Judith A. Smith and Judith K. Wolf
KEY CONCEPTS
![]() Ovarian cancer is denoted “the silent killer” because of the nonspecific signs and symptoms that contribute to the delay in diagnosis. The few patients who present with disease still confined to the ovary will have a 5-year survival rate greater than 90%, but most patients present with advanced disease and have a 5-year survival rate of 10% to 30%.
Ovarian cancer is denoted “the silent killer” because of the nonspecific signs and symptoms that contribute to the delay in diagnosis. The few patients who present with disease still confined to the ovary will have a 5-year survival rate greater than 90%, but most patients present with advanced disease and have a 5-year survival rate of 10% to 30%.
![]() Ovarian cancer is a sporadic disease with less than 10% of cases of ovarian cancer attributed to heredity. However, a history of two or more first-degree relatives with ovarian cancer increases a woman’s risk of developing ovarian cancer by greater than 50%.
Ovarian cancer is a sporadic disease with less than 10% of cases of ovarian cancer attributed to heredity. However, a history of two or more first-degree relatives with ovarian cancer increases a woman’s risk of developing ovarian cancer by greater than 50%.
![]() Considerable education efforts have been made to identify patients with the persistence, greater than 2 weeks, of nonspecific presenting symptoms of ovarian cancer including: abdominal pressure/pain, difficulty eating or feeling full quickly, urinary urgency/frequency, change in bowel habits, or unexplained vaginal bleeding.
Considerable education efforts have been made to identify patients with the persistence, greater than 2 weeks, of nonspecific presenting symptoms of ovarian cancer including: abdominal pressure/pain, difficulty eating or feeling full quickly, urinary urgency/frequency, change in bowel habits, or unexplained vaginal bleeding.
![]() CA-125 is a nonspecific antigen used as a tumor marker for diagnosis and monitoring epithelial ovarian carcinoma. If CA-125 is positive at the time of diagnosis, changes in CA-125 levels correlate with disease response and progression.
CA-125 is a nonspecific antigen used as a tumor marker for diagnosis and monitoring epithelial ovarian carcinoma. If CA-125 is positive at the time of diagnosis, changes in CA-125 levels correlate with disease response and progression.
![]() Although most patients will achieve a complete response to initial treatment, more than 50% of patients will have recurrence within the first 2 years. If recurrence is less than 6 months after completion of chemotherapy, tumor is defined to be platinum-resistant. The antitumor activity of second-line chemotherapy regimens is similar, and the choice of treatment for recurrent platinum-resistant ovarian cancer depends on residual toxicities, physician preference, and patient convenience. Participation in a clinical trial is also a reasonable option for these patients.
Although most patients will achieve a complete response to initial treatment, more than 50% of patients will have recurrence within the first 2 years. If recurrence is less than 6 months after completion of chemotherapy, tumor is defined to be platinum-resistant. The antitumor activity of second-line chemotherapy regimens is similar, and the choice of treatment for recurrent platinum-resistant ovarian cancer depends on residual toxicities, physician preference, and patient convenience. Participation in a clinical trial is also a reasonable option for these patients.
![]() Ovarian cancer is staged surgically with the International Federation of Gynecology and Obstetrics (FIGO) staging algorithm. Tumor debulking and total abdominal hysterectomy–bilateral oophorectomy surgery are the primary surgical interventions for ovarian cancer. After the completion of the staging and primary surgical treatment, the current standard of care is six cycles of a taxane/platinum-containing chemotherapy regimen.
Ovarian cancer is staged surgically with the International Federation of Gynecology and Obstetrics (FIGO) staging algorithm. Tumor debulking and total abdominal hysterectomy–bilateral oophorectomy surgery are the primary surgical interventions for ovarian cancer. After the completion of the staging and primary surgical treatment, the current standard of care is six cycles of a taxane/platinum-containing chemotherapy regimen.
![]() The interperitoneal (IP) route of chemotherapy administration has demonstrated a significant route of administration; however, it is dependent on appropriate patient selection.
The interperitoneal (IP) route of chemotherapy administration has demonstrated a significant route of administration; however, it is dependent on appropriate patient selection.
![]() A platinum-containing doublet chemotherapy regimen is the standard of care for the first recurrence of platinum-sensitive ovarian cancer.
A platinum-containing doublet chemotherapy regimen is the standard of care for the first recurrence of platinum-sensitive ovarian cancer.
![]() Despite recent advances, enrollment still primary treatment recommendation for patients with recurrent platinum-resistant ovarian cancer.
Despite recent advances, enrollment still primary treatment recommendation for patients with recurrent platinum-resistant ovarian cancer.
Ovarian cancer is a gynecologic cancer that usually arises from disruption or mutations in the epithelium of the ovary.1 It is associated with the highest mortality among the gynecologic cancers, primarily because most patients present with advanced disease. ![]() Ovarian cancer is denoted “the silent killer” because of the nonspecific signs and symptoms that often lead to a delay in diagnosis. Ovarian cancers often metastasize via the lymphatic and blood systems to the liver and/or lungs. Common complications of advanced and progressive ovarian cancer include ascites and small bowel obstruction. The few patients who present with disease still confined to the ovary will have a 5-year survival rate greater than 90%, but most patients present with advanced disease and have a 5-year survival rate of 10% to 30%. Primary treatment includes tumor-debulking surgery followed by six cycles of a taxane-platinum chemotherapy regimen. Although 70% of patients achieve an initial complete response to chemotherapy, more than 50% of these patients will have recurrence within the first 2 years from diagnosis.2
Ovarian cancer is denoted “the silent killer” because of the nonspecific signs and symptoms that often lead to a delay in diagnosis. Ovarian cancers often metastasize via the lymphatic and blood systems to the liver and/or lungs. Common complications of advanced and progressive ovarian cancer include ascites and small bowel obstruction. The few patients who present with disease still confined to the ovary will have a 5-year survival rate greater than 90%, but most patients present with advanced disease and have a 5-year survival rate of 10% to 30%. Primary treatment includes tumor-debulking surgery followed by six cycles of a taxane-platinum chemotherapy regimen. Although 70% of patients achieve an initial complete response to chemotherapy, more than 50% of these patients will have recurrence within the first 2 years from diagnosis.2
ETIOLOGY AND EPIDEMIOLOGY
It is estimated that 22,240 new cases of ovarian cancer were diagnosed, and 14,030 women died of the disease in 2013.3 Ovarian cancer is associated with the highest mortality rate among the gynecologic cancers and is the fifth leading cause of cancer-related deaths in women. Despite research efforts and recent advances, the mortality rate associated with ovarian cancer has not changed significantly over the past four decades. The high mortality rate is related to the insidious onset of nonspecific symptoms and the lack of adequate screening tools, which allows the disease to go undiagnosed until it has progressed beyond the pelvic cavity.
As with many other cancers, the risk of ovarian cancer increases with increasing age. A woman’s risk increases from 15.7 to 54 per 100,000 as her age advances from 40 to 79 years, and the median age at diagnosis is 59.3 Most cases of ovarian cancer are diagnosed during the peri- and postmenopausal phase of women’s reproductive life span.4
![]() Heredity accounts for less than 10% of all ovarian cancer cases. Family history is an important risk factor in the development of ovarian cancer. If one family member has a diagnosis of ovarian cancer, the associated lifetime risk is 9%, but this risk increases to greater than 50% if there are two or more first-degree relatives (e.g., her mother and sister) with a diagnosis of ovarian cancer or multiple cases of ovarian and breast cancer within the same family.1,2
Heredity accounts for less than 10% of all ovarian cancer cases. Family history is an important risk factor in the development of ovarian cancer. If one family member has a diagnosis of ovarian cancer, the associated lifetime risk is 9%, but this risk increases to greater than 50% if there are two or more first-degree relatives (e.g., her mother and sister) with a diagnosis of ovarian cancer or multiple cases of ovarian and breast cancer within the same family.1,2
BRCA1 and BRCA2 are the tumor suppressor genes thought to be involved in one or more pathways of DNA damage recognition and repair. The BRCA1 gene is located on chromosome 17q12–21, and the BRCA2 gene is located on chromosome 13q12–13. Both BRCA1 and BRCA2 mutations are associated with ovarian cancer. However, BRCA1 is more prevalent, being associated with 90% of inherited and 10% of sporadic cases of ovarian cancer.5 Patients with BRCA1-associated ovarian cancer are usually considerably younger than patients with BRCA2 mutations, with a mean age of 54 years.6 Patients usually present with advanced stage at diagnosis, and the BRCA1-linked ovarian cancers are more aggressive tumors that typically are serous histology, moderate to high grade. As BRCA1 and BRCA2 are thought to be involved in DNA damage or repair, their inactivation/mutations may be associated with an increased resistance of ovarian cancer cells to cytotoxic agents.
Hereditary breast and ovarian cancer syndrome is one of the two different forms of hereditary ovarian cancer and is associated with germline mutations in BRCA1 and BRCA2.5,7 The hereditary nonpolyposis colorectal cancer or Lynch syndrome is a familial syndrome with germline mutations causing defects in enzymes involved in DNA mismatch repair, which is associated with up to 12% of hereditary ovarian cancer cases.5 This syndrome is associated with mutations in DNA mismatch repair genes such as MSH2, MLH1, PMS1, and PMS2 and leads to microsatellite instability.
Hormone exposure, specifically estrogen, and reproductive history are also associated with the risk of developing ovarian cancer. Conditions that increase the total number of ovulations in women’s reproductive history, such as nulliparity, early menarche, or late menopause, are associated with an increasing risk for epithelial ovarian cancers.8,9 Conversely those conditions that limit ovulations are associated with a protective effect. Each time ovulation occurs, the ovarian epithelium is broken, followed by cellular repair. According to the incessant ovulation hypothesis, the risk of mutations and, ultimately, cancer increases each time the ovarian epithelium undergoes cell repair.
Finally, ovarian cancer is associated with certain dietary and environmental factors. A diet that is high in galactose, animal fat, and meat may increase the risk of ovarian cancer, whereas a vegetable-rich diet may decrease the risk of ovarian cancer.7,10 Although controversial, exogenous factors such as asbestos and talcum powder use in the perineal area are also associated with an increased risk of ovarian cancer.7,10
PATHOLOGY AND CLASSIFICATION
Ovarian carcinomas can be separated into three major entities: epithelial carcinomas, germ cell tumors, and stromal carcinomas. Most ovarian tumors (85% to 90%) are derived from the epithelial surface of the ovary.11 The classification of common epithelial tumors has been developed by the World Health Organization and the International Federation of Gynecology and Obstetrics (FIGO).12 The nomenclature considers cell type, location of the tumor, and the degree of the malignancy, which ranges from benign tumors to tumors of low malignancy to invasive carcinomas. Epithelial tumors classified as low malignancy (“borderline malignancy”) are characterized by epithelial papillae with atypical cell clusters, cellular stratification, nuclear atypia, and increased mitotic activity, and have a much better prognosis than those classified as invasive carcinomas. Malignant tumors are characterized by an infiltrative destructive growth pattern with malignant cells growing in a disorganized manner and dissection into stromal planes.
Invasive epithelial adenocarcinomas are characterized by histologic subtype and grade, which measures the degree of cellular differentiation. Although the histologic type of the tumor is not a significant prognostic factor, with the exception of clear cell, the histopathologic grade is an important prognostic factor. Undifferentiated tumors are associated with a poorer prognosis than those lesions that are considered to be well or moderately differentiated. A universal grading system for ovarian cancer was developed that combines mitotic score, nuclear atypia score, and architectural score based on the histologic pattern.13
The histologic subtypes of adenocarcinomas include papillary serous, mucinous, endometrioid, clear cell, mixed epithelial, transition-cell, and undifferentiated.2,4,13 Papillary serous adenocarcinoma is the most common type of epithelial ovarian cancer and accounts for about 46% of cases. The peak age of diagnosis ranges from 45 to 65 years with 63 years as the median age of diagnosis.14 Serous carcinomas typically display complex papillary and solid patterns and qualify as high-grade carcinomas. Endometrioid carcinomas are seen in women 40 to 50 years of age and comprise about 8% of ovarian carcinomas, of which about 6% are surface epithelial neoplasms.14 Endometrioid tumors are usually diagnosed as stage I disease and have a better prognosis than tumors with serous histology. Mucinous carcinomas occur in women between 40 and 70 years of age and account for about 36% of all ovarian cancers. The overall prognosis for mucinous carcinoma is better than for serous carcinoma because most patients present with stage I disease. Clear cell carcinoma comprises about 3% of ovarian carcinomas in women, with a mean age of 57 years. Although clear cell carcinoma is the least common ovarian neoplasm, it is most commonly associated with paraneoplastic-related hypercalcemia.14
Germ cell tumors of the ovary, including malignant teratoma and dysgerminomas, are rare, comprising about 2% to 3% of all ovarian cancers in Western countries with an increased incidence in black and Asian women.15,16These tumors are highly curable and affect primarily young women. In contrast to epithelial tumors, about 60% to 70% of germ cell tumors are stage I at diagnosis, which is related to earlier detection and response to symptoms in this younger patient population.16 Serum markers (human β-chorionic gonadotropin and α-fetoprotein) are helpful to confirm the diagnosis and monitor response to treatment.
Finally, ovarian sex cord-stromal tumors account for 7% of all ovarian cancers and tend to be diagnosed at stage I.12 Sex cord-stromal tumors are associated with hormonal effects, such as precocious puberty, amenorrhea, and postmenopausal bleeding. Because these tumors are rare, the optimal treatment of ovarian sex cord-stromal tumors is not clear. The current recommended standard of care is surgery followed by treatment with a platinum-based chemotherapy regimen.
Ovarian cancer is usually confined to the abdominal cavity, but spread can occur to the lung, liver, and, less commonly, the bone or brain. Disease is spread by direct extension, peritoneal seeding, lymphatic dissemination, or bloodborne metastasis. Lymphatic seeding is the most common pathway and frequently causes ascites.
SCREENING AND PREVENTION
Screening
Ovarian cancer is an uncommon disease with no known preinvasive component, which has made it difficult to screen patients to detect early disease. In addition, the risk factors for developing ovarian cancer are not well understood, which also makes it difficult to identify a high-risk group of individuals. At the present time, there are no effective screening tools for early detection of ovarian cancer. ![]() However, considerable education efforts have been made to help identify patients with the persistence (i.e., >2 weeks) of nonspecific presenting symptoms of ovarian cancer including: abdominal pressure/pain, difficulty eating or feeling full quickly, urinary urgency/frequency, change in bowel habits, or unexplained vaginal bleeding.
