Stephanie J. Doniger
HIGH-YIELD FACTS
• Dysrhythmias are classified according to rate, QRS width, and clinical stability.
• Sinus bradycardia in the neonate always requires aggressive evaluation and treatment.
• Infants with paroxysmal supraventricular tachycardia (PSVT) may present in a low output state with irritability, poor feeding, tachypnea, and diaphoresis.
• The presence of an accessory pathway is the most common mechanism for PSVT in the child, but is difficult to appreciate during PSVT. Digoxin may precipitate ventricular tachycardia (VT) and is only used under the supervision of a pediatric cardiologist.
• Atrial fibrillation or flutter associated with accessory pathway disease or hypertrophic cardiomyopathy (HC) puts a child at high risk for 1:1 conduction, ventricular tachycardia, and sudden death.
Disorders of rate and rhythm are fortunately rare in the pediatric population. The most common dysrhythmia is supraventricular tachycardia. Rhythm disturbances, such as sinus bradycardia, can be life-threatening, particularly in the neonate.
Dysrhythmias in children that are the result of cardiac lesions have a poorer prognosis than patients with a structurally normal heart. Noncardiac causes, such as hypoxia, electrolyte imbalance, toxins, and inflammatory disease, must be considered, as should cardioactive drugs, such as digoxin or over-the-counter cold remedies. Initial evaluation includes an electrocardiogram (ECG).
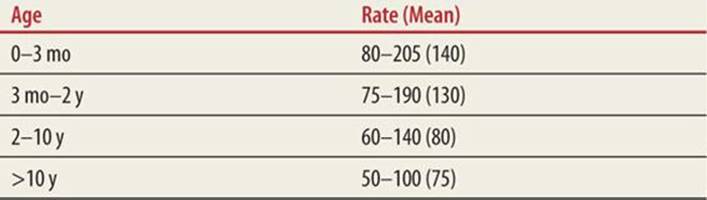
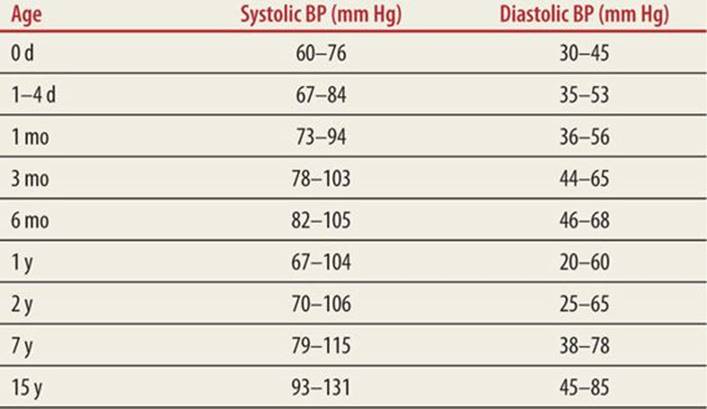
Age is an important consideration in the child presenting with a dysrhythmia. Age is also a factor in the clinical presentation of the dysrhythmia. The infant may present with poor feeding, tachypnea, irritability, or signs of a low output state. Caregivers often note that their baby is “not acting right.” The older child often presents with more specific symptoms, such as syncope from decreased cerebral blood flow, chest pain from decreased coronary blood flow, or palpitations. The ventricular rate in third-degree heart block may be adequate for the 2-month-old child but will not provide an adequate cardiac output for the child at age 12. Adolescents involved in competitive athletics with syncope, palpitations, or worrisome chest pain should be evaluated promptly. Normal ranges for heart rate and blood pressure are listed in Tables 42-1 and 42-2.
|
TABLE 42-1 |
Expected Heart Rates According to Age1 |
|
TABLE 42-2 |
Expected Systolic and Diastolic Blood Pressures According to Age |
The initial emergency management of dysrhythmias is dependent on three factors: rate, QRS width, and clinical stability. Decisions should be based on 12-lead ECG interpretation as single-lead monitor strips can be misleading. Rapid rates may appear supraventricular in origin in the child with tachycardia. Children tolerate most rhythm disturbances well, providing ample time for precise interpretation.
