OBJECTIVES
After studying this chapter, you should be able to:
![]() Describe the elements of the stretch reflex and how the activity of γ-motor neurons alters the response to muscle stretch.
Describe the elements of the stretch reflex and how the activity of γ-motor neurons alters the response to muscle stretch.
![]() Describe the role of Golgi tendon organs in control of skeletal muscle.
Describe the role of Golgi tendon organs in control of skeletal muscle.
![]() Describe the elements of the withdrawal reflex.
Describe the elements of the withdrawal reflex.
![]() Define spinal shock and describe the initial and long-term changes in spinal reflexes that follow transection of the spinal cord.
Define spinal shock and describe the initial and long-term changes in spinal reflexes that follow transection of the spinal cord.
![]() Describe how skilled movements are planned and carried out.
Describe how skilled movements are planned and carried out.
![]() Compare the organization of the central pathways involved in the control of axial (posture) and distal (skilled movement, fine motor movements) muscles.
Compare the organization of the central pathways involved in the control of axial (posture) and distal (skilled movement, fine motor movements) muscles.
![]() Define decerebrate and decorticate rigidity, and comment on the cause and physiologic significance of each.
Define decerebrate and decorticate rigidity, and comment on the cause and physiologic significance of each.
![]() Identify the components of the basal ganglia and the pathways that interconnect them, along with the neurotransmitters in each pathway.
Identify the components of the basal ganglia and the pathways that interconnect them, along with the neurotransmitters in each pathway.
![]() Explain the pathophysiology and symptoms of Parkinson disease and Huntington disease.
Explain the pathophysiology and symptoms of Parkinson disease and Huntington disease.
![]() Discuss the functions of the cerebellum and the neurologic abnormalities produced by diseases of this part of the brain.
Discuss the functions of the cerebellum and the neurologic abnormalities produced by diseases of this part of the brain.
INTRODUCTION
Somatic motor activity depends ultimately on the pattern and rate of discharge of the spinal motor neurons and homologous neurons in the motor nuclei of the cranial nerves. These neurons, the final common paths to skeletal muscle, are bombarded by impulses from an immense array of descending pathways, other spinal neurons, and peripheral afferents. Some of these inputs end directly on α-motor neurons, but many exert their effects via interneurons or via γ-motor neurons to the muscle spindles and back through the Ia afferent fibers to the spinal cord. It is the integrated activity of these multiple inputs from spinal, medullary, midbrain, and cortical levels that regulates the posture of the body and makes coordinated movement possible.
The inputs converging on motor neurons have three functions: they bring about voluntary activity, they adjust body posture to provide a stable background for movement, and they coordinate the action of the various muscles to make movements smooth and precise. The patterns of voluntary activity are planned within the brain, and the commands are sent to the muscles primarily via the corticospinal and corticobulbar systems. Posture is continually adjusted not only before but also during movement by information carried in descending brain stem pathways and peripheral afferents. Movement is smoothed and coordinated by the medial and intermediate portions of the cerebellum (spinocerebellum) and its connections. The basal ganglia and the lateral portions of the cerebellum (cerebrocerebellum) are part of a feedback circuit to the premotor and motor cortex that is concerned with planning and organizing voluntary movement.
This chapter considers two types of motor output: reflex (involuntary) and voluntary. A subdivision of reflex responses includes some rhythmic movements such as swallowing, chewing, scratching, and walking, which are largely involuntary but subject to voluntary adjustment and control.
GENERAL PROPERTIES OF REFLEXES
The basic unit of integrated reflex activity is the reflex arc. This arc consists of a sense organ, an afferent neuron, one or more synapses within a central integrating station, an efferent neuron, and an effector. The afferent neurons enter via the dorsal roots or cranial nerves and have their cell bodies in the dorsal root ganglia or in the homologous ganglia of the cranial nerves. The efferent fibers leave via the ventral roots or corresponding motor cranial nerves.
Activity in the reflex arc starts in a sensory receptor with a receptor potential whose magnitude is proportional to the strength of the stimulus (Figure 12–1). This generates all-or-none action potentials in the afferent nerve, the number of action potentials being proportional to the size of the receptor potential. In the central nervous system (CNS), the responses are again graded in terms of excitatory postsynaptic potentials (EPSPs) and inhibitory postsynaptic potentials (IPSPs) at the synaptic junctions. All-or-none responses (action potentials) are generated in the efferent nerve. When these reach the effector, they again set up a graded response. When the effector is smooth muscle, responses summate to produce action potentials in the smooth muscle, but when the effector is skeletal muscle, the graded response is adequate to produce action potentials that bring about muscle contraction. The connection between the afferent and efferent neurons is in the CNS, and activity in the reflex arc is modified by the multiple inputs converging on the efferent neurons or at any synaptic station within the reflex arc.
