Calcium and phosphate are fundamental intracellular components that participate in the functions of all cells. Half of circulating calcium is bound to serum albumin; the other half is composed of free calcium, calcium phosphates, citrates, and salts of other organic acids. Free, ionized calcium is physiologically active.
Plasma calcium levels are maintained within strict limits and are under the endocrine control of three hormones:
–Parathyroid hormone (PTH)
–Calcitriol (1,25-dihydroxycholecalciferol, or 1,25-(OH)2vitamin D3)
–Calcitonin
Note: PTH and calcitriol are the main hormones that control calcium levels. Calcitonin is not involved in maintaining calcium homeostasis on an acute basis. Its secretion is only of importance in hypercalcemic states. Fig. 26.1 provides an overview of calcium homeostasis.
Phosphate (PO43−) binds to Ca2+ and therefore decreases the amount of free, ionized Ca2+ in the blood. Phosphate levels are not regulated directly but rather occur as a secondary effect of calcium regulation (Fig. 26.2).
Physiological roles of calcium
Calcium is vital to normal body functioning, and its levels must be maintained within tight limits. Calcium is the major structural element of bones and teeth, where it is stored in the form of hydroxyapatite crystals [Ca10(PO4)6 (OH)2]. It is also involved in stabilization of membranes, secretion of neurotransmitters, muscle contraction, second messenger systems, vasodilation and vasoconstriction of blood vessels, activation of vitamin K–dependent clotting factors (II, VII, IX, and X), and the secretion of hormones (e.g., insulin).
Magnesium and bone metabolism
Magnesium is the fourth most abundant mineral in the body. About half of the body’s magnesium is stored in the hydroxyapatite crystals of bone, and the other half is intracellular. The levels of magnesium in the blood are well regulated and tend to follow those of calcium and phosphate. Magnesium has a role in bone turnover but less than calcium and phospate. Magnesium deficiency may result from severe diarrhea, alcohol abuse, drugs (e.g., diuretics), and diabetic ketoacidosis. It causes parethesias, seizures, arrhythmias, and tetany (due to accompanying hypocalcemia and hypokalemia). It is treated by replacement.
26.1 Hormones that Regulate Calcium Metabolism
Parathyroid Hormone
PTH is the major regulator of serum [Ca2+].
Synthesis
PTH is an 84 amino acid peptide synthesized and secreted by chief cells of the parathyroid gland.
Regulation of Secretion
PTH secretion is controlled by serum [Ca2+] via negative feedback. Secretion of PTH is stimulated by the following:
– ↓serum [Ca2+]
– ↑serum [PO43−]
– ↓serum [calcitriol]
Note: The effects of phosphate and calcitriol are indirect; they are due to the subsequent decrease in circulating free calcium levels caused by the increased phosphate levels.
Secretion of PTH is inhibited by decreases in serum [Mg2+].
Receptors and Cell Mechanisms
The actions of PTH on the kidney are via specific membrane receptors coupled to adenylate cyclase. Most of the cyclic adenosine monophosphate (cAMP) found in urine is generated by activated PTH receptors.
PTH activates specific membrane receptors on osteoblasts. PTH-activated osteoblasts stimulate osteoclasts via paracrine mechanisms, as there are no PTH receptors on osteoclasts. Activated osteoclasts demineralize bone via collagenases, hyaluronic acid, and acid phosphatases.
Actions
PTH targets the kidneys and bone to increase serum [Ca2+] and decrease serum [PO43−]. Its effects are first seen in the kidneys, as they are direct and occur at lower PTH concentrations. Conversely, the effects of PTH on bone are indirect and require a higher concentration of PTH.
Fig. 26.1 ![]() Calcium homeostasis.
Calcium homeostasis.
