Cholinoceptor-Activating & Cholinesterase-Inhibiting Drugs: Introduction
Cholinomimetic drugs are drugs that mimic acetylcholine by directly or indirectly activating the receptors with which acetylcholine interacts. The direct-acting agents combine with the cholinoceptor in the same way as acetylcholine. The indirect-acting cholinesterase inhibitors have many effects like those of the direct-acting agonists, but they act by inhibiting the enzyme that terminates the action of endogenous acetylcholine.
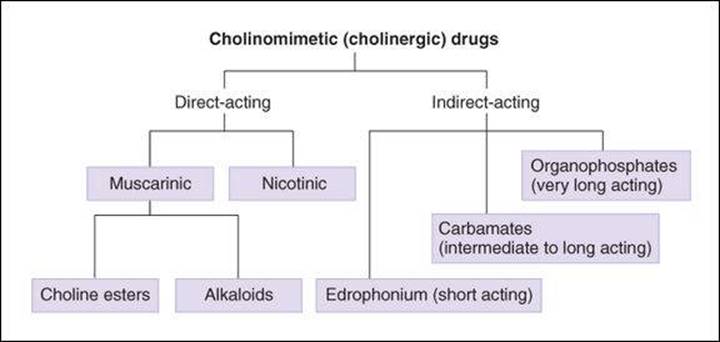
Drugs with acetylcholine-like effects (cholinomimetics) consist of 2 major subgroups on the basis of their mode of action (ie, whether they act directly at the acetylcholine receptor or indirectly through inhibition of cholinesterase). Drugs in the direct-acting subgroup are further subdivided on the basis of their spectrum of action (ie, whether they act on muscarinic or nicotinic cholinoceptors).
Acetylcholine may be considered the prototype that acts directly at both muscarinic and nicotinic receptors. Neostigmine is a prototype for the indirect-acting cholinesterase inhibitors.
High-Yield Terms to Learn
Choline ester A cholinomimetic drug consisting of choline (an alcohol) esterified with an acidic substance, (eg, acetic or carbamic acid); usually poorly lipid-soluble Cholinergic crisis The clinical condition of excessive activation of cholinoceptors; it may include skeletal muscle weakness as well as parasympathetic signs Cholinomimetic alkaloid A drug with weakly basic properties (usually of plant origin) whose effects resemble those of acetylcholine; usually lipid-soluble Cyclospasm Marked contraction of the ciliary muscle; maximum accommodation for close vision Direct-acting cholinomimetic A drug that binds and activates cholinoceptors; the effects mimic those of acetylcholine Endothelium-derived relaxing factor (EDRF) A potent vasodilator substance, largely nitric oxide (NO), that is released from vascular endothelial cells Indirect-acting cholinomimetic A drug that amplifies the effects of endogenous acetylcholine by inhibiting acetylcholinesterase Muscarinic agonist A cholinomimetic drug that binds muscarinic receptors and has primarily muscarine-like actions Myasthenic crisis In patients with myasthenia, an acute worsening of symptoms; usually relieved by increasing cholinomimetic treatment Nicotinic agonist A cholinomimetic drug that binds nicotinic receptors and has primarily nicotine-like actions Organophosphate An ester of phosphoric acid and an alcohol that inhibits cholinesterase Organophosphate aging A process whereby the organophosphate, after binding to cholinesterase, is chemically modified and becomes more firmly bound to the enzyme Parasympathomimetic A drug whose effects resemble those of stimulating the parasympathetic nerves
Direct-Acting Cholinomimetic Agonists
This class comprises a group of choline esters (acetylcholine, methacholine, carbachol, and bethanechol) and a second group of naturally occurring alkaloids (muscarine, pilocarpine, nicotine, lobeline). Newer drugs are occasionally introduced for special applications. The members differ in their spectrum of action (amount of muscarinic versus nicotinic stimulation) and in their pharmacokinetics (Table 7-1). Both factors influence their clinical use.
TABLE 7-1 Some cholinomimetics: spectrum of action and pharmacokinetics.
