MAJOR DRUG CLASSES
Antihelminths
Antiprotozoals
Drugs for intestinal and vaginal protozoa
Antimalarials
Antikinetoplastides
Therapeutic Overview
Parasitic infections are an important cause of morbidity throughout the world. Enteric parasites are prevalent in developing areas where sanitation and public health measures are poor. They intermittently cause epidemics in industrialized countries when they gain access to water or food supplies. An estimated 1.2 billion people, for example, are infected with the roundworm Ascaris lumbricoides worldwide, and hookworms are the leading cause of iron deficiency anemia in many areas. Arthropod-borne parasites are endemic in the tropics, and malaria poses a major health problem for residents of many tropical areas and for international travelers. More than 1 million deaths are attributed annually to malaria in sub-Saharan Africa alone. Trichomonas vaginalis is a common cause of vaginitis. Of the Kinetoplastida,Trypanosoma cruzi, the cause of Chagas’ disease, is endemic in Latin America; Trypanosoma brucei gambiense and Trypanosoma brucei rhodesiense cause sleeping sickness in Africa; and Leishmaniaspecies are present in widely scattered areas on every continent except Australia and Antarctica. Toxoplasma gondii is endemic worldwide. In industrialized countries parasitic diseases most commonly affect refugees, immigrants, military personnel, returning international travelers, and occasionally residents who have not traveled. Several protozoa have also emerged as important opportunistic pathogens in patients with acquired immunodeficiency syndrome (AIDS).
Prevention strategies and the major transmission pathways for protozoal infections are in the Therapeutic Overview Box.
CLASSIFICATION OF MAJOR PARASITIC GROUPS
There are two major groups of parasites: multicellular helminths (worms) and single-celled protozoa.
|
Abbreviations |
|
|
AIDS |
Acquired immunodeficiency syndrome |
|
CNS |
Central nervous system |
|
DNA |
Deoxyribonucleic acid |
|
G6PD |
Glucose-6-phosphate dehydrogenase |
|
GI |
Gastrointestinal |
|
IM |
Intramuscular |
|
IV |
Intravenous |
|
Therapeutic Overview |
|
Prevention Strategies |
|
Control disease vectors or reduce contact with them |
|
Improve hygiene and sanitation |
|
Vaccine development |
|
Drugs |
|
Transmission of Protozoal Infection |
|
Malaria—mosquitoes |
|
Leishmaniasis—sand flies |
|
African trypanosomiasis—tsetse flies |
|
Chagas’ disease—reduviid bugs |
|
Amebiasis—food, water |
|
Giardiasis—food, water |
|
Toxoplasmosis—cats, undercooked meats |
Helminths
Helminths have sophisticated organ systems and many have complex life cycles. Clinical manifestations of helminthic diseases are usually proportionate to the worm burden. Infections with light worm burdens are often asymptomatic, whereas heavy worm burdens can result in life-threatening disease. Exceptions occur when one or more helminths gain access to a critical organ such as the brain or an eye, or when an adult worm migrates into and obstructs the common bile duct, such as with A. lumbricoides. Helminths have finite life spans. Infestations resolve over time, unless there is autoinfection, as in the case of Strongyloides stercoralis or Hymenolepis nana, or the parasite has an extremely long life span, as in the case of Clonorchis sinensis. Eosinophilia is common when helminths migrate through tissue but may be absent after intestinal helminths have reached maturity in the bowel lumen.
Morphologically, helminths are composed of nematodes (roundworms) and platyhelminths (flatworms). Some roundworm species reside as adults in the human gastrointestinal (GI) tract, whereas others invade the body and migrate to specific organs. The platyhelminths include cestodes (tapeworms) and trematodes (flukes). Considering helminths in this manner is helpful clinically, because species in these groups frequently have similar life cycles, metabolic pathways, and susceptibilities to anthelmintic (or antihelminthic) medications.
Nematodes
Intestinal nematodes include the roundworm A. lumbricoides; the hookworms Ancylostoma duodenale and Necator americanus; Trichuris trichiura; S. stercoralis; and other species spread through feces. They are prevalent among persons living in conditions of poor hygiene. Such individuals, particularly children, are frequently infected with more than one species and often have high parasite burdens.Enterobius vermicularis (pinworm), which is common in industrialized countries, is spread among children after the mature female worm migrates from the rectum and deposits ova in the perianal area.
A number of nematode species reside outside the GI tract. The filariae Wuchereria bancrofti, Brugia malayi, and Loa loa are transmitted by mosquitoes, and Onchocerca volvulus is transmitted by black flies. Acute infections with W. bancrofti or B. malayi may be associated with lymphangitis, epididymitis, and fever. Chronic infections result in elephantiasis among residents of endemic areas. Adult O. volvulus produce microfilariae that cause inflammation in the skin and eyes. Other filariae that cause human disease include L. loa and M. perstans. Animal ascarids such as Toxocara species can produce visceral larva migrans, and animal hookworm species cause cutaneous larva migrans.
Platyhelminths
The cestodes, or tapeworms, live as adults in the GI tract of their definitive hosts and in cystic forms in organs of their intermediate hosts. Humans infected with Taenia saginata, the beef tape-worm, Taenia solium, the pork tapeworm, and Diphyllobothrium latum, the fish tapeworm, have ingested inadequately cooked infected meat or fish. With the exception of D. latum, which can compete with its host for vitamin B12 and on rare occasions results in symptomatic vitamin B12 deficiency, patients with adult tapeworms are asymptomatic or experience mild symptoms. Ova and proglottids are excreted in human feces.
Trematodes, or flukes, have complex life cycles involving snails. In the case of Schistosoma species, cercariae are released from snails into fresh water and enter humans through direct penetration of the skin after contact with infested fresh water. Schistosoma mansoni, Schistosoma japonicum, and Schistosoma mekongi undergo further development and reside as adults in venules of the GI tract, producing disease in the intestine and liver, whereas Schistosoma haematobium resides in venules of the urinary tract, resulting in damage to the ureters and bladder. Other trematode species encyst in secondary intermediate hosts such as fish or freshwater crustaceans, or on water plants. After they are ingested, trematodes excyst and develop in specific organs. Adult Paragonimus westermani reside in the lungs; C. sinensis, Opisthorchis viverrini, and Fasciola hepatica exist in the liver; and Fasciolopsis buski, Heterophyes heterophyes, Metagonimus yokogawai, and Nanophyetus salmincola are found in the intestine.