Calcium homeostasis is achieved by three main hormones, parathyroid hormone (from parathyroid gland), calcitonin (from parafollicular cells of the thyroid gland), and calcitriol (mainly produced in the kidneys). In low serum Ca2+ states, the actions of parathyroid hormone (PTH) and calcitriol predominate, causing increased Ca2+ uptake from the gut and bone and decreased renal Ca2+ excretion. In high serum Ca2+ states, the action of calcitonin predominates, causing decreased Ca2+ uptake from the gut, increased renal excretion, and storage of excess Ca2+ in bone. (UV, ultraviolet)

Kidneys
– PTH increases the reabsorption of Ca2+ in the distal tubule (primary effect), thereby reducing its excretion.
– PTH rapidly increases phosphodiuresis (urinary excretion of phosphate) by inhibiting phosphate reabsorption in the proximal tubule. This property prevents hyperphosphatemia secondary to the phosphate-liberating effect of PTH on bone. Enhanced clearance of phosphates increases Ca2+ ionization because of less formation of calcium phosphate.
– PTH increases the synthesis and activity of 1-α hydroxylase in the proximal tubule. This enzyme is needed for calcitriol synthesis.
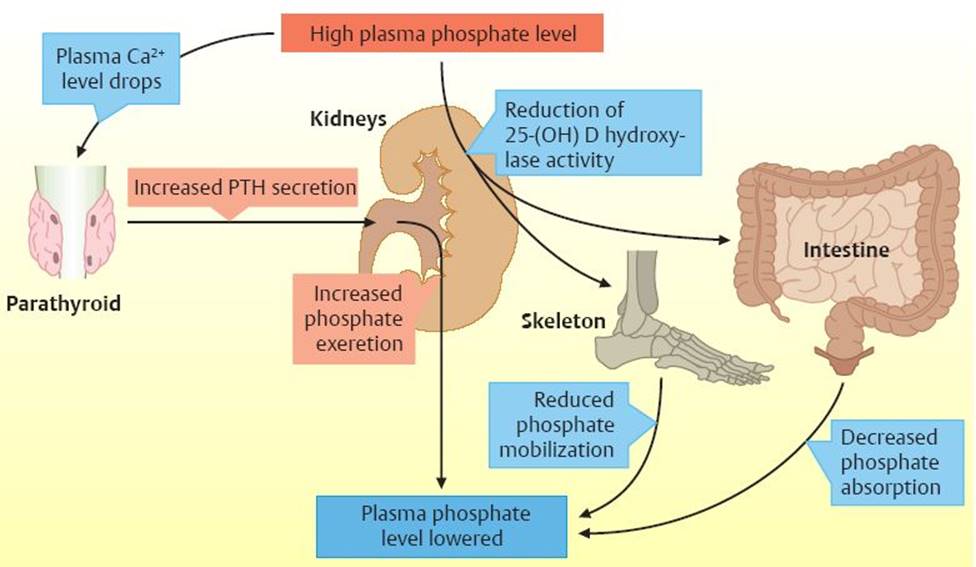
Fig. 26.2 ![]() Regulation of phosphate levels.
Regulation of phosphate levels.
Regulatory mechanisms of phosphate are closely tied to calcium homeostasis. High plasma phosphate levels reduce the level of free, unionized calcium, thus increasing PTH secretion. This causes increased renal phosphate excretion. High plasma phosphate levels also cause a reduction in 25-(OH) D hydroxylase activity, which lowers the formation of 1,25-(OH)2D3, causing decreased intestinal phosphate absorption and decreased phosphate release from bones. These homeostatic mechanisms allow plasma phosphate levels to be reduced without severely affecting calcium metabolism.
– PTH increases urinary cAMP (generated by activated renal PTH receptors).
– PTH increases the excretion of hydroxyproline (from the resorption of bone organic matrix).
Bone
–PTH liberates Ca2+ and PO43− from bone.
– PTH also releases hydroxyproline (from the organic matrix), magnesium, citrate, and osteocalcin from bone.
– PTH indirectly increases the resorption of both calcium and phosphate from bone by increasing calcitriol synthesis.
Intestines
– PTH indirectly increases intestinal Ca2+ absorption by increasing calcitriol synthesis.
Pathophysiology
Hypoparathyroidism. Table 26.1 summarizes hypoparathyroidism.

