Harry W. Lampiris, MD, & Daniel S. Maddix, PharmD
CASE STUDY
A 51-year-old alcoholic patient presents to the emergency department with fever, headache, neck stiffness, and altered mental status for 12 hours. Vital signs are blood pressure 90/55 mm Hg, pulse 120/min, respirations 30/min, temperature 40°C [104°F] rectal. The patient is minimally responsive to voice and does not follow commands. Examination is significant for a right third cranial nerve palsy and nuchal rigidity. Laboratory results show a white blood cell count of 24,000/mm3 with left shift, but other hematologic and chemistry values are within normal limits. An emergency CT scan of the head is normal. Blood cultures are obtained, and a lumbar puncture reveals the following cerebrospinal fluid (CSF) values: white blood cells 5000/mm3, red blood cells 10/mm3, protein 200 mg/dL, glucose 15 mg/dL (serum glucose 96 taken at same time). CSF Gram stain reveals gram-positive cocci in pairs. What is the most likely diagnosis in this patient? What organisms should be treated empirically? Are there other pharmacologic interventions to consider before initiating antimicrobial therapy?
The development of antimicrobial drugs represents one of the most important advances in therapeutics, both in the control or cure of serious infections and in the prevention and treatment of infectious complications of other therapeutic modalities such as cancer chemotherapy, immunosuppression, and surgery. However, evidence is overwhelming that antimicrobial agents are vastly overprescribed in outpatient settings in the United States, and the availability of antimicrobial agents without prescription in many developing countries has—by facilitating the development of resistance—already severely limited therapeutic options in the treatment of life-threatening infections. Therefore, the clinician should first determine whether antimicrobial therapy is warranted for a given patient. The specific questions one should ask include the following:
1.Is an antimicrobial agent indicated on the basis of clinical findings? Or is it prudent to wait until such clinical findings become apparent?
2.Have appropriate clinical specimens been obtained to establish a microbiologic diagnosis?
3.What are the likely etiologic agents for the patient’s illness?
4.What measures should be taken to protect individuals exposed to the index case to prevent secondary cases, and what measures should be implemented to prevent further exposure?
5.Is there clinical evidence (eg, from well-executed clinical trials) that antimicrobial therapy will confer clinical benefit for the patient?
Once a specific cause is identified based on specific microbiologic tests, the following further questions should be considered:
1.If a specific microbial pathogen is identified, can a narrower-spectrum agent be substituted for the initial empiric drug?
2.Is one agent or a combination of agents necessary?
3.What are the optimal dose, route of administration, and duration of therapy?
4.What specific tests (eg, susceptibility testing) should be undertaken to identify patients who will not respond to treatment?
5.What adjunctive measures can be undertaken to eradicate the infection? For example, is surgery feasible for removal of devitalized tissue or foreign bodies—or drainage of an abscess—into which antimicrobial agents may be unable to penetrate? Is it possible to decrease the dosage of immunosuppressive therapy in patients who have undergone organ transplantation? Is it possible to reduce morbidity or mortality due to the infection by reducing host immunologic response to the infection (eg, by the use of corticosteroids for the treatment of severe Pneumocystis jiroveci pneumonia or meningitis due to Streptococcus pneumoniae)?
![]() EMPIRIC ANTIMICROBIAL THERAPY
EMPIRIC ANTIMICROBIAL THERAPY
Antimicrobial agents are frequently used before the pathogen responsible for a particular illness or the susceptibility to a particular antimicrobial agent is known. This use of antimicrobial agents is called empiric (or presumptive) therapy and is based on experience with a particular clinical entity. The usual justification for empiric therapy is the hope that early intervention will improve the outcome; in the best cases, this has been established by placebo-controlled, double-blind prospective clinical trials. For example, treatment of febrile episodes in neutropenic cancer patients with empiric antimicrobial therapy has been demonstrated to have impressive morbidity and mortality benefits even though the specific bacterial agent responsible for fever is determined for only a minority of such episodes.
Finally, there are many clinical entities, such as certain episodes of community-acquired pneumonia, in which it is difficult to identify a specific pathogen. In such cases, a clinical response to empiric therapy may be an important clue to the likely pathogen.
Frequently, the signs and symptoms of infection diminish as a result of empiric therapy, and microbiologic test results become available that establish a specific microbiologic diagnosis. At the time that the pathogenic organism responsible for the illness is identified, empiric therapy is optimally modified to definitive therapy, which is typically narrower in coverage and is given for an appropriate duration based on the results of clinical trials or experience when clinical trial data are not available.
Approach to Empiric Therapy
Initiation of empiric therapy should follow a specific and systematic approach.
A. Formulate a Clinical Diagnosis of Microbial Infection
Using all available data, the clinician should determine that there is a clinical syndrome compatible with infection (eg, pneumonia, cellulitis, sinusitis).
B. Obtain Specimens for Laboratory Examination
Examination of stained specimens by microscopy or simple examination of an uncentrifuged sample of urine for white blood cells and bacteria may provide important immediate etiologic clues. Cultures of selected anatomic sites (blood, sputum, urine, cerebrospinal fluid, and stool) and nonculture methods (antigen testing, polymerase chain reaction, and serology) may also confirm specific etiologic agents.
C. Formulate a Microbiologic Diagnosis
The history, physical examination, and immediately available laboratory results (eg, Gram stain of urine or sputum) may provide highly specific information. For example, in a young man with urethritis and a Gram-stained smear from the urethral meatus demonstrating intracellular gram-negative diplococci, the most likely pathogen is Neisseria gonorrhoeae. In the latter instance, however, the clinician should be aware that a significant number of patients with gonococcal urethritis have negative Gram stains for the organism and that a significant number of patients with gonococcal urethritis harbor concurrent chlamydial infection that is not demonstrated on the Gram-stained smear.
D. Determine the Necessity for Empiric Therapy
Whether or not to initiate empiric therapy is an important clinical decision based partly on experience and partly on data from clinical trials. Empiric therapy is indicated when there is a significant risk of serious morbidity or mortality if therapy is withheld until a specific pathogen is detected by the clinical laboratory.
