Pamela J. Okada
Jennifer L. Bercaw-Pratt
HIGH-YIELD FACTS
• Ectopic pregnancy, as the leading cause of maternal mortality during the first half of pregnancy, should be considered in patients with abdominal pain and vaginal bleeding.
• Examination of the cervix may exacerbate hemorrhage in patients with placenta previa.
• Placental abruption may present with concealed hemorrhage or vaginal bleeding and typically includes vaginal bleeding, abdominal pain, uterine tenderness, and contractions. Ultrasound is not sensitive for its diagnosis.
• The relatively high prevalence of preeclampsia should warrant obtaining a routine blood pressure and consideration of screening for proteinuria.
• HELLP syndrome is defined as hemolysis, elevated liver enzymes, and low platelets (<100,000/mm3); lower platelet counts are associated with higher mortality.
• Shock may be difficult to diagnose in the pregnant patient as blood volume, heart rate, and respiratory rate are physiologically increased during pregnancy.
Although the teen pregnancy rate decreased 40% from 1990 to 2008 due to an increase in the use of contraception, a decrease in sexual activity, and effective pregnancy prevention programs, there are still more than 750,000 teen pregnancies a year.1–3 Pregnancy rates among non-Hispanic black and Hispanic teenagers remain disproportionately high.1 Pregnant teens are less likely to receive prenatal care and are more likely to partake in high-risk behaviors such as smoking and consumption of alcohol during pregnancy. Teen mothers are at risk of not completing their education and living in poverty as compared to their peers.
![]() VAGINAL BLEEDING IN PREGNANCY
VAGINAL BLEEDING IN PREGNANCY
Bleeding in the first trimester occurs in 25% of patients.4 Common etiologies include ectopic pregnancy, threatened abortion, spontaneous abortion, sexually transmitted infections, and trauma. In later trimesters the causes include placenta previa and placental abruption.
ECTOPIC PREGNANCY
An ectopic pregnancy is defined as the implantation of the blastocyst outside the endometrial lining of the uterine cavity. Ectopic pregnancy is an important cause of maternal morbidity and mortality during the first half of pregnancy in the United States. There is an overall incidence of 2% and an incidence of 1.8% in women presenting to the emergency department (ED) with either first trimester bleeding and/or abdominal pain.5 Higher rates of mortality in adolescents are largely due to delays in seeking care. Greater than 95% of ectopic pregnancies are located in the fallopian tube.5 Risk factors include prior ectopic pregnancy, prior tubal surgery, genital tract infections leading to pelvic inflammatory disease (PID), and intrauterine device.5
Most cases present within the first 8 weeks of gestation with abdominal pain or abnormal vaginal bleeding. Pelvic or abdominal pain and exquisite tenderness are the most common complaints. Others present with a late menstrual cycle and abnormal vaginal bleeding, which can be confused with menses. In the case of a ruptured ectopic with intra-abdominal hemorrhage and hypovolemia, the clinical presentation may include dizziness and/or presyncope.6
The classically described but rare presentation of ectopic pregnancy is tenderness on abdominal examination, shock, and an adnexal mass. More commonly, the abdominal examination may be unremarkable or there may be adnexal and/or cervical motion tenderness. Laboratory and radiographic modalities that aid in the diagnosis are listed in Table 100-1.5
|
TABLE 100-1 |
Diagnosis of Ectopic Pregnancy |

In an unruptured ectopic pregnancy, methotrexate, a folic acid antagonist, can be used for the medical management of an early ectopic pregnancy. Factors that favor improved success with methotrexate include hemodynamic stability without maternal hemorrhage, beta-HCG <15,000 mIU/mL, <3.5 cm gestational sac, and absence of fetal heart tones.5 Contraindications to medical management include unstable vital signs, maternal hemorrhage, tubal rupture, known liver or renal disease, or other contraindications to methotrexate; if contraindicated or not desired, then surgical management is appropriate. In a patient with a ruptured ectopic pregnancy, emergency management includes assurance of airway and breathing and cardiovascular stabilization. Rapid management of hypovolemic or hemorrhagic shock and early obstetric consultation for surgical management with transfer of care as necessary are critical.