Protozoa
Protozoa are composed of a single cell and can multiply in their human hosts. Theoretically, infection with only one cell can result in overwhelming disease. Protozoal species differ widely in their sensitivity to antiparasitic drugs, as discussed in the following text.
The vectors by which parasites spread are varied. Enteric pathogens are spread in fecally contaminated food and water, T. vaginalis is spread by intimate personal contact, whereas Plasmodium species, which cause malaria, are transmitted by anopheline mosquitoes, whose life cycle is depicted in Figure 52-1. Sporozoites are inoculated into the host when an infected female attempts to take a blood meal. The sporozoites travel to the liver through the circulation, invade hepatocytes, and develop within liver cells in 1 to 3 weeks. The erythrocytic stage, which is the only symptomatic stage, begins when merozoites are released from the liver and invade red blood cells. Plasmodium vivax and Plasmodium ovale, in contrast, can persist for months in the liver as hypnozoites before completing development and initiating symptomatic malaria.

FIGURE 52–1 The life cycle of Plasmodium species. Drugs used to treat infections are effective against erythrocytic or exoerythrocytic stages of the parasite.
The Kinetoplastida also are transmitted by arthropod vectors; T. cruzi by reduviid bugs that live in adobe dwellings in Latin America; T. brucei gambiense and T. brucei rhodesiense by tsetse flies in Africa; and Leishmania species by sand flies. They contain a unique mitochondrial structure, the kinetoplast.
Other diverse protozoa also produce human disease. T. gondii is spread in the feces of infected cats and in inadequately cooked, contaminated meat. Infection is often asymptomatic but can cause a mononucleosis-like syndrome, in utero infection resulting in birth defects or chorioretinitis, or encephalitis, particularly in persons with AIDS or other immune defects. Based on conserved structural proteins, Pneumocystis jiroveci is more closely related to fungi than protozoa, but its treatment is discussed here. The infection is ubiquitous and apparently spread by inhalation. P. jiroveci has emerged as an important cause of pneumonitis in persons with AIDS and occurs occasionally in others with abnormal T cell-mediated immunity.
Mechanisms of Action
Antihelminths
Albendazole sulfoxide, the primary metabolite of albendazole, and mebendazole bind to β-tubulin in susceptible nematodes and inhibit microtubule assembly, leading to disruption of microtubules and selective and irreversible inhibition of glucose uptake (Fig. 52-2). This results in depletion of glycogen stores, reduced formation of adenosine triphosphate, disruption of metabolic pathways, and ultimately parasitic death. Serum glucose concentrations are not affected in the human host.
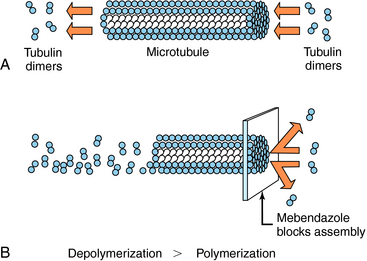
FIGURE 52–2 A, Under normal conditions, tubulin dimers are continually being polymerized and depolymerized from the ends of the microtubule. B, Albendazole and mebendazole can bind to β-tubulin and prevent polymerization, resulting in breakdown of microtubules.
Pyrantel pamoate, which is also active against several intestinal nematodes, acts as an agonist at nicotinic cholinergic receptors. Muscles of susceptible nematodes undergo depolarization and an increase in spike discharge frequency, leading to a short period of Ca++-dependent stimulation, resulting in irreversible paralysis. Pyrantel pamoate is also an acetylcholinesterase inhibitor. Affected helminths are unable to maintain their attachment in the intestinal lumen and are expelled from the body in the feces. Piperazine, an antihelminthic drug used to treat A. lumbricoides, paralyzes worms by hyperpolarization, and is therefore a mutual antagonist of pyrantel pamoate; the two drugs should not be administered concurrently.
Diethylcarbamazine is a piperazine derivative. The basis for its activity is uncertain, although microfilaria are paralyzed, perhaps by hyperpolarization of their musculature. Diethylcarbamazine also alters the microfilarial surface and may facilitate killing by the host’s immune responses. It also affects arachidonic acid metabolism and disrupts microtubule formation in the parasite.
Ivermectin is a macrocyclic lactone produced by Streptomyces avermitilis. It activates the opening of voltage-gated chloride channels that are found only in helminths and arthropods. The result is an influx of chloride ions and paralysis of the pharyngeal pumping motion in helminths.
Praziquantel is a heterocyclic pyrazine-isoquinoline derivative. It is rapidly taken up by tapeworms and flukes, but its precise mechanism of action is not known. Studies of the tapeworm Hymenolepis diminuta indicate that praziquantel releases Ca++ from endogenous stores, resulting in contraction and subsequent expulsion of the worm from the GI tract. In the schistosomes, praziquantel damages the tegument, causing intense vacuolation, exposure of sequestered schistosomal antigens, and increased permeability to Ca++, causing tetanic contraction and paralysis. Adult schistosomes are then swept back through the portal circulation to the liver, where they are destroyed by phagocytes. Figure 52-3 depicts the marked alterations in the schistosomal surface after drug exposure.
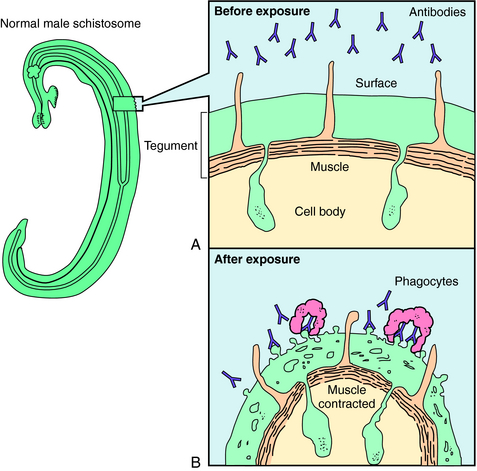
FIGURE 52–3 Before exposure to praziquantel, the schistosome is capable of avoiding antibodies directed toward surface and internally located antigens. A, Cross-section of the surface of a normal schistosome. After exposure to praziquantel, the muscles of the schistosome contract because of drug-induced influx of Ca++. B, Changes in the schistosome tegument include small holes and balloon-like structures and exposure of hidden parasite antigens, resulting in the binding of antibodies and phagocytes.
Niclosamide appears to uncouple oxidative phosphorylation in adult cestodes. The result is death of the worm, partial disintegration of the scolex and proximal portion, and expulsion of the remainder in the feces.