Multiple endocrine neoplasia type 1
Multiple endocrine neoplasia type 1 (MEN1) is a rare, inherited disorder that causes multiple tumors (usually benign) in the endocrine glands and duodenum. It affects men and women equally and is usually not detected until adulthood, when tumors start growing. The parathyroid glands are most commonly affected. Tumors here cause hyperparathyroidism, which leads to hypercalcemia and its symptoms. It also commonly affects the pancreas, causing gastrinomas (from excess gastrin secretion), which, in turn, cause ulcers. These ulcers are more sinister than normal gastric ulcers and are highly prone to perforate. Multiple gastrinomas causing ulcers is referred to as Zollinger–Ellison syndrome. Pancreatic tumors may also cause insulinomas (leading to hypoglycemia), glucagon excess (leading to diabetes), or vasoactive intestinal peptide (leading to watery diarrhea). Pituitary tumors may also occur, leading to derangement of its hormones. People with MEN1 are more likely to develop cancerous tumors in later life. MEN1 can be detected early by gene testing, and people affected have a 50% chance of passing the disease to their children. There is no cure for MEN1, but there are various drugs and surgical options available to treat the effects.
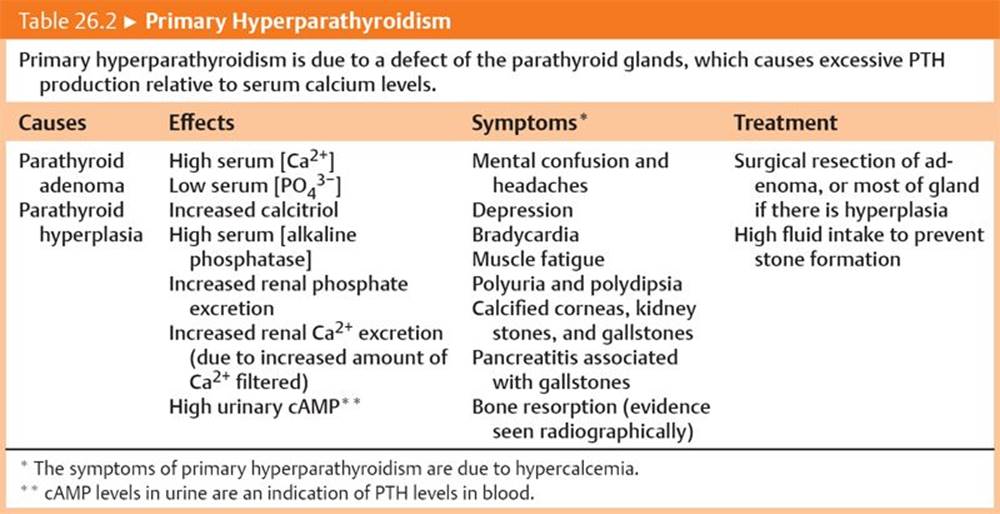
Hyperparathyroidism. Tables 26.2 and 26.3 summarize primary hyperparathyroidism and secondary hyperparathyroidism, respectively.

Alkaline phosphatase
Alkaline phosphatase (ALP) is an enzyme that is predominantly made in the liver and bone, but it may also be made in the kidneys and the intestines, as well as in the placenta during pregnancy. It acts to cleave phosphate groups from molecules, such as nucleotides and proteins. ALP is a by-product of osteoblastic activity, so levels of ALP are raised during periods of rapid bone growth (puberty), in bone diseases that cause bone turnover (e.g., Paget disease and osteomalacia), and during calcium derangement (e.g., hyperparathyroidism). Elevated ALP levels are also a sign of damage to liver cells. Tests that measure ALP are therefore used to detect liver and bone disease, but they lack specificity.