In other settings, empiric therapy may be indicated for public health reasons rather than for demonstrated superior outcome of therapy in a specific patient. For example, urethritis in a young sexually active man usually requires treatment for N gonorrhoeae and Chlamydia trachomatis despite the absence of microbiologic confirmation at the time of diagnosis. Because the risk of noncompliance with follow-up visits in this patient population may lead to further transmission of these sexually transmitted pathogens, empiric therapy is warranted.
E. Institute Treatment
Selection of empiric therapy may be based on the microbiologic diagnosis or a clinical diagnosis without available microbiologic clues. If no microbiologic information is available, the antimicrobial spectrum of the agent or agents chosen must necessarily be broader, taking into account the most likely pathogens responsible for the patient’s illness.
Choice of Antimicrobial Agent
Selection from among several drugs depends on host factors that include the following: (1) concomitant disease states (eg, AIDS, neutropenia due to the use of cytotoxic chemotherapy, organ transplantation, severe chronic liver or kidney disease) or the use of immunosuppressive medications; (2) prior adverse drug effects; (3) impaired elimination or detoxification of the drug (may be genetically predetermined but more frequently is associated with impaired renal or hepatic function due to underlying disease); (4) age of the patient; (5) pregnancy status; and (6) epidemiologic exposure (eg, exposure to a sick family member or pet, recent hospitalization, recent travel, occupational exposure, or new sexual partner).
Pharmacologic factors include (1) the kinetics of absorption, distribution, and elimination; (2) the ability of the drug to be delivered to the site of infection; (3) the potential toxicity of an agent; and (4) pharmacokinetic or pharmacodynamic interactions with other drugs.
Knowledge of the susceptibility of an organism to a specific agent in a hospital or community setting is important in the selection of empiric therapy. Pharmacokinetic differences among agents with similar antimicrobial spectrums may be exploited to reduce the frequency of dosing (eg, ceftriaxone, ertapenem, or daptomycin may be conveniently given once every 24 hours). Finally, increasing consideration is being given to the cost of antimicrobial therapy, especially when multiple agents with comparable efficacy and toxicity are available for a specific infection. Changing from intravenous to oral antibiotics for prolonged administration can be particularly cost-effective.
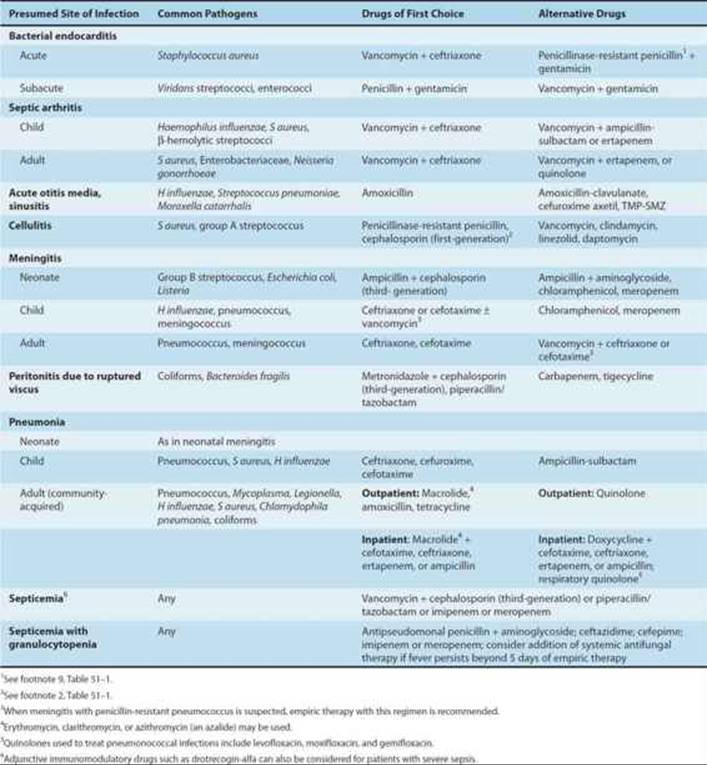
Brief guides to empiric therapy based on presumptive microbial diagnosis and site of infection are given in Tables 51–1 and 51–2.
TABLE 51–1 Empiric antimicrobial therapy based on microbiologic etiology.
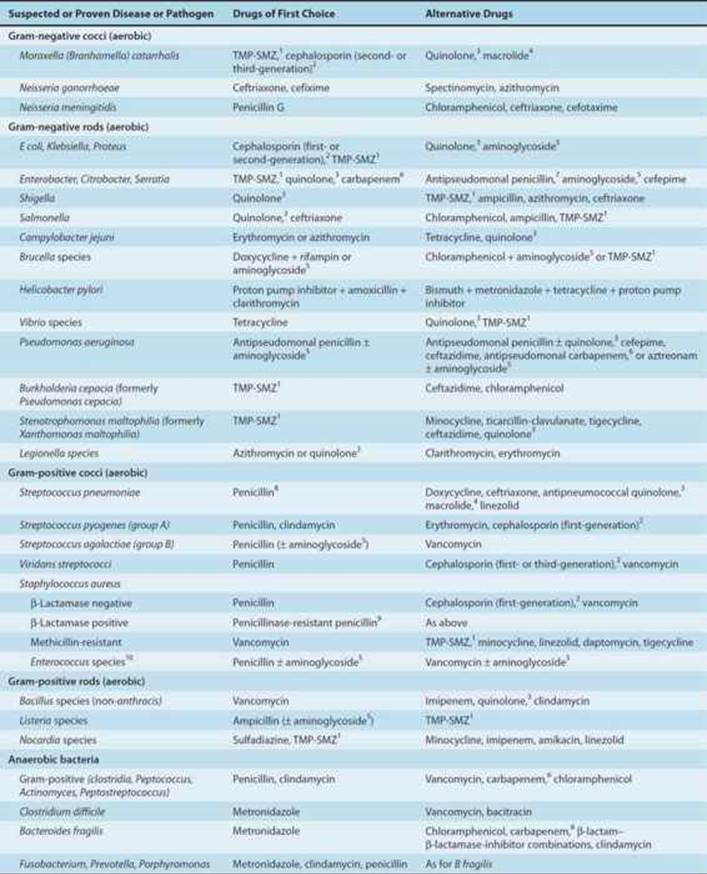
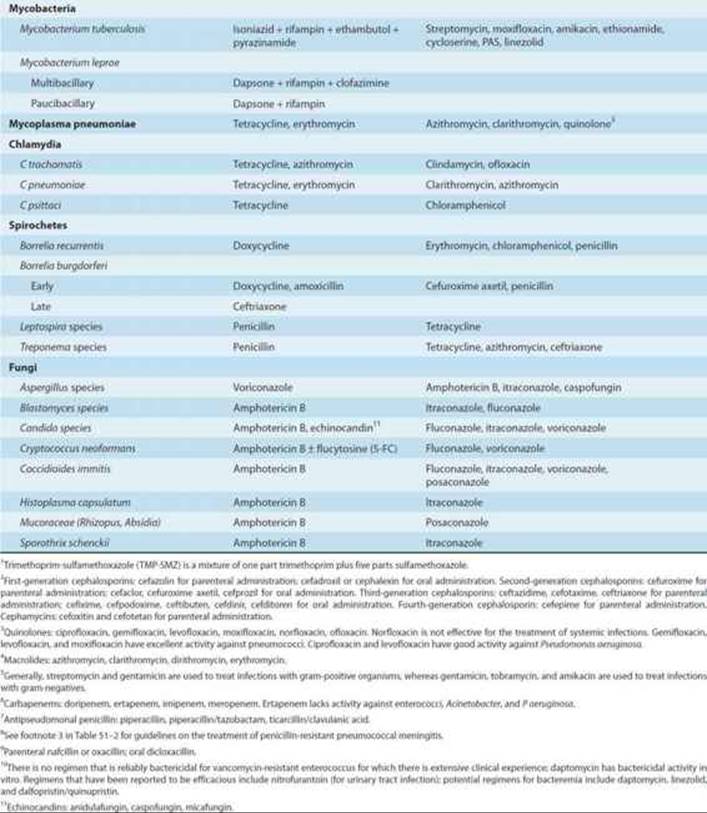
TABLE 51–2 Empiric antimicrobial therapy based on site of infection.
![]() ANTIMICROBIAL THERAPY OF INFECTIONS WITH KNOWN ETIOLOGY
ANTIMICROBIAL THERAPY OF INFECTIONS WITH KNOWN ETIOLOGY
INTERPRETATION OF CULTURE RESULTS
Properly obtained and processed specimens for culture frequently yield reliable information about the cause of infection. The lack of a confirmatory microbiologic diagnosis may be due to the following:
1.Sample error, eg, obtaining cultures after antimicrobial agents have been administered, inadequate volume or quantity of specimen obtained, or contamination of specimens sent for culture
2.Noncultivable or slow-growing organisms (Histoplasma capsulatum, Bartonella or Brucella species), in which cultures are often discarded before sufficient growth has occurred for detection
3.Requesting bacterial cultures when infection is due to other organisms