SLOW RATES
![]() SINUS BRADYCARDIA
SINUS BRADYCARDIA
Sinus bradycardia may be a manifestation of serious underlying disease or a normal physiologic variant; therefore, each child must be approached individually. Athletes commonly exhibit bradycardia which is a normal finding. Serious causes of bradycardia include hypoxia, hypoglycemia, hypothyroidism, hypo- or hyperkalemia, tension pneumothorax, cardiac tamponade, toxins, coronary or pulmonary thrombosis, or increased intracranial pressure. Sinus bradycardia can be a manifestation of calcium-channel blocker, β-blocker, or digoxin toxicity. Treating the underlying condition often corrects the rate. If the cause is unclear and oxygenation and ventilation are adequate, compressions should be initiated in an unstable patient. In a recent multihospital cohort study, pediatric patients who had chest compressions initiated for bradycardia and poor perfusion prior to the onset of pulselessness were more likely to survive to discharge.2 Epinephrine should be given (1:10,000, at 0.01 mg/kg IV/IO or 1:1000, at 0.1 mg/kg via the endotracheal tube). The maximum dose of epinephrine is 1 mg IV or 2.5 mg by the endotracheal route. High-dose epinephrine via the intravenous route is no longer recommended unless there is concern for β-blocker overdose.1 Atropine, 0.02 mg/kg (minimum dose 0.1 mg), is reserved for patients with either vagally mediated bradycardia or first- and second-degree heart blocks. It is important to note that small doses <0.1 mg may produce a paradoxic bradycardia. Thus, minimum dose is 0.1 mg and the maximum dose for children is 0.5 and 1.0 mg for adolescents.3–5
![]() ATRIOVENTRICULAR BLOCKS
ATRIOVENTRICULAR BLOCKS
Complete atrioventricular (AV) block may be congenital or acquired. Congenital blocks associated with structural disease, such as an AV canal, have a poorer prognosis than blocks associated with maternal collagen vascular disease. In the latter, maternal antibodies cause fibrosis and destruction of the conduction system. Complete AV block is suspected in utero in the setting of sustained fetal bradycardia, polyhydramnios, and congestive heart failure (CHF). Rates of 50 to 80 beats per minute (bpm) are typical in complete AV block. Symptoms are rate dependent and most patients with rates >50 bpm are rarely symptomatic. An alternative explanation for instability must be pursued in patients with heart rates approaching 80 bpm. Treatment of neonatal symptomatic bradycardia due to AV block includes control of CHF, isoproterenol, and temporary transcutaneous, transthoracic, or umbilical transvenous pacing.
Acquired third-degree block is associated with myocarditis, endocarditis, rheumatic fever, cardiomyopathy, Lyme disease, or tumor. Postoperative blocks are less common today because of advances in intraoperative mapping. Postoperative blocks may last for years or occur years after surgery. Unlike congenital third-degree block, QRS complexes are usually wide. Treatment is similar, except patients with syncope must be paced immediately.
![]() PACEMAKERS IN CHILDREN
PACEMAKERS IN CHILDREN
The indications for pediatric pacemakers differ from indications in adults. The most common is symptomatic bradycardia. Occasionally, an asymptomatic child with an extremely low heart rate needs pacing. Postoperative or acquired AV blocks require permanent pacing. Other indications include long QT syndrome (LQTS) and cardioinhibitory syncope lasting >10 seconds.
Advances in adult pacemakers have improved the management of children with dysrhythmias. Most permanent pediatric pacemakers are transvenous. Epicardial units are reserved for premature infants and those with right-to-left shunts. Choice of mode depends on disease. For example, children with sinus disease require atrial pacing. Ventricular pacing is unnecessary because AV conduction is normal. Children with congenital or acquired AV block require dual-chamber pacing. Isolated ventricular pacing is employed in the very young. Most units can be programmed to sense, demand, or inhibit at the atrial or ventricular level, depending on the needs of the child. Also, they may be programmed to sense motion or breathing.
Syncope or palpitations in a child with a pacemaker suggests malfunction. Chest radiography may reveal wire fracture or lead displacement. Most malfunctions are not mechanical and require external reprogramming. If the problem is not easily resolved, the patient should be admitted.
Temporary transvenous pacemakers are rarely necessary in children. Most temporary pacing is transcutaneous. Transthoracic pacing via pericardiocentesis catheter should be considered in the very unstable infant.
FAST RATES
![]() SINUS TACHYCARDIA
SINUS TACHYCARDIA
Sinus tachycardia is defined as a rate of sinus node discharge higher than normal for the patient’s age. In early versions of the PALS resuscitation guidelines, sinus tachycardia was considered a dysrhythmia. However, sinus tachycardia is a common, normal presentation and is often caused by such benign conditions as fever, dehydration, pain, and anxiety. Sinus tachycardia may also be associated with pathologic conditions such as anemia, sepsis, hypoxia, hyperthyroidism, and drug ingestions. More serious but less common causes include cardiac tamponade, tension pneumothorax, and thromboembolism. The treatment of sinus tachycardia is directed toward treating the underlying condition.6
![]() SUPRAVENTRICULAR TACHYCARDIA
SUPRAVENTRICULAR TACHYCARDIA
The most common dysrhythmia in the child is supraventricular tachycardia (SVT). SVT is a narrow complex tachycardia (QRS ≤0.09 seconds), and is differentiated from sinus tachycardia by its abrupt onset, rates >220 bpm, the absence of normal P waves (Fig. 42-1), or by little rate variation during stressful activities, such as phlebotomy. SVT occurs with an estimated incidence of 1/250 to 1/1000 children. The prognosis for most pediatric patients is excellent. Previously, the fatality rate was poorly defined for SVT. A recent study of 1755 patients with SVT reported a total of 4% fatalities. Only 1% of those deaths occurred without associated structural heart disease. Case fatality rate was higher in infants <1 month of age, in patients with structural heart disease, and in those with associated cardiomyopathy.7
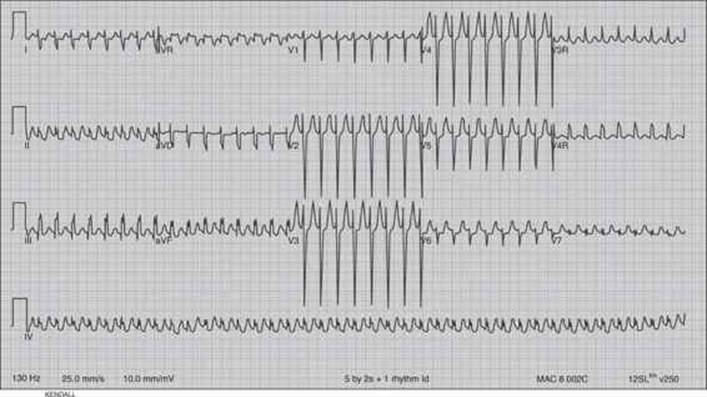
FIGURE 42-1. SVT, with concomitant right ventricular hypertrophy. This 4-year-old male was postoperative from repair of congenital heart disease (Fontan repair). He was eventually converted to normal sinus rhythm after multiple doses of adenosine.