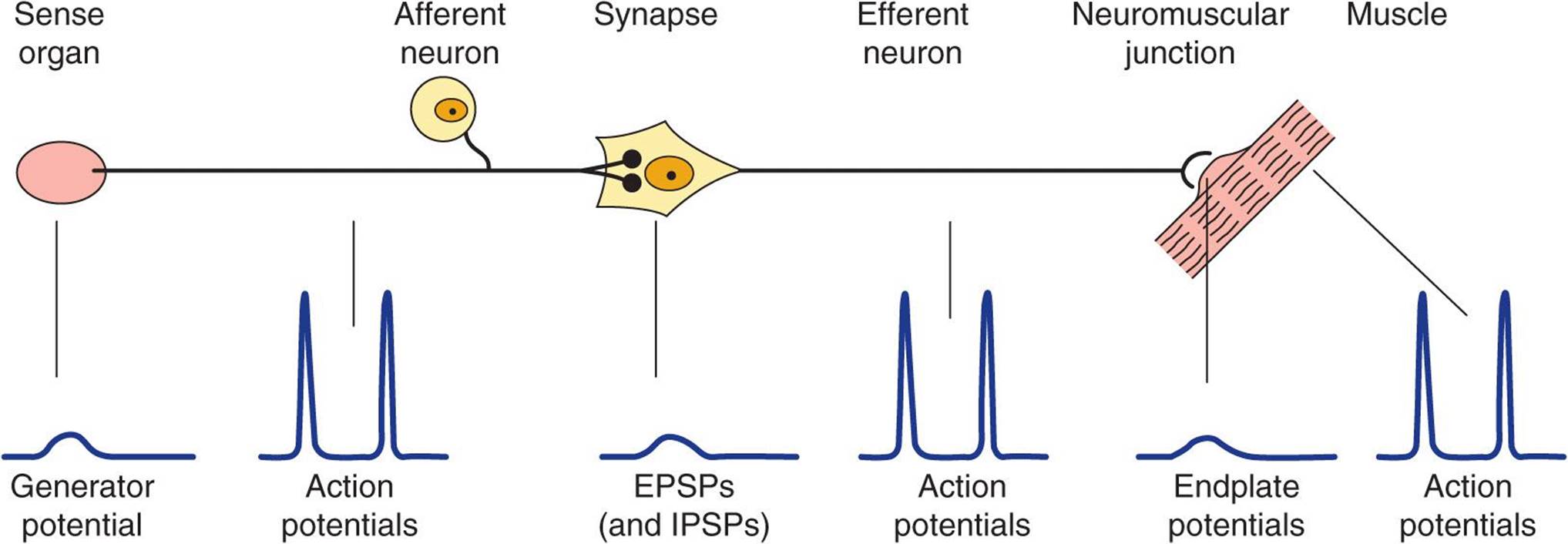
FIGURE 12–1 The reflex arc. Note that at the receptor and in the CNS a nonpropagated graded response occurs that is proportional to the magnitude of the stimulus. The response at the neuromuscular junction is also graded, though under normal conditions it is always large enough to produce a response in skeletal muscle. On the other hand, in the portions of the arc specialized for transmission (afferent and efferent nerve fibers, muscle membrane), muscle membrane), the responses are all-or-none action potentials.
The stimulus that triggers a reflex is generally very precise. This stimulus is called the adequate stimulus for the particular reflex. A dramatic example is the scratch reflex in the dog. This spinal reflex is adequately stimulated by multiple linear touch stimuli such as those produced by an insect crawling across the skin. The response is vigorous scratching of the area stimulated. If the multiple touch stimuli are widely separated or not in a line, the adequate stimulus is not produced and no scratching occurs. Fleas crawl, but they also jump from place to place. This jumping separates the touch stimuli so that an adequate stimulus for the scratch reflex is not produced.
Reflex activity is stereotyped and specific in that a particular stimulus elicits a particular response. The fact that reflex responses are stereotyped does not exclude the possibility of their being modified by experience. Reflexes are adaptable and can be modified to perform motor tasks and maintain balance. Descending inputs from higher brain regions play an important role in modulating and adapting spinal reflexes.