However, considerable education efforts have been made to help identify patients with the persistence (i.e., >2 weeks) of nonspecific presenting symptoms of ovarian cancer including: abdominal pressure/pain, difficulty eating or feeling full quickly, urinary urgency/frequency, change in bowel habits, or unexplained vaginal bleeding.
Pelvic examinations are noninvasive and well accepted and can detect large tumors with a sensitivity of 67% for detecting all tumors.15 However, because pelvic examinations cannot detect minimal or microscopic disease, they do not usually detect ovarian cancer until it is in an advanced stage. As a result of these limitations, routine pelvic examinations are not an effective screening tool and do not decrease overall mortality.15
Transvaginal ultrasound (TVUS) creates an image of the ovary by releasing sound waves. It can be used to evaluate the size and shape and to detect the presence of cystic or solid masses or abdominal fluid. TVUS can also evaluate blood flow within an ovarian mass. Normal ovarian size cutoff parameters range from 1.25 cm2 for women 55 to 59 years of age to 1.0 cm2 for women older than age 65 to 69 years.17,18 TVUS is sensitive in identifying ovarian lesions and abnormalities, but its use as a routine screening test is limited by a lack of specificity and an inability to detect peritoneal cancer or cancer in normal-size ovaries.19,20
Serum cancer antigen-125 (CA-125) is a nonspecific inflammatory antigen that can be elevated in numerous conditions associated with inflammation in the abdominal cavity. CA-125 has been extensively studied as a potential tumor marker for ovarian cancer based on the observation that CA-125 levels in a woman without ovarian cancer tend to stay the same or decrease over time, whereas levels associated with malignancy tend to gradually increase over time.19 However, CA-125 is a nonspecific test that can be elevated in a number of benign conditions, including other gynecologic conditions, such as endometriosis, and many nongynecologic conditions, such as diverticulitis and peptic ulcer disease. Because of these limitations, CA-125 levels are not recommended as a routine screening test for detection of ovarian cancer. Numerous other serologic markers such as carcinoembryonic antigen and lipid-associated sialic acid have been evaluated but cannot be recommended for routine screening for ovarian cancer.
The United States Preventive Services Task Force found fair evidence to support screening with CA-125 or TVUS and concluded that earlier detection would likely have a small effect, at best, on mortality from ovarian cancer.21Unfortunately, because of the low prevalence of ovarian cancer and the invasive nature of diagnostic testing after a positive screening test, the United States Preventive Services Task Force also found fair evidence that screening could likely lead to important harms. The United States Preventive Services Task Force concluded that the potential harms outweigh the potential benefits and recommended against any form of routine screening with CA-125 or TVUS for ovarian cancer.
In high-risk women, as defined by family history, most clinicians use a multimodality approach for ovarian cancer screening that includes an annual TVUS in combination with a CA-125 blood test every 6 months. Changes in CA-125 are monitored over time, and changes such as a persistent elevation or consistent increases in CA-125 levels in conjunction with TVUS abnormalities are evaluated further.
Prevention
It is difficult to make recommendations for prevention for the general population because ovarian cancer is a sporadic disease with no established risk factors. Noninvasive measures, such as chemoprevention, have demonstrated some benefit in decreasing the risk of developing ovarian cancer. Ovulation itself is considered a potential insult to the ovarian epithelium, increasing its susceptibility to damage and, ultimately, to cancer. Interventions or reproductive conditions associated with decreasing the number of ovulations, including multiparity, may have a protective effect for the prevention of ovarian cancer. However, the more invasive prevention interventions, such as prophylactic surgery and genetic screening, should be reserved for those women identified to be at high risk based on their heredity for developing ovarian cancer.
Chemoprevention
Although a number of agents have been investigated as chemoprevention of ovarian cancer, including oral contraceptives, aspirin, nonsteroidal antiinflammatory agents, and retinoids, none of these agents is currently accepted as standard treatment for the prevention of ovarian cancer. Oral contraceptives inhibit ovulation, which reduces the opportunity for potential for damage to the ovarian epithelium. When taken for longer than 10 years, oral contraceptives decrease the relative risk to less than 0.4.22,23 Because oral contraceptive use is associated with an increased risk of breast cancer, women with a family history of breast cancer are not candidates for this use of oral contraceptives as chemoprevention of ovarian cancer.22,23
Nonsteroidal antiinflammatory drugs, aspirin, and acetaminophen also have been suggested for use in the chemoprevention of different cancers, especially hereditary nonpolyposis colon cancer.24 Although the results of observational studies show that the use of nonsteroidal antiinflammatory drugs, aspirin, and acetaminophen reduces the risk of ovarian cancer, these findings have not been confirmed in prospective clinical studies. The proposed mechanism of these agents is the antiinflammatory effect on normal ovulation and inhibition of ovulation.24,25
Prophylactic Surgery
Prophylactic surgical interventions for the prevention of ovarian cancer are reserved for patients with a significant family history and/or with known genetic mutations such as BRCA1 and should be postponed until after childbearing is completed. The goal is to remove healthy, at-risk organs before any carcinogenic activity is initiated, ultimately reducing the risk of developing cancer. These surgeries include prophylactic oophorectomy or bilateral salpingo-oophorectomy and tubal ligation. These procedures will cause surgical menopause, which can be associated with severe hot flashes, vaginal dryness, sexual dysfunction, and increased risk for development of osteoporosis and heart disease in these women. Because of the potential impact on quality of life and increased health risks, prophylactic surgery is not recommended as a general prevention intervention for the general population.
Although prophylactic surgical interventions are associated with significant reduction in risk of developing ovarian cancer, patients who choose to have a prophylactic oophorectomy/bilateral salpingo-oophorectomy completed need to be informed that complete protection is not guaranteed.15,23,26 Although a 67% risk reduction has been shown, a potential 2% to 5% risk of primary peritoneal cancer remains.27,28 Primary peritoneal cancers have identical histology of ovarian tumors with diffuse involvement of peritoneal surfaces. Primary peritoneal cancers can often result from “seeding” during the prophylactic surgery. It is recommended for peritoneal washings to be completed during the prophylactic surgery to check for presence of peritoneal surfaces. If positive, then prophylactic surgery would change to staging and treatment surgery to determine extent of disease and remove any other possible lesions.
Tubal ligation is another procedure that can potentially reduce the risk for developing ovarian cancer. In a case-control study, Narod et al. reported that tubal ligation in BRCA-positive women was associated with a 63% reduction in risk of developing ovarian cancer.29 However, it is not recommended as a sole procedure in prophylaxis. The mechanism for its protective effect is not clear, but it has been proposed that tubal ligation may limit exposure of the ovary to environmental carcinogens.
Genetic Screening
Genetic screening should be considered for those women with a significant family history of ovarian cancer. Patients should be evaluated for the presence of genes such as BRCA1, BRCA2, or other genes such as those associated with hereditary nonpolyposis colorectal cancer or the hereditary breast ovarian cancer (hereditary breast and ovarian cancer syndrome) syndrome.29–32 Prior to genetic screening, appropriate patient/family counseling and genetic counseling should be available to help women prepare and deal with the health and psychosocial implications of the genetic screening results.
CLINICAL PRESENTATION
General
• Ovarian cancer is sometimes referred to as “the silent killer” because of the vague nonspecific signs and symptoms that contribute to the delay in diagnosis.
Symptoms
• The patient may complain of abdominal discomfort, nausea, dyspepsia, flatulence, bloating, fullness, early satiety, urinary frequency, change in bowel function (diarrhea or constipation), weight change, and digestive disturbances.
Signs
• Abdominal or pelvic mass may be palpable.
• Lymphadenopathy may be present.
• Vaginal bleeding may be irregular.
• Patient may have signs of ascites (abdominal distension, shifting, and dullness to percussion—may present like “pregnant abdomen”).
Laboratory Tests
• CA-125 may be elevated (normal level is less than 35 units/mL [35 kU/L]).
• Abnormalities in liver function tests may suggest hepatic involvement.
• Abnormalities in renal function tests may suggest compression of the renal system by the tumor.
CLINICAL PRESENTATION
Patients with early ovarian cancer are often asymptomatic and the ovarian mass is often detected incidentally during their annual pelvic examinations. Patients with ovarian cancer often present with nonspecific, vague symptoms such as abdominal bloating, pressure or pain, indigestion, or change in bowel movements.2,4,33 These symptoms can easily be confused with symptoms of common benign gastrointestinal disorders. Patients will often not seek medical attention until these symptoms become unrelenting and bothersome, which allows the disease to progress undetected. Patients with advanced disease may report symptoms such as pain, abdominal distension, and ascites.2,33
Several groups have partnered together to educate women about early signs and symptoms of ovarian cancer. Goff et al. recently developed a symptom index, based on a comparison of symptoms experienced in patients with ovarian cancer and a matched control group.34 Symptoms that were correlated with ovarian cancer were persistent or recurrent bloating, pelvic or abdominal pain, difficulty eating or feeling full quickly, and urinary symptoms (either urgency or frequency). The Gynecologic Cancer Foundation, Society of Gynecologic Oncologists, and American Cancer Society recommend that women who have any of those problems nearly every day for more than 2 weeks should see a gynecologist, especially if the symptoms are new and quite different from her usual state of health. Furthermore, healthcare professionals should keep ovarian cancer in the differential for women presenting with these persistent symptoms.
DIAGNOSIS
The diagnostic workup for suspected ovarian cancer includes a careful physical examination including a Papanicolaou (Pap) smear and a pelvic and rectovaginal examination.7 The presence of a pelvic mass that is unilateral or bilateral, solid, irregular, fixed, or nodular is highly suggestive of ovarian cancer. Unfortunately, by the time a pelvic mass can be palpitated on physical exam, the disease is already advanced beyond the pelvic cavity. A detailed family history should be taken, especially noting the number and pattern of first-degree relatives with malignancies.
A complete blood count, chemistry profile (including liver and renal function tests), and CA-125, carcinoembryonic antigen, and CA19–9 levels should be performed. ![]() Although CA-125 is a nonspecific antigen, it is the best current tumor marker for epithelial ovarian carcinoma. A normal CA-125 value is less then 35 units/mL (35 kU/L). If CA-125 is elevated at the time of diagnosis, changes in CA-125 levels correlate with tumor burden. Rising CA-125 levels are often associated with disease progression, but CA-125 can be elevated in various other conditions such as different phases of the menstrual cycle, diverticulitis, endometriosis, as well as other nongynecologic cancers. When a patient presents with an abdominal mass, it is important to rule out other cancers in the abdominal cavity. Carcinoembryonic antigen and CA19–9 are markers for other gastrointestinal cancers and may be helpful in the differential diagnosis.
Although CA-125 is a nonspecific antigen, it is the best current tumor marker for epithelial ovarian carcinoma. A normal CA-125 value is less then 35 units/mL (35 kU/L). If CA-125 is elevated at the time of diagnosis, changes in CA-125 levels correlate with tumor burden. Rising CA-125 levels are often associated with disease progression, but CA-125 can be elevated in various other conditions such as different phases of the menstrual cycle, diverticulitis, endometriosis, as well as other nongynecologic cancers. When a patient presents with an abdominal mass, it is important to rule out other cancers in the abdominal cavity. Carcinoembryonic antigen and CA19–9 are markers for other gastrointestinal cancers and may be helpful in the differential diagnosis.
Other diagnostic tests should include a transvaginal or abdominal ultrasonography, chest radiography, computed tomography, magnetic resonance imaging, or positron emission tomography scan. An upper GI series, IV pyelogram, cystoscopy, proctoscopy, or barium enema is sometimes indicated to confirm diagnosis and extent of disease.
TREATMENT
Ovarian Cancer
Desired Outcomes
The goals of treatment of ovarian cancer are dependent upon the FIGO stage at diagnosis. While ideally “treatment for cure” is desired, it is important to set realistic expectations for the patient. ![]() Most patients will achieve a complete response to the initial multimodality treatment, but over 50% of these patients will present with recurrent disease within the first 2 years after completion of treatment. Although overall survival has not significantly changed for ovarian cancer patients, the progression-free survival has improved, which translates to less time on chemotherapy and overall improvement in quality of life for these patients.
Most patients will achieve a complete response to the initial multimodality treatment, but over 50% of these patients will present with recurrent disease within the first 2 years after completion of treatment. Although overall survival has not significantly changed for ovarian cancer patients, the progression-free survival has improved, which translates to less time on chemotherapy and overall improvement in quality of life for these patients.
In patients who present with metastatic disease or are not surgical candidates, the goal of treatment becomes focused on alleviating symptoms and prolonging survival as long as quality of life is acceptable. In the setting of recurrent platinum-resistant ovarian cancer, the treatment goal is also focused on alleviating symptoms and prolonging survival as long as quality of life is acceptable. ![]() Phase I/investigational agent clinical trials should be considered because they may be the best treatment option for potential benefit due to the poor efficacy of the available chemotherapy agents for recurrent platinum-resistant ovarian cancer.
Phase I/investigational agent clinical trials should be considered because they may be the best treatment option for potential benefit due to the poor efficacy of the available chemotherapy agents for recurrent platinum-resistant ovarian cancer.
General Approach
![]() A multimodality approach that includes comprehensive surgery and chemotherapy is used for the initial treatment of ovarian cancer with curative intent. Although most patients will initially achieve a complete response, more than 50% will recur within the first 2 years.2,35 A clinical complete response to treatment is defined as no evidence of disease by physical examination or diagnostic tests and a normal CA-125 level.
A multimodality approach that includes comprehensive surgery and chemotherapy is used for the initial treatment of ovarian cancer with curative intent. Although most patients will initially achieve a complete response, more than 50% will recur within the first 2 years.2,35 A clinical complete response to treatment is defined as no evidence of disease by physical examination or diagnostic tests and a normal CA-125 level.