Humoral cancer–associated hypercalcemia
Humoral cancer–associated hypercalcemia is due to the secretion of PTH-related peptide (PTH-rp) by some malignant cancers, such as breast and lung cancer. Ectopic production of PTH itself in tumors is very rare. PTH-rp mimics the actions of PTH, leading to hypercalcemia, hypophosphatemia, and increased urinary phosphate excretion. PTH levels are low due to feedback inhibition from the high serum [Ca2+]. Ultimately, bone resorption occurs. Treatment of the underlying tumor will usually normalize calcium and phosphate levels.
Pseudohyperparathyroidism type 1a
Pseudohyperparathyroidism type 1a, or Albright osteodystrophy, is a disease in which there is resistance to PTH in the kidneys and bone due to a defective GS protein. This causes hypocalcemia, hyperphosphatemia, and increased PTH levels. People with this condition have short fourth and fifth meta-carpals, rounded facies, and the symptoms of hypoparathyroidism (convulsions, psychosis, tetany, paresthesias, numbness, laryngospasm, degeneration of teeth and nails, and cataracts). Unlike hypoparathyroidism, however, pseudohyperparathyroidism type 1a cannot be corrected by exogenous PTH. Treatment for pseudohyperparathyroidism type 1a involves correcting the hypocalcemia with oral calcium and calcitriol, or intravenous (IV) calcium if severe. This should suppress PTH secretion and avoid increased bone remodeling.
Chronic renal failure
In chronic renal failure, the kidneys are unable to perform the necessary α1-hydroxylation reactions to produce calcitriol, and they have a reduced capacity to excrete phosphate. This leads to secondary hyper-parathyroidism due to hypocalcemia and hyperphosphatemia. Derangement of bone remodeling occurs, which is referred to as renal osteodystrophy. The symptoms of renal osteodystrophy include bone and joint pain, bone deformation, and increased likelihood of bone fractures. Chronic renal failure requires hemodialysis several times per week until renal transplantation can occur. Renal osteodystrophy is treated by calcium and calcitriol, restricting dietary intake of phosphate, and by the administration of medication that binds phosphate, such as calcium carbonate or calcium acetate.
Calcitriol
Calcitriol (1,25-dihydroxycholecalciferol [1,25-(OH)2D3]) is the most active metabolite of vitamin D.
Synthesis
Calcitriol (1,25-(OH)2D3) is synthesized in the kidney from 25-hydroxycholecalciferol (25-(OH) D3 under the enzymatic control of 1-α hydroxylase (Fig. 26.3).
Regulation of secretion
Modulation of 1-α hydroxylase activity in the kidneys controls calcitriol synthesis and secretion. The factors that stimulate and inhibit 1-α hydroxylase activity are shown in Table 26.4.

Fig. 26.3 ![]() Vitamin D metabolism.
Vitamin D metabolism.
Ultraviolet B (UVB) light converts 7-dehydrocholesterol to cholecalciferol (vitamin D3). Ingested vitamin D is fat soluble and is transported to the liver in chylomicrons. All free vitamin D is transported in the blood and liver by a specific vitamin D–binding protein (DBP). DBPs are needed because steroid derivatives are hydrophobic. The liver converts vitamin D to 25-hydroxycholecalciferol [25-(OH) D], which is then transported to the kidneys, where it is converted to its active form, calcitriol, or 1,25-(OH)2D, under the influence of parathyroid hormone (via the enzyme 1-α-hydroxylase). The effects of this are increased mineralization of bone, increased calcium and phosphate reabsorption in the kidneys, and increased calcium absorption in the gut. Excess vitamin D is excreted into bile.

Actions
The principal role of calcitriol is to make calcium and phosphate available to extracellular fluid so that new bone can be mineralized.
Intestine
– Calcitriol increases synthesis of the protein calbindin. This is a specific Ca2+-binding protein that transports Ca2+ through cellular cytosols. It acts as a “shuttle” for Ca2+ during the absorptive process (Fig. 26.4).
– Calcitriol increases the permeability of the entire small intestine to Ca2+. This action facilitates passive absorption of Ca2+.
–Calcitriol increases the intestinal absorption of phosphate.
Bone
– Calcitriol mobilizes Ca2+ and phosphate from mature bone for the mineralization of new bone and osteoid tissue. The Ca2+ that is mobilized is also available to the extracellular space.