A summary of the observed effects and possible mechanisms of action of the major antihelminthic drugs is in Table 52-1.
TABLE 52–1 Observed Effects and Possible Mechanisms of Action of the Major Anthelmintic Drugs
|
Drug |
Observed Effects on Helminths |
Possible Mechanism of Action |
|
Albendazole |
Inhibition of glucose transport; depletion of glycogen stores, inhibition of fumarate reductase |
Binding to β-tubulin, prevents microtubule polymerization |
|
Mebendazole |
Inhibition of glucose transport; depletion of glycogen stores |
Binding to β-tubulin |
|
Pyrantel pamoate |
Muscles depolarize, increased spike wave activity, spastic paralysis |
Depolarizing neuromuscular blockade |
|
Diethylcarbamazine |
Hyperpolarization and paralysis of worm’s musculature; exposure of antigens, leading to antibody binding and attack by phagocytes |
Hyperpolarization and neuromuscular blockade |
|
Ivermectin |
Alters chloride currents, resulting in death of microfilariae |
Altered chloride channel function |
|
Praziquantel |
Depolarization of muscles, increased intracellular Ca++, displacement of schistosomes to the human liver, exposure of surface antigens, binding by antibody and phagocytes, tegument disruption |
Uncertain |
|
Niclosamide |
Uncouples phosphorylation; may inhibit anaerobic metabolism |
Uncertain |
Antiprotozoals
Metronidazole has a broad spectrum of activity against anaerobic bacteria and protozoa. It is activated when reduced by ferredoxins or their equivalents in protozoa or bacteria. The resultant products react with deoxyribonucleic acid (DNA) and other intracellular parasite constituents, causing damage and death. Tinidazole has a similar mechanism of action. Paromomycin, an aminoglycoside antibiotic (seeChapter 47), inhibits protein synthesis. Iodoquinol acts against E. histolytica cysts and, to a lesser extent, trophozoites by an unknown mechanism. Diloxanide furoate is directly amebicidal, and little is known about its mechanism of action also. Nitazoxanide has a broad spectrum of activity against protozoa and helminths and is approved for giardiasis and cryptosporidiosis in children. The mechanism involves inhibition of electron transport reactions essential to metabolism of anaerobic organisms. Furazolidone interferes with several bacterial enzyme systems, but its mechanism of action against G. lamblia is uncertain.
Chloroquine is concentrated in the hemoglobin-containing digestive vesicles of intraerythrocytic Plasmodium species. It inhibits the parasite’s heme polymerase that incorporates heme into an insoluble, nontoxic crystalline material. Chloroquine-resistant strains of P. falciparum transport chloroquine out of the intraparasitic compartment more rapidly than susceptible strains. Primaquine has activity against the exoerythrocytic stage of P. vivax and P. ovale and may interfere with electron transport or generate reactive O2 species. Quinine has been used to treat malaria for centuries. It is concentrated in the acidic food vacuoles of intracellular plasmodium and is thought to inhibit the activity of heme polymerase. Quinidine, the stereoisomer of quinine, presumably acts in the same manner. Mefloquine is an analog of quinine that produces swelling in the food vacuoles of intraerythrocytic plasmodium. Mefloquine may also form toxic complexes with heme. Atovaquone-proguanil (Malarone) is formulated as a fixed dose for prophylaxis and treatment of chloroquine-resistant P. falciparum malaria. Proguanil acts synergistically with atovaquone to inhibit mitochondrial electron transport, resulting in collapse of the mitochondrial membrane potential. It also inhibits dihydrofolate reductase-thymidylate synthase in Plasmodium ssp.
Atovaquone has activity against Plasmodium spp., Babesia spp., P. jiroveci, and T. gondii. It selectively inhibits electron transport, resulting in collapse of the mitochondrial membrane potential. It also inhibits pyrimidine biosynthesis, which is obligatorily coupled to electron transport in Plasmodium spp. Pyrimethamine binds to and irreversibly inhibits dihydrofolate reductase. It is approximately 1000-fold more active against plasmodium dihydrofolate reductase-thymidylate synthetase than against human dihydrofolate reductase. Pyrimethamine is often used with one of the sulfonamides to inhibit sequential steps in folate metabolism. Trimethoprim inhibits the dihydrofolate reductase of many bacteria and some protozoa and is frequently administered with sulfamethoxazole (see Chapter 48).Proguanil is metabolized to an active cyclic triazine metabolite that selectively inhibits plasmodium dihydrofolate reductase-thymidylate synthetase. Nifurtimox undergoes partial reduction followed by auto-oxidation, forming superoxide anion, hydrogen peroxide, and hydroxyl radicals that damage cell membranes and DNA. Eflornithine is an irreversible inhibitor of ornithine decarboxylase, the enzyme that catalyzes the rate-limiting step in polyamine synthesis. Although polyamines are essential for growth and differentiation of all cells, eflornithine has clinical activity only against T. brucei gambiense.
Pentamidine isethionate also has an unknown mechanism of action but may interfere with polyamine biosynthesis and inhibit topoisomerase II. Melarsoprol is an arsenical and reacts with sulfhydryl groups on proteins and inhibits many enzymes, including trypanothione. T. brucei rhodesiense and T. brucei gambiense have an unusual purine transporter that concentrates melarsoprol in the organisms.Sodium stibogluconate and meglumine antimonate are dosed on the basis of their pentavalent antimony content. They appear to affect bioenergetics in leishmania, inhibiting glycolysis and fatty acid β-oxidation.
A number of the antibiotics discussed in previous chapters have activity against some protozoa. Tetracycline, doxycycline, and clindamycin inhibit protein synthesis, and sulfonamides inhibit dihydropteroate synthetase and para-aminobenzoic acid binding to it. Amphotericin B is thought to act on leishmania as it does on susceptible fungi, by disrupting membranes (see Chapter 50). In addition, liposomal and lipid-associated amphotericin is selectively targeted to macrophages, the cells in which this parasite resides.
A summary of the possible mechanisms of action of the major antiprotozoal drugs is found in Table 52-2.