Chemotherapy regimens for ovarian cancer have evolved over the past several decades. Treatment regimens began with single-agent melphalan followed by single-agent cyclophosphamide. Shortly after cisplatin was introduced into clinical practice, it was added to cyclophosphamide, and this combination was the “standard of care” for more than a decade until the introduction of paclitaxel in the 1980s. Paclitaxel soon replaced cyclophosphamide, and paclitaxel plus cisplatin became the standard of care. Carboplatin was then substituted for cisplatin because of its improved toxicity profile, and paclitaxel plus carboplatin was adopted. During this same period, many researchers have conducted numerous clinical trials of intraperitoneal (IP) chemotherapy. In 2006, Armstrong and colleagues published the first IP therapy clinical trial to demonstrate a survival advantage over the standard IV regimen.36However, these advances in chemotherapy for the treatment of ovarian cancer have not yet translated into major changes in overall 5-year survival, which remains less than 20%.
Certain subgroups of patients have a better or worse response to chemotherapy. The histologic subtype of the tumor is a prognostic factor; clear cell histology is more likely to be poorly differentiated, faster growing, and have intrinsic drug resistance.2,37 However, the extent of residual disease, size larger than 1 cm, and tumor grade are better predictors of response to chemotherapy and overall survival.2
In general, younger patients have a better performance status and tolerate chemotherapy better than elderly patients. For unknown reasons, white women tend to have a worse prognosis and response to therapy as compared with women of other ethnic backgrounds.2,6,7
In patients with recurrent ovarian cancer, the goals of treatment are to relieve symptoms such as pain or discomfort from ascites, slow disease progression, and prevent serious complications such as small bowel obstructions.
Surgery
Surgery is the primary treatment intervention for ovarian cancer.37–41 Surgery may be curative for selected patients with limited stage IA disease.
Primary surgical treatment includes a total abdominal hysterectomy with bilateral salpingo-oophorectomy (TAH/BSO), omentectomy, and lymph node dissection (Fig. 110-1).37–41 The primary objective of the surgery is to optimally debulk the tumor to less than 1 cm of residual disease.42 Long-term followup studies confirm that residual disease smaller than 1 cm correlates with higher complete response rates to chemotherapy and longer overall survival as compared to patients with bulky residual disease (>1 cm).40,41
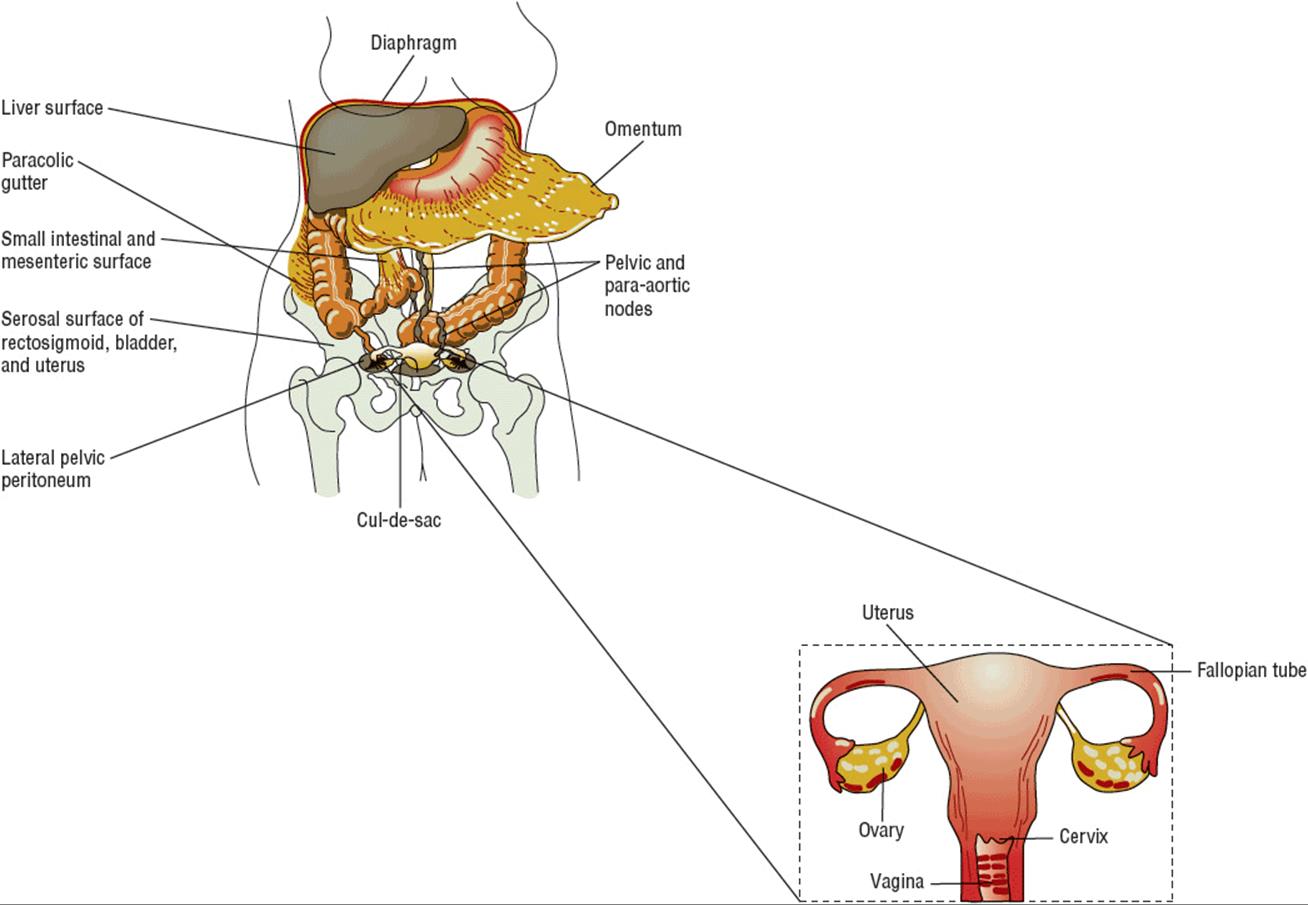
FIGURE 110-1 Staging laparotomy for ovarian cancer with diagram of female reproductive tract (uterus, fallopian tubes, ovaries, vagina). Dashed line box outlines what is removed during the total abdominal hysterectomy with bilateral salpingo-oophorectomy.
A comprehensive exploratory laparotomy is vital for the accurate confirmation of diagnosis and staging of ovarian cancer.37–39 ![]() Unlike other cancers that are typically diagnosed by biopsy or laboratory results and clinically staged by results from imaging tests, gynecologic cancers, such as ovarian cancer, are surgically diagnosed and then staged according to the FIGO staging algorithm (Fig. 110-2). The FIGO staging system requires a fairly extensive surgery by an experienced gynecologic oncologist. The skill of the surgeon has a significant effect on prognosis, with definitive benefit of a trained gynecologic oncologist performing surgery as compared with a gynecologist or general surgeon.43 The reasons for this approach include (a) pelvic tumors cannot be readily biopsied without risk of “tumor seeding,” which can increase the risk of recurrence, and (b) surgical staging takes into account the presence of microscopic disease in samples obtained by pelvic washing and lymph node dissection and read by a pathologist during the surgical procedure. It is recommended that the initial surgical staging and tumor-debulking surgery be completed by a trained gynecologic oncology surgeon when ovarian cancer is suspected to prevent understaging and to optimize overall outcome.44
Unlike other cancers that are typically diagnosed by biopsy or laboratory results and clinically staged by results from imaging tests, gynecologic cancers, such as ovarian cancer, are surgically diagnosed and then staged according to the FIGO staging algorithm (Fig. 110-2). The FIGO staging system requires a fairly extensive surgery by an experienced gynecologic oncologist. The skill of the surgeon has a significant effect on prognosis, with definitive benefit of a trained gynecologic oncologist performing surgery as compared with a gynecologist or general surgeon.43 The reasons for this approach include (a) pelvic tumors cannot be readily biopsied without risk of “tumor seeding,” which can increase the risk of recurrence, and (b) surgical staging takes into account the presence of microscopic disease in samples obtained by pelvic washing and lymph node dissection and read by a pathologist during the surgical procedure. It is recommended that the initial surgical staging and tumor-debulking surgery be completed by a trained gynecologic oncology surgeon when ovarian cancer is suspected to prevent understaging and to optimize overall outcome.44
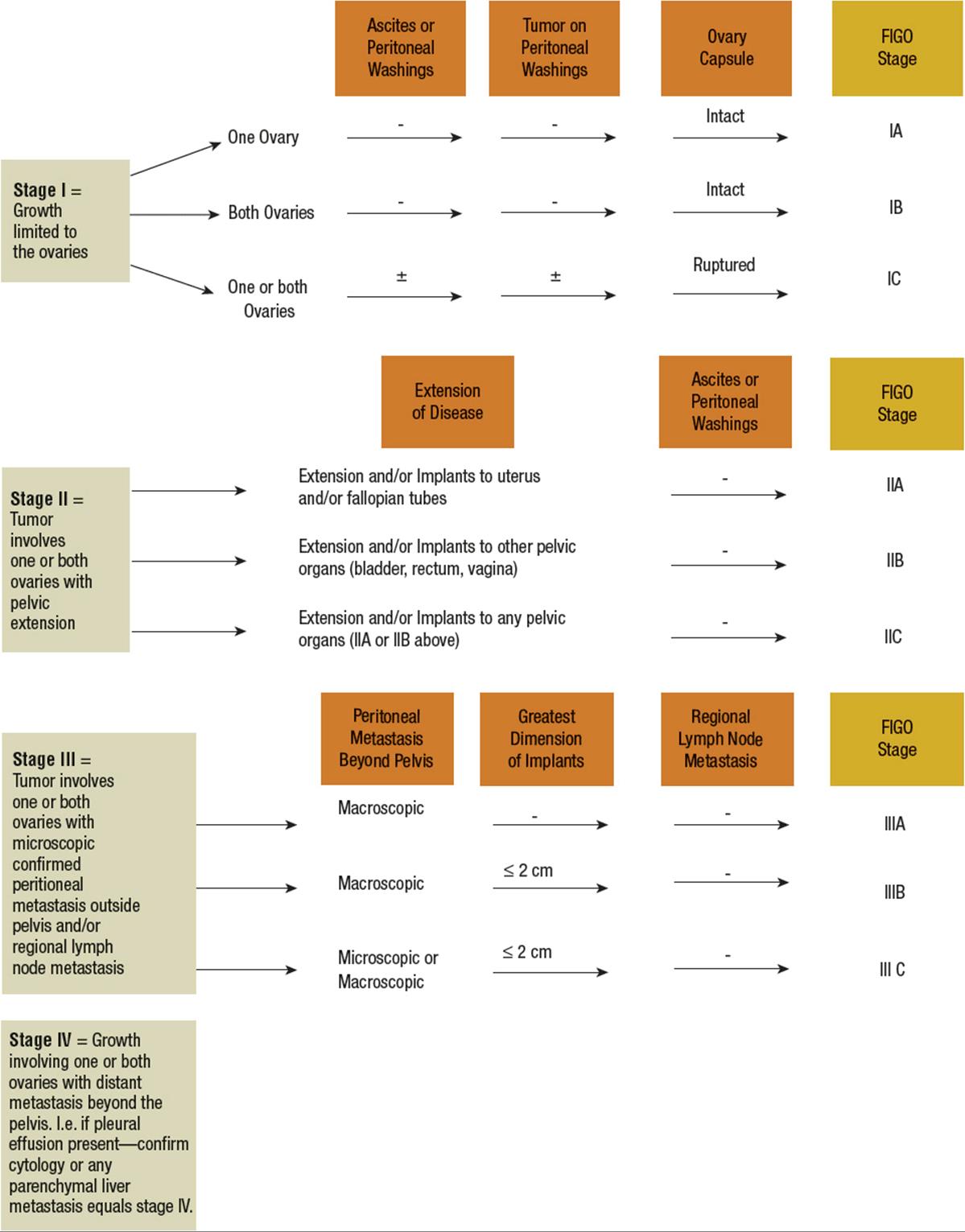
FIGURE 110-2 International Federation of Gynecology and Obstetrics (FIGO) staging algorithm.
Secondary cytoreduction or interval debulking is when surgery is performed after completion of some or all chemotherapy to remove residual disease. Some protocols include additional cycles of chemotherapy after the surgical procedure. The importance of cytoreduction before, during, or after chemotherapy is still controversial, but it has been recommended to facilitate response to chemotherapy and improve overall survival. Randomized trials of secondary surgical cytoreduction have reported conflicting results. In an older randomized trial, van der Burg et al. performed interval debulking surgery on 140 stage IIB to stage IV suboptimally debulked (<1 cm of residual disease) ovarian cancer patients after receiving three cycles of cisplatin plus cyclophosphamide.45 Patients then received an additional three cycles of these same drugs after surgery. Patients randomized to the nonsurgical treatment arm received six cycles of chemotherapy. Interval debulking surgery significantly prolonged overall and progression-free survival and reduced the risk of death by 33%. However, in a recently published study of 550 women with stage III or IV disease treated with primary cytoreductive surgery and three cycles of paclitaxel and cisplatin, patients randomized to receive secondary cytoreductive surgery followed by three more cycles of chemotherapy had similar progression-free survival and overall survival as compared with those randomized to receive three more cycles of chemotherapy alone.46
The overall effect of interval debulking is influenced by several factors, including initial response to chemotherapy, the amount of residual disease before and after second-look surgery, and the presence of microscopic residual disease. The results of recent trials suggest that secondary surgical cytoreduction does not prolong survival in patients who are treated with maximal primary cytoreductive surgery followed by appropriate postoperative chemotherapy.
“Second-look surgery” is an elective surgical procedure performed in patients who achieve a clinical complete response after primary chemotherapy to determine if any visible or microscopic disease is present in the peritoneal cavity. The benefit of “second-look laparotomy” to evaluate residual disease after completing chemotherapy remains controversial because it has been difficult to establish any impact on overall survival. It has questionable benefit because approximately 50% of those with a negative second look still relapsed.3 If visible or microscopic disease is detected during second look, then the clinician may decide to give additional chemotherapy. But if no visible or microscopic disease is detected during second look, the clinician may decide to observe and monitor the patient. Use of laparoscopic surgical techniques is controversial for initial surgery but is sometimes considered in debulking of recurrent or advanced disease when the intent is palliative rather than curative.40 In patients with recurrent disease, the goal of debulking surgery is to relieve symptoms associated with complications such as small bowel obstructions and to help improve the patient’s quality of life.