Drug Spectrum of Actiona
Pharmacokinetic Features Direct-acting Acetylcholine B Rapidly hydrolyzed by cholinesterase (ChE); duration of action 5-30 s; poor lipid solubility Bethanechol M Resistant to ChE; orally active, poor lipid solubility; duration of action 30 min to 2 h Carbachol B Like bethanechol Pilocarpine M Not an ester, good lipid solubility; duration of action 30 min to 2 h Nicotine N Like pilocarpine; duration of action 1-6 h; high lipid solubility Varenicline N Partial agonist at N receptors, high lipid solubility; duration 12-24 h Indirect-acting Edrophonium B Alcohol, quaternary amine, poor lipid solubility, not orally active; duration of action 5-15 min Neostigmine B Carbamate, quaternary amine, poor lipid solubility, orally active; duration of action 30 min to 2 h or more Physostigmine B Carbamate, tertiary amine, good lipid solubility, orally active; duration of action 30 min to 2 h Pyridostigmine B Carbamate-like neostigmine, but longer duration of action (4-8 h) Echothiophate B Organophosphate, moderate lipid solubility; duration of action 2-7 days Parathion B Organophosphate, high lipid solubility; duration of action 7-30 days
aB, both; M, muscarinic; N, nicotinic.
Classification
Muscarinic agonists are parasympathomimetic; that is, they mimic the actions of parasympathetic nerve stimulation in addition to other effects. Five subgroups of muscarinic receptors have been identified (Table 7-2), but the muscarinic agonists available for clinical use activate them nonselectively. Nicotinic agonists are classified on the basis of whether ganglionic or neuromuscular stimulation predominates; however, agonist selectivity is limited. On the other hand, relatively selective antagonists are available for the two nicotinic receptor types (Chapter 8).
TABLE 7-2 Cholinoceptor types and their postreceptor mechanisms.
Receptor Type G Protein Postreceptor Mechanisms M1
Gq
![]() IP3, DAG cascade
IP3, DAG cascade
M2
Gi
![]() cAMP synthesis M3
cAMP synthesis M3
Gq
![]() IP3, DAG cascade
IP3, DAG cascade
M4
Gi
![]() cAMP synthesis M5
cAMP synthesis M5
Gq
![]() IP3, DAG cascade
IP3, DAG cascade
NM
None Na+/K+ depolarizing current
NN
None Na+/K+ depolarizing current
cAMP, cyclic adenosine monophosphate; DAG, diacylglycerol; IP3 inositol-1,4,5-trisphosphate
Molecular Mechanisms of Action
Muscarinic Mechanism
Several molecular mechanisms of muscarinic action have been defined (Table 7-2). One involves Gq-protein coupling of M1 and M3 muscarinic receptors to phospholipase C, a membrane-bound enzyme, leading to the release of the second messengers, diacylglycerol (DAG) and inositol-1,4,5-trisphosphate (IP 3). DAG modulates the action of protein kinase C, an enzyme important in secretion, whereas IP3 evokes the release of calcium from intracellular storage sites, which results in contraction in smooth muscle. A second mechanism couples M2 muscarinic receptors to adenylyl cyclase through the inhibitory Gi-coupling protein. A third mechanism couples the same M2 receptors via the ![]()
![]() subunit of the G protein to potassium channels in the heart and elsewhere; muscarinic agonists facilitate opening of these channels. M4 and M5 receptors may be important in the central nervous system (CNS) but have not been shown to play major roles in peripheral organs.
subunit of the G protein to potassium channels in the heart and elsewhere; muscarinic agonists facilitate opening of these channels. M4 and M5 receptors may be important in the central nervous system (CNS) but have not been shown to play major roles in peripheral organs.
