Andrea T. Cruz
HIGH-YIELD FACTS
• Fever in the immunocompromised child is a medical emergency. Prompt evaluation and administration of antibiotics can decrease morbidity and mortality.
• Patients on chronic corticosteroids may also require stress dose steroids when febrile.
• Children with B cell immunodeficiencies are at risk for encapsulated pathogens. Children with T cell defects may present with chronic or severe viral infections, Pneumocystis jirovecii pneumonia, and recurrent bacterial infections. Children with phagocyte defects present with recurrent infections caused by pyogenic bacteria. Children with complement deficiencies present with encapsulated and pyogenic pathogens.
• Acute HIV infection should be suspected in the adolescent with fever, malaise, diffuse lymphadenopathy, pharyngitis, oral ulcers, and a maculopapular rash
The number of immunocompromised children seen in the emergency department (ED) is increasing due to improved detection of some primary immunodeficiencies (PIs) (Table 59-1) and to increased survival after diagnosis. This chapter will focus on infectious complications in immunocompromised children. See also Section 20 for hematologic and oncologic emergencies.
|
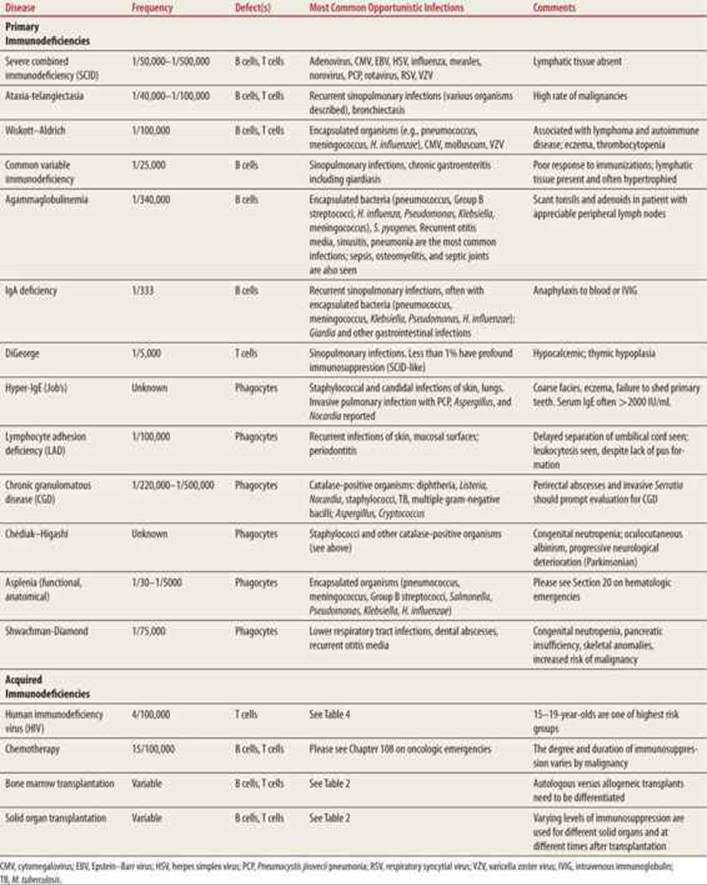
TABLE 59-1 |
The Most Common Immunodeficiencies and Immunocompromising Conditions in the United States |
SOLID ORGAN TRANSPLANT RECIPIENTS
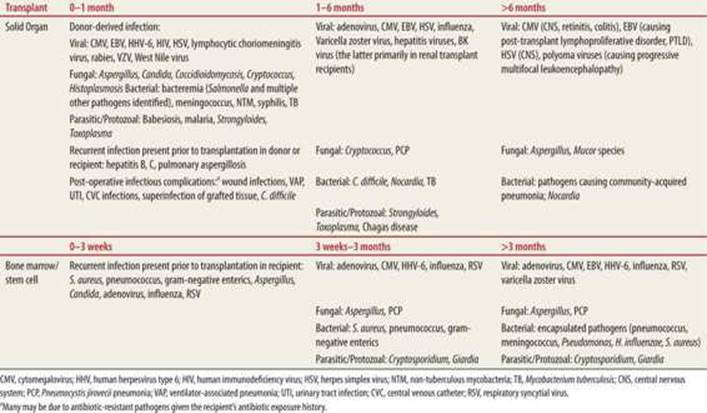
Certain infections are more common in varying time periods after solid organ transplant (SOT) (Table 59-2).1 Pathogens can be conceptualized as acquired from the donor, native flora that can reactivate or worsen after immunosuppression, postoperative infections, and newly acquired opportunistic infections (OIs) due to immunosuppression (Table 59-3).
