Na+ and Cl− reabsorption decreases from proximal tubules to Henle's loops to classic distal tubules to collecting tubules and ducts
Figure 35-2 summarizes the segmental distribution of Na+ reabsorption along the nephron. The proximal tubule reabsorbs the largest fraction of filtered Na+ (~67%). Because [Na+] in tubule fluid (or TFNa; see p. 733) remains almost the same as that in plasma (i.e., TFNa/PNa = 1.0) throughout the length of the proximal tubule, it follows that the [Na+] in the reabsorbate is virtually the same as that in plasma. Because Na+ salts are the dominant osmotically active solutes in the filtrate, reabsorption must be a nearly isosmotic process. ![]() N35-1
N35-1
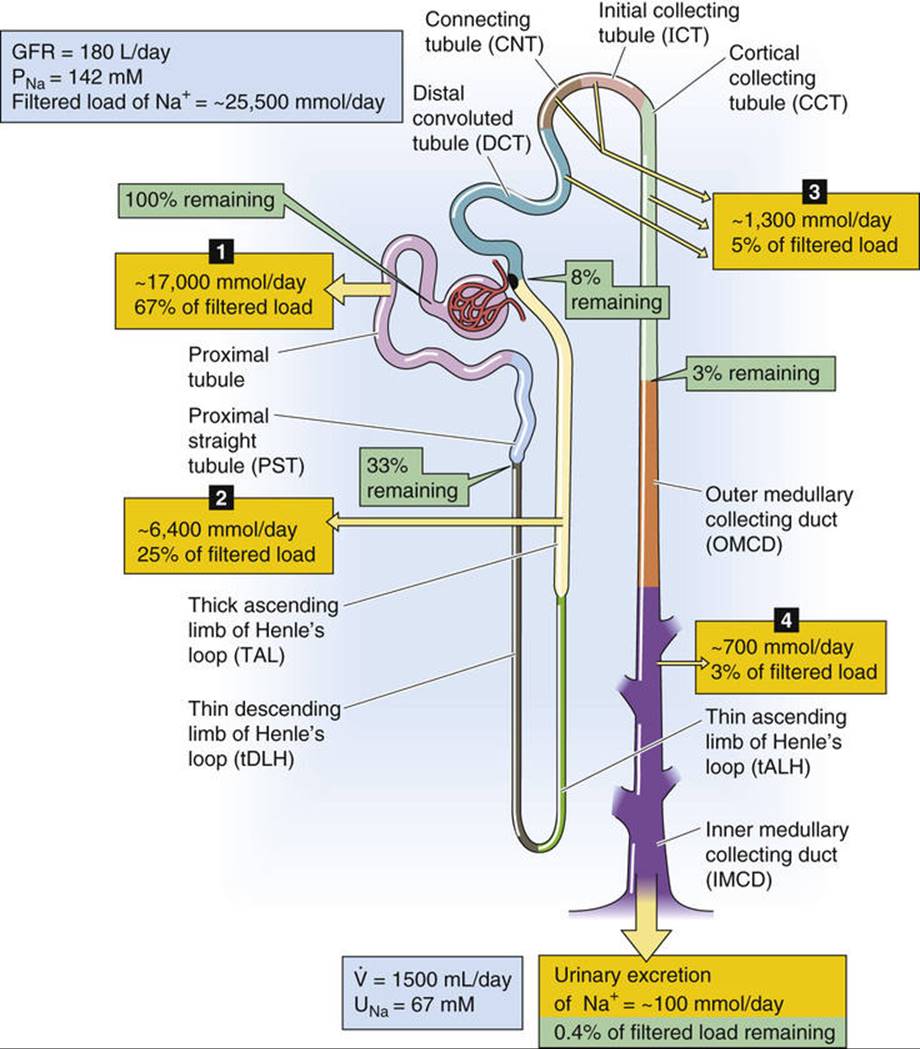
FIGURE 35-2 Estimates of renal handling of Na+ along the nephron. The numbered yellow boxes indicate the absolute amount of Na+—as well as the fraction of the filtered load—that various nephron segments reabsorb. The green boxes indicate the fraction of the filtered load that remains in the lumen at these sites. The values in the boxes are approximations. PNa, plasma sodium concentration; UNa, urine sodium concentration; ![]() , urine flow.