TABLE 52–2 Possible Mechanisms of Action of Major Antiprotozoal Drugs
|
Drug |
Possible Mechanism of Action |
|
Metronidazole |
Activated when reduced by ferredoxins, reacts with DNA and other parasite constituents |
|
Paromomycin |
Inhibits protein synthesis |
|
Nitazoxanide |
Inhibition of electron transfer reactions essential to the metabolism of anaerobic organisms |
|
Chloroquine |
Concentrated in hemoglobin-containing digestive vesicles, inhibits heme polymerase |
|
Quinine |
Concentrated in food vacuoles, probably inhibits heme polymerase |
|
Mefloquine |
Concentrated in food vacuoles, may form toxic complexes with heme |
|
Pyrimethamine |
Inhibits dihydrofolate reductase |
|
Sulfonamides |
Inhibit binding of p-aminobenzoic acid to dihydropteroate synthetase |
|
Nifurtimox |
Forms reactive O2 species that damage cell membranes and DNA |
|
Eflornithine |
Irreversibly inhibits ornithine decarboxylase, inhibits polyamine synthesis |
|
Melarsoprol |
Reacts with sulfhydryl groups, inhibits proteins |
Pharmacokinetics
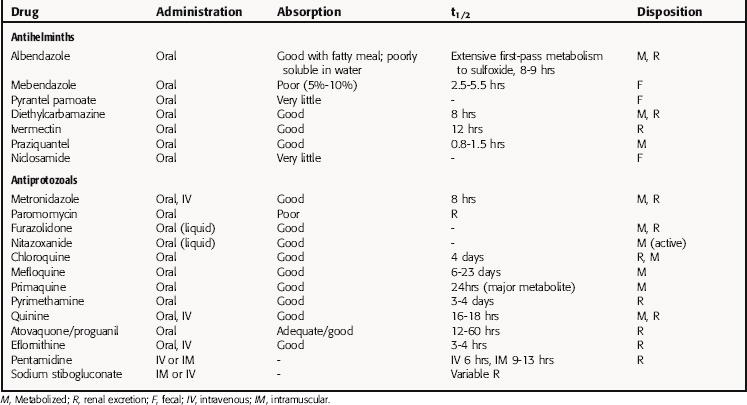
Pharmacokinetic parameters for selected antiparasitic drugs are listed in Table 52-3.
TABLE 52–3 Pharmacokinetic Parameters of Selected Antiparasitic Drugs
Antihelminths
All the antihelminths are absorbed orally, albeit to different extents. Albendazole is readily absorbed when taken with a fatty meal and undergoes extensive first-pass metabolism; only albendazole sulfoxide, which is responsible for systemic anthelmintic activity, is detectable in the serum. Sulfoxidation also occurs in the intestinal tract, and there is evidence of hepatobiliary recirculation. Albendazole sulfoxide reaches peak serum concentrations in 2 to 3 hours. Central nervous system (CNS) concentrations are 40% of those in serum; concurrent administration of dexamethasone increases the serum concentration by approximately half, which is advantageous for treatment of neurocysticercosis. The concentration of albendazole sulfoxide in echinococcal cysts is approximately 25% of that in serum. Elimination of albendazole and its metabolites is accomplished primarily by renal excretion.
Mebendazole is only slightly soluble in water and is poorly absorbed from the GI tract. These properties contribute to its low incidence of side effects but limit its effectiveness against tissue-dwelling helminths. Peak serum concentrations occur in 2 to 5 hours. Up to 10% of an orally administered dose is absorbed, metabolized, and excreted in the urine within 48 hours; the remainder is excreted unchanged in the feces.
Pyrantel pamoate and niclosamide are poorly absorbed and therefore act effectively against susceptible helminths in the lumen of the GI tract.
Ivermectin is rapidly absorbed after oral ingestion and reaches a serum peak after 4 to 5 hours. It is highly protein bound and has a t1/2 of 12 hours. It is eliminated by biliary excretion with enterohepatic circulation and tends to accumulate in adipose and hepatic tissues.
Praziquantel is well absorbed orally and reaches peak serum concentrations in 1 to 3 hours. There is extensive first-pass metabolism, generating inactive metabolites that are excreted primarily in the urine. Thus serum concentrations of praziquantel are increased in patients with moderate to severe liver impairment. Concentrations of praziquantel in cerebrospinal fluid are 15% to 20% of that of the serum.
Antiprotozoals
Metronidazole is rapidly and completely absorbed after oral administration and reaches peak plasma concentrations in 1 hour. More than half of the administered dose is metabolized in the liver, and the parent drug and metabolites are excreted in the urine.
Nitazoxanide is well absorbed orally and rapidly hydrolyzed to its active metabolite, tizoxanide, which undergoes conjugation to glucuronide. Maximal concentrations of these two metabolites are found within 1 to 4 hours. Tizoxanide is highly protein bound and is excreted in urine, bile, and feces, whereas the glucuronide is excreted in urine and bile. Furazolidone is well absorbed after oral administration and extensively metabolized, and more than 60% is excreted in the urine.
Chloroquine phosphate is well absorbed when taken orally, with peak serum concentrations reached in 3.5 hours. It is eliminated slowly after treatment is terminated. Approximately 50% of the drug is excreted unchanged in the urine, and the rest is metabolized in the liver.
Quinidine gluconate is administered by intravenous (IV) injection to patients with acute malaria who are unable to take oral medications.
Mefloquine is slowly and incompletely absorbed after oral administration, with peak serum concentrations after 7 to 24 hours. The drug is highly protein bound with a long half-life.
Proguanil is slowly but well absorbed after oral administration, with peak serum levels reached in 5 hours and an elimination half-life of 12 to 21 hours. The concentration in erythrocytes is approximately six times that in plasma.
Atovaquone is highly lipophilic, and administration with food enhances absorption by twofold. Plasma concentrations do not correlate with dose, and it is highly protein bound, with a half-life exceeding 60 hours. This is due to enterohepatic recycling with eventual fecal elimination. There is little excretion in urine.
Atovaquone/proguanil combinations have pharmacokinetics as described for the individual drugs.
Nifurtimox is well absorbed when taken orally, with a peak serum concentration observed in approximately 3.5 hours. It is rapidly metabolized.
Eflornithine can be administered orally or IV. Peak plasma concentrations are reached approximately 4 hours after oral administration. It is widely distributed in the body, including the CNS, with the bulk of drug excreted in the urine.
Pentamidine isethionate is administered via intramuscular (IM) or IV injection and has a plasma half-life of approximately 6 hours and a terminal elimination phase of approximately 12 days. It accumulates in tissue and is only slowly eliminated by the kidney.
Melarsoprol is administered intravenously, and a small but therapeutically significant amount enters the CNS. It is rapidly excreted in the feces.