The α-motor neurons that supply the extrafusal fibers in skeletal muscles are the efferent side of many reflex arcs. All neural influences affecting muscular contraction ultimately funnel through them to the muscles, and they are therefore called the final common pathway. Numerous inputs converge on α-motor neurons. Indeed, the surface of the average motor neuron and its dendrites accommodates about 10,000 synaptic knobs. At least five inputs go from the same spinal segment to a typical spinal motor neuron. In addition to these, there are excitatory and inhibitory inputs, generally relayed via interneurons, from other levels of the spinal cord and multiple long-descending tracts from the brain. All of these pathways converge on and determine the activity in the final common pathways.
MONOSYNAPTIC REFLEXES: THE STRETCH REFLEX
The simplest reflex arc is one with a single synapse between the afferent and efferent neurons, and reflexes occurring in them are called monosynaptic reflexes. Reflex arcs in which interneurons are interposed between the afferent and efferent neurons are called polysynaptic reflexes. There can be anywhere from two to hundreds of synapses in a polysynaptic reflex arc.
When a skeletal muscle with an intact nerve supply is stretched, it contracts. This response is called the stretch reflex or myotatic reflex. The stimulus that initiates this reflex is stretch of the muscle, and the response is contraction of the muscle being stretched. The sense organ is a small encapsulated spindlelike or fusiform shaped structure called the muscle spindle, located within the fleshy part of the muscle. The impulses originating from the spindle are transmitted to the CNS by fast sensory fibers that pass directly to the motor neurons that supply the same muscle. The neurotransmitter at the central synapse is glutamate. The stretch reflex is the best known and studied monosynaptic reflex and is typified by the knee jerk reflex (see Clinical Box 12–1).
CLINICAL BOX 12–1
Knee Jerk Reflex
Tapping the patellar tendon elicits the knee jerk, a stretch reflex of the quadriceps femoris muscle, because the tap on the tendon stretches the muscle. A similar contraction is observed if the quadriceps is stretched manually. Stretch reflexes can also be elicited from most of the large muscles of the body. Tapping on the tendon of the triceps brachii, for example, causes an extensor response at the elbow as a result of reflex contraction of the triceps; tapping on the Achilles tendon causes an ankle jerk due to reflex contraction of the gastrocnemius; and tapping on the side of the face causes a stretch reflex in the masseter. The knee jerk reflex is an example of a deep tendon reflex (DTR) in a neurological exam and is graded on the following scale: 0 (absent), 1 + (hypoactive), 2+ (brisk, normal), 3+ (hyperactive without clonus), 4+ (hyperactive with mild clonus), and 5+ (hyperactive with sustained clonus). Absence of the knee jerk can signify an abnormality anywhere within the reflex arc, including the muscle spindle, the Ia afferent nerve fibers, or the motor neurons to the quadriceps muscle. The most common cause is a peripheral neuropathy from such things as diabetes, alcoholism, and toxins. A hyperactive reflex can signify an interruption of corticospinal and other descending pathways that influence the reflex arc.
STRUCTURE OF MUSCLE SPINDLES
Each muscle spindle has three essential elements: (1) a group of specialized intrafusal muscle fibers with contractile polar ends and a noncontractile center, (2) large diameter myelinated afferent nerves (types Ia and II) originating in the central portion of the intrafusal fibers, and (3) small diameter myelinated efferent nerves supplying the polar contractile regions of the intrafusal fibers (Figure 12–2A). It is important to understand the relationship of these elements to each other and to the muscle itself to appreciate the role of this sense organ in signaling changes in the length of the muscle in which it is located. Changes in muscle length are associated with changes in joint angle; thus muscle spindles provide information on position (ie, proprioception).