, urine flow.
N35-1
The Proximal-Tubule Reabsorbate Is Slightly Hyperosmotic
Contributed by Emile Boulpaep, Walter Boron
Investigators have found that the lumen of the proximal tubule becomes slightly hypo-osmolar as the reabsorption of salt and water proceeds. Because the ultrafiltrate in Bowman's space is truly isosmotic with blood plasma, the reabsorbate (i.e., the fluid that the tubule reabsorbed) must be slightly hyperosmolar. Once the tubule has reabsorbed a slightly hyperosmolar solution, it has established an osmotic gradient along which water can flow by osmosis. The osmotic water permeability (Pf) of the proximal tubule is so high—owing to the extremely high expression of the water channel AQP1 in both the apical and basolateral membranes—that the H2O very nearly keeps up, so to speak, with the solutes, in an osmotic sense. A mathematical way of stating this fact is that the ratio of the NaCl reabsorptive flux (JNaCl) to the reabsorptive volume or H2O flux (JV) is nearly the same as the osmolality of the lumen:
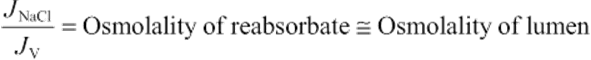 (NE 35-1)
(NE 35-1)
In mice lacking AQP1, the water permeability of the proximal tubule is so low that the diffusion of H2O can no longer keep up with the active reabsorption of NaCl, NaHCO3, and other solutes. As a result, the reabsorbate can become markedly hyperosmolar, and the fluid left behind becomes markedly hypo-osmolar. This situation is reminiscent of that in the TAL, which is sometimes called the diluting segment.
References
Schnermann J, Chou CL, Ma T, et al. Defective proximal tubular fluid reabsorption in transgenic aquaporin-1 null mice. Proc Natl Acad Sci U S A. 1998;95:9660–9664.
Vallon V, Verkman AS, Schnermann J. Luminal hypotonicity in proximal tubules of aquaporin-1-knockout mice. Am J Physiol Renal Physiol. 2000;278:F1030–F1033.
The loop of Henle reabsorbs a smaller but significant fraction of filtered Na+ (~25%). Because of the low water permeability of the thick ascending limb (TAL), this nephron segment reabsorbs Na+ faster than it reabsorbs water, so that [Na+] in the tubule fluid entering the distal convoluted tubule has decreased substantially (TFNa/PNa ≅ 0.45).
The classic distal tubule (see p. 729) and collecting ducts reabsorb smaller fractions of filtered Na+ and water than do more proximal segments. The segments between the distal convoluted tubule (DCT) and the cortical collecting tubule (CCT), inclusive, reabsorb ~5% of the filtered Na+ load under normal conditions. Finally, the medullary collecting duct reabsorbs ~3% of the filtered Na+ load. Although the distal nephron reabsorbs only small amounts of Na+, it can establish a steep transepithelial concentration gradient and can respond to several hormones, including mineralocorticoids and arginine vasopressin (AVP).
As discussed on pages 763–769, most nephron segments reabsorb greater amounts of Na+ when increased quantities of Na+ are delivered due to increased filtered load or inhibition of upstream Na+reabsorption.
The tubule reabsorbs Na+ via both the transcellular and the paracellular pathways
The tubule can reabsorb Na+ and Cl− via both transcellular and paracellular pathways (Fig. 35-3A). In the transcellular pathway, Na+ and Cl− sequentially traverse the apical and basolateral membranes before entering the blood. In the paracellular pathway, these ions move entirely by an extracellular route, through the tight junctions between cells. In the transcellular pathway, transport rates depend on the electrochemical gradients, ion channels, and transporters at the apical and basolateral membranes. However, in the paracellular pathway, transepithelial electrochemical driving forces and permeability properties of the tight junctions govern ion movements.