Sodium stibogluconate and meglumine antimonate are administered IM or IV daily for a period of 3 to 4 weeks. These compounds have biphasic kinetics, with a short first-phase half-life of 2 hours and a second-phase half-life of 1 to 3 days. Most is excreted in the urine.
Doxycycline, tetracycline, ciprofloxacin, and sulfonamides are well absorbed when taken orally. Their pharmacokinetics are discussed in detail in Chapters 47 and 48. Amphotericin B is discussed inChapter 50.
Relationship of Mechanisms of Action to Clinical Response
Antihelminths
Albendazole and mebendazole are active against the common intestinal nematodes: A. lumbricoides, the hookworms, T. trichiura, and E. vermicularis. The advantage of albendazole is that it can be administered as a single dose for these helminths, whereas mebendazole is given twice daily for 3 days. In persons with heavy T. trichiura infection, albendazole is administered daily for 3 days. Single-dose albendazole has been used successfully in mass treatment programs in developing areas, resulting in enhanced growth and development of children infected with intestinal helminths, but treatment must be repeated at intervals of approximately 4 months because of reinfection. Albendazole can be used to treat cutaneous larva migrans.
Mebendazole is recommended for patients with Trichinella spiralis, Trichostrongylus species, and Capillaria philippinensis. Albendazole may be effective for these infections, but clinical experience is limited. Steroids are administered concurrently to patients with trichinosis who experience severe symptoms. Neither single-dose albendazole nor mebendazole is reliably effective against S. stercoralis, but albendazole twice a day for 2 or more days can be used. Thiabendazole, a related benzimidazole, used to be the treatment of choice. Recent studies indicate that ivermectin is equally effective and better tolerated, and it is now the treatment of choice. Thiabendazole is no longer available in the United States.
Albendazole and praziquantel are recommended for the treatment of neurocysticercosis caused by the larvae of T. solium. Albendazole alone, with percutaneous aspiration and installation of a scolicidal agent followed by reaspiration or with surgical resection, is used for patients with echinococcosis.
Pyrantel pamoate is effective for treatment of E. vermicularis and Trichostrongylus species. It also has activity against hookworms and A. lumbricoides.
Diethylcarbamazine has long been recommended for treatment of W. bancrofti, B. malayi, L. loa, and tropical pulmonary eosinophilia. Acute allergic reactions after release of microfilarial antigens, which can be especially severe and include life-threatening encephalopathy in heavy L. loa infection, can be reduced by use of antihistamines or corticosteroids. Reactions to released microfilorid antigens are often severe in patients with onchocerciasis treated with this drug; therefore ivermectin is the drug of choice.
Ivermectin has an expanding spectrum of clinical applications in humans. Although widely used for veterinary parasitic diseases, currently its use in humans is in treatment of onchocerciasis and strongyloidiasis. Ivermectin is recommended for Onchocerca volvulus, which causes river blindness, because in killing the microfilariae in the skin and eye it elicits a less severe inflammatory response than diethylcarbamazine. Because ivermectin appears to have no effect on the viability or fecundity of adult worms, the patient may require additional treatment at 6- to 12-month intervals. Ivermectin can also be used to treat cutaneous larva migrans.
Praziquantel is active against all Schistosoma species and all but one of the human flukes, F. hepatica, for which treatment with triclabendazole or bithionol is effective. Both praziquantel and niclosamide are effective against adult T. saginata, T. solium, D. latum, and H. nana in the human GI tract. Praziquantel is preferable for the treatment of T. solium because it is active against larvae and adults and may prevent internal autoinfection, a theoretical possibility after niclosamide treatment. In the case of H. nana, praziquantel is effective as a single dose, because it kills cysticerci in the wall of the intestine and kills adult worms, whereas niclosamide kills only adult worms and must be administered for 7 days.
In most cases the chemotherapy of helminthic infections is effective and reasonably well tolerated. Drug resistance among the helminths has been reported but is infrequent. Some anthelmintics are available through pharmacies in the United States, whereas others must be obtained from the manufacturer or the Drug Service at the Centers for Disease Control and Prevention in Atlanta, Georgia. A number of other anthelmintic compounds, which are more toxic or less effective, can be obtained abroad.
Antiprotozoals
Intestinal and Vaginal Protozoa
G. lamblia, E. histolytica (major causes of diarrhea), and T. vaginalis live in anaerobic environments and are susceptible to metronidazole and tinidazole. Because these drugs are effective against E. histolytica trophozoites but do not eradicate cysts, a second “luminal” agent, such as paromomycin, iodoquinol, or diloxanide furoate, is administered concomitantly in the treatment of persons with symptomatic amebic infections. A luminal agent is used alone for the treatment of asymptomatic E. histolytica infection. Furazolidone or nitazoxanide is available in liquid formulations and is often used for the treatment of giardiasis in children.
Two other enteric protozoal pathogens, Cyclospora species and Isospora belli, are susceptible to trimethoprim-sulfamethoxazole. Cryptosporidium infections are usually self-limiting in immunocompetent persons but may be persistent and severe in those with AIDS. Treatment with nitazoxanide has been of benefit in immunocompetent patients with cryptosporidiosis and is approved for use in children.
Antimalarials
Efforts to prevent malaria focus on minimizing mosquito contact and the use of chemoprophylaxis. Persons traveling to areas where Plasmodium species remain sensitive to chloroquine should takechloroquine weekly, and people with intense or prolonged exposure to P. vivax or P. ovale should receive a course of primaquine after leaving the endemic area. For travelers to chloroquine-resistant areas, there are three choices of comparable efficacy: doxycycline, atovaquone/proguanil, and mefloquine. Doxycycline is least expensive but can cause untoward effects. It is administered daily starting 2 days before travel and continued for 4 weeks after exposure to kill parasites released into the blood after completing their incubation in the liver. The combination of atovaquone/proguanil is the most expensive but also the best tolerated. It is also administered 2 days before travel and for 1 week after leaving the endemic area. Mefloquine has the advantage of being used weekly but has neuropsychiatric effects in some individuals. It is started 1 to 2 weeks before departure and is continued for 4 weeks after leaving the endemic area. Recent data suggest that primaquine taken daily can be used for prophylaxis, but recipients must be screened to ensure that they are not glucose-6-phoposphate dehydrogenase (G6PD)-deficient. Neither doxycycline, atovaquone,proguanil, or primaquine can be used during pregnancy.