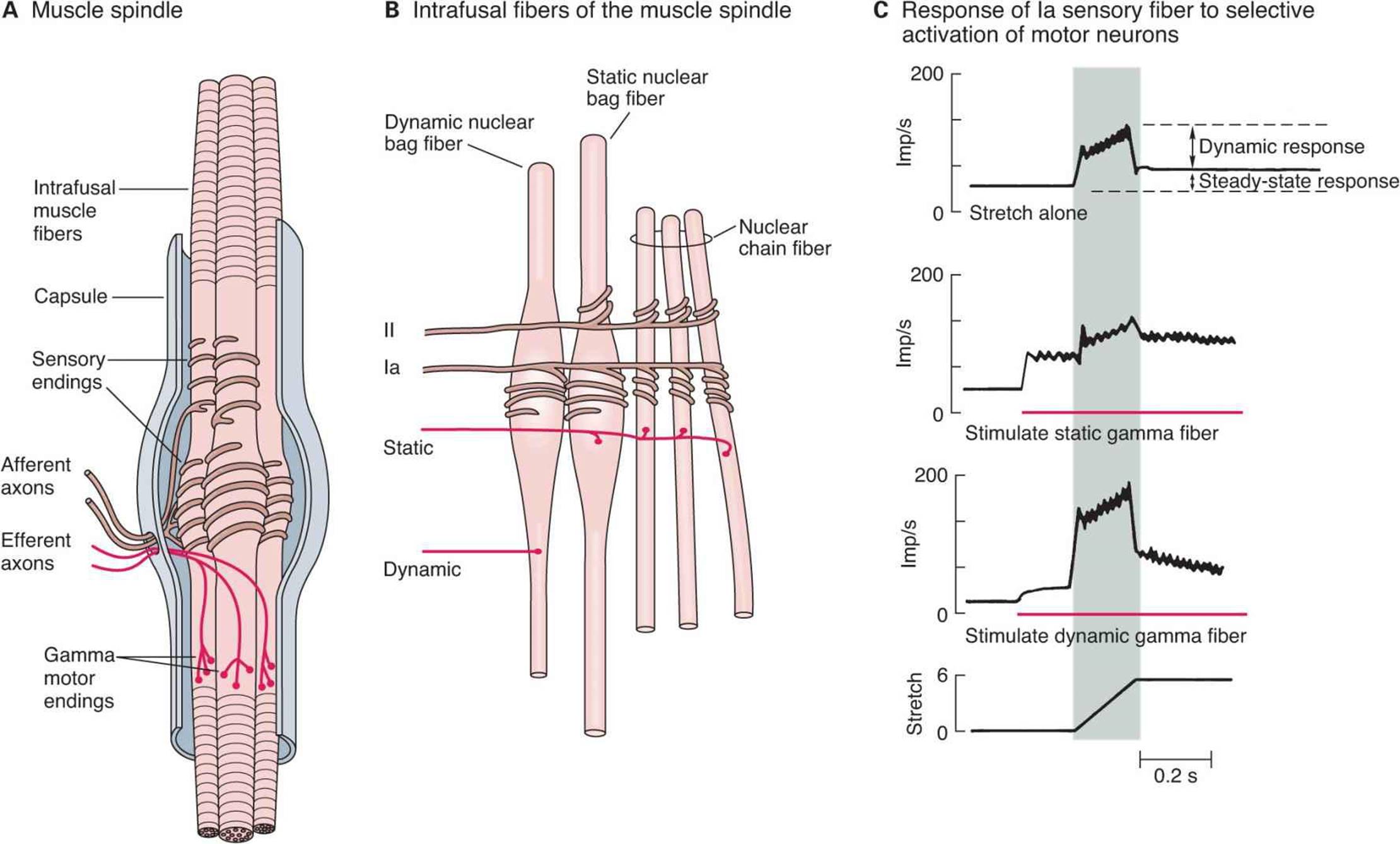
FIGURE 12–2 Mammalian muscle spindle. A) Diagrammatic representation of the main components of mammalian muscle spindle including intrafusal muscle fibers, afferent sensory fiber endings, and efferent motor fibers (γ-motor neurons). B) Three types of intrafusal muscle fibers: dynamic nuclear bag, static nuclear bag, and nuclear chain fibers. A single Ia afferent fiber innervates all three types of fibers to form a primary sensory ending. A group II sensory fiber innervates nuclear chain and static bag fibers to form a secondary sensory ending. Dynamic γ-motor neurons innervate dynamic bag fibers; static γ-motor neurons innervate combinations of chain and static bag fibers. C) Comparison of discharge pattern of Ia afferent activity during stretch alone and during stimulation of static or dynamic γ-motor neurons. Without γ-stimulation, Ia fibers show a small dynamic response to muscle stretch and a modest increase in steady-state firing. When static γ-motor neurons are activated, the steady-state response increases and the dynamic response decreases. When dynamic γ-motor neurons are activated, the dynamic response is markedly increased but the steady-state response gradually returns to its original level. (Adapted with permission from Brown MC, Matthews PBC: On the sub-division of the efferent fibres to muscle spindles into static and dynamic fusimotor fibres. In Andrew BL (editor): Control and Innervation of Skeletal Muscle, pp 18-31. Dundee, Scotland: University of St. Andrews, 1966.)
The intrafusal fibers are positioned in parallel to the extrafusal fibers (the regular contractile units of the muscle) with the ends of the spindle capsule attached to the tendons at either end of the muscle. Intrafusal fibers do not contribute to the overall contractile force of the muscle, but rather serve a pure sensory function. There are two types of intrafusal fibers in mammalian muscle spindles. The first type contains many nuclei in a dilated central area and is called a nuclear bag fiber (Figure 12–2B). There are two subtypes of nuclear bag fibers, dynamic and static. The second intrafusal fiber type, the nuclear chain fiber, is thinner and shorter and lacks a definite bag. Typically, each muscle spindle contains two or three nuclear bag fibers and about five nuclear chain fibers.