Symptoms of SVT in infants include poor feeding, tachypnea, and irritability. They may appear ill or septic. Approximately 50% present within the first year of life, and likely present in congestive heart failure. Infants usually present within the first 24 hours. There are two mechanisms to consider: AV reciprocating tachycardia or AV nodal reentry. Younger children are more likely to have accessory pathway tachycardia, which is very important in choosing treatment.
AV reciprocating tachycardia or accessory pathway tachycardia is most common in children. Conduction during SVT is usually orthodromic, with antegrade AV conduction and retrograde accessory pathway conduction (Fig. 42-2). Conduction during sinus rhythm can be via the accessory pathway, resulting in a short PR interval and appearance of a delta wave. This characterizes the Wolff–Parkinson–White (WPW) syndrome. Some accessory pathways only conduct retrograde during bouts of SVT and are termed “concealed” because they are not apparent on surface ECG.
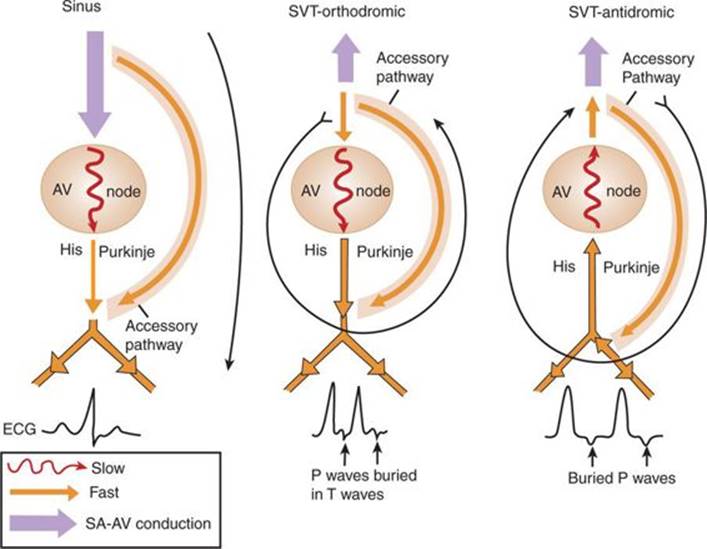
FIGURE 42-2. Diagrammatic representation of accessory pathway disease during sinus rhythm and SVT. Sinus—short PR, delta wave, characteristic of WPW; Orthodromic—fast retrograde conduction through accessory pathway leads to reentry. His–Purkinje conduction is normal, complexes are narrow. Retrograde P waves are abnormally directed and buried in T wave; Antidromic (rare)—fast antegrade conduction through accessory pathway leads to abnormal His–Purkinje conduction and wide complexes. Retrograde P waves are abnormally directed and buried in T waves.
The other mechanism, AV nodal reentry, is more common in adults, but may be responsible for one-third of cases of SVT in adolescents. Within the AV node, fast pathways with long refractory periods are blocked during a PAC, allowing for antegrade conduction down the slow tract. The impulse then propagates up the fast tract, initiating reentry. Distinguishing nodal from accessory pathway SVT is difficult during episodes of SVT. Negative P waves in II, III, and avF may indicate retrograde conduction through the accessory pathway but they are usually buried in the QRS complex. Pointed or peaked T waves suggest retrograde P waves. P waves are almost never seen in AV nodal reentry. Lack of delta wave during sinus rhythm does not rule out concealed accessory tracts. Information from parents may be helpful, but first episodes of SVT or unstudied children make diagnosis difficult in the ED.
Unstable SVT is treated with synchronized cardioversion, 0.5 to 1 J/kg, increasing to 2 J/kg as needed. Vagal maneuvers may convert stable patients. A bag containing ice and water is placed over the nose and forehead for intervals of 15 to 20 seconds. Another technique is to have the child blow into an occluded straw. Ocular pressure and nasogastric stimulation are discouraged. If vagal maneuvers fail, adenosine, 0.1 mg/kg followed by 0.2 mg/kg, is recommended. Given its short half-life, it should be administered through an IV closest to the heart. Recent case reports have suggested that the intraosseous route is an unreliable method of delivery.8 Adenosine terminates nodal and accessory pathway tachycardia by blocking adenosine receptors in the AV node and slowing conduction. Adenosine has a fairly wide safety margin because of its short half-life. Transient side effects include headache, flushing, and chest pain. Rhythm disturbances, such as atrial fibrillation, accelerated ventricular rhythm, and wide-complex tachycardia, may require resuscitation. Other side effects include bronchospasm, apnea, and asystole. Adenosine can be used in hypotension but should be avoided in the patient on theophylline.
Digitalis is commonly used to prolong AV nodal conduction and refractoriness of fast and slow tracts. A pediatric cardiologist should be consulted prior to administration as it can promote accessory pathway conduction and ventricular tachycardia (VT); therefore, it is best used in the well-known, stable patient with AV nodal reentry. It may take hours to work and if cardioversion is necessary, there is a risk of ventricular fibrillation.