![]() PLACENTA PREVIA
PLACENTA PREVIA
Placenta previa is defined as a placenta that overlies or is in close proximity to the internal cervical os (Table 100-2)7 and is seen in approximately 0.3% to 0.5% of births in the United States.7 It is the primary cause of painless third trimester bleeding. Risk factors include previous cesarean delivery or uterine surgery, smoking, increased maternal age, multiparity, cocaine use, and a multiple pregnancy.7 It is thought to be caused by scarring of the endometrium.7
|
TABLE 100-2 |
Classification of Placenta Previa |

The classic presentation of placenta previa is painless, bright red bleeding from the vagina during the late second or third trimester. The uterus usually remains soft; however, contractions may occur. Examination of the cervix may exacerbate hemorrhage with catastrophic results. Digital examination is contraindicated, and thus the use of ultrasonography is necessary to make the diagnosis. Transvaginal ultrasonography is more accurate than transabdominal ultrasonography for diagnosis but should be performed by an experienced individual with careful attention to not place the vaginal probe into the cervix. When placenta previa is diagnosed, especially in women with a history of previous cesarean delivery, placenta accreta, invasion of the previa into the myometrium, should be considered. The placenta can be evaluated by transvaginal ultrasonography or if needed by MRI for evidence of placenta accreta.
Management of known placenta previa with no acute bleeding in the ED is typically expectant and includes pelvic rest, limiting long-distance travel, and maintaining a safe hemoglobin level. Patients with hemorrhage should be stabilized with insertion of two large-bore IV catheters and given crystalloid fluids and blood transfusions, if needed. At time of delivery, cesarean delivery is required due to increased risk for hemorrhage. Delivery may ultimately lead to a hysterectomy. Thus, obstetric consultation and transfer to a facility capable of managing the mother and newly born should be initiated early as placenta previa is associated with both maternal and neonatal morbidity and mortality.7
![]() PLACENTAL ABRUPTION
PLACENTAL ABRUPTION
Placental abruption is the premature separation (partial or total) of a normally implanted placenta presenting with concealed hemorrhage or vaginal bleeding. It usually occurs during the latter half of pregnancy and is seen in 1% of all pregnancies.8 It has significant perinatal mortality with 119 deaths per 1,000 pregnancies usually due to preterm delivery.8 Maternal risk factors includes previous history of placenta abruption, elevated blood pressure (due to either chronic hypertension or preeclampsia), cocaine use, cigarette smoking, multiple gestation, premature rupture of membranes, oligohydramnios, and chorioamnionitis.8 The classic presentation of placental abruption typically includes vaginal bleeding, abdominal pain, uterine tenderness, and contractions. The amount of bleeding does not correlate with the severity of abruption. In more severe cases of placental abruption, severe hemorrhage, uterine tetany, maternal hypotension, coagulopathy, fetal distress, and fetal death can be seen.8 Ultrasonography is insensitive and unreliable in the diagnosis of placental abruption; in most mild cases, the clinical diagnosis is made and confirmed postpartum on inspection of the placenta.
The management of a patient with suspected placenta abruption includes stabilizing the airway, 100% oxygen, two large-bore IV catheters, fluid resuscitation, and careful monitoring of the mother and fetus. Laboratory evaluation includes a CBC including a platelet count, coagulation studies, fibrinogen, and type and cross for matched blood.8 Early obstetric consultation and transfer to facility capable of caring for the mother and newly born must be initiated early.