There are two kinds of sensory endings in each spindle, a single primary (group Ia) ending and up to eight secondary (group II) endings (Figure 12-2B). The Ia afferent fiber wraps around the center of the dynamic and static nuclear bag fibers and nuclear chain fibers. Group II sensory fibers are located adjacent to the centers of the static nuclear bag and nuclear chain fibers; these fibers do not innervate the dynamic nuclear bag fibers. Ia afferents are very sensitive to the velocity of the change in muscle length during a stretch (dynamic response); thus they provide information about the speed of movements and allow for quick corrective movements. The steady-state (tonic) activity of group Ia and II afferents provide information on steady-state length of the muscle (static response). The top trace in Figure 12–2C shows the dynamic and static components of activity in a Ia afferent during muscle stretch. Note that they discharge most rapidly while the muscle is being stretched (shaded area of graphs) and less rapidly during sustained stretch.
The spindles have a motor nerve supply of their own. These nerves are 3–6 μm in diameter, constitute about 30% of the fibers in the ventral roots, and are called γ-motor neurons. There are two types of γ-motor neurons: dynamic, which supply the dynamic nuclear bag fibers and static, which supply the static nuclear bag fibers and the nuclear chain fibers. Activation of dynamic γ-motor neurons increases the dynamic sensitivity of the group Ia endings. Activation of the static γ-motor neurons increases the tonic level of activity in both group Ia and II endings, decreases the dynamic sensitivity of group Ia afferents, and can prevent silencing of Ia afferents during muscle stretch (Figure 12–2C).
CENTRAL CONNECTIONS OF AFFERENT FIBERS
Ia fibers end directly on motor neurons supplying the extrafusal fibers of the same muscle (Figure 12–3). The time between the application of the stimulus and the response is called the reaction time. In humans, the reaction time for a stretch reflex such as the knee jerk is 19–24 ms. Weak stimulation of the sensory nerve from the muscle, known to stimulate only Ia fibers, causes a contractile response with a similar latency. Because the conduction velocities of the afferent and efferent fiber types are known and the distance from the muscle to the spinal cord can be measured, it is possible to calculate how much of the reaction time was taken up by conduction to and from the spinal cord. When this value is subtracted from the reaction time, the remainder, called the central delay, is the time taken for the reflex activity to traverse the spinal cord. The central delay for the knee jerk reflex is 0.6–0.9 ms. Because the minimum synaptic delay is 0.5 ms, only one synapse could have been traversed.
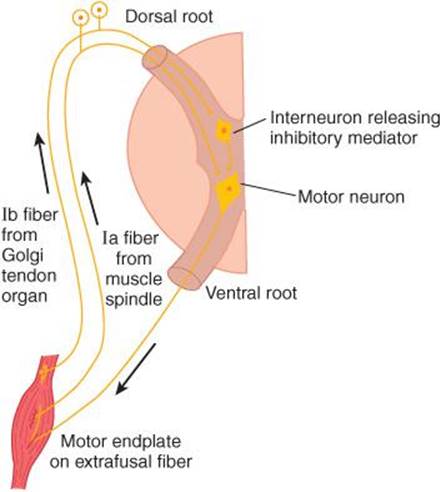
FIGURE 12–3 Diagram illustrating the pathways responsible for the stretch reflex and the inverse stretch reflex. Stretch stimulates the muscle spindle, which activates Ia fibers that excite the motor neuron. Stretch also stimulates the Golgi tendon organ, which activates Ib fibers that excite an interneuron that releases the inhibitory mediator glycine. With strong stretch, the resulting hyperpolarization of the motor neuron is so great that it stops discharging.
FUNCTION OF MUSCLE SPINDLES
When the muscle spindle is stretched, its sensory endings are distorted and receptor potentials are generated. These in turn set up action potentials in the sensory fibers at a frequency proportional to the degree of stretching. Because the spindle is in parallel with the extrafusal fibers, when the muscle is passively stretched, the spindles are also stretched, referred to as “loading the spindle.” This initiates reflex contraction of the extrafusal fibers in the muscle. On the other hand, the spindle afferents characteristically stop firing when the muscle is made to contract by electrical stimulation of the α-motor neurons to the extrafusal fibers because the muscle shortens while the spindle is unloaded (Figure 12–4).