Verapamil is not routinely used and is reserved for children >2 to 3 years of age. Hypotension, cardiovascular collapse, and death have occurred with its use in infants since it can cause myocardial depression, hypotension, and even cardiac arrest. Older children with stable but recalcitrant SVT may respond to IV verapamil, 0.1 to 0.3 mg/kg slowly. Calcium chloride, 10 mg/kg and IV saline should be available to treat hypotension.
β-Blockers such as propranolol or esmolol may be used with caution, as propranolol may cause hypotension, tachycardia, or ventricular fibrillation. If the above measures fail or SVT resumes, procainamide may be useful. Procainamide is preferred in narrow complex tachycardia thought to be ventricular. In the 2009 PALS guidelines, it was introduced as a possible therapy for refractory SVT.9 A 15 mg/kg bolus is given over 20 to 30 minutes, watching for hypotension or prolongation of the QRS complex. Propranolol, 0.1 mg/kg IV, is useful in WPW or other accessory pathway diseases. Esmolol dosage is 0.1 to 0.5 mg/kg IV over 1 minute, and then 0.05 mg/kg/min, titrated as noted in PALS protocols. Amiodarone, 5 mg/kg administered over 20 to 60 minutes, is another option. However, it should not be given in conjunction with procainamide because of the risk of refractory hypotension or increased prolongation of the QRS interval. β-Blockers, calcium-channel blockers, and digoxin should be avoided in SVT or atrial fibrillation/flutter with a wide complex (i.e., WPW) as these agents can increase the transmission through the bypass tract and induce ventricular fibrillation.
Any infant with new onset SVT should be hospitalized and have structural heart disease ruled out. Infants are likely to have accessory pathway disease and require further therapy. A child with immediate recurrence is at higher risk for repeat episodes or recalcitrant SVT and may require surgical ablation.
ATRIAL FLUTTER AND FIBRILLATION
Atrial flutter and fibrillation are rare in children, occurring in those with congenital heart disease, rheumatic fever, or dilated cardiomyopathy. Patients with atrial flutter or fibrillation, in combination with an accessory pathway or hypertrophic cardiomyopathy, are at high risk for sudden death. Unstable patients are cardioverted with 0.5 J/kg. Overdrive pacing 10 to 20 bpm faster than the flutter rate may also be effective. Cardioversion may be the only option. Patients with long-standing atrial disease associated with a diseased sinus node are at risk for bradycardia or asystole on termination. Pacing must be available. Consultation with a pediatric cardiologist is helpful if one chooses drug therapy, as the choices vary based on left ventricular function and whether WPW is suspected or known. The long-term prognosis of children with congenital heart disease and atrial fibrillation or flutter may depend on the elimination of all flutter activity.
![]() PREMATURE VENTRICULAR CONTRACTIONS
PREMATURE VENTRICULAR CONTRACTIONS
Premature ventricular contractions (PVCs) in the infant and young child are rare. Unifocal PVCs begin appearing in healthy children during adolescence. The patient is usually asymptomatic and has a normal physical examination, chest x-ray, and ECG. Unusual morphology, such as multifocal PVCs, coupling, or the “R on T” phenomenon, is rarely a cause for emergency intervention in the asymptomatic child with normal QT interval. Continuous ECG monitor may define and quantify the PVCs. PVCs that diminish during exercise or stress are benign and require no therapy. Patients with myocarditis, cardiomyopathy, congenital heart disease, or who are postoperative from cardiac surgery are at greater risk and may require treatment. Syncope or exercise-induced PVCs may also require therapy. Lidocaine, procainamide, or amiodarone may be useful using guidelines similar for VT (see Ventricular Tachycardia section).
![]() ACCELERATED IDIOVENTRICULAR RHYTHM
ACCELERATED IDIOVENTRICULAR RHYTHM
Accelerated idioventricular rhythm (AIVR) is a benign pediatric dysrhythmia that has the appearance of VT. Rates are rarely faster than 150 bpm. AIVR begins gradually with fusion beats and is a monomorphic, wide-complex rhythm that originates from an accelerated ventricular focus. Diagnosis can be difficult in the new patient. Patients with AIVR are stable. Unstable wide-complex tachycardia is not AIVR and should be converted immediately. AIVR rarely responds to medication but can be a warning of a residual hemodynamic abnormality associated with corrected congenital heart disease.
![]() VENTRICULAR TACHYCARDIA
VENTRICULAR TACHYCARDIA
Ventricular tachycardia (VT) is rare in children and can be confused with other forms of tachycardia. It is distinguished from SVT by wide QRS complexes, >0.09 seconds, depending on age. Complexes as narrow as 0.06 seconds have been noted in infantile VT (Fig. 42-3). Wide complexes can be seen in SVT but are rare. Antegrade accessory pathway conduction can result in wide and bizarre-looking QRS complexes that simulate VT. Rates averaging 250 bpm may be helpful in distinguishing VT from AIVR, with average rates of 150. AV dissociation with P wave and QRS independence can also help distinguish VT from SVT.