4.Not recognizing the need for special media or isolation techniques (eg, charcoal yeast extract agar for isolation of Legionella species, shell-vial tissue culture system for rapid isolation of cytomegalovirus)
Even in the setting of a classic infectious disease for which isolation techniques have been established for decades (eg, pneumococcal pneumonia, pulmonary tuberculosis, streptococcal pharyngitis), the sensitivity of the culture technique may be inadequate to identify all cases of the disease.
GUIDING ANTIMICROBIAL THERAPY OF ESTABLISHED INFECTIONS
Susceptibility Testing
Testing bacterial pathogens in vitro for their susceptibility to antimicrobial agents is extremely valuable in confirming susceptibility, ideally to a narrow-spectrum nontoxic antimicrobial drug. Tests measure the concentration of drug required to inhibit growth of the organism (minimal inhibitory concentration [MIC]) or to kill the organism (minimal bactericidal concentration [MBC]). The results of these tests can then be correlated with known drug concentrations in various body compartments. Only MICs are routinely measured in most infections, whereas in infections in which bactericidal therapy is required for eradication of infection (eg, meningitis, endocarditis, sepsis in the granulocytopenic host), MBC measurements occasionally may be useful.
Specialized Assay Methods
A. Beta-Lactamase Assay
For some bacteria (eg, Haemophilus species), the susceptibility patterns of strains are similar except for the production of β lactamase. In these cases, extensive susceptibility testing may not be required, and a direct test for β lactamase using a chromogenic β-lactam substrate (nitrocephin disk) may be substituted.
B. Synergy Studies
Synergy studies are in vitro tests that attempt to measure synergistic, additive, indifferent, or antagonistic drug interactions. In general, these tests have not been standardized and have not correlated well with clinical outcome. (See section on Antimicrobial Drug Combinations for details.)
MONITORING THERAPEUTIC RESPONSE: DURATION OF THERAPY
The therapeutic response may be monitored microbiologically or clinically. Cultures of specimens taken from infected sites should eventually become sterile or demonstrate eradication of the pathogen and are useful for documenting recurrence or relapse. Follow-up cultures may also be useful for detecting superinfections or the development of resistance. Clinically, the patient’s systemic manifestations of infection (malaise, fever, leukocytosis) should abate, and the clinical findings should improve (eg, as shown by clearing of radiographic infiltrates or lessening hypoxemia in pneumonia).
The duration of definitive therapy required for cure depends on the pathogen, the site of infection, and host factors (immunocompromised patients generally require longer courses of treatment). Precise data on duration of therapy exist for some infections (eg, streptococcal pharyngitis, syphilis, gonorrhea, tuberculosis, and cryptococcal meningitis). In many other situations, duration of therapy is determined empirically. For recurrent infections (eg, sinusitis, urinary tract infections), longer courses of antimicrobial therapy or surgical intervention are frequently necessary for eradication.
Clinical Failure of Antimicrobial Therapy
When the patient has an inadequate clinical or microbiologic response to antimicrobial therapy selected by in vitro susceptibility testing, systematic investigation should be undertaken to determine the cause of failure. Errors in susceptibility testing are rare, but the original results should be confirmed by repeat testing. Drug dosing and absorption should be scrutinized and tested directly using serum measurements, pill counting, or directly observed therapy.