Nicotinic Mechanism
The mechanism of nicotinic action has been clearly defined. The nicotinic acetylcholine ACh receptor is located on a channel protein that is selective for sodium and potassium. When the receptor is activated, the channel opens and depolarization of the cell occurs as a direct result of the influx of sodium, causing an excitatory postsynaptic potential (EPSP). If large enough, the EPSP evokes a propagated action potential in the surrounding membrane. The nicotinic receptors on sympathetic and parasympathetic ganglion cells (NN, also denoted NG) differ slightly from those on neuromuscular end plates (NM).
Tissue and Organ Effects
The tissue and organ system effects are summarized in Table 7-3. Note that vasodilation (and decreased blood pressure) is not a parasympathomimetic response (ie, it is not evoked by parasympathetic nerve discharge, even though directly acting cholinomimetics cause vasodilation). This action results from the release of endothelium-derived relaxing factor (EDRF; nitric oxide and possibly other substances) in the vessels, mediated by uninnervated muscarinic receptors on the endothelial cells. Note also that decreased blood pressure evokes the baroreceptor reflex, resulting in strong compensatory sympathetic discharge to the heart. As a result, injections of small to moderate amounts of direct-acting muscarinic cholinomimetics often cause tachycardia, whereas parasympathetic (vagal) nerve discharge to the heart causes bradycardia. Another effect seen with cholinomimetic drugs but not with parasympathetic nerve stimulation is thermoregulatory sweating; this is a sympathetic cholinergic effect (see Chapter 6).
TABLE 7-3 Effects of cholinomimetics on major organ systems.
Organ Responsea
CNS Complex stimulatory effects, for example, nicotine (elevation of mood, alerting, addiction), physostigmine (convulsions); excessive concentrations may cause coma Eye Sphincter muscle of iris Contraction (miosis) Ciliary muscle Contraction (accommodation for near vision), cyclospasm Heart Sinoatrial node Decrease in rate (negative chronotropy), but note important reflex response in intact subject (see text) Atria Decrease in contractile force (negative inotropy); decrease in refractory period Atrioventricular node Decrease in conduction velocity (negative dromotropy), increase in refractory period Ventricles Small decrease in contractile force Blood vessels Dilation via release of EDRF from endothelium Bronchi Contraction (bronchoconstriction) Gastrointestinal tract Motility Increase in smooth muscle contraction, peristalsis Sphincters Decrease in tone, relaxation Urinary bladder Detrusor Increase in contraction Trigone and sphincter Relaxation; voiding Skeletal muscle Activation of neuromuscular end plates, contraction Glands (exocrine) Increased secretion (thermoregulatory sweating, lacrimation, salivation, bronchial secretion, gastrointestinal glands)
aOnly the direct effects are indicated; homeostatic responses to these direct actions may be important (see text).
EDRF, endothelium-derived relaxing factor (primarily nitric oxide).
The tissue and organ level effects of nicotinic ganglionic stimulation depend on the autonomic innervation of the organ involved. The blood vessels are dominated by sympathetic innervation; therefore, nicotinic receptor activation results in vasoconstriction mediated by sympathetic postganglionic nerve discharge. The gut is dominated by parasympathetic control; nicotinic drugs increase motility and secretion because of increased parasympathetic postganglionic neuron discharge. Nicotinic neuromuscular end plate activation by direct-acting drugs results in fasciculations and spasm of the muscles involved. Prolonged activation results in paralysis (see Chapter 27), which is an important hazard of exposure to nicotine-containing and organophosphate insecticides.
Clinical Use
Several clinical conditions benefit from an increase in cholinergic activity, including glaucoma, Sjogren's syndrome, and loss of normal PANS activity in the bowel and bladder. See the Drug Summary Table at the end of this chapter. Direct-acting nicotinic agonists have no therapeutic applications except in smoking cessation and producing skeletal muscle paralysis (succinylcholine, Chapter 27). Indirect-acting agents are used when increased nicotinic activation is needed at the neuromuscular junction (see later discussion). Nicotine is still used, however, as an insecticide. Varenicline is a newer nicotinic agonist with partial agonist properties. It appears to reduce craving in persons addicted to nicotine.