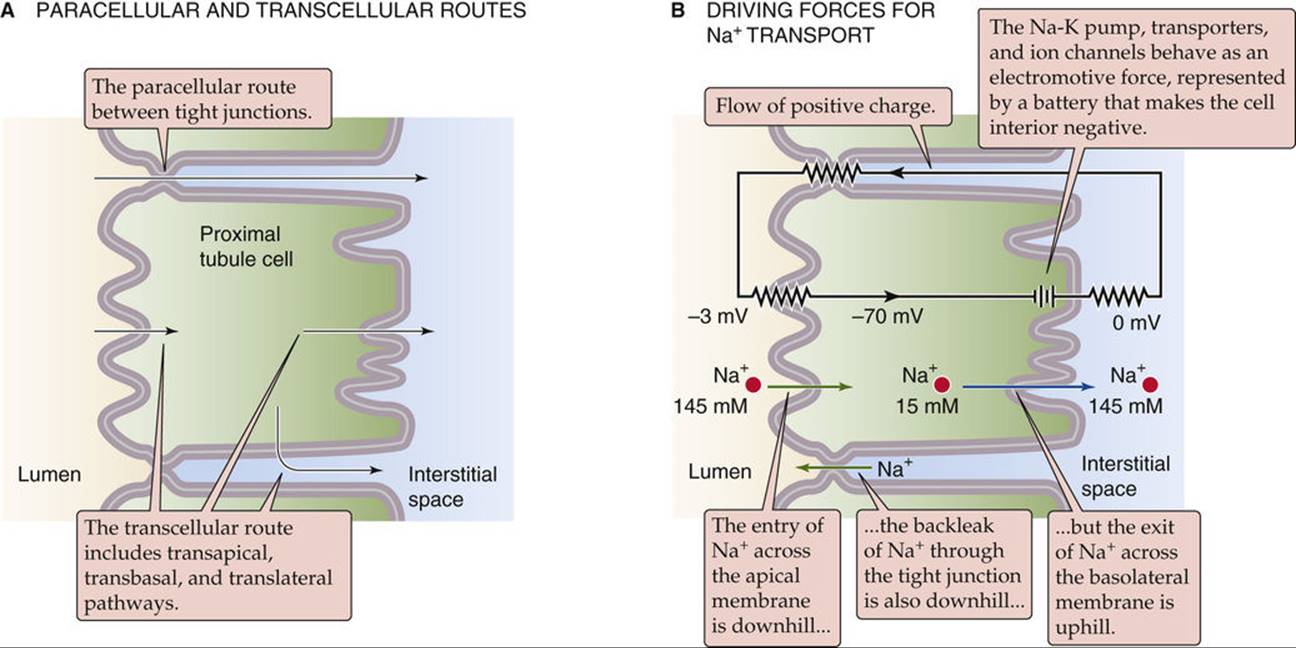
FIGURE 35-3 Transcellular and paracellular mechanisms of Na+ and Cl− reabsorption. The example in B illustrates the electrochemical driving forces for Na+ in the early proximal tubule. The equivalent circuit demonstrates that the flow of positive charge across the apical membrane slightly depolarizes the apical membrane (−67 mV) relative to the basolateral membrane (−70 mV).
Transcellular Na+ Reabsorption
The basic mechanism of transcellular Na+ reabsorption is similar in all nephron segments and is a variation on the classic two-membrane model of epithelial transport (see pp. 137–138). The first step is the passive entry of Na+ into the cell across the apical membrane. Because the intracellular Na+ concentration ([Na+]i) is low and the cell voltage is negative with respect to the lumen, the electrochemical gradient is favorable for passive Na+ entry across the apical membrane (see Fig. 35-3B). However, different tubule segments use different mechanisms of passive Na+ entry across the apical membrane. The proximal tubule, the TAL, and the DCT all use a combination of Na+-coupled cotransporters and exchangers to move Na+ across the apical membrane; however, in the cortical and medullary collecting ducts, Na+ enters the cell through epithelial Na+ channels (ENaCs).
The second step of transcellular Na+ reabsorption is the active extrusion of Na+ out of the cell across the basolateral membrane (see Fig. 35-3B). This Na+ extrusion is mediated by the Na-K pump (see pp. 115–117), which keeps [Na+]i low (~15 mM) and [K+]i high (~120 mM). Because the basolateral membrane is primarily permeable to K+, it develops a voltage of ~70 mV, with the cell interior negative with respect to the interstitial space. Across the apical membrane, the cell is negative with respect to the lumen. The magnitude of the apical membrane voltage may be either lower or higher than that of the basolateral membrane, depending on the nephron segment and its transport activity.