The clinical data should be reviewed to determine whether the patient’s immune function is adequate and, if not, what can be done to maximize it. For example, are adequate numbers of granulocytes present and is undiagnosed immunodeficiency, malignancy, or malnutrition present? The presence of abscesses or foreign bodies should also be considered. Finally, culture and susceptibility testing should be repeated to determine whether superinfection has occurred with another organism or whether the original pathogen has developed drug resistance.
ANTIMICROBIAL PHARMACODYNAMIC
The time course of drug concentration is closely related to the antimicrobial effect at the site of infection and to any toxic effects. Pharmacodynamic factors include pathogen susceptibility testing, drug bactericidal versus bacteriostatic activity, drug synergism, antagonism, and postantibiotic effects. Together with pharmacokinetics, pharmacodynamic information permits the selection of optimal antimicrobial dosage regimens.
Bacteriostatic versus Bactericidal Activity
Antibacterial agents may be classified as bacteriostatic or bactericidal (Table 51–3). For agents that are primarily bacteriostatic, inhibitory drug concentrations are much lower than bactericidal drug concentrations. In general, cell wall-active agents are bactericidal, and drugs that inhibit protein synthesis are bacteriostatic.
TABLE 51–3 Bactericidal and bacteriostatic antibacterial agents.
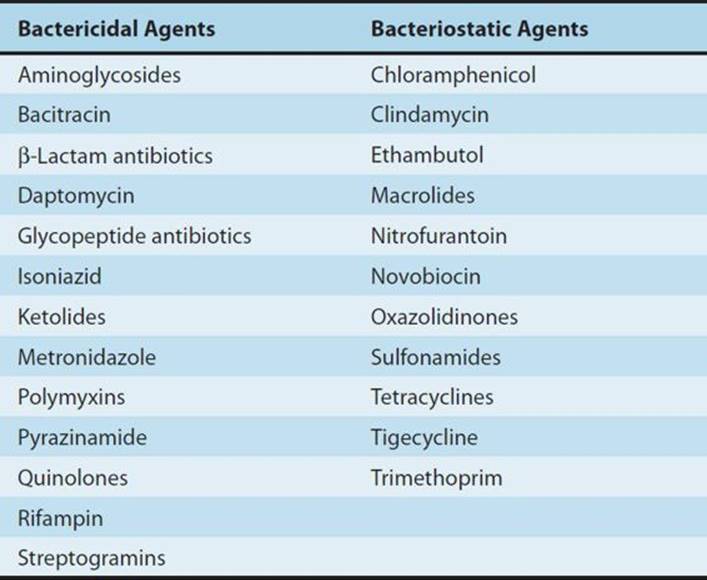
The classification of antibacterial agents as bactericidal or bacteriostatic has limitations. Some agents that are considered to be bacteriostatic may be bactericidal against selected organisms. On the other hand, enterococci are inhibited but not killed by vancomycin, penicillin, or ampicillin used as single agents.
Bacteriostatic and bactericidal agents are equivalent for the treatment of most infectious diseases in immunocompetent hosts. Bactericidal agents should be selected over bacteriostatic ones in circumstances in which local or systemic host defenses are impaired. Bactericidal agents are required for treatment of endocarditis and other endovascular infections, meningitis, and infections in neutropenic cancer patients.
Bactericidal agents can be divided into two groups: agents that exhibit concentration-dependent killing (eg, aminoglycosides and quinolones) and agents that exhibit time-dependent killing (eg, β lactams and vancomycin). For drugs whose killing action is concentration-dependent, the rate and extent of killing increase with increasing drug concentrations. Concentration-dependent killing is one of the pharmacodynamic factors responsible for the efficacy of once-daily dosing of aminoglycosides. For drugs whose killing action is time-dependent, bactericidal activity continues as long as serum concentrations are greater than the MBC.
Postantibiotic Effect
Persistent suppression of bacterial growth after limited exposure to an antimicrobial agent is known as the postantibiotic effect (PAE). The PAE can be expressed mathematically as follows:
PAE = T - C
where T is the time required for the viable count in the test (in vitro) culture to increase tenfold above the count observed immediately before drug removal and C is the time required for the count in an untreated culture to increase tenfold above the count observed immediately after completion of the same procedure used on the test culture. The PAE reflects the time required for bacteria to return to logarithmic growth.
Proposed mechanisms include (1) slow recovery after reversible nonlethal damage to cell structures; (2) persistence of the drug at a binding site or within the periplasmic space; and (3) the need to synthesize new enzymes before growth can resume. Most antimicrobials possess significant in vitro PAEs (≥ 1.5 hours) against susceptible gram-positive cocci (Table 51–4). Antimicrobials with significant PAEs against susceptible gram-negative bacilli are limited to carbapenems and agents that inhibit protein or DNA synthesis.
TABLE 51–4 Antibacterial agents with in vitro postantibiotic effects ≥ 1.5 hours.
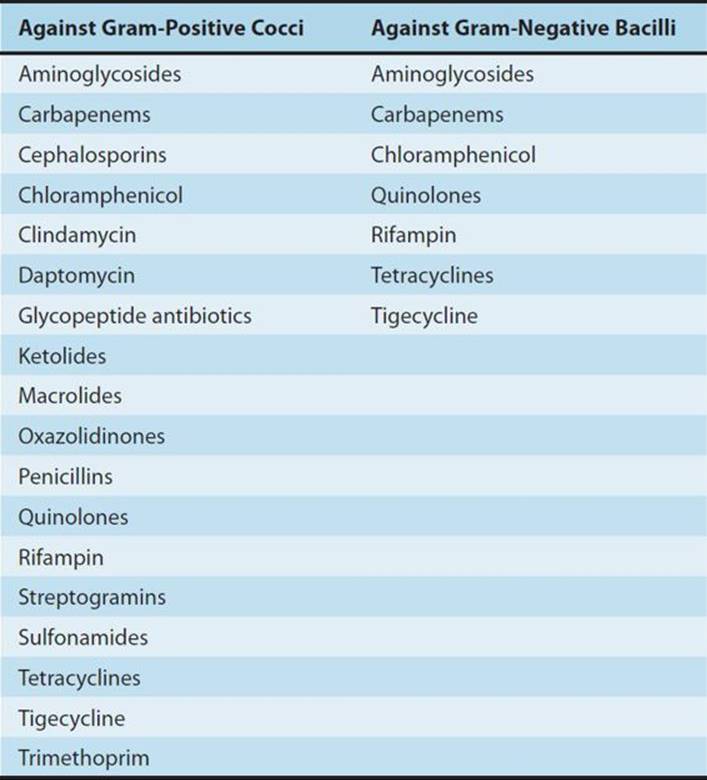
In vivo PAEs are usually much longer than in vitro PAEs. This is thought to be due to postantibiotic leukocyte enhancement (PALE) and exposure of bacteria to subinhibitory antibiotic concentrations. The efficacy of once-daily dosing regimens is in part due to the PAE. Aminoglycosides and quinolones possess concentration-dependent PAEs; thus, high doses of aminoglycosides given once daily result in enhanced bactericidal activity and extended PAEs. This combination of pharmacodynamic effects allows aminoglycoside serum concentrations that are below the MICs of target organisms to remain effective for extended periods of time.