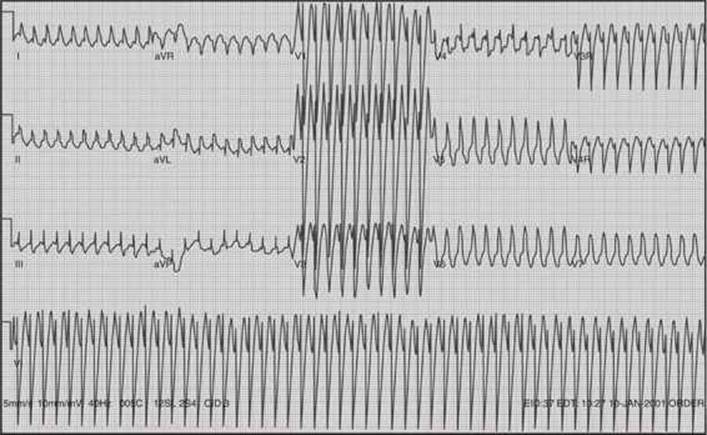
FIGURE 42-3. Ventricular tachycardia. This is an example of an extraordinarily fast ventricular tachycardia with a heart rate of almost 300 bpm.
Idiopathic VT is occasionally encountered in a child who is completely asymptomatic and has a normal heart. It is usually not treated. Serious causes, such as electrolyte disturbance, toxins, or myocarditis, should be considered. Causes can include structural heart disease, tumor, cardiomyopathy, or LQTS. Recurrent exercise-induced syncope is often because of VT. The initial workup may be negative. A search for a small myocardial tumor, early cardiomyopathy, or occult myocarditis may be necessary. EPS or biopsy may be necessary to guide treatment.
Regardless of etiology, unstable wide-complex tachycardia should be cardioverted synchronously with 0.5 to 1 J/kg. Amiodarone, 5 mg/kg over 1 hour, may be given for patients with stable VT. If amiodarone is unavailable, lidocaine may be considered, 1 mg/kg bolus, followed by a 15 to 50 μg/kg/min infusion. In addition, procainamide may be useful for wide-complex tachycardia of uncertain origin because of its effect both above and below the AV node. An initial dose of 10 to 15 mg/kg over 30 to 45 minutes is followed by 20 to 80 μg/kg/min drip. Adenosine is safe and may be useful in the rare case of SVT with aberrancy. It should be used only in stable situations when other traditional therapies have failed.
![]() VENTRICULAR FIBRILLATION
VENTRICULAR FIBRILLATION
Ventricular fibrillation is an uncommon rhythm in the pediatric population, but is life-threatening. Out-of-hospital ventricular fibrillation arrests vary by setting and age; they are uncommon in infants and children (4%–5%) and occur more frequently in adolescents (15%).10 Causes include postoperative complications from congenital heart disease repair, severe hypoxemia, hyperkalemia, medications (digitalis, quinidine, catecholamines, and anesthesia), myocarditis, and myocardial infarction. The hallmark of ventricular fibrillation is chaotic irregular ventricular contractions without circulation to the body rhythm. The ECG reveals a rapid rate, with bizarre QRS complexes with varying sizes and configurations.
In patients with unwitnessed arrests, CPR is beneficial immediately prior to defibrillation. Giving CPR prior to defibrillation increases survival rates from 4% to 22%.11 The majority of pediatric arrests are secondary to respiratory events. Therefore, previous resuscitation of children is focused on respiratory events rather than compressions and defibrillation, as has been traditionally emphasized in the resuscitation of adults. According to the 2009 PALS guidelines, patients should receive compressions immediately and defibrillation within less than 3 minutes of a shockable rhythm (ventricular fibrillation, pulseless VT). In a recent study, pediatric residents did not deliver compressions and defibrillation according to these guidelines. They also had errors in operating the defibrillator, presumably because they had less experience in actually discharging the defibrillator.12 The dosages of defibrillation are now 2 J/kg followed by 4 J/kg for subsequent dosages, regardless of the type of defibrillator. Although the optimal pediatric defibrillation dose remains unknown, a higher defibrillation dose of greater than 4 J/kg is not recommended.13 It is important to note that stacked shocks are no longer recommended. A single shock is recommended followed by CPR largely because of the prolonged period of time to administer three shocks. Do not interrupt CPR until five cycles or 2 minutes for a pulse/rhythm check (Fig. 42-4). More specifically, the treatment of each rhythm disturbance can be classified according to the tachycardia algorithm (Fig. 42-5). The presence or absence of a pulse determines which arm of the algorithm to initiate.
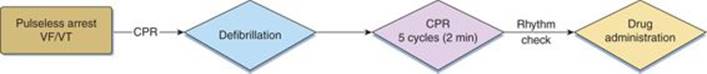
FIGURE 42-4. Sequence of resuscitation in pulseless arrest with ventricular fibrillation (VF) and ventricular tachycardia (VT).