PREECLAMPSIA
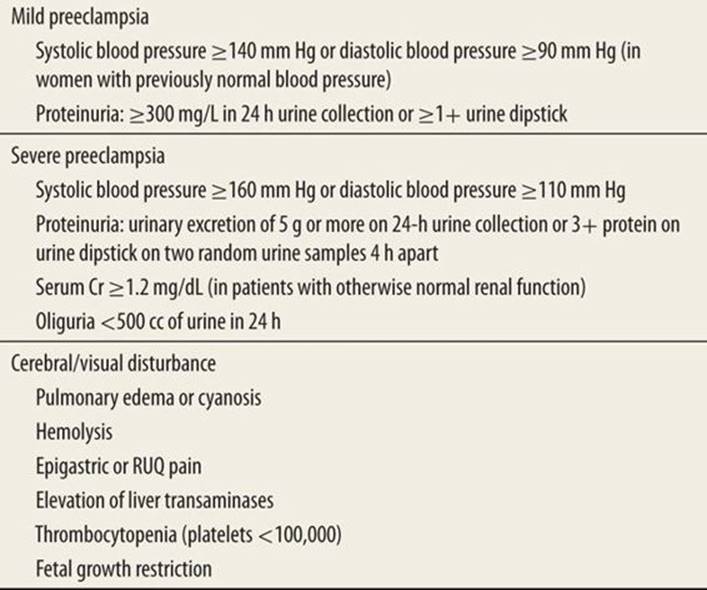
Preeclampsia is of unknown etiology but complicates 2% to 7% of all pregnancies.9 It is defined as hypertension with a blood pressure of 140/90 mm Hg or greater associated with proteinuria at greater than 20 weeks gestation. In mild preeclampsia, the patient is asymptomatic with blood pressure <160/110 mm Hg and otherwise normal physical examination and liver function and coagulation studies. In severe preeclampsia, blood pressures may exceed 160/110 mm Hg and may be associated with visual blurring, headache, upper abdominal pain, coagulopathy, hyperreflexia, or disseminated intravascular coagulation (Table 100-3).10
|
TABLE 100-3 |
Mild and Severe Preeclampsia Diagnostic Criteria |
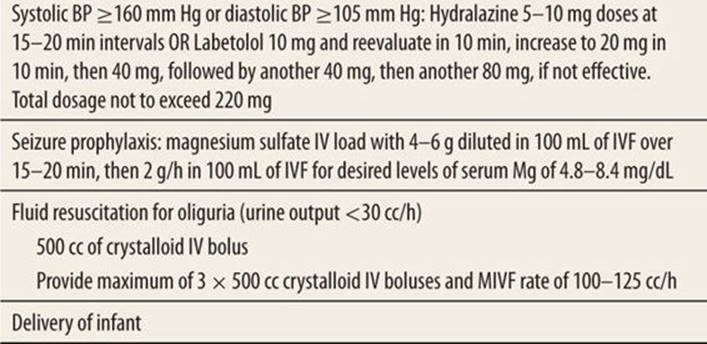
Ultimately, the most effective therapy is delivery of the infant. The treatment goal is to prevent maternal seizures and permanent end-organ damage as well as to protect the health of the infant. Therapy includes controlling seizures with magnesium sulfate, lowering blood pressure with labetalol or hydralazine, and delivery of the infant (Table 100-4).11 Additional laboratory studies such as a CBC with platelet count, liver function transaminases, and creatinine level should be obtained to assess maternal organ injury. Monitor urine output closely. A cranial head computed tomography (CT) or MRI should be considered if there is a change in mental status, presence of seizures, or lateralizing neurologic signs.
|
TABLE 100-4 |
Treatment for Severe Preeclampsia |
ECLAMPSIA
Eclampsia is the occurrence of seizures that cannot be attributed to other causes in a pregnant woman with preeclampsia. Associated complications include placenta abruption, disseminated intravascular coagulopathy, pulmonary edema, acute renal failure, aspiration pneumonia, and cardiopulmonary arrest.12 Perinatal death rate ranges from 5.6% to 11.8%.12
The provider should prioritize preventing maternal injury and supporting respiratory and cardiovascular function while initiating immediate obstetrical support/consultation. Women who develop eclampsia should otherwise be treated in a similar fashion as preeclampsia with magnesium sulfate, antihypertensive medications, and delivery of the infant.