|
TABLE 59-2 |
Most Common Infections in Transplant Recipients as a Function of Time Since Transplant |
|
TABLE 59-3 |
The Most Common Pathogens seen in Immunocompromised Children, with Emphasis on Differences in Clinical Manifestations, Diagnosis, and Treatmenta |
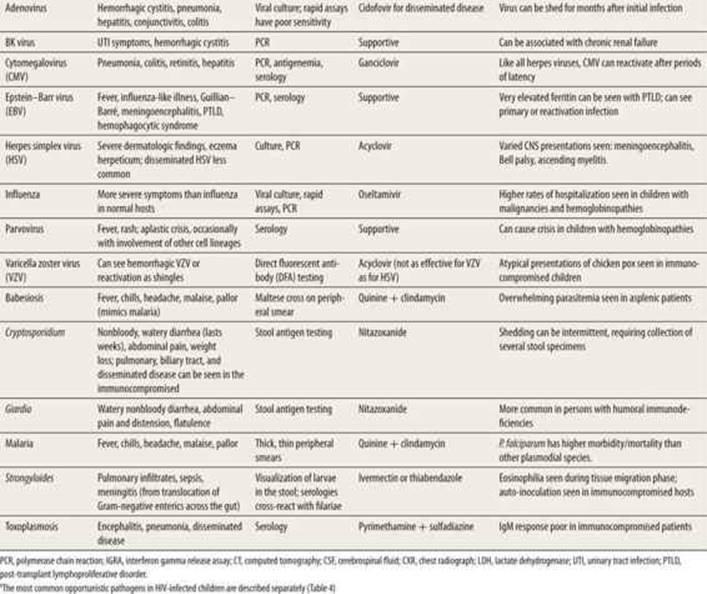
Recurrent pneumonia caused by typical or atypical pathogens (e.g., adenovirus, cytomegalovirus [CMV]) can be seen following lung transplantation. UTIs have been described almost half of children after renal transplantation, commonly with Escherichia coli and Klebsiella. BK virus and other polyoma viruses can cause hematuria or pyuria in renal transplant recipients and can be associated with chronic renal failure and graft loss. Colitis due to viral pathogens (CMV, herpes simplex virus [HSV]) or Clostridium difficile can be seen in small bowel transplant recipients. Bacterial infection of the transplanted organ is less common in heart and liver transplant recipients.
Bloodstream infections (BSI) in the post-transplant period can be secondary to CVC-associated infections, immunosuppression, or from translocation across leaky intestinal walls. In lung transplantation, BSIs are most common in the first month after SOT with the most common organisms being CONS, Pseudomonas, and Candida species.2 Although BSIs are also most common early after heart transplantation, gram-negative bacteria predominate over gram-positive organisms; fungal, anaerobic, or cryptococcal BSI account for <10% of cases. Liver transplant recipients are at higher risk of ascending cholangitis and should be covered for gram-positive, gram-negative, and anaerobic pathogens.3
Viral infections contribute to morbidity, mortality, and graft loss in SOT recipients.4 The most common virus in the post-transplant period is CMV. Treatment with valganciclovir or ganciclovir in the immediate post-transplant period does not prevent all cases of post-transplant disease or establishment of viral latency. SOT recipients have higher morbidity and mortality with influenza, and graft loss can be seen.5 Rapid viral testing has poor sensitivity mandating empiric therapy for SOT recipients with an influenza-like illness during influenza season,6 regardless of the rapid test result. Adenovirus can cause hemorrhagic cystitis, pneumonitis, hepatitis, and colitis. The most dreaded post-transplant virus is Epstein–Barr Virus (EBV), which can cause organ-specific pathology (hepatitis, pneumonitis, colitis, marrow dysfunction) or post-transplant lymphoproliferative disease (PTLD). SOT recipients have a risk of malignancy 5 to 10 times higher than the general population, most caused by EBV-associated malignancies, including hemophagocytic syndrome, lymphomas, and leiomyosarcomas.4 PTLD symptoms are protean and include constitutional symptoms, isolated or disseminated lymphadenopathy, tonsillar hypertrophy, sore throat, vomiting, diarrhea, GI bleeding, or organ failure. Intussusception in a SOT recipient should trigger an evaluation for PTLD, as the lead point for the intussusception may be GI lymphoid hyperplasia. Cytopenias and elevated ferritin and triglycerides commonly are seen.