PHARMACOKINETIC CONSIDERATIONS
Route of Administration
Many antimicrobial agents have similar pharmacokinetic properties when given orally or parenterally (ie, tetracyclines, trimethoprim-sulfamethoxazole, quinolones, metronidazole, clindamycin, rifampin, linezolid, and fluconazole). In most cases, oral therapy with these drugs is equally effective, is less costly, and results in fewer complications than parenteral therapy.
The intravenous route is preferred in the following situations: (1) for critically ill patients; (2) for patients with bacterial meningitis or endocarditis; (3) for patients with nausea, vomiting, gastrectomy, ileus, or diseases that may impair oral absorption; and (4) when giving antimicrobials that are poorly absorbed following oral administration.
Conditions That Alter Antimicrobial Pharmacokinetics
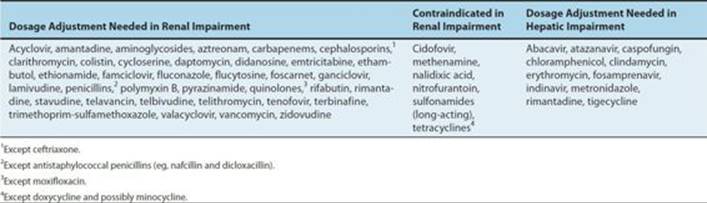
Various diseases and physiologic states alter the pharmacokinetics of antimicrobial agents. Impairment of renal or hepatic function may result in decreased elimination. Table 51–5 lists drugs that require dosage reduction in patients with renal or hepatic insufficiency. Failure to reduce antimicrobial agent dosage in such patients may cause toxic effects. Conversely, patients with burns, cystic fibrosis, or trauma may have increased dosage requirements for selected agents. The pharmacokinetics of antimicrobials is also altered in the elderly (see Chapter 60), in neonates (see Chapter 59), and in pregnancy.
TABLE 51–5 Antimicrobial agents that require dosage adjustment or are contraindicated in patients with renal or hepatic impairment.
Drug Concentrations in Body Fluids
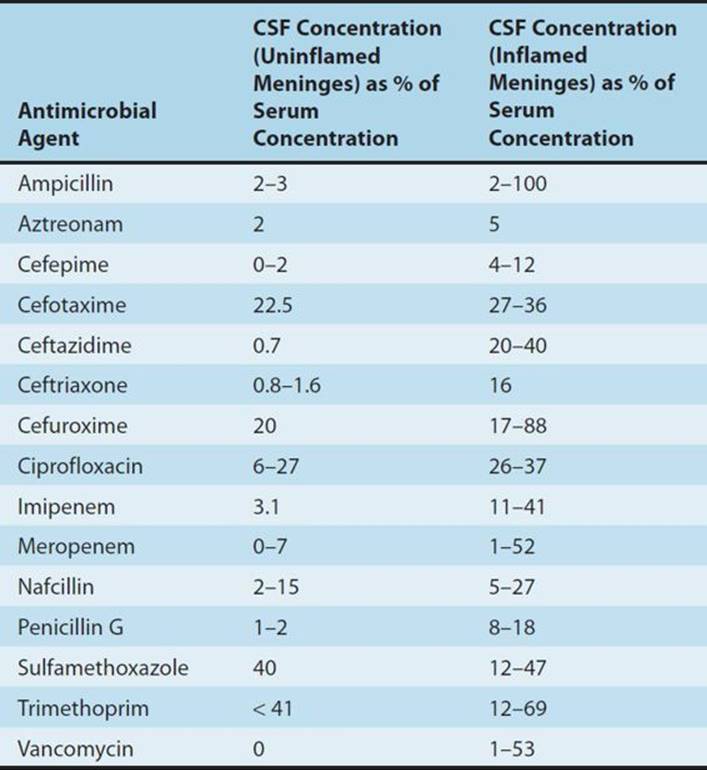
Most antimicrobial agents are well distributed to most body tissues and fluids. Penetration into the cerebrospinal fluid is an exception. Most do not penetrate uninflamed meninges to an appreciable extent. In the presence of meningitis, however, the cerebrospinal fluid concentrations of many antimicrobials increase (Table 51–6).
TABLE 51–6 Cerebrospinal fluid (CSF) penetration of selected antimicrobials.
Monitoring Serum Concentrations of Antimicrobial Agents
For most antimicrobial agents, the relation between dose and therapeutic outcome is well established, and serum concentration monitoring is unnecessary for these drugs. To justify routine serum concentration monitoring, it should be established (1) that a direct relationship exists between drug concentrations and efficacy or toxicity; (2) that substantial interpatient variability exists in serum concentrations on standard doses; (3) that a small difference exists between therapeutic and toxic serum concentrations; (4) that the clinical efficacy or toxicity of the drug is delayed or difficult to measure; and (5) that an accurate assay is available.
In clinical practice, serum concentration monitoring is routinely performed on patients receiving aminoglycosides or vancomycin. Flucytosine serum concentration monitoring has been shown to reduce toxicity when doses are adjusted to maintain peak concentrations below 100 mcg/mL.