Kidney
– Calcitriol promotes the reabsorption of Ca2+ and PO43− at physiological concentrations.
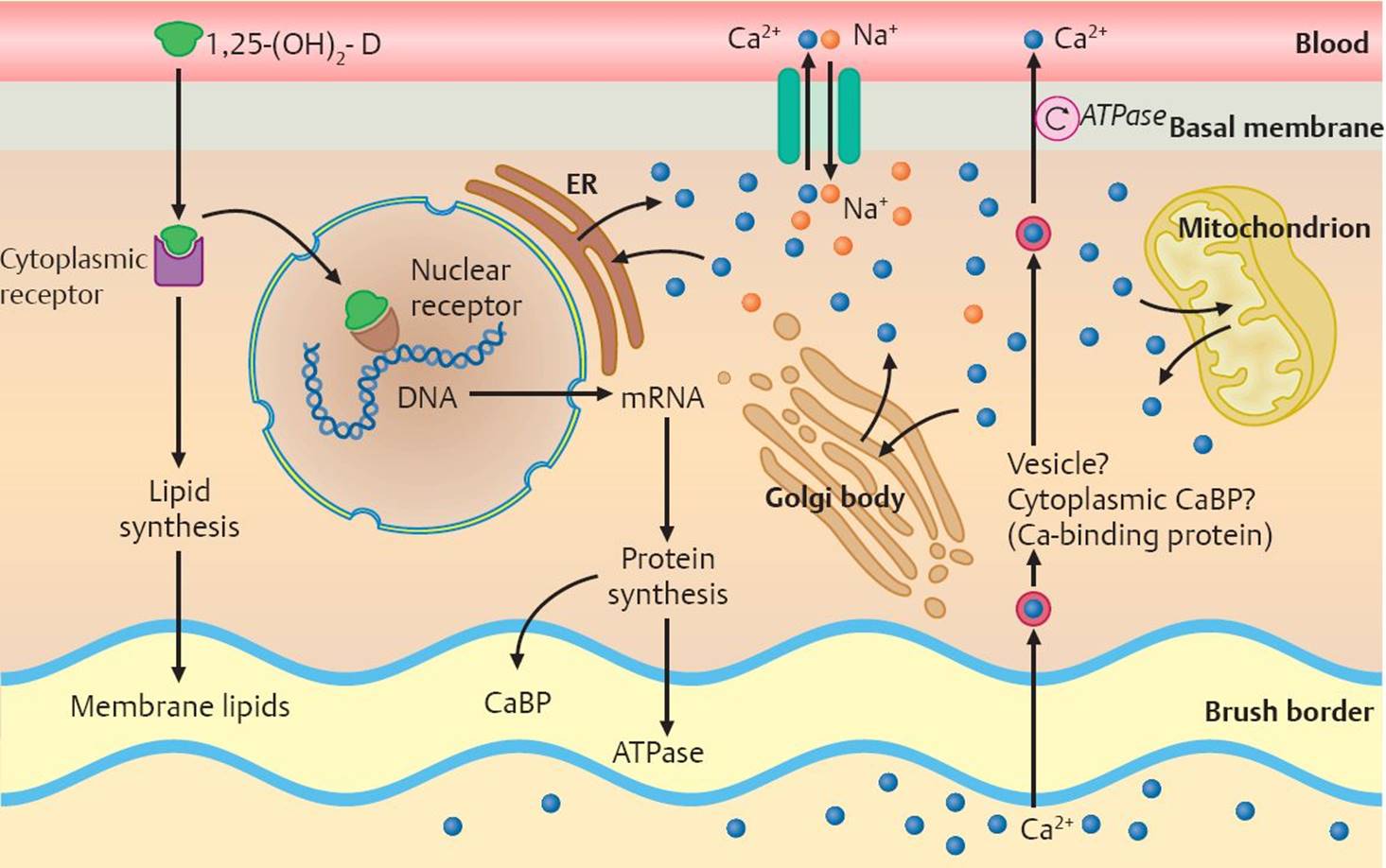
Fig. 26.4 ![]() Induction of calcium absorption.