Toxicity
The signs and symptoms of overdosage are readily predicted from the general pharmacology of acetylcholine.
Muscarinic Toxicity
These effects include CNS stimulation (uncommon with choline esters and pilocarpine), miosis, spasm of accommodation, bronchoconstriction, excessive gastrointestinal and genitourinary smooth muscle activity, increased secretory activity (sweat glands, airway, gastrointestinal tract, lacrimal glands), and vasodilation. Transient bradycardia occurs, followed by reflex tachycardia if the drug is administered as an intravenous bolus; reflex tachycardia occurs otherwise. Muscarine and similar alkaloids are found in certain mushrooms (Inocybe species and Amanita muscaria) and are responsible for the short-acting form of mushroom poisoning, which is characterized by nausea, vomiting, and diarrhea. (The much more dangerous and potentially lethal form of mushroom poisoning from Amanita phalloides and related species involves initial vomiting and diarrhea but is followed by hepatic and renal necrosis. It is not caused by muscarinic agonists but by amanitin and phalloidin, RNA polymerase inhibitors.)
Nicotinic Toxicity
Toxic effects include ganglionic stimulation and block and neuromuscular end plate depolarization leading to fasciculations and then paralysis. CNS toxicity includes stimulation (including convulsions) followed by depression.
Skill Keeper: Drug Metabolism
(See Chapter 4)
Acetylcholine is metabolized in the body by hydrolysis of the ester bond. Is this a phase I or phase II metabolic reaction? The Skill Keeper Answer appears at the end of the chapter.
Indirect-Acting Agonists
Classification and Prototypes
Hundreds of indirect-acting cholinomimetic drugs have been synthesized in 2 major chemical classes: carbamic acid esters (carbamates) and phosphoric acid esters (organophosphates). Neostigmine is a prototypic carbamate, whereas parathion, an important insecticide, is a prototypic organophosphate. A third class has only one clinically useful member— edrophonium is an alcohol (not an ester) with a very short duration of action.
Mechanism of Action
Both carbamate and organophosphate inhibitors bind to cholinesterase and undergo prompt hydrolysis. The alcohol portion of the molecule is then released. The acidic portion (carbamate ion or phosphate ion) is released much more slowly, and this retained portion prevents the binding and hydrolysis of endogenous acetylcholine, thus amplifying acetylcholine effects wherever the transmitter is released.
Carbamates
After hydrolysis, the carbamate residue is released by cholinesterase over a period of 2-8 h.
Organophosphates
Organophosphates are long-acting drugs; they form an extremely stable phosphate complex with the enzyme. After initial hydrolysis, the phosphoric acid residue is released over periods of days to weeks. Recovery is due in part to synthesis of new enzyme.
Effects
By inhibiting cholinesterase, these agents cause an increase in the concentration, half-life, and actions of acetylcholine in synapses where acetylcholine is released physiologically. Therefore, the indirect agents have muscarinic or nicotinic effects depending on which organ system is under consideration. Cholinesterase inhibitors do not have significant actions at uninnervated sites where acetylcholine is not normally released (eg, vascular endothelial cells).
Clinical Use
The clinical applications of the indirect-acting cholinomimetics are predictable from a consideration of the organs and the diseases that benefit from an amplification of cholinergic activity. The effects are summarized in the Drug Summary Table. Carbamates, which include neostigmine, physostigmine, pyridostigmine , and ambenonium , are used far more commonly in therapeutics than are organophosphates. The treatment of myasthenia is especially important. (Because myasthenia has important autoimmune aspects, treatment may also include thymectomy and immunosuppressant drugs.) Rivastigmine , a carbamate, and several other cholinesterase inhibitors are used exclusively in Alzheimer's disease. A portion of their action may be due to other, unknown mechanisms. Their effects are significant but modest and temporary. However, lacking any better therapy, these drugs are frequently used in this devastating condition. Some carbamates (eg, carbaryl) are used in agriculture as insecticides. Two organophosphates used in medicine are malathion (a scabicide), and metrifonate (an antihelminthic agent).
