STACEY R. ROSE
DEFINITION
Jaundice is the yellow discoloration of the skin, mucous membranes, and sclerae caused by elevated serum levels of bilirubin, a by-product of heme breakdown. Bilirubin is a lipophilic pigment and must bind to plasma albumin to be transported to the liver. It is then taken up by hepatocytes for conjugation with solubilizing sugars to form bilirubin diglucuronides (and, less commonly, monoglucuronides), which can be excreted into bile. Several factors can cause jaundice. While many of these processes are pathologic, physiologic jaundice in neonates, a benign process, accounts for the vast majority of clinically encountered jaundice in pediatrics.
CAUSE AND FREQUENCY
Jaundice may be secondary to an unconjugated or conjugated hyperbilirubinemia (Table 15-1). In general, unconjugated hyperbilirubinemia results from overproduction of bilirubin, impaired uptake of bilirubin by hepatocytes, or impaired conjugation. A primary cause of overproduction is hemolysis, which is usually associated with other laboratory abnormalities including anemia, elevated LDH, low haptoglobin, and increased reticulocyte count. Reduced uptake of bilirubin can be due to poor blood flow to the liver, certain drugs, and various inherited diseases. Impaired bilirubin conjugation is usually due to various inherited disorders, such as Gilbert syndrome or Crigler-Najjar, where the enzymes needed for conjugation are deficient.
TABLE 15-1. Common causes of jaundice in infants and children.
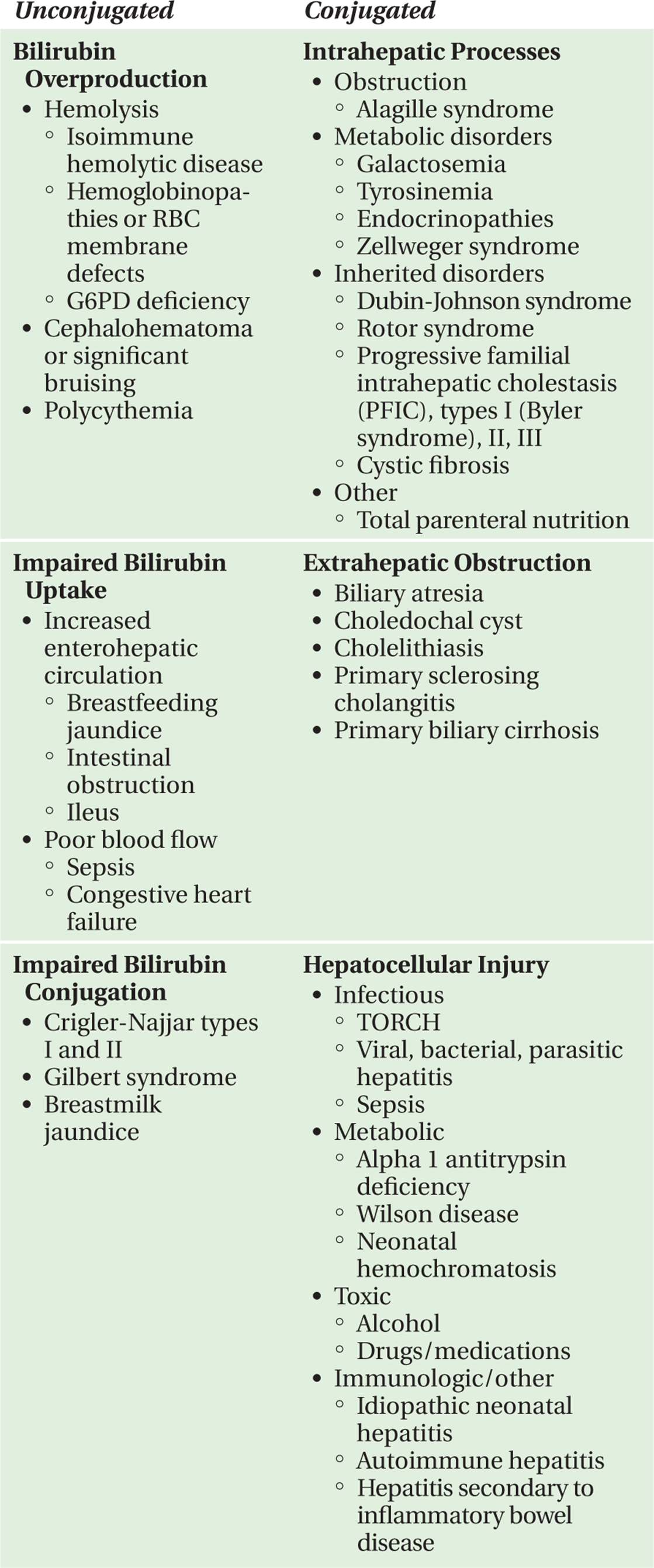
Conjugated hyperbilirubinemia occurs secondary to cholestatic conditions or direct hepatocellular injury. Cholestasis results from intra- or extrahepatic impairments in bile flow, and is accompanied by elevations in alkaline phosphatase and/or gamma-glutamyl transpeptidase (GGT). The differential diagnosis of cholestatic jaundice in the older child differs from diseases that present early in life. Young infants are more likely to have congenital anatomic anomalies, such as biliary atresia, or inborn metabolic disorders such as galactosemia. In contrast, older children are more likely to experience acquired or secondary liver diseases, such as cholelithiasis or sclerosing cholangitis. There is often an overlap between syndromes causing cholestasis and those resulting in hepatocellular injury. Generally, hepatocellular injury is accompanied by elevations in the transaminases (AST and ALT). Additionally, synthetic liver dysfunction, evidenced by hypoalbuminemia as well as prolonged coagulation measurements, may also be present. There are many causes of hepatocellular injury including infectious, autoimmune or toxic hepatitis, or metabolic processes such as alpha-1 antitrypsin deficiency.