HELLP SYNDROME
HELLP syndrome is defined as hemolysis (microangiopathic hemolytic anemia), elevated liver enzymes, and low platelets (<100,000/mm3). HELLP syndrome is a part of the spectrum of preeclampsia. Hypertension can be absent in 12% to 18% of cases, and proteinuria may be absent in 13% of cases.13 Complications such as pulmonary edema, acute renal failure, disseminated intravascular coagulopathy, placenta abruption, liver failure, acute respiratory distress, sepsis, and stroke lead to a 1% mortality rate; perinatal death can occur in 7% to 20% of cases.13 Treatment is the same as preeclampsia and requires immediate obstetrical support.
DEEP VEIN THROMBOSIS AND PULMONARY EMBOLISM
The risk for developing a deep vein thrombosis (DVT) and/or subsequent pulmonary embolism (PE) is five times greater during pregnancy and the puerperium.14 Of the approximate 1 in 1000 pregnancies complicated by thrombotic events, up to 15% result in PE; PE is the primary cause of direct maternal death (rate of 15% with PE) during this period.15,16
Venous stasis, blood vessel trauma, and hypercoagulability contribute to the increased thrombotic events during this period. Venous stasis is compounded by compression of the inferior vena cava and pelvic veins by a gravid uterus.14 Hypercoagulability is due to physiologic changes during pregnancy, which include increase in circulating clotting factors and decreasing amounts of protein S.14 The left lower extremity is the most common site for a DVT; however, the pelvic veins are the most common source for PE.15 PE is preceded by symptomatic DVT in less than half of the cases; one-half of these events occur during pregnancy and the other half occur in the puerperium.14Specific risk factors for thrombotic events include cesarean section and operative vaginal delivery, advancing maternal age, thrombophilias and antiphospholipid antibodies, obesity, prolonged bed rest, trauma, and infection.
The clinical presentation of DVT includes leg pain or discomfort especially on the left, swelling, tenderness, increased temperature and edema, and/or lower abdominal pain. Symptomatic DVTs present similarly in pregnant or postpartum women as they do in nonpregnant women and are particularly difficult to diagnose clinically as leg swelling and discomfort are common features of normal pregnancy. DVTs can also be asymptomatic. DVT can be diagnosed by compression ultrasonography with or without color Doppler, or by venography or magnetic resonance venography if compression ultrasonography does not provide a definite diagnosis.
PE arises from DVT, but many DVTs are not recognized prior to the occurrence of a PE. Recognizing the risk factors for venous thromboembolism, timely diagnosis, and appropriate treatment are critical to good outcomes in the pregnant patient (see Table 100-5 for risk factors for PE).17 Signs and symptoms of PE may vary and the diagnosis requires a high degree of suspicion. Dyspnea and tachypnea are the most common symptoms (in up to 90% of cases), but hemoptysis (in less than 10% of cases), chest pain, cough, and tachycardia are also seen in the clinical presentation of PE.18 Arterial blood gas, chest radiograph, and ECG findings will be seen only variably. Ventilation/perfusion (V/Q) lung scan is safe throughout pregnancy; however, it has low specificity. The current first-line diagnostic test is a spiral chest CT. In pregnancy, D-dimer can be increased secondarily to the physiologic changes in the coagulation system, especially if there is preeclampsia. Thus, the use of serum D-dimer for the diagnosis of DVT or PE is not presently recommended.
|
TABLE 100-5 |
Risk Factors for Pulmonary Embolism |

When a DVT or PE is suspected clinically, treatment with unfractionated heparin or low-molecular-weight heparin should be started until the diagnosis is excluded, unless anticoagulation is contraindicated (Table 100-6).14,19 In patients with PE, analgesia for pleuritic pain, oxygen, and early obstetric consultation and transfer is recommended.