OIs seen in SOT recipients include many pathogens seen in other immunocompromised children (Tables 59-2 and 59-3). These may include vaccine-preventable diseases if the child was unable to respond to vaccinations received in the pretransplant period. Although Pneumocystis (PCP) is associated with HIV-infection, transplant recipients accounted for almost 40% of PCP cases in one large series.4 Finally, children who have received SOT are at higher risk for autoimmune disease.7
The initial ED evaluation of the febrile SOT child should include a CBC, blood cultures (peripheral and from all lumens if child has a central line), rapid viral testing, chest radiograph, and chemistries. Evaluation should be targeted based on the kind of transplant, degree of immunosuppression, time since transplantation (Table 59-2), results of pretransplant serologies, and post-transplant infections. Broad-spectrum antibiotics should be initiated. A third-generation cephalosporin or a semi-synthetic, antipseudomonal penicillin (e.g., piperacillin-tazobactam) would be reasonable. The latter would be of particular use in liver transplant recipients, who are at risk for polymicrobial ascending cholangitis.
PRIMARY IMMUNODEFICIENCIES
PIs can involve isolated or combined defects in various arms of the immune response. The most common PIs include B cell defects, and the most common organisms seen in these patients are encapsulated bacteria, as seen in asplenic patients. OIs begin as maternal antibodies wane in infants, and can include bacteremia and upper and lower respiratory tract disease. T cell (cellular immune) defects present with prolonged or severe viral illnesses, PCP,8mycobacteria, and fungi. Antibody response can be impaired in patients with T cell defects, making these children at higher risk for some of the same bacterial pathogens seen in children with B cell defects. The complement system targets antigens on the surface of pathogens for destruction by phagocytosis. The best-known complement defect is in the terminal portion of the complement cascade (C5–C9), predisposing to meningococcal disease. Phagocyte defects are associated with invasive disease from an early age with S. aureus, Serratia, Pseudomonas, Aspergillus, and Candida. Phagocyte defects should be suspected in any patient who presents with invasive Serratia, even if this is the first documented infection.9
The low prevalence and heterogenous nature of PIs results in few large case series describing infectious etiologies. The most common reasons for hospitalization are gastroenteritis (Giardia, Candida, and Salmonella) and pneumonia.10
Initial diagnostic evaluation should include a CBC, blood culture, rapid viral studies (or PCR), and chest radiograph. For children with diarrhea, fecal white cells, bacterial and viral stool culture, C. difficile, and Cryptosporidium/Giardia antigen testing should be considered. Cryptosporidium also should be in the differential diagnosis of a PI patient with cholangitis. If PCP is considered, elevated lactate dehydrogenase (LDH) may be the first supportive finding. PCR, when available, has excellent negative predictive value for PCP.11 Broad-spectrum antibiotics targeting the most common bacteria isolated from each class of PI should be promptly begun, tailored to prior culture results. Knowledge of the patient’s prophylactic therapy is helpful to potentially narrow the differential diagnosis and to select empiric therapy. For example, if a child is receiving penicillin prophylaxis and presents with pneumonia, ampicillin should not be part of the empiric regimen given the risk of resistance to this class of drugs.
HIV
HIV-infected children may present to the ED with acute HIV infection, OIs (Table 59-4)12 in undiagnosed HIV, or with established disease. The diagnosis of acute HIV infection should be considered in the adolescent who present to the ED with fever, pharyngitis, oral ulcers, and disseminated lymphadenopathy. The presentation mimics mononucleosis, and often occurs 4 to 8 weeks after the infection, during the “window period” where serologies are negative. Diagnosis is established by polymerase chain reaction (PCR). A CBC may show leukopenia, lymphopenia, and thrombocytopenia. The diagnosis can be made in previously undiagnosed children who have harbored the virus for longer periods of time when they present with failure to thrive, chronic diarrhea, or recurrent upper and lower respiratory tract infections. Examination findings may include nontender parotid enlargement, splenomegaly, disseminated lymphadenopathy, and cachexia. OI types are associated with the CD4 cell count (Table 59-4).