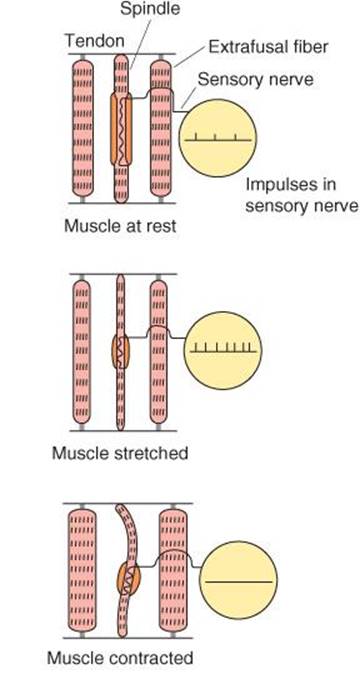
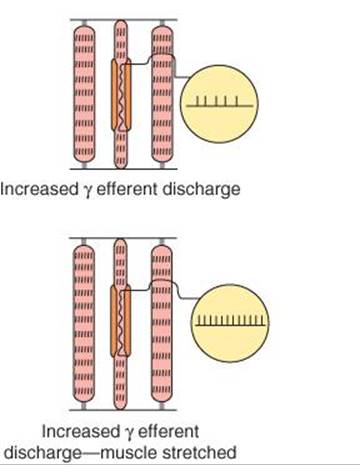
FIGURE 12–4 Effect of various conditions on muscle spindle discharge. When the whole muscle is stretched, the muscle spindle is also stretched and its sensory endings are activated at a frequency proportional to the degree of stretching (“loading the spindle”). Spindle afferents stop firing when the muscle contracts (“unloading the spindle”). Stimulation of γ-motor neurons cause the contractile ends of the intrafusal fibers to shorten. This stretches the nuclear bag region, initiating impulses in sensory fibers. If the whole muscle is stretched during stimulation of the γ-motor neurons, the rate of discharge in sensory fibers is further increased.
The muscle spindle and its reflex connections constitute a feedback device that operates to maintain muscle length. If the muscle is stretched, spindle discharge increases and reflex shortening is produced. If the muscle is shortened without a change in γ-motor neuron discharge, spindle afferent activity decreases and the muscle relaxes.
Dynamic and static responses of muscle spindle afferents influence physiological tremor. The response of the Ia sensory fiber endings to the dynamic (phasic) as well as the static events in the muscle is important because the prompt, marked phasic response helps to dampen oscillations caused by conduction delays in the feedback loop regulating muscle length. Normally a small oscillation occurs in this feedback loop. This physiologic tremor has low amplitude (barely visible to the naked eye) and a frequency of approximately 10 Hz. Physiological tremor is a normal phenomenon which affects everyone while maintaining posture or during movements. However, the tremor would be more prominent if it were not for the sensitivity of the spindle to velocity of stretch. It can become exaggerated in some situations such as when we are anxious or tired or because of drug toxicity. Numerous factors contribute to the genesis of physiological tremor. It is likely dependent on not only central (inferior olive) sources but also peripheral factors including motor unit firing rates, reflexes, and mechanical resonance.
EFFECTS OF γ-MOTOR NEURON DISCHARGE
Stimulation of γ-motor neurons produces a very different picture from that produced by stimulation of the α-motor neurons. Stimulation of γ-motor neurons does not lead directly to detectable contraction of the muscles because the intrafusal fibers are not strong enough or plentiful enough to cause shortening. However, stimulation does cause the contractile ends of the intrafusal fibers to shorten and therefore stretches the nuclear bag portion of the spindles, deforming the endings, and initiating impulses in the Ia fibers (Figure 12–4). This in turn can lead to reflex contraction of the muscle. Thus, muscles can be made to contract via stimulation of the α-motor neurons that innervate the extrafusal fibers or the γ-motor neurons that initiate contraction indirectly via the stretch reflex.
If the whole muscle is stretched during stimulation of the γ-motor neurons, the rate of discharge in the Ia fibers is further increased (Figure 12–4). Increased γ-motor neuron activity thus increases spindle sensitivity during stretch.
In response to descending excitatory input to spinal motor circuits, both α- and γ-motor neurons are activated. Because of this “α–γ coactivation,” intrafusal and extrafusal fibers shorten together, and spindle afferent activity can occur throughout the period of muscle contraction. In this way, the spindle remains capable of responding to stretch and reflexively adjusting α-motor neuron discharge.
CONTROL OF γ-MOTOR NEURON DISCHARGE
The γ-motor neurons are regulated to a large degree by descending tracts from a number of areas in the brain that also control α-motor neurons (described below). Via these pathways, the sensitivity of the muscle spindles and hence the threshold of the stretch reflexes in various parts of the body can be adjusted and shifted to meet the needs of postural control.