Edrophonium is used for the rapid reversal of nondepolarizing neuromuscular blockade (Chapter 27), in the diagnosis of myasthenia, and in differentiating myasthenic crisis from cholinergic crisis in patients with this disease. Because cholinergic crisis can result in muscle weakness like that of myasthenic crisis, distinguishing the 2 conditions may be difficult. Administration of a short-acting cholinomimetic, such as edrophonium, will improve muscle strength in myasthenic crisis but weaken it in cholinergic crisis.
Toxicity
In addition to their therapeutic uses, some indirect-acting agents (especially organophosphates) have clinical importance because of accidental exposures to toxic amounts of pesticides. The most toxic of these drugs (eg, parathion) can be rapidly fatal if exposure is not immediately recognized and treated. After standard protection of vital signs (see Chapter 58), the antidote of first choice is the antimuscarinic agent atropine , but this drug has no effect on the nicotinic signs of toxicity. Nicotinic toxicity is treated by regenerating active cholinesterase. Immediately after binding to cholinesterase, most organophosphate inhibitors can be removed from the enzyme by the use of regenerator compounds such as pralidoxime (see Chapter 8), and this may reverse both nicotinic and muscarinic signs. If the enzyme-phosphate binding is allowed to persist, however, aging (a further chemical change) occurs and regenerator drugs can no longer remove the inhibitor. Treatment is described in more detail in Chapter 8.
Because of their toxicity and short persistence in the environment, organophosphates are used extensively in agriculture as insecticides and antihelminthic agents; examples are malathion and parathion. Some of these agents (eg, malathion, dichlorvos) are relatively safe in humans because they are metabolized rapidly to inactive products in mammals (and birds) but not in insects. Some are prodrugs (eg, malathion, parathion) and must be metabolized to the active product (malaoxon from malathion, paraoxon from parathion). The signs and symptoms of poisoning are the same as those described for the direct-acting agents, with the following exceptions: vasodilation is a late and uncommon effect; bradycardia is more common than tachycardia; CNS stimulation is common with organophosphate and physostigmine overdosage and includes convulsions, followed by respiratory and cardiovascular depression. The spectrum of toxicity can be remembered with the aid of the mnemonic DUMBBELSS (diarrhea, urination, miosis, bronchoconstriction, bradycardia, excitation [of skeletal muscle and CNS], lacrimation, and salivation and sweating).
Skill Keeper Answer: Drug Metabolism
(See Chapter 4)
The esters acetylcholine and methacholine are hydrolyzed by acetylcholinesterase. Hydrolytic drug metabolism reactions are classified as phase I.
Checklist
When you complete this chapter, you should be able to:
![]() List the locations and types of acetylcholine receptors in the major organ systems (CNS, autonomic ganglia, eye, heart, vessels, bronchi, gut, genitourinary tract, skeletal muscle, exocrine glands).
List the locations and types of acetylcholine receptors in the major organ systems (CNS, autonomic ganglia, eye, heart, vessels, bronchi, gut, genitourinary tract, skeletal muscle, exocrine glands).
![]() Describe the second messengers involved and the effects of acetylcholine on the major organs.
Describe the second messengers involved and the effects of acetylcholine on the major organs.
![]() List the major clinical uses of cholinomimetic agonists.
List the major clinical uses of cholinomimetic agonists.
![]() Describe the pharmacodynamic differences between direct-acting and indirect-acting cholinomimetic agents.
Describe the pharmacodynamic differences between direct-acting and indirect-acting cholinomimetic agents.
![]() Relate the different pharmacokinetic properties of the various choline esters and cholinomimetic alkaloids to their chemical properties.
Relate the different pharmacokinetic properties of the various choline esters and cholinomimetic alkaloids to their chemical properties.
![]() List the major signs and symptoms of (1) organophosphate insecticide poisoning and (2) acute nicotine toxicity.
List the major signs and symptoms of (1) organophosphate insecticide poisoning and (2) acute nicotine toxicity.