Radiation
Radiation has a limited role in the management of ovarian cancer. Use of radiation for treatment of early stage disease has had no benefit or impact on overall survival.47 Radiation therapy is most beneficial for palliation of symptoms in patients with recurrent pelvic disease, often associated with small bowel obstructions. The two forms of radiation therapy used in ovarian cancer are external beam whole-abdominal irradiation and intraperitoneal isotopes such as phosphorus-32 (32P). Alleviation of symptoms with external beam whole-abdominal irradiation is associated with a significant improvement in the patient’s quality of life. The recommended dose ranges from 35 to 45 Gy (3500 to 4500 rad), depending on the treatment history and ability to tolerate radiation treatments.
First-Line Chemotherapy
The mainstay of ovarian cancer treatment is chemotherapy. It is used as a component of first-line treatment after completion of surgery and is the primary modality of treatment for recurrent ovarian cancer.
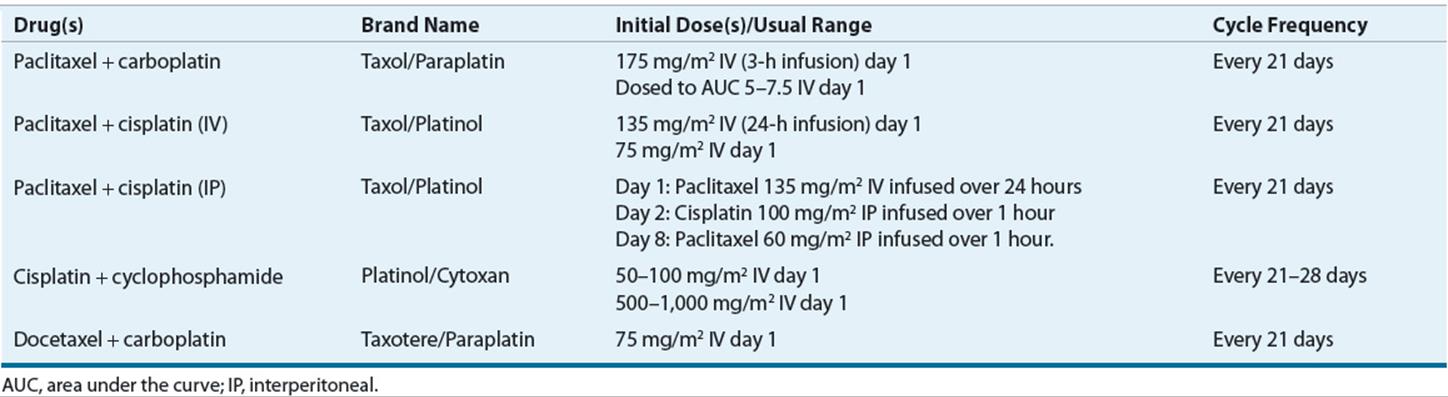
Systemic chemotherapy with a taxane and platinum regimen following optimal surgical debulking is the standard of care for treatment of epithelial ovarian cancer (Fig. 110-3). Table 110-1 summarizes the chemotherapeutic regimens used as the initial treatment of newly diagnosed epithelial ovarian cancer. More than 60 randomized, controlled clinical trials have evaluated combination chemotherapy regimens for the treatment of advanced ovarian cancer, and a meta-analysis of these trials confirmed the efficacy of platinum and taxane regimens over other regimens.48,49
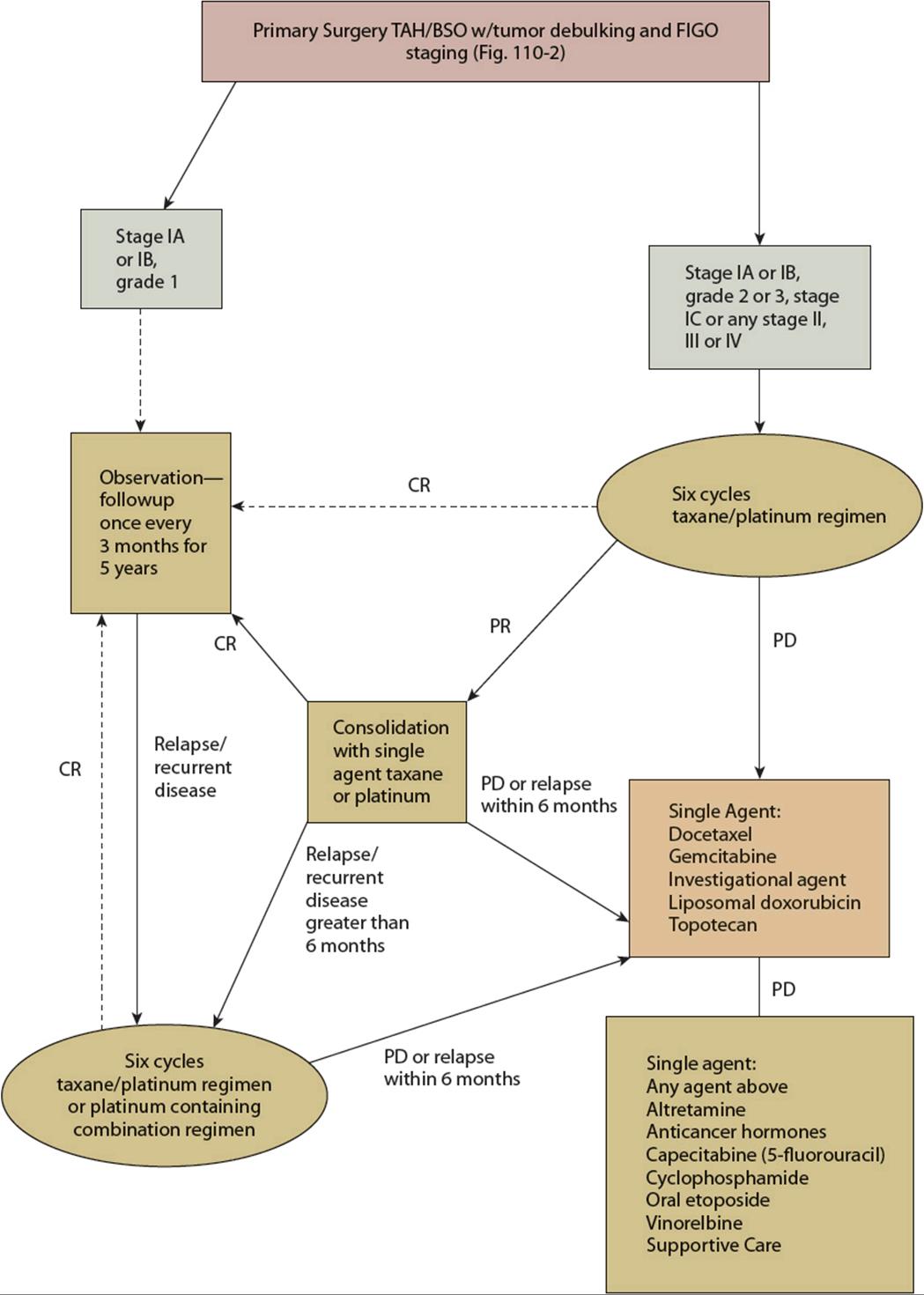
FIGURE 110-3 Management of newly diagnosed, refractory, and progressive epithelial ovarian cancer. All recommendations are category 2A unless otherwise indicated. (CR, complete response; PD, progression of disease; PR, partial response; TAH/BSO, total abdominal hysterectomy/bilateral salpingo-oophorectomy; USO, unilateral salpingo-oophorectomy.)
TABLE 110-1 Initial Chemotherapeutic Regimens of Epithelial Ovarian Cancer
Historically, single-agent alkylating agents such as melphalan, and later cyclophosphamide, were used for the treatment of advanced ovarian cancer until the introduction of cisplatin in the 1970s. Combination chemotherapy regimens containing cisplatin and cyclophosphamide achieved higher response rates and overall survival than regimens without cisplatin in patients with advanced ovarian cancer.50 Based on the results of these trials, the combination of cisplatin plus cyclophosphamide remained the standard of care for the treatment of ovarian cancer until the early 1990s.
The next major advance in the therapy of advanced ovarian cancer occurred with the introduction of paclitaxel into chemotherapy regimens. McGuire et al. reported the results of a Gynecologic Oncology Group (GOG)-111 study that found the combination of paclitaxel 135 mg/m2 over 24 hours and cisplatin 75 mg/m2 achieved higher response rates and longer survival than did cyclophosphamide 750 mg/m2and cisplatin 75 mg/m2 in patients with newly diagnosed, suboptimally debulked, stages III and IV ovarian cancer.51 Survival improved significantly in the paclitaxel arm, with an increase in median progression-free survival (18 months vs. 13 months) and overall survival (38 months vs. 24 months). Neutropenia, alopecia, and peripheral neuropathy were more severe in the paclitaxel plus cisplatin group. Similar results were reported in a large European-Canadian Intergroup Phase III randomized trial study (OV10) that also confirmed superior response rates with the paclitaxel 135 mg/m2 over 24 hours with cisplatin 75 mg/m2 regimen as compared with cyclophosphamide 750 mg/m2 with cisplatin 75 mg/m2 regimen.52 Based on the results of these studies, paclitaxel plus cisplatin was widely adopted and became the accepted standard of care.
The availability of carboplatin led to clinical trials to evaluate whether carboplatin could be substituted for cisplatin, which would spare patients from the significant neurotoxicity and nephrotoxicity associated with cisplatin. Several prospective randomized comparisons of carboplatin plus paclitaxel versus cisplatin plus paclitaxel in patients with advanced ovarian cancer have been conducted.53–56 The results of these trials show that carboplatin plus paclitaxel is equally efficacious and better tolerated than cisplatin and paclitaxel. In the GOG-158 study, 840 previously untreated patients with optimally resected stage III disease (no residual tumor nodule >1 cm) were randomized to carboplatin (area under the curve [AUC] = 7.5) plus paclitaxel 175 mg/m2 over 3 hours, or cisplatin 75 mg/m2 plus paclitaxel 135 mg/m2 over 24 hours administered every 21 days for six cycles.53,55 The results of that trial showed no difference in progression-free survival between the two treatment arms with a median time-to-progression of 19.4 months in the paclitaxel plus cisplatin arm versus 20.7 months in the paclitaxel plus carboplatin arm. As expected, the incidence of leukopenia, fever, gastrointestinal toxicity, and metabolic toxicity was higher in patients in the cisplatin arm, whereas patients in the carboplatin arm experienced more thrombocytopenia and pain. Although the incidence of neurotoxicity was similar in the two treatment arms, it was more severe in the paclitaxel plus cisplatin arm. The results of this study showed that the substitution of carboplatin for cisplatin in the regimen does not compromise efficacy and improves tolerability. These findings were confirmed in two other large randomized, controlled studies.55,56 Based on these results, paclitaxel plus carboplatin became the accepted standard of care.
Other clinical trials have evaluated the use of docetaxel as a substitute for paclitaxel. In the Scottish Randomized Trial in Ovarian Cancer (SCOTROC), Vasey et al. compared carboplatin (AUC = 5) combined with either docetaxel (75 mg/m2 over 1 hour) or paclitaxel (175 mg/m2 over 3 hours) administered every 21 days for six cycles as first-line chemotherapy for stages IC to IV epithelial ovarian cancer.57 The results of this study showed that the substitution of docetaxel for paclitaxel does not compromise efficacy and improves tolerability, particularly neurotoxicity. These findings were not confirmed in another randomized, controlled trial. However, based on the results of this study the combination of docetaxel plus carboplatin is considered a reasonable treatment option for patients with advanced ovarian cancer. Six cycles of paclitaxel plus carboplatin following tumor debulking surgery remain the current standard of care for treatment of advanced ovarian cancer.
Although the choice of taxane or platinum agent does not appear to have a major effect on antitumor activity, weekly paclitaxel administration (“dose density”) may be superior to administration every 3 weeks.58–60 In a phase III trial conducted in Japan, Katsumata et al. reported that patients randomized to six cycles of dose-dense weekly paclitaxel plus carboplatin every 3 weeks had longer progression-free survival as compared to the standard paclitaxel plus carboplatin every 3 weeks.60 Overall survival at 3 years was also significantly longer in patients who received the dose-dense regimen (72% vs. 65%, P = 0.03). However, over 42% of the patients who received the dose-dense regimen dropped out of the study before completing six cycles because of treatment-related toxicities. A confirmatory GOG phase III trial is ongoing to confirm these results and address concerns regarding the feasibility of the dose-dense regimen in a larger group of patients as well as the elderly population.
IP chemotherapy was initially employed as palliative care in the management of ascites and uncontrolled intraabdominal tumors. In the late 1970s, IP chemotherapy administration as a primary treatment intervention was initiated based on the rationale that exposure of the tumor to high drug concentrations would increase tumor drug uptake by passive diffusion and ultimately cancer cell death.61 The increase in AUC exposure in the peritoneal cavity was demonstrated, but the correlative increase in drug uptake in tumor tissue has yet to be validated in any preclinical or clinical study.