Induction of calcium absorption.
1,25-(OH)2D3 is necessary for the active intestinal absorption of calcium. In the cytosol, 1,25-(OH)2D is thought to bind to a cytoplasmic receptor before being transferred to a DNA-associated nuclear receptor. This process induces the formation of several proteins, including calcium-binding protein (CaBP), an ATPase, alkaline phosphatase, and phytase. At the same time, lipid synthesis is increased, altering membrane lipids. The exact mechanism by which calcium is transported from the brush border of the intestines to the basal membrane and its absorption into the bloodstream is unknown.
Pathophysiology
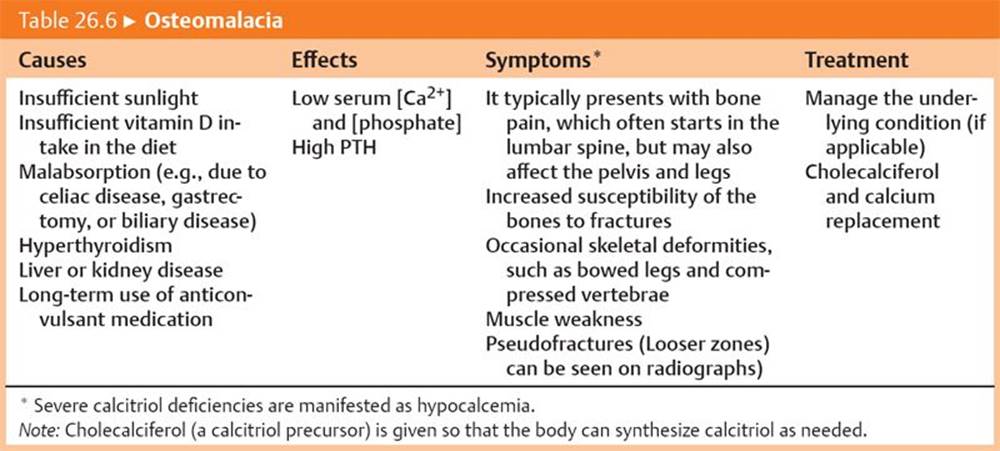
Reduced calcitriol production or resistance to calcitriol produces rickets in infants/children (Table 26.5) and osteomalacia in adults (Table 26.6).

Sarcoidosis and vitamin D hypersensitivity
Sarcoidosis is a multisystem granulomatous inflammatory disease that mainly affects the lungs, lymph nodes, eyes, and skin. It is thought to be autoimmune in origin (delayed-type hypersensitivity reaction [type IV]). Sarcoidosis may be associated with vitamin D hypersensitivity. There is increased 1-α hydroxylase activity in macrophages within the granulomas, causing calcitriol activation. This extrarenal production of calcitriol causes toxicity, producing symptoms such as lethargy and fatigue, irritability, cognitive disturbances, memory loss, and a metallic taste. Hypercalcemia and its accompanying symptoms may also be present, but the compensatory reduction in PTH hormone may prevent hyper-calcemia. This vitamin D hypersensitivity reaction may also occur in some cancers, including lymphomas and sarcomas.
Calcitonin
Synthesis
Calcitonin is a 32 amino acid peptide that is synthesized and secreted by the parafollicular cells (or C cells) of the thyroid gland.
Regulation of Secretion
Secretion of calcitonin is stimulated by high serum [Ca2+] and is inhibited by normal or low serum [Ca2+].
Actions
– The main role of calcitonin is to inhibit bone resorption, which reduces liberation of the calcium and phosphate, thereby indirectly lowering serum Ca2+ levels. It also prevents acute hypercalcemia due to dietary intake postprandially (after eating).
Bone
– Calcitonin limits the resorption of Ca2+ from bone and thereby protects bone mass. It has no effect on bone formation.
Kidneys
– Calcitonin increases the urinary excretion of Ca2+, PO43−, Na+, and K+ and reduces the excretion of Mg2+.