Paracellular Na+ Reabsorption
The basic mechanism of paracellular Na+ transport is similar among nephron segments: the transepithelial electrochemical gradient for Na+ drives transport. However, both the transepithelial voltage (Vte) and luminal [Na+] vary along the nephron (Table 35-1). As a result, the net driving force for Na+ is positive—favoring passive Na+ reabsorption—only in the S2 and S3 segments of the proximal tubule and in the TAL. In the other segments, the net driving force is negative—favoring passive Na+ diffusion from blood to lumen (“backleak”). In addition to undergoing purely passive, paracellular reabsorption in the S2 and S3 segments and TAL, Na+ can move uphill from lumen to blood via solvent drag across the tight junctions. In this case, the movement of H2O from the lumen to the lateral intercellular space—energized by the active transport of Na+ into the lateral intercellular space—also sweeps Na+ and Cl− in the same direction.
TABLE 35-1
Transepithelial Driving Forces for Na+
|
LUMINAL [Na+] |
TRANSEPITHELIAL CHEMICAL DRIVING FORCE*† |
TRANSEPITHELIAL VOLTAGE (ELECTRICAL DRIVING FORCE)†‡ |
TRANSEPITHELIAL ELECTROCHEMICAL DRIVING FORCE† |
|
|
Proximal tubule, S1 |
142 mM |
0 mV |
−3 mV |
−3 mV |
|
Proximal tubule, S3 |
142 mM |
0 mV |
+3 mV |
+3 mV |
|
TAL |
100 mM |
−9 mV |
+15 mV |
+6 mV |
|
DCT |
70 mM |
−19 mV |
−5 to +5 mV |
−24 to −14 mV |
|
CCT |
40 mM |
−34 mV |
−40 mV |
−74 mV |
*The chemical driving force is calculated assuming a plasma [Na+] of 142 mM and is given in mV.
†A negative value promotes passive Na+ movement from blood to lumen (i.e., backleak or secretion), whereas a positive value promotes passive Na+ movement from lumen to blood (i.e., reabsorption).
‡A negative value indicates that the lumen is negative with respect to the blood.
Nephron segments also vary in their leakiness to Na+ ions. This leakiness is largely a function of the varying ionic conductance of the paracellular pathway between cells across the tight junction, due to the expression of different claudins. In general, the leakiness of the paracellular pathway decreases along the nephron from the proximal tubule (the most leaky) to the papillary collecting ducts. However, even the tightest renal epithelia have what might be regarded as only a moderate degree of tightness compared with truly “tight” epithelia, such as in those the skin, the gastric mucosa, and the urinary bladder (see pp. 136–137).
The leakiness of an epithelium has serious repercussions for the steepness of the ion gradients that the epithelium can develop and maintain. For both Na+ and Cl−, the ability of specific nephron segments to establish large concentration gradients correlates with the degree of tightness, which limits the backflux of ions between cells. Thus, the luminal fluid attains much lower concentrations of Na+ and Cl− in the distal nephron than it does in the proximal tubule.
An important consequence of a highly leaky paracellular pathway is that it provides a mechanism by which the basolateral membrane voltage can generate a current that flows through the tight junctions and charges up the apical membrane, and vice versa (see Fig. 35-3B). For example, hyperpolarization of the basolateral membrane leads to a hyperpolarization of the apical membrane. A consequence of this paracellular electrical coupling is that the apical membrane of a leaky epithelium, such as the proximal tubule, has a membrane voltage that is negative (−67 mV in Fig. 35-3B) and close to that of the basolateral membrane (−70 mV in Fig. 35-3B), whereas one would expect that, based on the complement of channels and ion gradients at the apical membrane, the apical membrane would have a far less negative voltage. A practical benefit of this crosstalk is that it helps couple the activity of the basolateral electrogenic Na-K pump to the passive entry of Na+ across the apical membrane. If the Na-K pump rate increases, not only does [Na+]i decrease, enhancing the chemical Na+ gradient across the apical membrane, but also the basolateral membrane hyperpolarizes (i.e., the cell becomes more negative with respect to the blood). Electrical coupling translates this basolateral hyperpolarization to a concomitant apical hyperpolarization, thus also enhancing the electrical gradient favoring apical Na+ entry.