid correction can result in acidification of CSF (w/ worsening symptoms); volume overload; development of alkalemia, hypocalcemia, and respiratory depression