|
TABLE 100-6 |
Treatment of Acute PE |

TRAUMA IN PREGNANCY
Trauma in pregnancy is the most common cause of nonobstetrical morbidity and mortality: motor vehicle crashes followed by assaults, falls, burns, and drowning are the primary causes.20 Anatomic and physiologic changes in pregnancy that alter the approach to a pregnant patient with trauma include an increased respiratory rate, heart rate and cardiac output, a relative hypotension, an increased blood volume by 50%, and a relative decrease in hematocrit. The priorities of resuscitation follow Advanced Trauma Life Support guidelines. As compared to the nonobstetric patient, pregnant women are at higher risk for aspiration from delayed gastric emptying thus lowering the threshold for definitive protection of the airway. Oxygen saturation should always be optimized.20 Placement of a chest tube thoracostomy should be 1 to 2 intercostal spaces higher than usual.20 Hemodynamic instability and shock can be difficult to diagnose because maternal blood volume is increased. As in all trauma cases, two large-bore IV catheters are necessary. Deflection of the uterus to the left side, once the airway is secured, is important to limit aortocaval compression and to improve venous return.20 In addition to the mother’s vital signs, fetal heart tones should be monitored; fetal distress may be the first sign of maternal hemodynamic instability and shock. Early obstetric and surgical consultation is essential in maximizing outcomes.
Laboratory tests may include a CBC, type and cross matching, urinalysis, serum bicarbonate or blood gas analysis, and coagulation profile.20 Radiographs should not be withheld because of concerns of radiation exposure to the fetus. Plain radiographs of the cervical spine, chest, and pelvis can be obtained while shielding the uterus. CT of the head and chest are generally safe. As a general rule, avoid abdominal CTs in early pregnancy and consider other diagnostic modalities such as ultrasound or diagnostic peritoneal lavage (DPL). Ultrasonography can locate free intraperitoneal fluid, guide for DPL, document fetal heart status and viability, establish gestational age and presentation, localize the placenta, and aid in diagnosing placental abruption.
Disposition after trauma varies with severity of injury and gestational age. Generally, patients who are older than 20 weeks with blunt abdominal trauma should be monitored for at least 4 to 6 hours as evidence of fetal distress requires urgent obstetric assistance and may require emergency cesarean section. Emergency cesarean delivery aids in maternal resuscitation and perfusion by eliminating the aortocaval compression produced by the gravid uterus; and in the first 5 minutes of maternal cardiopulmonary arrest or death, aids in fetal survival.20
RH SENSITIZATION
Rh sensitization occurs when an Rh-negative mother is exposed to Rh-positive fetal blood. It can occur after delivery, obstetrical procedures, trauma, threatened or spontaneous miscarriages, therapeutic abortions, or ectopic pregnancy. Anytime there is mixing of maternal and fetal blood, anti-D immune globulin (RhoGAM) is indicated at a dose of 50 μg intramuscularly (IM) prior to 12 weeks gestational age or 300 μg IM after 12 weeks gestational age.21
EMERGENCY CONTRACEPTION
In 2006, 82% of adolescent pregnancies were unplanned.22 Emergency contraception (EC) is defined as a drug or device used to prevent pregnancy after sexual intercourse or after recognition of contraception failure. In the United States, the two most common and available methods include levonorgestrel 1.5 mg (Plan B One Step® or Next Choice One Dose™) and copper IUD containing 380 mm of copper (ParaGard®), both of which can be used up to 5 days after sexual intercourse. Levonorgestrel is noted to have improved effectiveness if used within the first 48 hours. Because nausea and vomiting can be significant side effects, if vomiting is experienced within 1 to 2 hours after taking Plan B, the dose should be repeated; other side effects include irregular bleeding, dizziness, fatigue, breast tenderness, headache, and abdominal pain.23 The copper IUD does not have reduced effectiveness between 48 and 120 hours after sexual intercourse. Placement of an IUD as an EC has the same risk factors as in a person using it for conventional contraception. Plan B became available without need of prescription in August 2006. EC is intended as a back-up method of contraception and should be used sparingly.