![]() IP chemotherapy has demonstrated a benefit in the first-line treatment of patients with optimally debulked advanced-stage ovarian cancer.49,62–64 In the most recent trial, Armstrong et al. reported the results of the GOG-172 study, which evaluated 415 patients randomized to receive either the combination regimen of paclitaxel 135 mg/m2 over 24 hours and cisplatin 75 mg/m2 or a new combination regimen that included paclitaxel 135 mg/m2 IV infused over 24 hours followed by cisplatin 100 mg/m2 IP infused over 1 hour on day 2, and then paclitaxel 60 mg/m2 IP infused over 1 hour on day 8.36 Both treatment regimens were given once every 21 days for a total of six cycles. Patients randomized to the IP chemotherapy arm had a 5.5-month increase in the median progression-free survival and a 15.9-month increase in overall survival.36
IP chemotherapy has demonstrated a benefit in the first-line treatment of patients with optimally debulked advanced-stage ovarian cancer.49,62–64 In the most recent trial, Armstrong et al. reported the results of the GOG-172 study, which evaluated 415 patients randomized to receive either the combination regimen of paclitaxel 135 mg/m2 over 24 hours and cisplatin 75 mg/m2 or a new combination regimen that included paclitaxel 135 mg/m2 IV infused over 24 hours followed by cisplatin 100 mg/m2 IP infused over 1 hour on day 2, and then paclitaxel 60 mg/m2 IP infused over 1 hour on day 8.36 Both treatment regimens were given once every 21 days for a total of six cycles. Patients randomized to the IP chemotherapy arm had a 5.5-month increase in the median progression-free survival and a 15.9-month increase in overall survival.36
Since the publication of this study, there has been a resurgence of interest in the use of IP chemotherapy despite the limitation that only 42% of the patients on the IP treatment arm were able to complete the planned six cycles as a result of significantly more toxicity, including pain, fatigue, myelosuppression, gastrointestinal, metabolic, and neurotoxicity.36,63,65,66 Because only 42% of patients were able to complete the planned six courses of IP chemotherapy, some experts have questioned whether the route of administration was an important contributing factor in the observed differences in overall survival.36The significant increase in systemic toxicity, primarily neurotoxicity, has led to the question of whether IP carboplatin could be substituted for IP cisplatin. Although these platinum agents have demonstrated equal efficacy when administered IV to ovarian cancer patients, based on the concept that drug passively diffuses into the tumor, the difference in molecular size of cisplatin versus carboplatin makes it difficult to extrapolate IP activity of cisplatin to carboplatin.
Clinical Controversy…
The use of interperitoneal (IP) chemotherapy as first-line treatment of advanced ovarian cancer has been recommended by the National Comprehensive Cancer Network (NCCN) guidelines. Most clinical trials have used platinum agents given IP until the Gynecologic Oncology Group (GOG)-172 trial that incorporated IP paclitaxel. Many clinicians are concerned about how to manage hypersensitivity reactions to either platinum or taxane agents when administered IP.
The National Comprehensive Cancer Network (NCCN) 2013 guidelines recommend that IP chemotherapy be considered and offered to appropriate patients as first-line treatment of optimally debulked, ≥1 cm residual disease, ovarian cancer.67 The National Cancer Institute also released a position statement in January 2006 supporting the role of IP chemotherapy as first-line treatment for advanced ovarian cancer.68 ![]() Because of the significant toxicities associated with IP therapy, only carefully selected patients should receive IP therapy. Ideal candidates for IP therapy are younger patients with good performance status, minimal comorbidities, adequate renal and liver function, and optimally debulked disease without significant bowel resection.62,66
Because of the significant toxicities associated with IP therapy, only carefully selected patients should receive IP therapy. Ideal candidates for IP therapy are younger patients with good performance status, minimal comorbidities, adequate renal and liver function, and optimally debulked disease without significant bowel resection.62,66
In patients who are poor surgical candidates because of comorbidities or bulky tumors, neoadjuvant chemotherapy can be given prior to any surgical interventions.69 In patients with bulky disease, the goal of neoadjuvant chemotherapy is to reduce tumor burden to make surgery more feasible and optimal tumor debulking more likely. The typical regimen used in neoadjuvant chemotherapy is three cycles of a taxane combined with a platinum agent followed by surgery. After surgery, patients usually receive another three to six cycles, depending on their response to chemotherapy. In patients who are poor candidates for surgery because of comorbidities, the, primary intent of neoadjuvant chemotherapy is to relieve symptoms and slow disease progression. In this setting, palliative chemotherapy alone has not been curative for patients with advanced ovarian cancer.69 If tolerated, these patients will receive the standard taxane plus platinum chemotherapy regimen once every 3 to 4 weeks. Another option for palliative neoadjuvant chemotherapy, especially in elderly patients, is single-agent carboplatin once every 4 weeks.
Neoadjuvant Chemotherapy
Neoadjuvant chemotherapy is first-line treatment for patients who are poor surgical candidates or patients with bulky or significant tumor burden.35 The neoadjuvant chemotherapy regimen typically includes a combination of taxane with platinum agent is administered every 21 to 28 days as tolerated with intent to reduce tumor burden to point where it potentially could be surgically resected and ideally optimally debulking during surgery.36 After surgery, patient will receive another three to six cycles depending on response to chemotherapy. The role of neoadjuvant chemotherapy for all patients presenting with advance ovarian cancer is being revisited in ongoing GOG clinical trials.
Consolidation Therapy
If patients do not achieve a clinical complete response after completion of six cycles of taxane-platinum regimen, then consolidation chemotherapy should be considered in an attempt to achieve a complete response (see Fig. 110-3). If the patient has a partial response to first-line chemotherapy, as measured by a greater than 50% decline in CA-125 (as compared with the presurgery level) or tumor regression, the cancer is still considered sensitive to the regimen. The typical regimens for consolidation chemotherapy are the taxane plus platinum regimen or single-agent therapy with either a taxane or platinum agent.70 If the patient had a poor response to taxane and platinum, then alternative second-line agents can be considered.67 Additional cycles of chemotherapy are given until complete response is achieved. Another alternative in the setting of no or minimal measurable disease after completion of primary chemotherapy is to just observe the patient and provide supportive care as indicated until disease progresses, then reinitiate chemotherapy at that time.67
Because the initial clinical complete response observed in first-line treatment has not been durable, optimization of first-line therapy is under investigation. Numerous options have been evaluated, including the use of additional cycles or maintenance chemotherapy and dose intensity.
Maintenance Chemotherapy
Maintenance chemotherapy is similar to consolidation chemotherapy except maintenance chemotherapy is given to those patients who have achieved a clinically complete response. The primary differences between consolidation and maintenance chemotherapy are the types of agents used and duration of therapy. Consolidation therapy usually consists of more aggressive combination regimens, whereas maintenance chemotherapy usually consists of single agents given less frequently (i.e., once monthly) to minimize adverse effects. The goal of maintenance chemotherapy is to eliminate any residual microscopic disease that may be present to extend progression-free and overall survival.
Maintenance chemotherapy has gained popularity after the publication of the results of the collaborative Southwest Oncology Group (SWOG) and GOG 178 study that compared single-agent paclitaxel 175 mg/m2 over 3 hours once every 21 days for 3 additional cycles versus an additional 12 cycles.71,72 Eligible patients had to have been in complete clinical remission after at least five to six cycles of a taxane-platinum regimen. This study was closed after the interim analysis by the SWOG Safety Monitoring Committee because patients receiving the additional 12 cycles had longer progression-free survival than those receiving 3 cycles of single-agent paclitaxel (28 vs. 21 months). After the results were reported, many patients randomized to the three-cycle arm chose to receive nine additional cycles of paclitaxel, which reduced the ability of the trial to show a difference in overall survival.73 Because this study was closed early and did not demonstrate an overall survival benefit, another randomized, controlled trial through the GOG was initiated to confirm the improvement in progression-free survival and to attempt to determine the impact on overall survival. Until these confirmatory trials are completed, the role of maintenance chemotherapy is controversial in the management of advanced ovarian cancer patients. Maintenance chemotherapy is listed as an option in the 2013 NCCN guidelines (2B recommendation).67
High-Dose Chemotherapy with Hematopoietic Stem Cell Transplantation
High-dose chemotherapy with autologous or allogeneic hematopoietic stem cell transplantation (HSCT) is an option for selected patients with chemosensitive disease, few comorbidities, and good performance status. Although high response rates have been reported in patients with recurrent ovarian cancer treated with autologous HSCT, the duration of response is usually short and few patients have experienced long-term progression-free survival.74,75Allogeneic HSCT has also been evaluated in recurrent ovarian cancer to induce an immune response against the tumor (“graft-versus-tumor” effect).
Based on the activity of autologous HSCT in recurrent ovarian cancer, Goncalves et al. evaluated the modality for first-line treatment of patients with optimally debulked ovarian cancer. In this multicenter phase II study, 34 patients received two cycles of high-dose cyclophosphamide-epirubicin once every 21 days followed by two cycles of high-dose carboplatin (days 42 and 98).76 Each dose of high-dose carboplatin was followed by hematopoietic stem cell infusion. The results of this study failed to show an improvement in the rate of pathologic complete response with upfront autologous HSCT as compared with standard taxane plus platinum chemotherapy. Additional studies are ongoing to determine the role of autologous or allogeneic HSCT in the treatment of advanced ovarian cancer.
Treatment of Recurrent Disease
![]() Although most patients will achieve a complete response to initial treatment, most patients will eventually have recurrence of their disease within the first 2 years. When a patient relapses, the prognostic factors are similar to the factors after initial surgery except that the disease-free interval—defined as the length of time that has lapsed since the completion of chemotherapy—should be considered to determine if the tumor is likely to be drug resistant to agents used in first-line treatment, which included platinum and taxanes agents. If recurrence occurs less than 6 months after completion of chemotherapy, or if the patient progresses during platinum-based chemotherapy, the tumor is defined as platinum-resistant. Patients with platinum-sensitive disease generally have a better prognosis than platinum-resistant patients.
Although most patients will achieve a complete response to initial treatment, most patients will eventually have recurrence of their disease within the first 2 years. When a patient relapses, the prognostic factors are similar to the factors after initial surgery except that the disease-free interval—defined as the length of time that has lapsed since the completion of chemotherapy—should be considered to determine if the tumor is likely to be drug resistant to agents used in first-line treatment, which included platinum and taxanes agents. If recurrence occurs less than 6 months after completion of chemotherapy, or if the patient progresses during platinum-based chemotherapy, the tumor is defined as platinum-resistant. Patients with platinum-sensitive disease generally have a better prognosis than platinum-resistant patients.
If the patient has a clinical complete response to first-line chemotherapy and the recurrence occurs more than 6 months after chemotherapy is completed, the tumor is considered platinum-sensitive. ![]() In patients with platinum-sensitive ovarian cancer the standard of care is to treat the first recurrence with a doublet, platinum-containing chemotherapy regimen. Table 110-2 summarizes some of the chemotherapeutic regimens used in the treatment of recurrent or refractory ovarian cancer. Because the chemotherapy agents used for second-line treatment of recurrent or refractory platinum-resistant disease have similar response rates that average less than 30%, the selection of the agent is dependent on multiple factors including the toxicity profile of the agent, physician preference, patient performance status, residual toxicities, and patient convenience (see Fig. 110-3). In this setting, the intent of treatment is to prolong survival and alleviate symptoms, not necessarily to achieve another “complete response” to chemotherapy.
In patients with platinum-sensitive ovarian cancer the standard of care is to treat the first recurrence with a doublet, platinum-containing chemotherapy regimen. Table 110-2 summarizes some of the chemotherapeutic regimens used in the treatment of recurrent or refractory ovarian cancer. Because the chemotherapy agents used for second-line treatment of recurrent or refractory platinum-resistant disease have similar response rates that average less than 30%, the selection of the agent is dependent on multiple factors including the toxicity profile of the agent, physician preference, patient performance status, residual toxicities, and patient convenience (see Fig. 110-3). In this setting, the intent of treatment is to prolong survival and alleviate symptoms, not necessarily to achieve another “complete response” to chemotherapy. ![]() Because of poor response rates of the available agents, participation in a clinical trial of an investigational agent is often recommended for patients with recurrent platinum-resistant ovarian cancer.
Because of poor response rates of the available agents, participation in a clinical trial of an investigational agent is often recommended for patients with recurrent platinum-resistant ovarian cancer.
TABLE 110-2 Single-Agent Chemotherapeutic Regimens for Recurrent or Refractory Ovarian Cancer
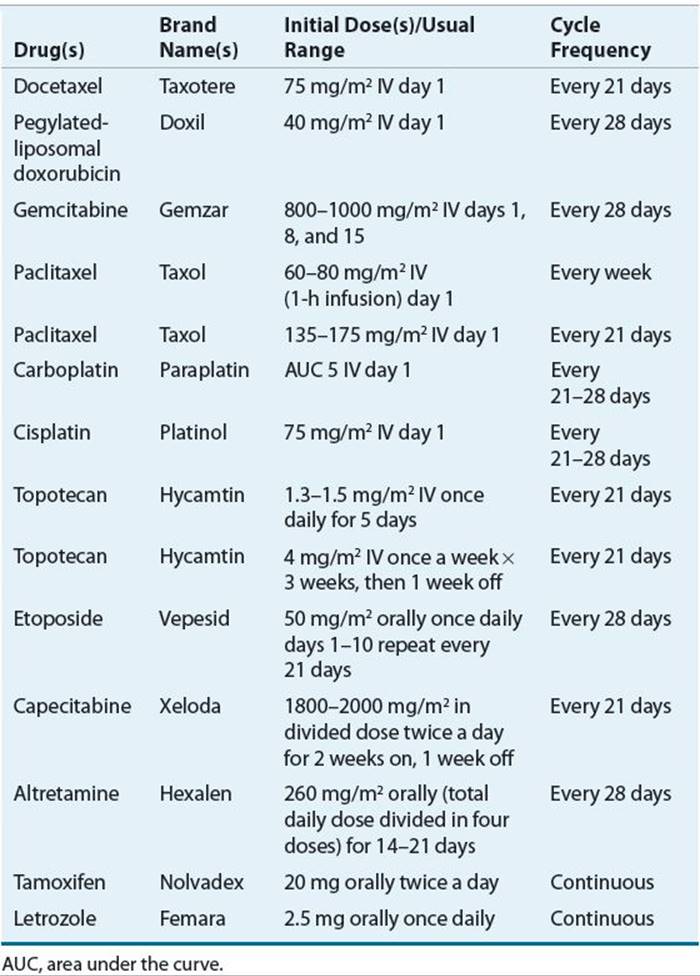
Platinum-Sensitive Disease
![]() Retreatment with a platinum-containing regimen should be considered in patients with platinum-sensitive disease. The International Collaborative Ovarian Neoplasm 4 and Arbeitsgemeinschaft Gynaekologische randomized 802 patients with recurrent platinum-sensitive ovarian cancer to either single-agent platinum, a non–taxane-platinum combination, or a taxane plus platinum combination.75Patients treated with the paclitaxel plus platinum regimen had significantly longer progression-free (29 vs. 24 months) and overall survival (hazard ratio = 0.82 [95% CI 0.69 to 0.97]) as compared with the other two treatment arms.77,78 Although the taxane–platinum combination was clearly superior in this European study, it is difficult to extrapolate these results to patients treated in the United States because of differences in first-line treatment. At the time that International Collaborative Ovarian Neoplasm 4 (ICON4) was conducted, the standard of care in Europe for first-line treatment was single-agent carboplatin, so most patients enrolled in this study had no prior exposure to a taxane agent.77 However, the standard of care in the United States has been a taxane-platinum combination since the early 1990s. Confirmatory data are needed to evaluate whether combination regimens would also be more beneficial in these patients for treatment of recurrent ovarian cancer.