Other factors also influence γ-motor neuron discharge. Anxiety causes an increased discharge, a fact that probably explains the hyperactive tendon reflexes sometimes seen in anxious patients. In addition, unexpected movement is associated with a greater efferent discharge. Stimulation of the skin, especially by noxious agents, increases γ-motor neuron discharge to ipsilateral flexor muscle spindles while decreasing that to extensors and produces the opposite pattern in the opposite limb. It is well known that trying to pull the hands apart when the flexed fingers are hooked together facilitates the knee jerk reflex (Jendrassik’s maneuver), and this may also be due to increased γ-motor neuron discharge initiated by afferent impulses from the hands.
RECIPROCAL INNERVATION
When a stretch reflex occurs, the muscles that antagonize the action of the muscle involved (antagonists) relax. This phenomenon is said to be due to reciprocal innervation. Impulses in the Ia fibers from the muscle spindles of the protagonist muscle cause postsynaptic inhibition of the motor neurons to the antagonists. The pathway mediating this effect is bisynaptic. A collateral from each Ia fiber passes in the spinal cord to an inhibitory interneuron that synapses on a motor neuron supplying the antagonist muscles. This example of postsynaptic inhibition is discussed in Chapter 6, and the pathway is illustrated in Figure 6–6.
INVERSE STRETCH REFLEX
Up to a point, the harder a muscle is stretched, the stronger is the reflex contraction. However, when the tension becomes great enough, contraction suddenly ceases and the muscle relaxes. This relaxation in response to strong stretch is called the inverse stretch reflex. The receptor for the inverse stretch reflex is in the Golgi tendon organ (Figure 12–5). This organ consists of a netlike collection of knobby nerve endings among the fascicles of a tendon. There are 3–25 muscle fibers per tendon organ. The fibers from the Golgi tendon organs make up the Ib group of myelinated, rapidly conducting sensory nerve fibers. Stimulation of these Ib fibers leads to the production of IPSPs on the motor neurons that supply the muscle from which the fibers arise. The Ib fibers end in the spinal cord on inhibitory interneurons that in turn terminate directly on the motor neurons (Figure 12–3). They also make excitatory connections with motor neurons supplying antagonists to the muscle.
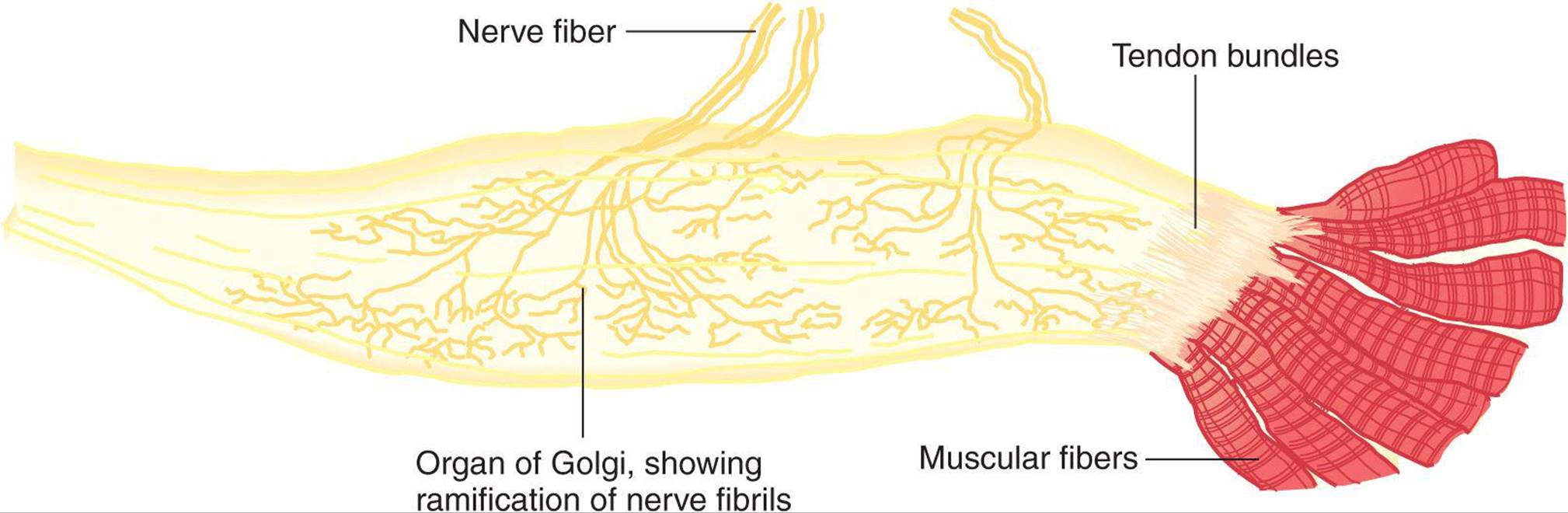
FIGURE 12–5 Golgi tendon organ. This organ is the receptor for the inverse stretch reflex and consists of a netlike collection of knobby nerve endings among the fascicles of a tendon. The innervation is the Ib group of myelinated, rapidly conducting sensory nerve fibers. (Reproduced with permission from Gray H [editor]: Gray’s Anatomy of the Human Body, 29th ed. Lea & Febiger, 1973.)