|
TABLE 59-4 |
The Most Common Opportunistic Infections seen in Children Infected with Human Immunodeficiency Virus (HIV) |
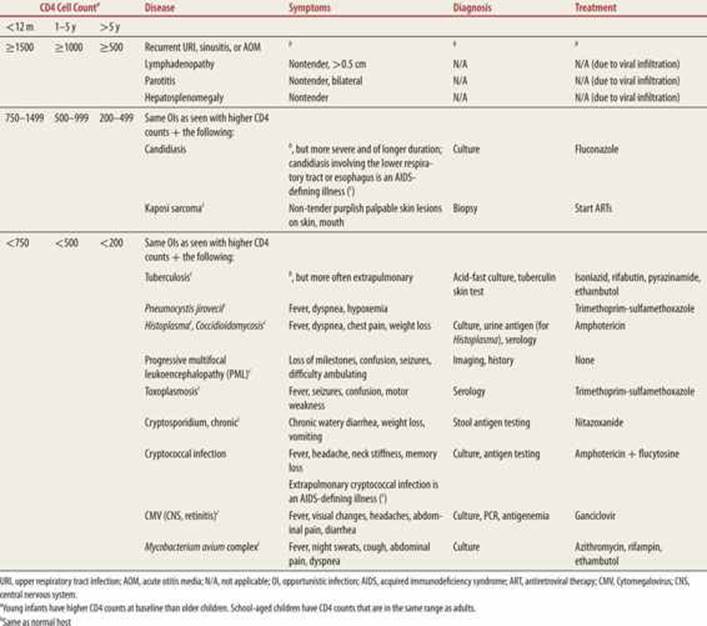
In the child already receiving antiretroviral (ART) therapy, knowledge of the most recent CD4 count, adherence with antiretrovirals (ARVs), and receipt of prophylactic medications may help narrow the differential diagnosis. With good adherence, OIs are less common, but these children may present for ART adverse effects. The most common side effects with nucleoside reverse transcriptase inhibitors (NRTIs) are hepatic toxicity, marrow suppression, and hypersensitivity reactions. Non-NRTIs can be associated with hepatotoxicity and rashes. Protease inhibitors are associated with hepatotoxicity, hyperglycemia, rashes, and fat maldistribution.
One opportunistic infection that must be considered in any HIV-infected patient presenting with respiratory symptoms and/or radiographic infiltrates is PCP. PCP can cause subacute pneumonia, quiet tachypnea, and hypoxemia. The classic radiographic finding is a diffuse reticulonodular pattern. Serum LDH is usually very high. Prompt initiation of high-dose trimethoprim sulfamethoxazole can be life-saving. For patients with a Pao2 of <70 mm Hg on room air, adjunctive corticosteroids should be considered.
REFERENCES
1. Anaissie D. Overview of infections following hematopoietic cell transplantation. In: Basow DS, ed. UpToDate. Waltham, MA: 2012.
2. Danziger-Isakov LA, Sweet S, Delamorena M, Huddleston CB, Mendeloff E, Debaun MR. Epidemiology of bloodstream infections in the first year after pediatric lung transplantation. Pediatr Infect Dis J.2005;24:324–330.
3. Florescu DF, Qiu F, Langnas AN, et al. Bloodstream infections during the first year after pediatric small bowel transplantation. Pediatr Infect Dis J. 2012;31:700–704.
4. Allen U, Green M. Prevention and treatment of infectious complications after solid organ transplantation in children. Pediatr Clin N Am. 2010;57:459–479.
5. Cordero E, Perez-Romero P, Moreno A, et al. Pandemic influenza A (H1N1) virus infection in solid organ transplant recipients: impact of viral and non-viral co-infection. Clin Microbiol Infect. 2012;18: 67–73.
6. Ison MG, de Jong MD, Gilligan KJ, et al: End points for testing influenza antiviral treatments for patients at high risk of severe and life-threatening disease. J Infect Dis. 2010;201: 1654–1662.
7. Kulikowska A, Boslaugh SE, Huddleston CB, Ghandi SK, Gumbiner C, Canter CE. Infectious, malignant, and autoimmune complications in pediatric heart transplant recipients. J Pediatr. 2008;152: 671–677.
8. Saltzman RW, Albin S, Russo P, Sullivan KE. Clinical conditions associated with PCP in children. Pediatr Pulmonol. 2012;47:510–516.
9. Ben-Ari J, Wolach O, Gavrieli R, Wolach B. Infections associated with chronic granulomatous disease: linking genetics to phenotypic expression. Expert Rev Anti-Infect Ther. 2012;10:881–894.
10. Mamishi S, Eghbali AN, Rezaei N, Abolhassani H, Parvanech N, Aghamohammadi A. A single center 14 years study of infectious complications leading to hospitalization of patients with primary antibody deficiencies. Braz J Infect Dis. 2010;14:351–355.
11. Mühlethaler K, Bägli-Stuber K, Wasmer S, et al. Quantitative PCR to diagnose Pneumocystis pneumonia in immunocompromised non-HIV patients. Eur Respir J. 2012;39:971–978.
12. Centers for Disease Control and Prevention. 1994 revised classification system for human immunodeficiency virus infection in children less than 13 years of age. MMWR Recomm Rep. 1994;43:1–19.