![]() MANAGEMENT OF ANTIMICROBIAL DRUG TOXICITY
MANAGEMENT OF ANTIMICROBIAL DRUG TOXICITY
Owing to the large number of antimicrobials available, it is usually possible to select an effective alternative in patients who develop serious drug toxicity (Table 51–1). However, for some infections there are no effective alternatives to the drug of choice. For example, in patients with neurosyphilis who have a history of anaphylaxis to penicillin, it is necessary to perform skin testing and desensitization to penicillin. It is important to obtain a clear history of drug allergy and other adverse drug reactions. A patient with a documented antimicrobial allergy should carry a card with the name of the drug and a description of the reaction. Cross-reactivity between penicillins and cephalosporins is less than 10%. Cephalosporins may be administered to patients with penicillin-induced maculopapular rashes but should be avoided in patients with a history of penicillin-induced immediate hypersensitivity reactions. On the other hand, aztreonam does not cross-react with penicillins and can be safely administered to patients with a history of penicillin-induced anaphylaxis. For mild reactions, it may be possible to continue therapy with use of adjunctive agents or dosage reduction.
Adverse reactions to antimicrobials occur with increased frequency in several groups, including neonates, geriatric patients, renal failure patients, and AIDS patients. Dosage adjustment of the drugs listed in Table 51–5 is essential for the prevention of adverse effects in patients with renal failure. In addition, several agents are contraindicated in patients with renal impairment because of increased rates of serious toxicity (Table 51–5). See the preceding chapters for discussions of specific drugs.
![]() ANTIMICROBIAL DRUG COMBINATIONS
ANTIMICROBIAL DRUG COMBINATIONS
RATIONALE FOR COMBINATION ANTIMICROBIAL THERAPY
Most infections should be treated with a single antimicrobial agent. Although indications for combination therapy exist, antimicrobial combinations are often overused in clinical practice. The unnecessary use of antimicrobial combinations increases toxicity and costs and may occasionally result in reduced efficacy due to antagonism of one drug by another. Antimicrobial combinations should be selected for one or more of the following reasons:
1.To provide broad-spectrum empiric therapy in seriously ill patients.
2.To treat polymicrobial infections (such as intra-abdominal abscesses, which typically are due to a combination of anaerobic and aerobic gram-negative organisms, and enterococci). The antimicrobial combination chosen should cover the most common known or suspected pathogens but need not cover all possible pathogens. The availability of antimicrobials with excellent polymicrobial coverage (eg, β-lactamase inhibitor combinations or carbapenems) may reduce the need for combination therapy in the setting of polymicrobial infections.
3.To decrease the emergence of resistant strains. The value of combination therapy in this setting has been clearly demonstrated for tuberculosis.
4.To decrease dose-related toxicity by using reduced doses of one or more components of the drug regimen. The use of flucytosine in combination with amphotericin B for the treatment of cryptococcal meningitis in non–HIV-infected patients allows for a reduction in amphotericin B dosage with decreased amphotericin B-induced nephrotoxicity.
5.To obtain enhanced inhibition or killing. This use of antimicrobial combinations is discussed in the paragraphs that follow.
SYNERGISM & ANTAGONISM
When the inhibitory or killing effects of two or more antimicrobials used together are significantly greater than expected from their effects when used individually, synergism is said to result. Synergism is marked by a fourfold or greater reduction in the MIC or MBC of each drug when used in combination versus when used alone. Antagonism occurs when the combined inhibitory or killing effects of two or more antimicrobial drugs are significantly less than observed when the drugs are used individually.
Mechanisms of Synergistic Action
The need for synergistic combinations of antimicrobials has been clearly established for the treatment of enterococcal endocarditis. Bactericidal activity is essential for the optimal management of bacterial endocarditis. Penicillin or ampicillin in combination with gentamicin or streptomycin is superior to monotherapy with a penicillin or vancomycin. When tested alone, penicillins and vancomycin are only bacteriostatic against susceptible enterococcal isolates. When these agents are combined with an aminoglycoside, however, bactericidal activity results. The addition of gentamicin or streptomycin to penicillin allows for a reduction in the duration of therapy for selected patients with viridans streptococcal endocarditis. Some evidence exists that synergistic combinations of antimicrobials may be of benefit in the treatment of gram-negative bacillary infections in febrile neutropenic cancer patients and in systemic infections caused by Pseudomonas aeruginosa.
Other synergistic antimicrobial combinations have been shown to be more effective than monotherapy with individual components. Trimethoprim-sulfamethoxazole has been successfully used in the treatment of bacterial infections and P jiroveci (carinii) pneumonia.* β-Lactamase inhibitors restore the activity of intrinsically active but hydrolyzable β lactams against organisms such as Staphylococcus aureus and Bacteroides fragilis. Three major mechanisms of antimicrobial synergism have been established:
1.Blockade of sequential steps in a metabolic sequence: Trimethoprim-sulfamethoxazole is the best-known example of this mechanism of synergy (see Chapter 46). Blockade of the two sequential steps in the folic acid pathway by trimethoprim-sulfamethoxazole results in a much more complete inhibition of growth than achieved by either component alone.
2.Inhibition of enzymatic inactivation: Enzymatic inactivation of β-lactam antibiotics is a major mechanism of antibiotic resistance. Inhibition of β lactamase by β-lactamase inhibitor drugs (eg, sulbactam) results in synergism.
3.Enhancement of antimicrobial agent uptake: Penicillins and other cell wall-active agents can increase the uptake of aminoglycosides by a number of bacteria, including staphylococci, enterococci, streptococci, and P aeruginosa. Enterococci are thought to be intrinsically resistant to aminoglycosides because of permeability barriers. Similarly, amphotericin B is thought to enhance the uptake of flucytosine by fungi.
Mechanisms of Antagonistic Action
There are few clinically relevant examples of antimicrobial antagonism. The most striking example was reported in a study of patients with pneumococcal meningitis. Patients who were treated with the combination of penicillin and chlortetracycline had a mortality rate of 79% compared with a mortality rate of 21% in patients who received penicillin monotherapy (illustrating the first mechanism set forth below).
The use of an antagonistic antimicrobial combination does not preclude other potential beneficial interactions. For example, rifampin may antagonize the action of anti-staphylococcal penicillins or vancomycin against staphylococci. However, the aforementioned antimicrobials may prevent the emergence of resistance to rifampin.