Chloroquine is recommended for treatment of acute infections with P. ovale, Plasmodium malariae, and chloroquine-sensitive strains of P. vivax and P. falciparum. Primaquine is administered to patients with P. vivax and P. ovale to prevent relapses. Oral Proguanil or quinine plus doxycycline or tetracycline is used for treatment of chloroquine-resistant P. falciparum malaria. Clindamycin is used in place of doxycycline with quinine in children or pregnant women. Mefloquine used at full treatment doses is effective but has frequent side effects. Halofantrine is used in Europe and Africa to treat persons with chloroquine-resistant P. falciparum, but it has potentially serious side effects, including sudden death.
The recommendations for prophylaxis and treatment of malaria are reviewed regularly and are available from the Centers for Disease Control and Prevention (www.cdc.gov/travel) and in The Medical Letter on Drugs and Therapeutics (www.medicalletter.org).
Antikinetoplastides
The drug currently recommended for the treatment of Chagas’ disease, nifurtimox, is administrated over long periods of time and has substantial toxicity. Benznidazole is used in Latin America. These drugs are not effective in patients with chronic chagasic cardiomyopathy, megaesophagus, or megacolon.
Pentamidine isethionate is used for treatment of patients with the hemolymphatic stage of T. brucei gambiense, and suramin is used for T. brucei rhodesiense. Melarsoprol is used in patients with CNS disease but is highly toxic. Eflornithine, also known as the resurrection drug, is effective for the treatment of T. brucei gambiense in both the hemolymphatic and later CNS stages of infection, but supplies are very limited.
Liposomal amphotericin B is the only drug approved for treatment of visceral leishmaniasis in the United States. The pentavalent antimonials sodium stibogluconate and meglumine antimoniate have historically been used for treatment of visceral and cutaneous leishmaniasis around the world, but resistance is increasing. Miltefosine, an orally administered drug, is now considered the treatment of choice for visceral leishmaniasis in India. Amphotericin B deoxycholate and pentamidine isethionate are more toxic alternatives for leishmaniasis.
Treatment of Other Protozoal Diseases
Treatment of symptomatic toxoplasmosis consists of pyrimethamine and a short-acting sulfonamide, such as sulfadiazine. Leucovorin (discussed in Chapter 54) is administered concurrently to prevent bone marrow suppression. The combination of pyrimethamine and clindamycin is also effective and frequently used in patients with AIDS. The macrolide spiramycin has been used to treat women infected withT. gondii during pregnancy.
The drug of choice for P. jerovici is trimethoprim-sulfamethoxazole. There is, however, a very high incidence of adverse reactions to sulfonamides in patients with AIDS, and it is often necessary to use alternatives, such as pentamidine isethionate, trimethoprim plus dapsone, atovaquone, or primaquine plus clindamycin. Prophylactic administration of trimethoprim-sulfamethoxazole, dapsone, atovaquone, or pentamidine aerosol is effective in reducing recurrences and preventing disease in AIDS patients with low CD4+ counts.
Pharmacovigilance: Side Effects, Clinical Problems, and Toxicity
Antihelminths
Albendazole is usually well tolerated when given as a single dose for treatment of intestinal helminthic infections. Diarrhea, abdominal discomfort, and drug-elicited migration of A. lumbricoides occur in a few cases. High-dose, prolonged therapy for echinococcal infections is occasionally complicated by alopecia, hepatocellular injury, or reversible bone marrow suppression.
Mebendazole is well tolerated when used to treat intestinal nematodes. When administered at high doses for prolonged periods, as in the treatment of echinococcal liver cysts, it can produce alopecia, dizziness, transient bone marrow suppression with neutropenia, and hepatocellular injury. Pyrantel pamoate has minimal toxicity at the recommended dose.
Diethylcarbamazine may cause headache, malaise, dizziness, nausea, and vomiting. Acute psychotic events have been reported. Of greater concern is the Mazzotti reaction, resulting from lysis of O. volvulus microfilariae and release of their antigens. The manifestations can include pruritus, fever, wheezing, tachycardia, and hypotension. For these reasons ivermectin is the drug of choice for persons with onchocerciasis. Ocular sequelae include chorioretinitis and uveitis. Encephalopathy has been noted in patients with heavy L. loa infections treated with ivermectin or diethylcarbamazine. In patients with W. bancrofti or B. malayi, localized swelling or nodules can develop along lymphatics, and transient lymphedema or a hydrocele may be observed after diethylcarbamazine treatment. Ivermectin is generally well tolerated but on occasion may also trigger an inflammatory or Mazzotti-type reaction, resulting from the release of onchocercal antigens. Toxicity may include fever, pruritus, tender lymph nodes, headache, and arthralgias. Hypotension has been reported on rare occasions.
Praziquantel is frequently also associated with side effects such as dizziness, headache, lassitude, nausea, vomiting, and abdominal pain, but side effects are usually mild and transient. Allergic reactions can occur and are usually attributed to release of worm antigens. Urticarial reactions have been associated with the treatment of P. westermani. Increased intracranial pressure has been observed among some patients treated for neurocysticercosis. Praziquantel and albendazole are contraindicated in persons with ocular cysticercosis or cysticerci in the spinal cord, because destruction of the cysticercus can result in irreparable inflammatory damage.
Niclosamide is well tolerated except for occasional side effects of dizziness, lightheadedness, abdominal pain, loss of appetite, diarrhea, and nausea.
Antiprotozoals
Metronidazole administration is commonly associated with GI complaints such as nausea, vomiting, diarrhea, and a metallic taste. Neurotoxicity including dizziness, vertigo, and numbness is rare but is a basis for discontinuation of treatment. Metronidazole has a disulfiram-like effect, and patients undergoing treatment should abstain from alcohol while taking this drug. Tinidazole has a similar spectrum of untoward effects but is generally better tolerated.
Side effects associated with the use of paromomycin consist of GI disturbances and diarrhea. Because it belongs to the aminoglycoside class of antibiotics, the small percentage of paromomycin that is absorbed can produce ototoxicity and renal toxicity, particularly in persons with preexisting renal disease.
Iodoquinol is contraindicated in persons sensitive to iodine. It occasionally causes rash, anal pruritus, acne, slight enlargement of the thyroid gland, nausea, and diarrhea.
Diloxanide furoate administration is occasionally associated with mild side effects of nausea, vomiting, diarrhea, flatulence, pruritus, and urticaria.