REFERENCES
1. Ventura SJ, Curtin SC, Abma JC, Henshaw SK. Estimated pregnancy rates and rates of pregnancy outcomes for the United States, 1990–2008. Natl Vital Stat Rep. 2012;60:1.
2. Santelli JS, Lindberg LD, Finer LB, Singh S. Explaining recent declines in adolescent pregnancy in the United States: The contributions of abstinence and improved contraceptive use. Am J Pub Health. 2007;97:150.
3. Kost K, Henshaw S. US teenage pregnancies, births, abortions, 2008: national trends by age, race, and ethnicity. http://www.guttmacher.org/pubs/USTPtrends08.pdf. Accessed May 20, 2014.
4. Hasan, R, Baird DD, Herring AH, Olshan AF, Jonsson Funk ML, Hartmann KE. Patterns and predictors of vaginal bleeding in the first trimester of pregnancy. Ann Epidemiol. 2010;20:524.
5. ACOG Practice Bulletin Number 94. Medical management of ectopic pregnancy. Obstet Gynecol. 2008;111:1479.
6. Mukul LV, Teal SB. Current management of ectopic pregnancy. Obstet Gynecol Clin N Am. 2007;34:403.
7. Oyelese Y, Smulian JC. Placenta previa, placenta accreta, and vasa previa. Obstet Gynecol. 2006;107:927.
8. Oyelese Y, Ananth CV. Placental abruption. Obstet Gynecol. 2006; 108:1005.
9. Sibai BM. Diagnosis and management of gestational hypertension and preeclampsia. Obstet Gynecol. 2003;102:181.
10. ACOG Practice Bulletin Number 33. Diagnosis and management of preeclampsia and eclampsia. Obstet Gynecol. 2002;99:159.
11. Cunningham FG, Leveno KJ, Bloom SL, et al. Pregnancy hypertension. In: Cunningham FG, Leveno KJ, Bloom SL, et al., eds. Williams Obstetrics. New York, NY: McGraw-Hill, 2010:706.
12. Sibai BM. Diagnosis, prevention, and management of eclampsia. Obstet Gynecol. 2005;105:402.
13. Barton JR, Sibai BM. Diagnosis and management of hemolysis, elevated liver enzymes, and low platelets syndrome. Clin Perinatol. 2004;31:807.
14. ACOG Practice Bulletin Number 123. Thromboembolism in pregnancy. Obstet Gynecol. 2011;118:718.
15. Gherman RB, Goodwin TM, Leung B, et al. Incidence, clinical characteristics, and timing of objectively diagnosed venous thromboembolism during pregnancy. Obstet Gynecol. 1999;94:730.
16. Rutherford SE, Phelan JP. Deep venous thrombosis and pulmonary embolism in pregnancy. Obstet Gynecol Clin N Am. 1991;18:345.
17. Greer IA. Prevention and management of venous thromboembolism in pregnancy. Clin Chest Med. 2003;24:123.
18. Greer IA, Thomson AJ. Management of venous thromboembolism in pregnancy. Best Pract Res Clin Obstet Gynaecol. 2001;15:583.
19. Lowe GD. Treatment of venous thrombotic embolism. Bailleres Clin Obstet Gynaecol. 1997;11:511.
20. Brown HL. Trauma in pregnancy. Obstet Gynecol. 2009;114:147.
21. ACOG Practice Bulletin Number 75. Management of alloimmunization during pregnancy. Obstet Gynecol. 2006;108:457.
22. Finer LB, Zolna MR. Unintended pregnancy in the United States: incidence and disparities, 2006. Contraception. 2011;84:478.
23. ACOG Practice Bulletin Number 69. Emergency contraception. Obstet Gynecol. 2010;115:1100.