Two major mechanisms of antimicrobial antagonism have been established:
1.Inhibition of cidal activity by static agents: Bacteriostatic agents such as tetracyclines and chloramphenicol can antagonize the action of bactericidal cell wall-active agents because cell wall-active agents require that the bacteria be actively growing and dividing.
2.Induction of enzymatic inactivation: Some gram-negative bacilli, including enterobacter species, P aeruginosa, Serratia marcescens, and Citrobacter freundii, possess inducible β lactamases. β-Lactam antibiotics such as imipenem, cefoxitin, and ampicillin are potent inducers of β-lactamase production. If an inducing agent is combined with an intrinsically active but hydrolyzable β lactam such as piperacillin, antagonism may result.
![]() ANTIMICROBIAL PROPHYLAXIS
ANTIMICROBIAL PROPHYLAXIS
Antimicrobial agents are effective in preventing infections in many settings. Antimicrobial prophylaxis should be used in circumstances in which efficacy has been demonstrated and benefits outweigh the risks of prophylaxis. Antimicrobial prophylaxis may be divided into surgical prophylaxis and nonsurgical prophylaxis.
Surgical Prophylaxis
Surgical wound infections are a major category of nosocomial infections. The estimated annual cost of surgical wound infections in the United States is more than $1.5 billion.
The National Research Council (NRC) Wound Classification Criteria have served as the basis for recommending antimicrobial prophylaxis. NRC criteria consist of four classes (see Box: National Research Council [NRC] Wound Classification Criteria).
The Study of the Efficacy of Nosocomial Infection Control (SENIC) identified four independent risk factors for postoperative wound infections: operations on the abdomen, operations lasting more than 2 hours, contaminated or dirty wound classification, and at least three medical diagnoses. Patients with at least two SENIC risk factors who undergo clean surgical procedures have an increased risk of developing surgical wound infections and should receive antimicrobial prophylaxis.
Surgical procedures that necessitate the use of antimicrobial prophylaxis include contaminated and clean-contaminated operations, selected operations in which postoperative infection may be catastrophic such as open heart surgery, clean procedures that involve placement of prosthetic materials, and any procedure in an immunocompromised host. The operation should carry a significant risk of postoperative site infection or cause significant bacterial contamination.
General principles of antimicrobial surgical prophylaxis include the following:
1.The antibiotic should be active against common surgical wound pathogens; unnecessarily broad coverage should be avoided.
2.The antibiotic should have proved efficacy in clinical trials.
3.The antibiotic must achieve concentrations greater than the MIC of suspected pathogens, and these concentrations must be present at the time of incision.
4.The shortest possible course—ideally a single dose—of the most effective and least toxic antibiotic should be used.
5.The newer broad-spectrum antibiotics should be reserved for therapy of resistant infections.
6.If all other factors are equal, the least expensive agent should be used.
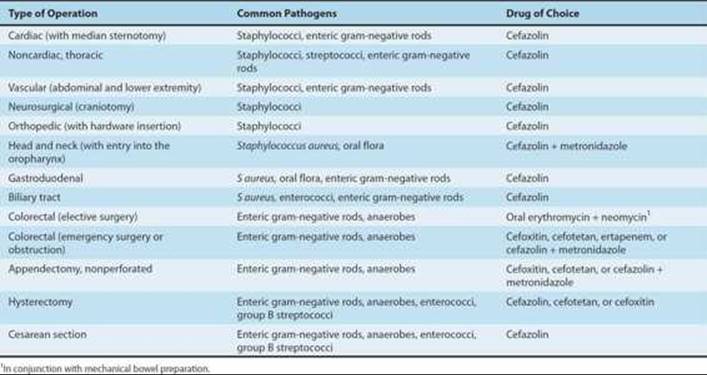
The proper selection and administration of antimicrobial prophylaxis are of utmost importance. Common indications for surgical prophylaxis are shown in Table 51–7. Cefazolin is the prophylactic agent of choice for head and neck, gastroduodenal, biliary tract, gynecologic, and clean procedures. Local wound infection patterns should be considered when selecting antimicrobial prophylaxis. The selection of vancomycin over cefazolin may be necessary in hospitals with high rates of methicillin-resistant S aureus or S epidermidis infections. The antibiotic should be present in adequate concentrations at the operative site before incision and throughout the procedure; initial dosing is dependent on the volume of distribution, peak levels, clearance, protein binding, and bioavailability. Parenteral agents should be administered during the interval beginning 60 minutes before incision. In cesarean section, the antibiotic is administered after umbilical cord clamping. For many antimicrobial agents, doses should be repeated if the procedure exceeds 2–6 hours in duration. Single-dose prophylaxis is effective for most procedures and results in decreased toxicity and antimicrobial resistance.
TABLE 51–7 Recommendations for surgical antimicrobial prophylaxis.
National Research Council (NRC) Wound Classification Criteria
Clean: Elective, primarily closed procedure; respiratory, gastrointestinal, biliary, genitourinary, or oropharyngeal tract not entered; no acute inflammation and no break in technique; expected infection rate ≤ 2%.
Clean contaminated: Urgent or emergency case that is otherwise clean; elective, controlled opening of respiratory, gastrointestinal, biliary, or oropharyngeal tract; minimal spillage or minor break in technique; expected infection rate ≤ 10%.
Contaminated: Acute nonpurulent inflammation; major technique break or major spill from hollow organ; penetrating trauma less than 4 hours old; chronic open wounds to be grafted or covered; expected infection rate about 20%.
Dirty: Purulence or abscess; preoperative perforation of respiratory, gastrointestinal, biliary, or oropharyngeal tract; penetrating trauma more than 4 hours old; expected infection rate about 40%.
Improper administration of antimicrobial prophylaxis leads to excessive surgical wound infection rates. Common errors in antibiotic prophylaxis include selection of the wrong antibiotic, administering the first dose too early or too late, failure to repeat doses during prolonged procedures, excessive duration of prophylaxis, and inappropriate use of broad-spectrum antibiotics.