Retreatment with a platinum-containing regimen should be considered in patients with platinum-sensitive disease. The International Collaborative Ovarian Neoplasm 4 and Arbeitsgemeinschaft Gynaekologische randomized 802 patients with recurrent platinum-sensitive ovarian cancer to either single-agent platinum, a non–taxane-platinum combination, or a taxane plus platinum combination.75Patients treated with the paclitaxel plus platinum regimen had significantly longer progression-free (29 vs. 24 months) and overall survival (hazard ratio = 0.82 [95% CI 0.69 to 0.97]) as compared with the other two treatment arms.77,78 Although the taxane–platinum combination was clearly superior in this European study, it is difficult to extrapolate these results to patients treated in the United States because of differences in first-line treatment. At the time that International Collaborative Ovarian Neoplasm 4 (ICON4) was conducted, the standard of care in Europe for first-line treatment was single-agent carboplatin, so most patients enrolled in this study had no prior exposure to a taxane agent.77 However, the standard of care in the United States has been a taxane-platinum combination since the early 1990s. Confirmatory data are needed to evaluate whether combination regimens would also be more beneficial in these patients for treatment of recurrent ovarian cancer.
Clinical Controversy…
In patients with recurrent ovarian cancer that is platinum-sensitive, some clinicians will recommend retreatment with a chemotherapy regimen including a platinum agent. Other clinicians suggest that the platinum-free interval for these patients should be extended and will recommend that recurrent disease first be treated with a non-platinum regimen (i.e., liposomal doxorubicin) and reserve the platinum agent until the next relapse.
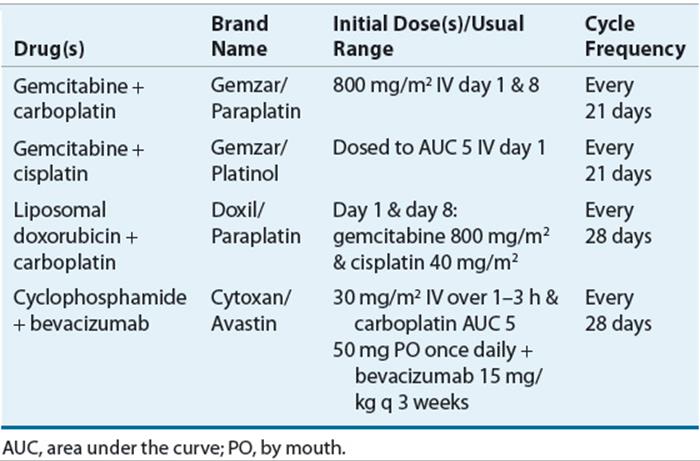
The 2013 NCCN guidelines recommend the combination of platinum agent with gemcitabine, liposomal doxorubicin, or paclitaxel for treatment of platinum-sensitive recurrent ovarian cancer67 (Table 110-3). In addition, the combination of gemcitabine plus cisplatin has demonstrated improvement in progression-free survival78 (see Table 110-3). Carboplatin alone or any of the second-line agents is recommended for patients with platinum-sensitive disease who are unable to tolerate additional combination chemotherapy regimens because of residual toxicity or poor performance status.67,79
TABLE 110-3 Combination Chemotherapy Regimens for Platinum-Sensitive Recurrent Ovarian Cancer
Platinum-Resistant Disease
Frequently patients present with recurrent drug-resistant disease after initial platinum-based therapy and cytoreductive surgery.80,81 Patients who progress on a platinum agent or have no response are considered “platinum-refractory,” whereas those patients who have recurrence within 6 months of completing a platinum-containing regimen are considered “platinum-resistant.”79 The 2013 NCCN guidelines list many possible treatment options for recurrent platinum-resistant or refractory ovarian carcinoma.67 The optimal chemotherapeutic agent or regimen in the treatment of platinum-resistant disease is currently unclear. Ideally, the agent should be active in ovarian cancer and non–cross-resistant with taxanes or platinum agents. Regrettably, the response rate is low for all of the agents in platinum-refractory or resistant ovarian cancer.82 Patients should typically be evaluated for response after treatment with at least three cycles of the chemotherapy agent or regimen. Because partial responses are rare, stable disease with relief of symptoms is considered a treatment success. If no response is observed, then an alternative chemotherapy regimen may be selected. Because all the potential agents have similar efficacy, the selection of agents and sequence used for treatment as the patient progresses will vary based on residual toxicity, dosing schedule, patient convenience, and physician preference.
Topotecan, an analog of the plant alkaloid 20(S)-camptothecin, is active in patients with metastatic ovarian cancer and is non–cross-resistant with platinum-based chemotherapy.83 Preclinical studies suggest that protracted schedules of administration with low doses achieve the greatest antitumor response.84 Topotecan has demonstrated activity in phase II trials as second-line and salvage therapy in patients who have relapsed after, or progressed during, platinum-based therapy.83–85 A randomized phase III trial compared topotecan and paclitaxel in patients with advanced ovarian cancer who had failed one platinum-based regimen.86 Patients were randomized to receive topotecan 1.5 mg/m2 per day as a 30-minute infusion for 5 days repeated every 21 days or paclitaxel 175 mg/m2 as a 3-hour infusion every 21 days. The overall response rate was 21% and 13% for the topotecan- and paclitaxel-treated groups, respectively. The median time-to-progression for topotecan-treated patients (32 weeks) was not significantly different from that for paclitaxel-treated patients (20 weeks). Median survival was 61 weeks in the topotecan-treated group and 43 weeks in the paclitaxel-treated group. Topotecan was well tolerated with minimal nonhematologic toxicities.83,85,86
Pegylated liposomal doxorubicin is one of the primary agents used for second-line therapy of recurrent ovarian cancer.87–89 The drug tends to be better tolerated than topotecan, which is important for heavily pretreated patients with advanced disease. A large, randomized phase III study compared pegylated liposomal doxorubicin 50 mg/m2 every 4 weeks to topotecan 1.5 mg/m2 per day for 5 days repeated every 21 days in patients who failed first-line platinum therapy.86 A total of 474 patients were randomized, 239 to pegylated liposomal doxorubicin and 235 to topotecan. The overall response rates for the pegylated liposomal doxorubicin and topotecan groups were 20% and 17%, respectively. Overall survival tended to favor pegylated liposomal doxorubicin, with a median of 108 weeks versus 71 weeks for topotecan. Differences in toxicity were observed between the arms, with more hematologic toxicity occurring in the topotecan arm and more palmar–plantar erythrodysesthesia (PPE) in the pegylated liposomal doxorubicin arm. However, the incidence of PPE has decreased in current clinical practice because the standard dose of pegylated liposomal doxorubicin used currently, 40 mg/m2, is less than the dose that was used in the initial clinical trials and approved by the FDA.90,91
Gemcitabine, a novel pyrimidine antimetabolite, is also widely used in the treatment of recurrent platinum-resistant ovarian cancer. Although the overall response rate is only about 13% to 22% with single-agent gemcitabine in patients with platinum-refractory recurrent ovarian cancer, an additional 16% to 50% of patients have stable disease for a median of 7 months.92,93 The main toxicities include myelosuppression, fatigue, myalgia, and skin rash. Because of its non–cross-resistant activity and in vivo synergy with platinum agents, gemcitabine is being evaluated in doublet regimens in patients with refractory disease and with carboplatin/taxane regimens in previously untreated patients.93 The combination of gemcitabine with taxanes has demonstrated response rates from 36% to 90%, which if confirmed, are extremely encouraging.93
Other agents that have shown an overall response rate of 10% to 25% in patients with recurrent ovarian cancer include altretamine, etoposide, capecitabine, tamoxifen, letrozole, vinorelbine, and oxaliplatin.94Response rates tend to be higher in the platinum-sensitive subgroups. Most of these agents are available in oral formulations, which allows for outpatient administration in the palliative care setting.
Although there are no therapeutic guidelines for the selection of agents for the treatment of recurrent platinum-resistant ovarian cancer, the three most commonly used agents in clinical practice include pegylated liposomal doxorubicin, gemcitabine, and topotecan. These agents have demonstrated efficacy when used as a single agent and in combination with other agents. A phase II GOG study is ongoing to help define the optimal chemotherapy combination for treatment of recurrent or refractory platinum-resistant ovarian cancer. Selection of chemotherapy for treatment of recurrent disease is ultimately based on the patient’s residual toxicities, scheduling and convenience, and physician preference.
Additional research continues to identify new agents and new targets for the treatment of ovarian cancer. Because platinum agents and taxanes have been identified as the most active classes of agents for treatment of ovarian cancer, drug development has focused on new platinum derivatives, taxanes and taxane analogs, and agents that exert cytotoxic activity by interacting with DNA directly. Specifically, new cytotoxic agents such as trabectedin, pemetrexed, and epothilones are currently being evaluated in clinical trials.
Biologic and Targeted Agents
Monoclonal antibodies such as bevacizumab and cetuximab and small-molecule tyrosine kinase inhibitors such as sunitinib, gefitinib, or sorafenib, are being evaluated to be incorporated into first-line and recurrent treatment regimens for ovarian cancer.95–99 Although the biologic agents as single agents have not demonstrated significant activity, the results of several clinical trials show that the addition of agents such as bevacizumab into first-line and maintenance regimens improves progression-free survival. However, the impact on overall survival is controversial.
Bevacizumab
Bevacizumab is a recombinant humanized monoclonal antibody that targets vascular endothelial growth factor (VEGF), a key mediator of angiogenesis. In the setting of recurrent disease, single-agent bevacizumab produces a response rate similar to other therapies of 16% to 21%.100,101 Response rates with combinations of bevacizumab range from 15% to 80%.98,100–110 However, these phase II trials have also reported a higher risk of bowel perforation in patients treated with bevacizumab-containing regimens.100,101,111 Bevacizumab should therefore not be given to patients who have had recent bowel surgery or a history of significant bowel resections. Recent efforts have focused on the integration of bevacizumab into first-line treatment regimens. Phase II studies confirmed the safety and feasibility of 6 cycles of paclitaxel, carboplatin plus bevacizumab, given every 3 weeks, followed by maintenance bevacizumab once every 3 weeks for 1 year.107,108 Based on these encouraging results, the GOG initiated a confirmatory phase III study comparing six cycles of standard paclitaxel plus carboplatin to six cycles of the same regimen with bevacizumab to determine whether bevacizumab improves the efficacy of paclitaxel plus carboplatin. The duration of maintenance bevacizumab remains controversial. The results of these trials should determine the role of bevacizumab in the treatment of ovarian cancer.
Clinical Controversy…
Although bevacizumab has demonstrated some progression-free survival advantages when used in combination, its effect on overall survival is not clear. Therefore, it is not clear that the benefits justify the high cost of bevacizumab. As a result, health insurance companies do not consistently reimburse providers for bevacizumab when used for the treatment of ovarian cancer.
Targeted Agents
Tyrosine kinase inhibitors such as sorafenib, sunitinib, pazopanib, and cediranib inhibit angiogenesis by specifically targeting the VEGF receptor (VEGFR). When given as single agents, tyrosine kinase inhibitors have demonstrated some antitumor activity in ovarian cancer.112,113 Ongoing trials have focused on combination regimens with cytotoxic agents for first-line treatment and also treatment of recurrent ovarian cancer. Another interesting targeted agent is VEGF Trap (aflibercept), a fusion protein that targets VEGF-A. Aflibercept has been beneficial in the treatment of malignant ascites and is currently being incorporated into first-line regimens. Epidermal growth factor receptor (EGFR) inhibitors such as erlotinib have not demonstrated activity either alone or combined with chemotherapy or bevacizumab for the treatment of ovarian cancer.109 The newer classes of targeted therapies such as platelet-derived growth factor (PDGF) inhibitors and poly-ADP-ribose polymerase (PARP) inhibitors are being investigated in ongoing clinical trials.114
PERSONALIZED PHARMACOTHERAPY
Current research efforts are focused on identifying biomarkers which are predictive of response in ovarian cancer. The primary focus has been on response to first-line treatment agents, paclitaxel and platinum and the multidrug resistance (MDR) pathway, specifically ABC-transport protein P-glycoprotein (Pgp).115
Epigenetics is a potential source of drug resistance. Epigenetic changes are heritable changes outside of the “traditional” DNA coding sequence. Aberrant DNA methylation and histone acetylation are epigenetic events which can silence tumor suppression genes required for apoptosis or DNA repair and therefore lead to resistance. The acetylation of histones is required for active genes and deacetylation occurs in silenced genes. Histone acetyltransferases (HATs) add acetyl groups and histone deacetylases (HDACs) remove acetyl groups.116
Ovarian cancers upregulate a variety of factors involved in DNA repair, angiogenesis, proliferation, and migration; they also downregulate mismatch-repair (MMR), cell adhesion, and apoptotic genes.116Tumorigenesis can induce hypermethylation or hypomethylation, which leads to chromosomal instability.117 Hypermethylation or deacetylation has been shown to silence specific genes such as hMLH1, which leads to tumor formation and progression in the ovaries. Deacetylation of p21, a cell cycle regulator, can occur in ovarian carcinomas. Epigenetics may downregulate Apaf-1 and p16 while potentially upregulating MDR1. Finally, resistance to a platinum and taxane regimen may be associated with hMLH1 methylation.116
Genomic information is being gathered to help overcome resistance such as finding amplified or deleted sequences or determining whether single nucleotide polymorphisms (SNPs) are the cause of resistance. Proteomics can also be used to identify mechanisms of resistance by finding over- or underexpressed proteins. These methods could lead to personalized pharmacotherapy if any of these biomarkers are predictive of drug response or resistance.118
While most chemotherapy drugs used to treat ovarian cancer are dosed according to body surface area (BSA), carboplatin dosing is personalized based on each individual’s renal function with the Calvert formula: carboplatin dose = AUC × (glomerular filtration rate [GFR] + 25).119 When it was originally developed and validated, measured GFR was used in the Calvert equation. However, the estimated creatinine clearance (CLcr) is now used in clinical practice in place of measured GFR. Despite more than 30 years of clinical use, it is still not clear which equation to use to estimate CLcr and the best method to estimate CLcr in certain patient subgroups. The use of personalized carboplatin dose has reduced potential toxicity such as thrombocytopenia, neuropathy, and nephrotoxicity.119 Personalized dosing of carboplatin is one of the reasons why it is often the preferred platinum agent over cisplatin for primary treatment for ovarian cancer.56
EVALUATION OF THERAPEUTIC OUTCOMES
During chemotherapy patients may experience numerous side effects such as nausea and vomiting, myelosuppression, neuropathy, and changes in organ function. Patients receiving a taxane or platinum chemotherapy regimen should be monitored for signs of hypersensitivity or infusion-related reactions. Patients treated with paclitaxel often experience infusion-related reactions, which have been attributed to the polyethoxylated castor oil (Cremophor) diluent. Premedications including an H1-blocker, H2-blocker, and steroid should be administered prior to each chemotherapy administration to prevent hypersensitivity reactions. If a patient has a reaction, increasing the duration of the infusion from 3 to 6 hours may help with infusion-related reactions. For patients with a true taxane allergy, paclitaxel desensitization can be attempted with 24 hours of premedications (H1-blocker, H2-blocker, and steroids) followed by paclitaxel given as a titrated infusion (1:1000 → 1:100 → 1:10 → full dose) over 8 hours. With repeated exposure (i.e., seven cycles or more) to carboplatin, patients can develop a delayed hypersensitivity reaction. A similar protocol can be used for carboplatin desensitization.