Nitazoxanide is very well tolerated. On rare occasions, the eyes may appear yellow, and the urine may be similarly discolored.
Chloroquine is relatively well tolerated when used for malaria treatment or prophylaxis. The side effects are dose-related and reversible and include headache, nausea, vomiting, blurred vision, dizziness, and fatigue. When high doses are used, as in the treatment of rheumatologic diseases, serious and permanent retinal damage may occur, and chloroquine is contraindicated in persons with retinal disease, psoriasis, or porphyria. Children are especially sensitive to chloroquine, and cardiopulmonary arrest has occurred after accidental overdoses and in adults attempting suicide.
Primaquine is also relatively well tolerated, although abdominal discomfort and nausea occur in some persons. The major toxicity is hemolysis in persons with G6PD deficiency. Primaquine is contraindicated during pregnancy because intrauterine hemolysis can occur in a G6PD-deficient fetus. Neutropenia, GI disturbances, and methemoglobinemia have been reported.
Quinine has the poorest therapeutic index among antimalarial drugs. Common side effects include tinnitus, decreased hearing, headache, dysphoria, nausea, vomiting, and mild visual disturbances. Quinine therapy has been associated with severe hypoglycemia in persons with heavy P. falciparum infections as a result of the parasite’s use of glucose and the quinine-mediated release of insulin from the pancreas, which responds to intravenous administration of glucose. Rare complications include allergic skin rashes, pruritus, agranulocytosis, hepatitis, and massive hemolysis in patients with P. falciparummalaria, which has been termed “blackwater fever.” Quinine causes respiratory paralysis in persons with myasthenia gravis, stimulates uterine contractions, and may produce abortion but has been used successfully to treat serious cases of malaria during pregnancy. Quinidine gluconate, the stereoisomer of quinine, is also a Class 1A antiarrhythmic drug (see Chapter 22). It decreases ventricular ectopy, affects cardiac conduction, and prolongs the QTc interval. Life-threatening dysrhythmias can occur but are rare. Hypotension may result if the drug is administered too rapidly.
Mefloquine is relatively well tolerated when used for prophylaxis, but CNS side effects have limited its use. Side effects are more frequent and severe in patients receiving higher doses. Neuropsychiatric reactions such as seizures, acute psychosis, anxiety neurosis, and other disturbances occur in a small percentage of individuals but can be severe, and mefloquine should not be used in persons with a history of epilepsy or psychiatric disturbances.
Pyrimethamine is generally well tolerated. Blood dyscrasias, rash, vomiting, and seizures are rare. Bone marrow suppression sometimes occurs when high doses are used, but it can be prevented by concurrent administration of folinic acid.
Proguanil can cause GI signs and symptoms with occasional nausea, diarrhea, urticaria, or oral ulceration when administered in low doses.
Atovaquone is generally well tolerated but has been associated with GI side effects including nausea, vomiting, and diarrhea. It has also been reported to cause skin rash and pruritus.
Atovaquone/proguanil combination is the best tolerated of all medications available for prophylaxis against chloroquine-resistant P. falciparum. Side effects include those of both proguanil and atovaquone. Asymptomatic transient elevations in liver enzymes have also been reported.
Nifurtimox is often toxic. Side effects include anorexia, vomiting, weight loss, memory loss, sleep disorders, tremor, paresthesias, weakness, and polyneuritis.
Pentamidine isethionate toxicity is common, including GI complaints, dizziness, flushing, hypotension, renal damage, and blood dyscrasias. A major adverse reaction is hypoglycemia caused by acute damage to β-cells of the pancreatic islets, resulting in insulin release and the long-term consequence of insulin-dependent diabetes mellitus.
Melarsoprol is extremely toxic, which limits its use to patients with CNS involvement by T. brucei rhodesiense. Myocardial toxicity, albuminuria, hypertension, abdominal pain, vomiting, and peripheral neuropathy are all side effects. Approximately 10% of recipients develop allergic encephalitis, which may be fatal.
Eflornithine is tolerated much better than other antitrypanosomal medications. Its side effects include flatulence, nausea, vomiting, diarrhea, anemia, leukopenia, and thrombocytopenia. On rare occasions, diplopia, dizziness, cutaneous hypersensitivity reactions, hearing loss, or seizures may occur.
Sodium stibogluconate and meglumine antimonate have frequent side effects, but they usually do not prevent completion of therapy. Chemical pancreatitis is common, and recipients also frequently experience myalgias, arthralgias, fatigue, and nausea. Nonspecific ST-T wave changes are observed on the electrocardiogram. Untoward effects are more common in persons with renal failure. Side effects of antibiotics and amphotericin B are discussed in Chapter 46 through 48 and 50.
An overview of the adverse effects associated with the different antiparasitic drugs is summarized in the Clinical Problems Box.
CLINICAL PROBLEMS
Antihelminthic Drugs
|
Albendazole and Mebendazole |
GI discomfort, alopecia, bone marrow suppression, hepatic injury |
|
Diethylcarbamazine |
Central and GI effects, Mazzotti reaction |
|
Ivermectin |
Inflammatory reactions |
|
Praziquantel |
GI side effects, allergic reactions caused by release of helminthic antigens |
Antiprotozoal Drugs
|
Metronidazole |
GI side effects, neurotoxicity, alcohol intolerance |
|
Paromomycin |
GI side effects, ototoxicity, renal toxicity |
|
Chloroquine |
Headache, nausea, vomiting, blurred vision, retinal damage |
|
Primaquine |
GI side effects, hemolysis in people with glucose-6-phosphate deficiency |
|
Quinine |
Tinnitus, decreased hearing, headache, dysphoria, GI side effects, visual disturbances |
|
Mefloquine |
Neuropsychiatric reactions |
|
Pyrimethamine |
Rare blood dyscrasias, rash, vomiting, seizures, shock |
New Horizons
Antihelminths
Ivermectin has emerged as the treatment of choice for onchocerciasis and S. stercoralis. Ivermectin also has activity against A. lumbricoides, T. trichiura, and E. vermicularis. However, it has only limited activity against the hookworm N. americanus. Its effectiveness against some ectoparasites also has been documented, and its clinical indications are likely to expand in the future.