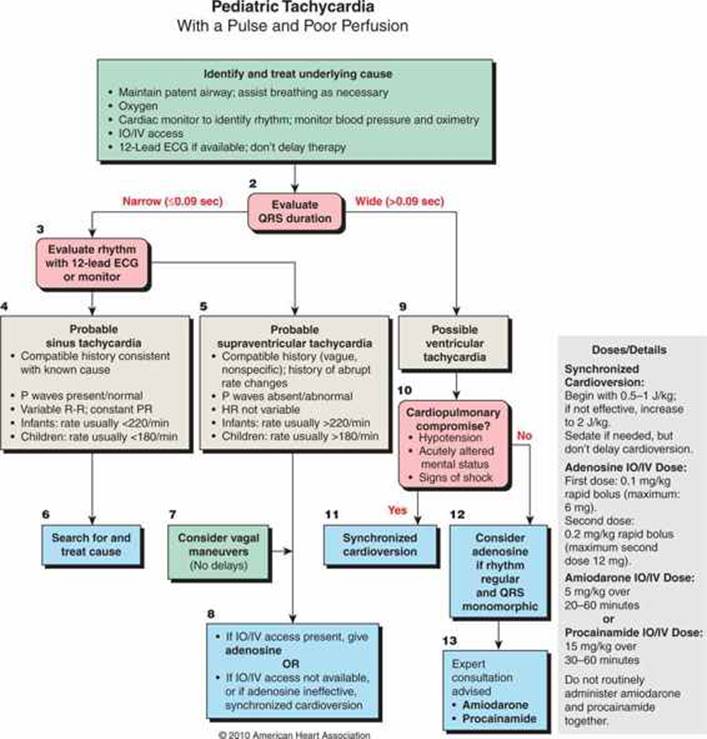
FIGURE 42-5. Tachycardia algorithm. (Reproduced with permission from Kleinman ME, Chameides L, Schexnayder SM, et al: Part 14: pediatric advanced life support: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care, Circulation. 2010 Nov 2;122(18 Suppl 3):S876–S908.)
For the most part, the algorithm drug dosages remain the same in the updated 2009 AHA recommendations. Drug delivery should not interrupt CPR. The timing of drug delivery is less important than minimizing interruption of chest compressions. Amiodarone is the preferred drug for treatment for pulseless arrest, since it is more effective. Lidocaine is only recommended when amiodarone is unavailable.14 It is important to note that amiodarone and procainamide should not be administered together as they can lead to severe hypotension and prolongation of the QT interval. High-dose epinephrine (1:1000 concentration via IV) is not recommended in any age group, and is actually associated with a worse outcome, especially in cases of asphyxia.3–5 The standard recommended dose is 0.01 mg/kg IV/IO for all doses, which correlates to 0.1 mL/kg. Although the preferred routes of administration are intravenous or intraosseous, it may be given via the endotracheal tube (ETT) when such access is unable to be obtained (0.1 mg/kg ETT). In exceptional cases, such as β-blocker overdose, high-dose epinephrine may be considered. Magnesium sulfate at a dose of 25 to 50 mg/kg (maximum 2 g) should be given for torsades de pointes.
In the community, automated external defibrillators (AEDs) have increased survival rates. There is sufficient evidence to show that AEDs can safely be used for those older than 1 year. In a sudden witnessed collapse, use the AED as soon as it becomes available. However, if the collapse is unwitnessed, CPR should be performed for five cycles or 2 minutes, prior to the use of the AED. Use pediatric AED pads and energy levels in those 1 to 8 years of age.5 If the pediatric dose is unavailable, the adult dose is a reasonable alternative.
OTHER CARDIAC CONDITIONS ASSOCIATED WITH DYSRHYTHMIAS
![]() LONG QT SYNDROME (LQTS)
LONG QT SYNDROME (LQTS)
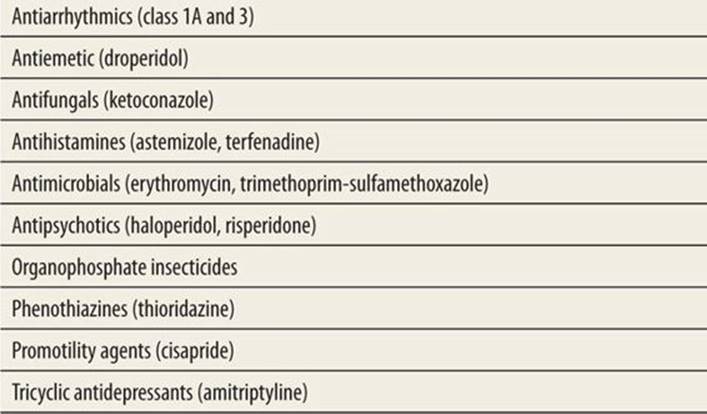
Long QT syndrome (LQTS) has an incidence of 1:2000,15 and should be considered in any patient presenting with a suggestive history, including first-degree relatives of known LQTS carrier, family history of syncope, seizures, or sudden death, a sibling with sudden infant death syndrome, seizure of unknown etiology, unexplained near-drowning. Other risk factors include congenital deafness and bradycardia in infants. Jervell and Lange-Nielsen first described the association of syncope, sudden death, deafness, and long QT interval in 1957. In 1963, Romano described the syndrome in normal-hearing patients. Congenital LQTS is an inherited syndrome characterized by paroxysmal VT and torsades de pointes. It can be emotionally induced or stress related and can progress to ventricular fibrillation and sudden death. Acquired QT prolongation associated with drugs (Table 42-3), anorexia nervosa, bulimia, and electrolyte derangements can also predispose to dysrhythmia. The congenital form is the most common form affecting the child.
|
TABLE 42-3 |
Drugs that Prolong the QT Interval |
The QT interval is a fluid measurement, and is influenced by physiologic and metabolic states, and exhibits diurnal variation. In order to account for the normal physiologic shortening of the QT interval that occurs with increasing heart rate, the corrected QT interval (QTc) is calculated using the Bazett formula: QTc = QT/√RR. For greatest accuracy, the QT and preceding RR intervals should be measured for three consecutive beats and averaged. The current practice identifies a QTc ≥ 460 ms as prolonged. A QTc value between 440 and 470 ms is borderline and warrants additional assessment.16 Although ECGs automatically calculate the QT and QTc, a manual calculation of the QTcshould be performed in those patients with a suggestive history, as the computer calculation often is inaccurate. Measurement of the QT interval with Lead II is generally accepted as being the most accurate. If the diagnosis of LQTS is suspected but the screening ECG is not diagnostic, increasing sympathetic activity such as with vagal maneuvers may trigger abnormalities on ECG. These abnormalities include QT-interval prolongation, prominent U waves, T-wave alternans, and ventricular dysrhythmias (Fig. 42-6).