Ovarian cancer patients receive multiple courses of chemotherapy that can have varying effects on kidney and liver function, often with a delayed onset. Appropriate laboratory tests should be ordered to assess organ function so that chemotherapy doses can be adjusted as indicated. Patients on platinum-containing regimens can often experience electrolyte wasting, so patients should be monitored for electrolyte replacement, IV or oral, as indicated. The use of myeloid growth factors should be considered to prevent treatment delays and/or dose reductions. Prevention of nausea and vomiting, both acute and delayed, is critical for patients receiving emetogenic chemotherapy regimens such as paclitaxel plus carboplatin.
During initial taxane plus platinum chemotherapy, a CA-125 level should be obtained with each cycle and monitored for at least a 50% reduction in CA-125 after completion of four cycles, which is related to an improved prognosis. Patients who achieve a complete response after completion of first-line treatment should have followup once every 3 months, including CA-125, physical examination, pelvic examination, and appropriate diagnostic scans (i.e., computed tomography, magnetic resonance imaging, or positron emission tomography), which should be evaluated for presence of disease. In addition to routine followup examinations, clinicians should monitor for resolution of any residual chemotherapy-related side effects, including neuropathies, nephrotoxicity, ototoxicity, myelosuppression, and nausea and vomiting.
In the progressive disease or recurrent setting, CA-125 levels can still be used to monitor for response and should be checked with each cycle, although no change in therapy is recommended until after completion of at least three cycles of the second-line chemotherapy. In addition to laboratory monitoring, appropriate diagnostic scans (i.e., computed tomography, magnetic resonance imaging, or positron emission tomography) should be done once every three cycles. Patients need to be monitored with each cycle of chemotherapy to evaluate for new or persistent toxicities such as neuropathies, fluid retention, PPE, myelosuppression, and nausea and vomiting. Another precaution to keep in mind for patients with significant ascites, the “dry weight” or an adjusted body weight should be used for dosing chemotherapy.
Eventually, most ovarian cancer patients will progress through all chemotherapy regimens and investigational treatment options, after which the best supportive care measures should be provided to maintain patient comfort and quality of life. A plan to treat common complications of progressive ovarian cancer, including thrombosis, ascites, uncontrollable pain, and small bowel obstruction should be developed. The primary goal at the end of life for patients with progressive ovarian cancer is to provide any measures necessary to maintain patient comfort and quality of life.
ABBREVIATIONS

REFERENCES
1. Bast RC, Brewer M, Zou, C, et al. Prevention and early detection of ovarian cancer: Mission impossible? Recent Results Cancer Res 2007;174:91–100.
2. Heintz APM, Odicino F, Maisonneuve P, et al. Carcinoma of the ovary. FIGO 6th annual report on the results of treatment in gynecological cancer. Int J Gynaecol Obstet 2006;95(suppl):S161–S192.
3. Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin 2013;63:11–30.
4. Colomob N, VanGorp T, Parma G, et al. Ovarian cancer. Crit Rev Oncol Hematol 2006;60:159–179.
5. Lux MP, Fashing PA, Beckmann MW. Hereditary breast and ovarian cancer: Review and future perspectives. J Mol Med 2006;84:16–28.
6. Pavelka JC, Li AJ, Karlan BY. Hereditary ovarian cancer-assessing risk and prevention strategies. Obstet Gynecol Clin North Am 2007;34:651–665.
7. Lancaster JM, Powell CB, Kauff ND, et al. Society of Gynecologic Oncologists Education Committee statement on risk assessment for inherited gynecologic cancer predispositions. Gynecol Oncol 2007;107:159–162.
8. RimMertens-Waler I, Baxter RC, Marsh DJ. Gonadotropin signaling in epithelial ovarian cancer. Cancer Letters 2012;324:152–159.
9. Aletti GC, Gallenberg MM, Cliby WA, et al. Current management strategies for ovarian cancer. Mayo Clin Proc 2007;82:751–770.
10. Permuth-Wey J, Sellers TA. Epidemiology of ovarian cancer. Methods Mol Biol 2009;472:413–437.
11. Bhoola S, Hoskins WJ. Diagnosis and management of epithelial ovarian cancer. Obstet Gynecol 2006;107:1399–1410.
12. Cannistra SA, Gershenson, DM, Recht A. Ovarian cancer, fallopian tube carcinoma, and peritoneal carcinoma. In: Devita VT, Hellman S, Rosenberg SA, eds. Cancer: Principles and Practice of Oncology. 8th ed. Philadelphia: Lippincott Williams & Wilkins, 2008:1568–1592.
13. Silverberg SG, Histopathologic grading of ovarian carcinoma: A review and proposal. Int J Gynecol Pathol 2000;19:7–15.
14. Longuespee R, Boyon C, Desmons A, et al. Ovarian cancer molecular pathology. Cancer Metastat Rev 2012;31:713–732.
15. Krygiou M, Tsoumpou I, Martin-Hirsch P, et al. Ovarian cancer screening. Anticancer Res 2006;26:4793–4801.
16. Patterson DM, Rustin GJS. Controversies in the management of germ cell tumours of the ovary. Curr Opin Oncol 2006;18:500–506.
17. Sherman ME, Lacey JV, Buys SS, et al. Ovarian volume: Determinants and associations with cancer among postmenopausal women. Cancer Epidemiol Biomarkers Prev 2006;15:1550–1554.
18. Pavlik EJ, DePriest PD, Gallion HH, et al. Ovarian volume related to age. Gynecol Oncol 2000;77:410–412.
19. Edwards BK, Brown ML, Wingo PA, et al. Annual report to the nation on the status of cancer 1975–2002 featuring population-based trends in cancer treatment. J Natl Cancer Inst 2005;97:1407–1427.
20. Munkarah A, Chatterjee M, Tainsky MA. Update on ovarian cancer screening. Curr Opin Obstet Gynecol 2007;19:22–26.
21. Moyer VA. U.S. Preventive Services Task Force. Screening for ovarian cancer: Recommendation statement. Ann Intern Med 2012;157:900–904.
22. Chu CS, Rubin SC. Screening for ovarian cancer in the general population. Best Pract Res Clin Obstet Gynaecol 2006;20:307–320.
23. Dann JL, Zorn KK. Strategies for ovarian cancer prevention. Obstet Gynecol Clin North Am 2007;34:667–686.
24. Harris RE, Beebe-Donk J, Doss H, et al. Aspirin, ibuprofen, and other non-steroidal anti-inflammatory drugs in cancer prevention: A critical review of non-selective COX-2 blockade. Oncol Rep 2005;13:559–583.
25. Tavani A, Gallus S, La Vecchia C, et al. Aspirin and ovarian cancer: An Italian case-control study. Ann Oncol 2000;11:1171–1173.
26. Domchek SM, Rebbeck TR. Prophylactic oophorectomy in women at increased cancer risk. Curr Opin Obstet Gynecol 2007;19:27–30.
27. Meeuwissen PAM, Seynaeve C, Brekelmans CTM, et al. Outcome of surveillance and prophylactic salpingo-oophorectomy in asymptomatic women at high risk for ovarian cancer. Gynecol Oncol 2005;97:476–482.
28. Finch A, Beiner M, Lubinski J, et al. Salpingo-oophorectomy and the risk of ovarian, fallopian tube, and peritoneal cancers in women with a BRCA1 or BRCA2 mutation. JAMA 2006;296:185–192.
29. Narod SA, Sun P, Ghadirian P, et al. Tubal ligation and risk of ovarian cancer in carriers of BRCA1 and BRCA2 mutations: A case control study. Lancet 2001;357:1467–1470.
30. Lux MP, Fashing PA, Beckmann MW. Hereditary breast and ovarian cancer: Review and future perspectives. J Mol Med 2006;84:16–28.
31. Coukos G. Gene therapy for ovarian cancer. Oncology 2001;15:1197–1208.
32. Tait DL, Obermiller PS, Hatmaker AR, Relin-Frazier S, Holt JT. Ovarian cancer BRCA1 gene therapy: Phase I and II trial differences in immune response and vector stability. Clin Cancer Res 1999;5:1708–1714.
33. Goff BA, Mandel LS, Melancon CH, Muntz HG. Frequency of symptoms of ovarian cancer in women presenting to primary care clinics. JAMA 2004;291:2705–2712.
34. Goff BA, Mandel LS, Drescher CW, et al. Development of an ovarian cancer symptom index. Cancer 2007;109:221–227.
35. Fung-Kee-Fung M, Oliver T, Elit L, Oza A, Kirte HW. Optimal chemotherapy treatment for women with recurrent ovarian cancer. Curr Oncol 2007;14:195–208.
36. Armstrong DK, Bundy B, Wenzel L, et al. Intraperitoneal cisplatin and paclitaxel in ovarian cancer. N Engl J Med 2006;354:34–43.
37. Cooper A, DePriest P. Surgical management of women with ovarian cancer. Semin Oncol 2007;34:226–233.
38. Schorge JO, McCann C, Del Carmen M. Surgical debulking of ovarian cancer: What difference does it make? Rev Obstet Gynecol 2010;3:111–117.
39. Stratton JF, Tidy JA, Paterson MEL. The surgical management of ovarian cancer. Cancer Treat Rev 2001;27:111–118.
40. Ibeanu OA, Bristow RW. Predicting the outcome of cytoreductive surgery for advanced ovarian cancer: a review. Int J Gynecol Cancer 2010;20(suppl 1):S1–S11.
41. Hoffman MS, Griffin D, Tebes S, et al. Sites of bowel resected to achieve optimal ovarian cancer cytoreduction: Implications regarding surgical management. Am J Obstet Gynecol 2005;193:582–588.
42. Bhoola S, Hoskins WJ. Diagnosis and management of epithelial ovarian cancer. Obstet Gynecol 2006;107:1399–1410.
43. Mayer AR, Chambers SK, Graves E, et al. Ovarian cancer staging: Does it require a gynecologic oncologist? Gynecol Oncol 1992;47:223–227.
44. Nguyen HN, Averette HE, Hoskins W, et al. National survey of ovarian carcinoma, V: The impact of physician’s specialty on patients’ survival. Cancer 1993;72:3663–3670.
45. Bristo RE, Eisenhauer EL, Santillan An, Chi DS. Delaying the primary surgical effort for advanced ovarian cancer: A systematic review of neoadjuvant chemotherapy and interval cytoreduction. Gynecol Oncol 2007;140:480–490.
46. Martinek IE, Kehoe S. When should surgical cytoreduction in advanced ovarian cancer take place? J Oncol 2010. DOI:10.1155/2010/852028.
47. Lorusso D, Mancini M, Di Rocco R, Fontanelli R, Raspagliesi F. The role of secondary surgery in recurrent ovarian cancer. Int J Surg Oncol 2012, in press.
48. Bohra U. Recent advances in management of epithelial ovarian cancer. Apollo Med 2012;9:212–218.
49. Kyrgiou M, Salanti G, Pavlidis N, Paraskevaidis E, Ioannidis JPA. Survival benefits with diverse chemotherapy regimens for ovarian cancer: Meta-analysis or multiple treatments. J Natl Cancer Inst 2006;98:1655–1663.
50. Ozols R. Systemic therapy for ovarian cancer: Current status and new treatments. Semin Oncol 2006;33(suppl 6):S3–S11.
51. McGuire WP, Hoskins WJ, Brady MF, et al. Cyclophosphamide and cisplatin compared with paclitaxel and cisplatin in patients with stage III and stage IV ovarian cancer. N Engl J Med 1996;334:1–6.
52. Piccart MJ, Bertelsen K, Stuart G, et al. Long-term follow-up confirms a survival advantage of the paclitaxel-cisplatin regimen over the cyclophosphamide-cisplatin combination in advanced ovarian cancer. Int J Gynecol Cancer 2003;13(suppl 2):144–148.
53. Bookman MA, Greer BE, Ozols RF. Optimal therapy of advanced ovarian cancer: Carboplatin and paclitaxel vs. cisplatin and paclitaxel (GOG 158) and an update on GOG0 182-ICON5. Int J Gynecol Cancer 2003;136:735–740.
54. Ozols RF, Bundy BN, Green BE, et al. Phase III trial of carboplatin and paclitaxel compared with cisplatin and paclitaxel in patients with optimally resected stage III ovarian cancer: A Gynecologic Oncology Group Study. J Clin Oncol 2003;21:3194–3200.
55. du Bois A, Luck HJ, Meier W, et al. A randomized clinical trial of cisplatin/paclitaxel versus carboplatin/paclitaxel as first-line treatment of ovarian cancer. J Natl Cancer Inst 2003;95:1320–1329.