Therapeutic Uses
– Calcitonin is used clinically in cases of hypercalcemia and hyperphosphatemia because it decreases blood levels of these minerals while protecting bone mass from breakdown.
– Calcitonin also inhibits the loss of bone mass in aging, so it is used to treat postmenopausal women for osteoporosis (bone thinning).
Table 26.7 provides a summary of the effects of PTH, calcitriol, and calcitonin in calcium homeostasis.

26.2 Integrated Regulation of Calcium and Phosphate in the Body
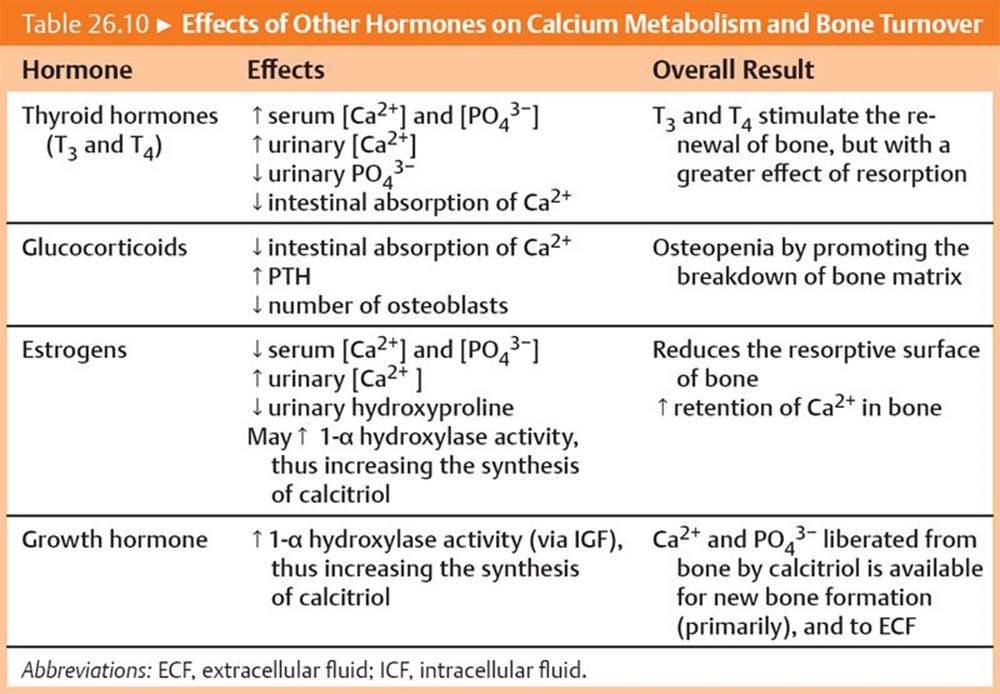
The main hormonal events and effects of low serum Ca2+ and high serum Ca2+ are summarized in Tables 26.8 and 26.9, respectively. The actions of other hormones on calcium homeostasis are summarized in Table 26.10.


Clinical signs of hypocalcemia
There are two specific clinical signs of hypocalcemia, Chvostek sign and Trousseau sign. Chvostek sign is twitching of the facial muscles after tapping on the facial nerve. Trousseau sign is carpopedal spasm elicited by inflating a blood pressure cuff above systolic pressure for 3 minutes. The carpopedal spasm includes flexion of the wrist, flexion of the metacarpophalangeal joints, extension of the interphalangeal joints, and adduction of the thumbs and fingers. It may be accompanied by paresthesias and fasciculations. Trousseau sign is an earlier and more sensitive sign of hypocalcemia than Chvostek sign.
Calcium balance in pregnancy
Pregnancy and lactation are two periods of potential high calcium loss. To maintain calcium balance, there is increased Ca2+ absorption from the intestine during pregnancy due to enhanced levels of calcitriol and plasma-binding proteins. PTH is also elevated during pregnancy, but increased calcitonin levels ensure that bone mass is protected.