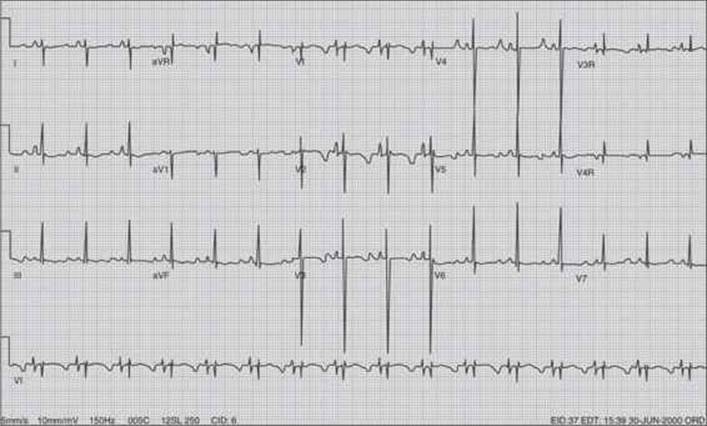
FIGURE 42-6. Long QT without associated heart block. Markedly prolonged QT interval calculated with the Bazett formula6: QTc = QT/(square root of the preceding RR interval); QTc = 0.452/(square root of 0.612); QTc = 578 ms; of note, for improved accuracy average three consecutive RR intervals and QT intervals, where each small box = 0.04 ms.
Patients presenting with LQTS may require emergency intervention. Patients presenting with an episode of polymorphic VT or torsades de pointes of unknown etiology should receive IV magnesium sulfate (25–50 mg/kg, maximum 2 g). Serum electrolytes and a toxicology screen should be obtained. β-Blockers may be useful in suppressing catecholamine surges and further dysrhythmic activity. In those patients with torsades owing to prolonged QT, they may worsen acutely, whereas those with normal QT improve. Patients with recurrent VT may require temporary transcutaneous ventricular pacing.
Any patient with a compatible history, borderline prolongation of the QT interval (440–470 ms) with symptoms, or identified prolonged QT syndrome should be referred to a cardiologist for further management. Interestingly, up to one-third of patients having a borderline QTc after having a presyncopal or syncopal event had normalization of values on follow-up ECG.17 Admission is limited to those who are symptomatic or have cardiovascular compromise. Holter monitoring may be helpful in capturing a prolonged QT interval not apparent on a resting ECG. Therapy is aimed at reducing sympathetic activity of the heart, either pharmacologically or surgically. β-Blockers are generally recommended as the initial therapy of choice and effectively eradicate dysrhythmias in 60% of patients, and decrease mortality from 71% in untreated patient to 6% in those who are treated.18 The most commonly used are propranolol (2–4 mg/kg/d, maximum 60 mg/d) and nadolol (0.5–1 mg/kg/d, maximum 2.5 mg/kg/d). Patients with severe asthma in whom β-blockers are contraindicated may be candidates for pacer therapy. Approximately 20% of patients remain refractory to treatment with β-blockers, and continue to experience recurrent syncope. Alternative therapies include left-sided cervicothoracic sympathetic ganglionectomy, and implantation of automatic implantable cardiac defibrillators.
Once a patient is diagnosed with LQTS, an ECG should be performed on all other family members. All affected individuals, regardless of age, should be restricted from competitive sports, but not necessarily recreational sports. Patients should be educated to avoid triggering factors, such as certain medications, loud noises, emotionally stressful situations, and dehydration. Because of the high risk of unexpected cardiac events, family members and close friends should be instructed in CPR and even consider purchasing a home AED.
![]() POSTOPERATIVE DYSRHYTHMIAS
POSTOPERATIVE DYSRHYTHMIAS
Following congenital heart surgery, 15%–48% of children develop a postoperative arrhythmia. This is largely caused by myocardial ischemia or injury, high catecholamine levels, and electrolyte disturbances.19 The most common dysrhythmia is junctional ectopic tachycardiac (JET). JET is a narrow complex tachycardia that usually occurs in conjunction with ventriculoatrial dissociation. The mechanism is unknown but one theory involves the mechanical stretching or trauma to the AV node during surgery. It is more common after repair of the right ventricular outflow tract or a VSD closure. It is generally a self-limited arrhythmia with a recent study reporting a mortality of 1.8%, lower than historically reported as 5%–10%.20 The recommended antiarrhythmic agent is amiodarone for acute and long-term suppression.21
![]() HYPERTROPHIC CARDIOMYOPATHY
HYPERTROPHIC CARDIOMYOPATHY
Hypertrophic cardiomyopathy (HC) is characterized by a hypertrophied, nondilated left ventricle. Symptoms include chest pain, dyspnea, syncope, or sudden death. Clinical presentation varies. Some patients are asymptomatic. The majority of symptomatic adults suffer from CHF secondary to insufficient diastolic filling. Dysrhythmias include atrial fibrillation and ventricular tachyarrhythmia, the leading causes of sudden death. Outflow obstruction is rare. Mortality rate of 3% is probably overestimated due to sampling bias and may lead to an overly aggressive approach to therapy. This is especially true of the asymptomatic individual. However, pediatric HC tends to be more serious, with mortality rate in infants approaching 6%. Other risk factors include advanced symptoms at diagnosis, LV dysfunction, and a family history of sudden death. Atrial fibrillation is especially dangerous in the HC patient. The ECG may provide a clue to the diagnosis. Look for narrow Q waves in leads 1 and avL combined with increased voltage in V2 and V5.