56. Neijt JP, Engelholm SA, Tuxen MK, et al. Exploratory phase III study of paclitaxel and cisplatin versus paclitaxel and carboplatin in advanced ovarian cancer. J Clin Oncol 2000;18:3084–3092.
57. Vasey PA, Jayson GC, Gordon A, et al. Phase III randomized trial of docetaxel-carboplatin versus paclitaxel-carboplatin as first-line chemotherapy for ovarian carcinoma. J Natl Cancer Inst 2004;96:1682–1691.
58. Fennelly D, Aghajanian C, Shapiro F, et al. Phase I and pharmacologic study of paclitaxel administered weekly in patients with relapsed ovarian cancer. J Clin Oncol 1997;15:187–192.
59. Markman M, Blessing J, Rubin SC, et al. Phase II trial of weekly paclitaxel (80 mg/m2) in platinum and paclitaxel-resistant ovarian and primary peritoneal cancers: A Gynecologic Group Study. Gynecol Oncol 2006;101:436–440.
60. Katsumata N, Yasuda M, Takahashi F, et al. Dose-dense paclitaxel once a week in combination with carboplatin every 3 weeks for advanced ovarian cancer: A phase 3, open-label, randomized controlled trial. Lancet 2009;374:1331–1338.
61. Fujiwara K, Armstrong D, Morgan M, Markman M. Principles and practice of intraperitoneal chemotherapy for ovarian cancer. Int J Gynecol Cancer 2007;17:1–20.
62. Jaaback K, Johnson N. Intraperitoneal chemotherapy for the initial management of primary epithelial ovarian cancer. Cochrane Database Syst Rev 2006;3:1–28.
63. Markman M, Bundy BN, Alberts DS, et al. Phase III trial of standard-dose intravenous cisplatin in small-volume stage III ovarian carcinoma: An intergroup study of the gynecologic oncology group, southwestern oncology group, and eastern cooperative oncology group. J Clin Oncol 2001;19:1001–1007.
64. Alberts DS, Liu PY, Hannigan EV, et al. Intraperitoneal cisplatin plus intravenous cyclophosphamide versus intravenous cisplatin plus intravenous cyclophosphamide for stage III ovarian cancer. N Engl J Med 1996;335:1950–1955.
65. Walker JL, Armstrong DK, Huang HQ, et al. Intraperitoneal catheter outcomes in a phase III trial of intravenous versus intraperitoneal chemotherapy in optimal stage III ovarian and primary peritoneal cancer: A Gynecologic Oncology Group Study. Gynecol Oncol 2006;100:27–32.
66. Markman M, Walker JL. Intraperitoneal chemotherapy of ovarian cancer: A review, with a focus on practical aspects of treatment. J Clin Oncol 2006;24:988–994.
67. National Comprehensive Cancer Network (NCCN) Practice Guidelines in Oncology—Ovarian Cancer, V1.2013. 2013, http://www.nccn.org.
68. NCI Clinical Announcement. Intraperitoneal Chemotherapy for Ovarian Cancer. January 5, 2006, http://ctep.cancer.gov/highlights/clin_annc_010506.pdf.
69. Salzberg M, Thurlimann B, Bonnefois H, et al. Current concepts of treatment strategies in advanced or recurrent ovarian cancer. Oncology 2005;68:293–298.
70. Gadducci A, Cosio S, Conte PF, Genazzani AR. Consolidation and maintenance treatments for patients with advanced epithelial ovarian cancer in complete response after first-line chemotherapy: A review of the literature. Crit Rev Oncol Hematol 2005;55:153–166.
71. Markman M, Liu PY, Wilczynski S, et al. Phase III randomized trial of 12 versus 3 months of maintenance paclitaxel in patients with advanced ovarian cancer who attained a clinically-defined complete response to platinum/paclitaxel-based chemotherapy: A Southwest Oncology Group and Gynecology Oncology Group trial. J Clin Oncol 2003;21:2460–2465.
72. Gadducci A, Cosio S, Conte PF, Genazzani AR. Consolidation and maintenance treatments for patients with advanced epithelial ovarian cancer in complete response after first-line chemotherapy: A review of the literature. Crit Rev Oncol Hematol 2005;55:153–166.
73. Markman M. Unresolved issues in the chemotherapeutic management of gynecologic malignancies. Semin Oncol 2006;33(suppl 6):S33–S38.
74. Mulder PO, Willemse PH, Aalders JG, et al. High-dose chemotherapy with autologous bone marrow transplantation in patients with refractory ovarian cancer. Eur J Cancer Clin Oncol 1989;25:645–649.
75. Stiff PJ, Veum-Stone J, Lazarus HM, et al. High-dose chemotherapy and autologous stem cell transplantation for ovarian cancer: An autologous blood and marrow transplant registry report. Ann Intern Med 2000;133:504–515.
76. Goncalves A, Delva R, Fabbro M, et al. Post-operative sequential high-dose chemotherapy with haematopoietic stem cell support as front-line treatment in advanced ovarian cancer: A multicenter study. Bone Marrow Transplant 2006;37:651–659.
77. ICON and AGO Collaborators. Paclitaxel plus platinum-based chemotherapy versus conventional platinum-based chemotherapy in women with relapsed ovarian cancer: The ICON4/AGO-OVAR-2.2 trial. Lancet 2003;361:2099–2106.
78. Bozas G, Bamias A, Koutsoukou, et al. Biweekly gemcitabine and cisplatin in platinum resistant/refractory, paclitaxel pre-treated, ovarian and peritoneal carcinoma. Gynecol Oncol 2007;104:580–585.
79. Gronlund B, Hogdall C, Hansen HH, Engelholm SA. Performance status rather than age is the key prognostic factor in second-line treatment of elderly patients with epithelial ovarian carcinoma. Cancer 2002;94:1961–1967.
80. Fung-Kee-Fung M, Oliver T, Elit L, et al. Optimal chemotherapy treatment for women with recurrent ovarian cancer. Curr Oncol 2007;14:195–208.
81. Inciura A, Simavicius A, Juozaityte E, et al. Comparison of adjuvant and neoadjuvant chemotherapy in the management of advanced ovarian cancer: A retrospective study of 574 patients. BMC Cancer 2006;6:153.
82. Herzog TJ, Pothuri B. Ovarian cancer: A focus on management of recurrent disease. Nat Clin Pract Oncol 2006;3:604–611.
83. Swisher EM, Mutch DG, Rader JS, et al. Topotecan in platinum- and paclitaxel-resistant ovarian cancer. Gynecol Oncol 1997;66:480–486.
84. Markman M. Topotecan: An important new drug in the management of ovarian cancer. Semin Oncol 1997;24:S5–S11.
85. ten Bokkel Huinink W, Gore M, Carmichael J, et al. Topotecan versus paclitaxel for the treatment of recurrent epithelial ovarian cancer. J Clin Oncol 1997;15:2183–2193.
86. Creemers GJ, Bolis G, Gore M, et al. Topotecan, an active drug in the second-line treatment of epithelial ovarian cancer: Results of a large European phase II study. J Clin Oncol 1996;14:3056–3061.
87. Muggia FM, Hainsworth JD, Jeffers S, et al. Phase II study of liposomal doxorubicin in refractory ovarian cancer: Antitumor activity and toxicity modification by liposomal encapsulation. J Clin Oncol 1997;15:987–993.
88. Gordon AN, Cranai CO, Rose PG, et al. Phase II study of liposomal doxorubicin in platinum- and paclitaxel refractory epithelial ovarian cancer. J Clin Oncol 2000;18:3093–3100.
89. Gordon AN, Fleagle JT, Guthrie D, et al. Recurrent epithelial ovarian carcinoma: A randomized phase III trial of pegylated liposomal doxorubicin versus topotecan. J Clin Oncol 2001;19:3312–3322.
90. Wilailk S, Linasmita V. A study of pegylated liposomal doxorubicin in platinum-refractory epithelial ovarian cancer. Oncology 2004;67:183–186.
91. Drake RD, Lin WM, King M, Farrar D, Miller DS, Coleman RL. Oral dexamethasone attenuates Doxil-induced palmer-plantar erythrodysesthesias in patients with recurrent gynecologic malignancies. Gynecol Oncol 2004;94:320–324.
92. Lund B, Hansen P, Theilade K, et al. Phase II study of gemcitabine (2′, 2′-difluorodeoxycytidine in previously treated ovarian cancer patients. J Natl Cancer Inst 1994;6:1530–1533.
93. Poveda A. Gemcitabine in patients with ovarian cancer. Cancer Treat Rev 2005;31(suppl 4):S29–S37.
94. Cannistra SA, Bast RC, Berek JS, et al. Progress in the management of gynecologic cancer: Consensus summary statement. J Clin Oncol 2003;21(suppl):129S–132S.
95. Ozols RF. Systemic therapy for ovarian cancer: Current status and new treatments. Semin Oncol 2006;33(suppl 6): S3–S11.
96. Kurzeder C, Sauer G, Deissler H. Molecular targets of ovarian carcinomas with acquired resistance to platinum/taxane chemotherapy. Curr Cancer Drug Targets 2006;6:207–227.
97. Cannistra SA, Bast RC, Berek JS, et al. Progress in the management of gynecologic cancer: Consensus summary statement. J Clin Oncol 2003;21(suppl):129s–132s.
98. Garcia AA, Hirte H, Fleming G, et al. Phase II Clinical Trial of Bevacizumab and Low-Dose Metronomic Oral Cyclophosphamide in Recurrent Ovarian Cancer: A Trial of the California, Chicago, and Princess Margaret Hospital Phase II Consortia. J Clin Oncol 2008;26:76–82.
99. Cohn DE, Valmadre S, Resnick KE, et al. Bevacizumab and weekly taxane chemotherapy demonstrates activity in refractory ovarian cancer. Gynecol Oncol 2006;102:134–139.
100. Cannistra SA, Matulonis UA, Penson RT, et al. Phase II study of bevacizumab in patients with platinum-resistant ovarian cancer or peritoneal serous cancer. J Clin Oncol 2007;25:5180–5186.
101. Burger RA, Sill MW, Monk BJ, Greer BE, Sorvosky JI. Phase II trial of bevacizumab in persistent or recurrent epithelial ovarian cancer or primary peritoneal cancer: A gynecologic oncology group study. J Clin Oncol 2007;25:5165–5171.
102. Monk BJ, Han E, Josephs-Cowan CA, Pugmire G, Burger RA. Salvage bevacizumab (rhuMAB VEGF)-based therapy after multiple prior cytotoxic regimens in advanced refractory epithelial ovarian cancer. Gynecol Oncol 2006;102:140–144.
103. Wright JD, Hagemann A, Rader JS, et al. Bevacizumab combination therapy in recurrent, platinum-refractory, epithelial ovarian carcinoma. Cancer 2006;107:83–89.
104. Chura JC, Van Iseghem K, Downs LS Jr, Carson IF, Judson PI. Bevacizumab plus cyclophosphamide in heavily pretreated patients with recurrent ovarian cancer. Gynecol Oncol 2007;107:326–330.
105. Simpkins F, Belinson JI, Rose PG, et al. Avoiding bevacizumab related gastrointestinal toxicity for recurrent ovarian cancer by careful patient screening. Gynecol Oncol 2007;107:118–123.
106. McGonigle KF, Muntz HG, Vuky J, et al. Combined weekly topotecan and biweekly bevacizumab in women with platinum-resistant ovarian, peritoneal, or fallopian tube cancer: Results of a phase 2 study. Cancer 2011;117:3731–3740.
107. Penson RT, Dizon DS, Cannistra SA, et al. Phase II study of carboplatin, paclitaxel, and bevacizumab with maintenance bevacizumab as first-line chemotherapy for advanced müllerian tumors. J Clin Oncol 2009;28:154–159.
108. Micha JP, Goldstein BH, Rettenmaier MA, et al. A phase II study of outpatient first-line paclitaxel, carboplatin, and bevacizumab for advanced-stage epithelial ovarian, peritoneal, and fallopian tube cancer. Int J Gynecol Cancer 2007;17:771–776.
109. Nimeiri HS, Oza AM, Morgan RJ, et al. Efficacy and safety of bevacizumab plus erlotinib for patients with recurrent ovarian, primary peritoneal, and fallopian tube cancer: A trial of the Chicago, PMH, and California Phase II consortia. Gynecol Oncol 2008;110:49–55.
110. Azad NS, Posadas EM, Kwitkowski, et al. Combination targeted therapy with sorafenib and bevacizumab results in enhanced toxicity and antitumor activity. J Clin Oncol 2008;26:3709–3714.
111. Richardson DL, Backes FJ, Seamon LG, et al. Combination gemcitabine, platinum, and bevacizumab for the treatment of recurrent ovarian cancer. Gynecol Oncol 2008;111:461–466.
112. Matulonis UA, Berlin S, Ivy P, et al. Cediranib, an inhibitor of vascular endothelial growth factor receptor kinases, in an active drug in recurrent epithelial ovarian, fallopian tube and peritoneal cancer. J Clin Oncol 2009;27:5601–5606.
113. Biagi JJ, Oza AM, Chalchal HI, et al. A phase II study of sunitinib in patients with recurrent epithelial ovarian and primary peritoneal carcinoma: An NCIC Clinical Trials Group Study. Ann Oncol 2011;22:335–340.
114. Banerjee S, Kaye S. The role of targeted therapy in ovarian cancer. Eur J Cancer 2011;47(suppl 3):S116–S130.
115. Marsh S, Paul J, King CR, Gifford G, McLeod H, Brown R. Pharmacogenetic assessment of toxicity and outcome after platinum plus taxane chemotherapy in ovarian cancer: The Scottish Randomized Trial in Ovarian Cancer. J Clin Oncol 2007;25:4528–4535.
116. Glasspool RM, Teodoridis JM, Brown R. Epigenetics as a mechanism driving polygenic clinical drug resistance. Br J Cancer 2006;94:1087–1092.
117. Kanai Y. Alterations of DNA methylation and clinicopathological diversity of human cancers. Pathol Int 2008;58:544–558.
118. Schiavone MB, Bashir S, Herzog TJ. Biologic therapies and personalized medicine in gynecologic malignancies. Obstet Gynecol Clin North Am 2012;39:131–144.
119. Calvert AH, Newell DR, Gumbrell LA, et al. Carboplatin dosage: Prospective evaluation of a simple formula based on renal function. J Clin Oncol 1989;7:1748–1756.