Drug Summary Table: Cholinoceptor-Activating & Cholinesterase-Inhibiting Drugs
Subclass Mechanism of Action Clinical and Other Applications Pharmacokinetics Toxicities, Interactions Direct-acting, muscarinic agonists Bethanechol Activates muscarinic (M) receptors; increases IP 3 and DAG
Bladder and bowel atony, for example, after surgery or spinal cord injury Oral, IM activity Poor lipid solubility: does not enter CNS; not active in eye after topical application Duration: 0.3-2 h All parasympathomimetic effects: cyclospasm, diarrhea, urinary urgency, plus vasodilation, reflex tachycardia, and sweating Pilocarpine Same as bethanechol; may also activate EPSP via M receptors in ganglia Sjögren's syndrome (increases salivation); was used in glaucoma (causes miosis, cyclospasm) Oral, IM activity Good lipid solubility, topical activity in eye Similar to bethanechol but may cause vasoconstriction via ganglionic effect Muscarine Same as bethanechol Alkaloid found in mushrooms Low lipid solubility but readily absorbed from gut Mushroom poisoning of fast-onset type Direct-acting, nicotinic agonists Nicotine Activates all nicotinic (N) receptors; opens Na-K channels in ganglia and neuromuscular end plates Smoking cessation (also used as insecticide) High lipid solubility, absorbed by all routes For smoking cessation: usually used as gum or transdermal patch Duration: 4-6 h
Generalized ganglionic stimulation: hypertension, tachycardia, nausea, vomiting, diarrhea
Major overdose: convulsions, paralysis, coma
Varenicline A partial agonist at N receptors Smoking cessation High lipid solubility, oral activity Duration: ~12 h Hypertension, sweating, sensory disturbance, diarrhea, polyuria, menstrual disturbance Succinylcholine N-receptor agonist, moderately selective for neuromuscular (NM) end plate NM relaxation (see Chapter 27)
Highly polar, used IV Duration: 5-10 min Initial muscle spasms and postoperative pain Prolonged action in persons with abnormal butyrylcholinesterase Indirect-acting, alcohol Edrophonium Inhibitor of cholinesterase; amplifier of endogenously released ACh Reversal of NM block by nondepolarizing drugs; diagnosis of myasthenia gravis
Highly polar; used IV
Duration: 5-10 min
Increased parasympathetic effects, especially nausea, vomiting, diarrhea, urinary urgency Indirect-acting, carbamate Neostigmine Like edrophonium plus small direct nicotinic agonist action Reversal of NM block, treatment of myasthenia
Moderately polar but orally active Duration: 2-4 h Like edrophonium but longer duration Pyridostigmine Like edrophonium Treatment of myasthenia Moderately polar but orally active Duration: 4-8 h Like edrophonium but longer duration Physostigmine Like edrophonium Reversal of severe atropine poisoning (IV); occasionally used in acute glaucoma (topical)
Lipid soluble; can be used topically in the eye
Duration: 2-4 h
Like edrophonium but longer duration plus CNS effects: seizures Indirect-acting, organophosphate Parathion Like edrophonium Insecticide only Duration: days to weeks Highly lipid-soluble Highly dangerous insecticide; causes all parasympathetic effects plus muscle paralysis and coma Malathion Like edrophonium
Insecticide and scabicide (topical)
Duration: days
Highly lipid-soluble but metabolized to inactive products in mammals and birds Much safer insecticide than parathion Sarin, tabun, others Like parathion Nerve gases; terrorist threat Like parathion but more rapid action Rapidly lethal Indirect-acting, for Alzheimer's disease Rivastigmine, galantamine, donepezil, tacrine (obsolete) Cholinesterase inhibition plus variable other poorly understood effects Alzheimer's disease Lipid soluble, enter CNS Half-lives: 1.5-70 h Nausea, vomiting
ACh, acetylcholine; DAG, diacylglycerol; EPSP, excitatory postsynaptic potential; IP3 inositol-1,4,5-trisphosphate.