Signs of HC include late systolic murmur and paradoxical splitting of S2. LV or septal hypertrophy on ECG is a poor prognostic sign. Echocardiogram is diagnostic. Therapy depends on the clinical manifestation. β-Blockers are the mainstay of therapy for CHF, effective in relieving dyspnea and chest pain. β-Blockers have no effect on rates of sudden death. Atrial fibrillation should be controlled because of its association with rapid ventricular rate and decreased outflow. Amiodarone may be effective but has also been associated with sudden death in some symptomatic patients. Implantable defibrillators may be preferred. Dual-chamber pacing and surgical myectomy may be necessary for significant outflow obstruction.
SUMMARY
Children, fortunately, rarely have dysrhythmias, and can remain stable for longer periods of time. The clinician needs to assess the patient for stability, for type of rhythm disturbance, and for the appropriate therapy.
REFERENCES
1. Perondi M, Reis A, Paiva E, Nadkarni V, Berg R. A comparison of high-dose and standard-dose epinephrine in children with cardiac arrest. N Engl J Med. 2004;350(17):1708–1709.
2. Donoghue A, Berg R, Hazinski M, Praestgaard A, Roberts K, Nadkarni V. Cardiopulmonary resuscitation for bradycardia with poor perfusion versus pulseless cardiac arrest. Pediatrics. 2009;124:1541–1548.
3. ECCC. American Heart Association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care: part 13: neonatal resuscitation guidelines. Circulation. 2005;112:188–195.
4. ECCC. Highlights of the 2005 American Heart Association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Currents. 2006;15(4):23–27.
5. Ralston M, Hazinski M, Zaritsky A, Schexnayder S, Kleinman M. Pediatric assessment. In: Association AH, ed. Pediatric Advanced Life Support Provider Manual. Dallas, TX: American Heart Association; 2006.
6. Jat KR, Lodha R, Kabra S. Arrhythmias in children. Indian J Pediatr. 2011;78(2):211–218.
7. Salerno J, Garrison M, Larison C, Seslar S. Case fatality in children with supraventricular tachycardia in the United States. PACE. 2011;34:832–836.
8. Goodman I, Lu C. Intraosseous infusion is unreliable for adenosine in the treatment of supraventricular tachycardia. Pediatr Emerg Care. 2012;28(1):47–48.
9. Gupta S, Taneja L. PALS update 2010. Indian Pediatrics. 2012;49:789–792.
10. Topjian A, Nadkarni V, Berg R. Cardiopulmonary resuscitation in children. Curr Opin Critical Care. 2009;15:203–208.
11. Wik L, Kramer-Johansen J, Myklebust H, et al. Quality of cardiopulmonary resuscitation during out-of-hospital cardiac arrest. JAMA. 2005;293:299–304.
12. Hunt E, Vera J, Diener-West M, et al. Delays and errors in cardiopulmonary resuscitation and defibrillation by pediatric residents during simulated cardiopulmonary arrests. Resuscitation. 2009;80:819–825.
13. Meaney P, Nadkarni V, Atkins D, et al. Effect of defibrillation energy dose during in-hospital pediatric cardiac arrest. Pediatrics. 2010;127:e16–e23.
14. Dorian P, Cass D, Schwartz B, Cooper R, Gelaznikas R, Barr A. Amiodarone as compared with lidocaine for shock-resistant ventricular fibrillation. N Engl J Med. 2002;346:884–890.
15. Komarlu R, Beerman L, Freeman D, Arora G. Fetal and neonatal presentation of long QT syndrome. PACE. 2012;35:e87–e90.
16. Ackerman M. The long QT syndrome: ion channel diseases of the heart. Mayo Clin Proc. 1998;73:250–269.
17. Dorn CV, Johnson J, Taggart N, Thorkelson L, Ackerman M. QTc values among children and adolescents presenting to the emergency department. Pediatrics. 2011;128:e1395–e1401.
18. Schwartz P. Idiopathic long QT syndrome: progress and questions. Am Heart J. 1985;109:399–411.
19. Bar-Cohen Y, Silka M. Management of postoperative arrhythmias in pediatric patients. Curr Treat Options Cardiovasc Med. 2012;14:443–454.
20. Makhoul M, Oster M, Fischbach P, Das S, Deshoande S. Junctional ectopic tachycardia after congenital heart surgery in the current surgical era. Pediatr Cardiol. 2013;34(2):370–374.
21. Lane R, Nguyen K, Nieman J, Bolte R, Etheridge S, Gausche-Hill M. Amiodarone for the emergency care of children. Pediatr Emerg Care. 2010;26(5):382–389.