Antimalarials
The emergence of multidrug-resistant P. falciparum has stimulated the search for new forms of treatment and prophylaxis for malaria. Among the exciting new compounds are the artemisinin derivatives, which include artesunate. These were identified in studies of quinghaosu, the Chinese herbal treatment for malaria derived from the wormwood plant Artemisia annua. Artemisinin and other artemisinin relatives are endoperoxide-containing compounds. In the presence of intraparasitic iron, these drugs may be converted into free radicals and other intermediates that alkylate specific malaria proteins. Artesunate and its derivatives have been used successfully to treat acute P. falciparum malaria in areas where mefloquine- and quinine-resistant strains are endemic. In many instances artemisinin derivatives are used concurrently with mefloquine or another antimalarial drug. Currently available artemisinin derivatives are not useful prophylactically because of their short half-lives and concern about neurotoxicity with long-term use. Other anti-malarials are being studied in combinations to determine their additive or synergistic effects and whether they may be useful against drug-resistant isolates.
Antikinetoplastides
Drugs used to treat Chagas’ disease are toxic and variably effective in eradicating T. cruzi. Better drugs are needed. With respect to African sleeping sickness, eflornithine is effective and reasonably well tolerated in treatment of T. brucei gambiense, but economic and logistical factors have limited its production, and supplies are very limited. Drugs recommended for treatment of the hemolymphatic and CNS stages of T. brucei rhodesiense have substantial, at times life-threatening toxicity.
Pentavalent antimonials remain the treatment of choice for leishmaniasis in many areas despite their toxicity and reports of clinical failures and resistance. Liposomal and lipid-associated amphotericin preparations are effective for visceral leishmaniasis. They are theoretically attractive because they target macrophages, the only cells infected by Leishmania species. Unfortunately, these preparations are expensive and must be given parenterally. The most exciting recent advance in this area has been the development of miltefosine, a phosphocholine analog that is administered orally. It is currently the drug of choice for treating visceral leishmaniasis in India, where resistance to sodium stibogluconate is common. Although it has GI and liver side effects, these are seldom severe enough to require discontinuation of therapy. In time, this drug may become more widely used.
TRADE NAMES
(In addition to generic and fixed-combination preparations, the following trade-named materials are some of the other compounds used for parasitic diseases around the world.)
|
Antihelminths |
Antimalarials |
|
Albendazole (Albenza) |
Atovaquone and proguanil (Malarone) |
|
Bithionol (Bitin) |
Chloroquine (Aralen) |
|
Diethylcarbamazine (Hetrazan) |
Doxycycline (Vibramycin) |
|
Ivermectin (Stromectol) |
Mefloquine (Lariam) |
|
Mebendazole (Vermox) |
Primaquine |
|
Niclosamide (Yomesan) |
Quinine |
|
Praziquantel (Biltricide) |
Quinidine |
|
Proguanil (Paludrine) |
|
|
Pyrantel pamoate (Antiminth) |
|
|
Pyrimethamine (Daraprim) |
|
|
Antiprotozoals |
Antikinetoplastides |
|
Clindamycin (Cleocin) |
Amphotericin B (lipid associated) |
|
Diloxanide furoate (Furamide) |
Melarsoprol (Arsobal) |
|
Furazolidone (Furoxone) |
Pentamidine isethionate (Pentam 300) |
|
Iodoquinol (Yodoxin) |
Trimethoprim-Sulfamethoxazole (Bactrim, Septra) |
|
Metronidazole (Flagyl) |
Stibocluconate sodium (Pentostam) |
|
Nitazoxanide (Alinia) |
|
|
Pentamidine isethionate (Pentam 300) |
|
|
Paromomycin (Humatin) |
|
|
Pyrimethamine (Daraprim) |
|
|
Spiramycin (Rovamycine) |
|
|
Tinidazole (Tindamax) |
|
|
Trimethoprim-Sulfamethoxazole (Bactrim, Septra) |
FURTHER READING
Griffith et al. 2007 Griffith KS, Lewis LS, Mali S, Parise ME. Treatment of malaria in the United States. JAMA. 2007;297:2264-2277.
Anonymous. Drugs for parasitic infections. Med Lett. 2007;49(Suppl):1-15.
Pearson RD. Antiparasitic drugs. In Mandell GL, Bennett JE, Dolin R, editors: Principles and practice of infectious diseases, ed 5, Philadelphia: Churchill Livingstone, 2005.
Centers for et al. 2007 Centers for Disease Control and Prevention. Traveler’s Health: 2007, US Department of Health and Human Resources, Public Health Service. wwwn.cdc.gov/travel.
SELF-ASSESSMENT QUESTIONS
1. A 5-year-old boy has developed localized erythema and severe itching on his feet. He appears malnourished and lethargic despite reportedly eating a well-balanced diet. His mother reports that he typically plays in the yard and sandbox barefooted. Fecal samples reveal evidence of Necator americanus. Which of the following is the best treatment for this patient?
A. Diethylcarbamazine
B. Ivermectin
C. Mebendazole
D. Metronidazole
E. Praziquantal
2. While traveling overseas, a 35-year-old man and his wife eat pork prepared by a street vendor, which appears slightly undercooked. Within the next few weeks, they experience vague abdominal discomfort and generalized weakness. They both notice strange things in their bowel movements and take a sample to the physician, who identifies them as proglottids. Which of the following is the best treatment for these patients?
A. Albendazole
B. Bithionol
C. Mebendazole
D. Metronidazole
E. Praziquantal
3. A 30-year-old woman reports an uncomfortable, yellow, vaginal discharge, and physical examination reveals vulvar erythema and edema. A vaginal saline wet mount demonstrates the herky-jerky motion of a trichomonad. A 7-day regimen of metronidazole is prescribed, yet 1 month later, the woman returns with similar symptoms. The organism is again observed on vaginal wet mount. Which of the following is the most likely explanation for the recurrence of her illness?
A. A cervical neoplasm
B. A new vaginal infection with the fungus Candida krusei
C. Evolution of a metronidazole-resistant strain of trichomonas
D. Failure to treat the infected partner with the metronidazole regimen
E. An unusual infection with another resistant trichomonas species
4. Why is Chloroquine ineffective in treating recurrent episodes of fever and headache caused by infection with Plasmodium ovale?
A. It does not cross the blood-brain barrier.
B. It does not eradicate the dormant hepatic forms of the organism.
C. It does not kill infected mosquitoes upon their next blood meal.
D. It is an ineffective blood schizonticide.
E. It is an ineffective gametocide.
5. Which of the following drugs has a major side effect of hemolysis in persons with G6PD deficiency?
A. Chloroquine
B. Doxycycline
C. Mefloquine
D. Primaquine
E. Pyrimethamine