Nonsurgical Prophylaxis
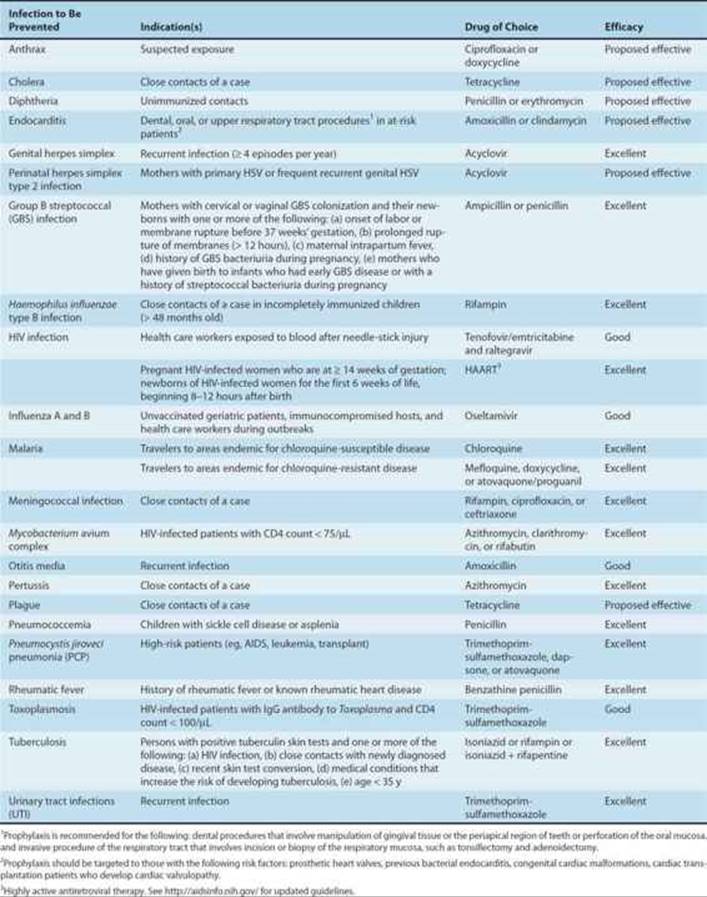
Nonsurgical prophylaxis includes the administration of antimicrobials to prevent colonization or asymptomatic infection as well as the administration of drugs following colonization by or inoculation of pathogens but before the development of disease. Nonsurgical prophylaxis is indicated in individuals who are at high risk for temporary exposure to selected virulent pathogens and in patients who are at increased risk for developing infection because of underlying disease (eg, immunocompromised hosts). Prophylaxis is most effective when directed against organisms that are predictably susceptible to antimicrobial agents. Common indications and drugs for nonsurgical prophylaxis are listed in Table 51–8.
TABLE 51–8 Recommendations for nonsurgical antimicrobial prophylaxis.
REFERENCES
American Thoracic Society: Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med 2005;171:388.
Baddour LM et al: Infective endocarditis: Diagnosis, antimicrobial therapy, and management of complications. Circulation 2005;111:3167.
Baron EJ et al: Guide to utilization of the microbiology laboratory for diagnosis of infectious diseases: 2013 recommendations by the Infectious Diseases Society of America (IDSA) and the American Society for Microbiology (ASM). Clin Infect Dis 2013;57:e22.
Blumberg HM et al: American Thoracic Society/Centers for Disease Control and Prevention/Infectious Diseases Society of America: Treatment of tuberculosis. Am J Respir Crit Care Med 2003;167:603.
Boucher HW: 10 × ‵20 Progress—Development of new drugs active against gram-negative bacilli: An update from the Infectious Diseases Society of America. Clin Infect Dis 2013;56:1685.
Bratzler DW et al: Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am J Health Syst Pharm 2013;70:195.
Gonzales R et al: Principles of appropriate antibiotic use for treatment of acute respiratory tract infections in adults: Background, specific aims, and methods. Ann Intern Med 2001;134:479.
Gruchalla RS et al: Antibiotic allergy. N Engl J Med 2006;354:601.
Jones RN, Pfaller MA: Bacterial resistance: A worldwide problem. Diagn Microbiol Infect Dis 1998;31:379.
Kaye KS, Kaye D: Antibacterial therapy and newer agents. Infect Dis Clin North Am 2009;23:757.
Mandell LA et al: Infectious Diseases Society of America/American Thoracic Society Consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis 2007;44:S27.
Mazuski JE: Surgical infections. Surg Clin North Am 2009;89:295.
National Nosocomial Infections Surveillance (NNIS) System Report, Data Summary from January 1992–June 2004, issued October 2004. Am J Infect Control 2004;32:470.
Panel on Opportunistic Infections in HIV-Infected Adults and Adolescents: Guidelines for the prevention and treatment of opportunistic infections in HIV-infected adults and adolescents: Recommendations from the Centers for Disease Control and Prevention, the National Institutes of Health, and the HIV Medicine Association of the Infectious Diseases Society of America. Available at: http://aidsinfo.nih.gov/contentfiles/lvguidelines/adult_oi.pdf. Accessed August 18, 2013.
Sexually transmitted diseases treatment guidelines 2010. Centers for Disease Control and Prevention. MMWR Morb Mortal Wkly Rep 2010;59(RR-12):1.
Simons FE: Anaphylaxis. J Allergy Clin Immunol 2010;125(Suppl 2):S161.
Spellberg B et al: The future of antibiotics and resistance. N Engl J Med 2013;368:299.
Tunkel AR et al: Practice guidelines for the management of bacterial meningitis. Clin Infect Dis 2004;39:1267.
Wilson W et al: Prevention of infective endocarditis: Guidelines from the American Heart Association. Circulation 2007;116:1736.
CASE STUDY ANSWER
The most likely diagnosis for this patient is Streptococcus pneumoniae meningitis, the most common bacterial cause of meningitis in adults. Other possible microbiologic etiologies include Neisseria meningitidis, Listeria monocytogenes, and enteric gram-negative bacilli. Intravenous antimicrobials to which local strains of these organisms are sensitive should be started while awaiting culture and sensitivity results. In addition, the use of dexamethasone has been demonstrated to reduce mortality in adults with pneumococcal meningitis in conjunction with appropriate antimicrobial therapy.