QUESTIONS TO ASK AND WHY
• Is the elevated bilirubin level all unconjugated? Is the process a conjugated hyperbilirubinemia?
—Separating a total bilirubin measurement into its conjugated and unconjugated components is a critical step in the evaluation of hyperbilirubinemia in a child. Conjugated hyperbilirubinemia is present when the conjugated fraction is at least 1.5 mg/dL or accounts for greater than 15% of the total bilirubin measurement. Conjugated hyperbilirubinemia is abnormal and merits prompt evaluation, particularly in infants, since diseases like biliary atresia require urgent therapy. An increased unconjugated bilirubin level suggests a very different differential diagnosis and may also be a medical emergency when levels are very high since unconjugated bilirubin is able to cross the blood-brain barrier and directly injures the brain.
Sometimes the terms “direct” and “indirect” bilirubin are used interchangeably with the terms “conjugated” and “unconjugated.” The former terms derive from the van den Bergh reaction in which the conjugated bilirubin component is measured directly (by colorimetric analysis after reaction with a diazo compound). In the van den Bergh assay’s next step, the addition of methanol allows for a measurement of total bilirubin; the unconjugated fraction is then determined—indirectly—by subtracting the conjugated bilirubin level from the total bilirubin level. Of note, measurement of the direct bilirubin fraction detects not only bilirubin di- and mono-glucuronides, but also “delta” bilirubin, which forms when conjugated bilirubin seeps retrograde into the serum and binds covalently to albumin. Because of the delta component’s long half-life, the “direct fraction” can remain deceptively elevated even as a conjugated hyperbilirubinemia improves.
• Does the jaundiced baby have other concerning physical findings?
—A significant unconjugated hyperbilirubinemia can result from the accelerated breakdown of red blood cells secondary to a cephalohematoma or extensive bruising. A newborn afflicted with a TORCH infection might have microcephaly, growth retardation, hepatosplenomegaly, chorioretinitis, or rash. A heart murmur is often heard in children with Alagille syndrome, while a baby with Zellweger syndrome will be hypotonic and dysmorphic. Additionally, the presence of hypotonia, opsithotonus (backward arching of the trunk), or retrocollis (backward arching of the neck) on neurologic examination should raise immediate concerns for acute bilirubin encephalopathy or kernicterus.
• Is there a family history of jaundice?
—Many of the disorders that present with jaundice are heritable. Alpha-1-antitrypsin deficiency, Crigler-Najjar syndromes type I and II, galactosemia, and tyrosinemia are just a few of the auto-somal recessive diseases associated with neonatal jaundice. However, Alagille syndrome is an autosomal dominant disorder (but with variable reentrance and expressivity). The inheritance of glucose-6-phosphate (G6PD) deficiency is X-linked but so highly polymorphic that it should be considered in the evaluation of boys and girls alike.
• Were there changes in the child’s diet, or other new “exposures,” that preceded the onset of jaundice?
—Deficiencies in the metabolism of galactose or fructose can lead to jaundice in infants. Likewise, children with G6PD deficiency can have hemolytic crises triggered by certain foods (e.g., fava beans), medications (e.g., sulfa drugs), and other compounds (e.g., mothballs).
• Does the baby have risk factors for severe neonatal hyperbilirubinemia?
—Risk factors for neonatal jaundice include intrauterine and perinatal complications such as gestational diabetes, prematurity, blood group incompatibility, or birth trauma resulting in extra-vascular blood collections. Other independent risk factors for the infant include ethnicity (East Asian, Native American, and others), polycythemia, acidosis, hypoalbuminemia, exclusive breastfeeding, urinary tract infection or sepsis, and a long, heterogeneous list of genetic disorders.
SUGGESTED READINGS
1. Abrams SH, Shulman RJ. Causes of neonatal cholestasis. UpToDate. www.uptodate.com/contents/causes-of-neonatal-cholestasis. Updated September 1, 2010. Accessed September 22, 2011.
2. American Academy of Pediatrics Subcommittee on Hyperbilirubinemia. American Academy of Pediatrics Clinical Practice Guideline: Management of hyperbilirubinemia in the newborn infant 35 or more weeks of gestation. Pediatrics. 2004;114:297-316.
3. Dennery PA, Seidman DS, Stevenson DK. Neonatal hyperbilirubinemia. N Engl J Med. 2001;344:581-590.
4. Maisels MJ. Neonatal jaundice. Pediatr Rev. 2006;27:443-453.
5. Maisels MJ, McDonagh AF. Phototherapy for neonatal jaundice. N Engl J Med. 2008;358:920-928.