Because the Golgi tendon organs, unlike the spindles, are in series with the muscle fibers, they are stimulated by both passive stretch and active contraction of the muscle. The threshold of the Golgi tendon organs is low. The degree of stimulation by passive stretch is not great because the more elastic muscle fibers take up much of the stretch, and this is why it takes a strong stretch to produce relaxation. However, discharge is regularly produced by contraction of the muscle, and the Golgi tendon organ thus functions as a transducer in a feedback circuit that regulates muscle force in a fashion analogous to the spindle feedback circuit that regulates muscle length.
The importance of the primary endings in the spindles and the Golgi tendon organs in regulating the velocity of the muscle contraction, muscle length, and muscle force is illustrated by the fact that section of the afferent nerves to an arm causes the limb to hang loosely in a semiparalyzed state. The interaction of spindle discharge, tendon organ discharge, and reciprocal innervation determines the rate of discharge of α-motor neurons (see Clinical Box 12–2).
CLINICAL BOX 12–2
Clonus
A characteristic of states in which increased γ-motor neuron discharge is present is clonus. This neurologic sign is the occurrence of regular, repetitive, rhythmic contractions of a muscle subjected to sudden, maintained stretch. Only sustained clonus with five or more beats is considered abnormal. Ankle clonus is a typical example. This is initiated by brisk, maintained dorsiflexion of the foot, and the response is rhythmic plantar flexion at the ankle. The stretch reflex–inverse stretch reflex sequence may contribute to this response. However, it can occur on the basis of synchronized motor neuron discharge without Golgi tendon organ discharge. The spindles of the tested muscle are hyperactive, and the burst of impulses from them discharges all the motor neurons supplying the muscle at once. The consequent muscle contraction stops spindle discharge. However, the stretch has been maintained, and as soon as the muscle relaxes it is again stretched and the spindles stimulated. Clonus may also occur after disruption of descending cortical input to a spinal glycinergic inhibitory interneuron called the Renshaw cell. This cell receives excitatory input from α-motor neurons via axon collaterals (and in turn it inhibits the same). In addition, cortical fibers activating ankle flexors contact Renshaw cells (as well as type Ia inhibitory interneurons) that inhibit the antagonistic ankle extensors. This circuitry prevents reflex stimulation of the extensors when flexors are active. Therefore, when the descending cortical fibers are damaged (upper motor neuron lesion), the inhibition of antagonists is absent. The result is repetitive, sequential contraction of ankle flexors and extensors (clonus). Clonus may be seen in patients with amyotrophic lateral sclerosis, stroke, multiple sclerosis, spinal cord damage, epilepsy, liver or kidney failure, and hepatic encephalopathy.
THERAPEUTIC HIGHLIGHTS
Since there are numerous causes of clonus, treatment centers on the underlying cause. For some individuals, stretching exercises can reduce episodes of clonus. Immunosuppressants (eg, azathioprine and corticosteroids), anticonvulsants (eg, primidone and levetiracetam), and tranquilizers (eg, clonazepam) have been shown to be beneficial in the treatment of clonus. Botulinum toxin has also been used to block the release of acetylcholine in the muscle, which triggers the rhythmic muscle contractions.
MUSCLE TONE
The resistance of a muscle to stretch is often referred to as its tone or tonus. If the motor nerve to a muscle is severed, the muscle offers very little resistance and is said to be flaccid. A hypertonic (spastic) muscle is one in which the resistance to stretch is high because of hyperactive stretch reflexes. Somewhere between the states of flaccidity and spasticity is the ill-defined area of normal tone. The muscles are generally hypotonic when the rate of γ-motor neuron discharge is low and hypertonic when it is high.
When the muscles are hypertonic, the sequence of moderate stretch → muscle contraction, strong stretch → muscle relaxation is clearly seen. Passive flexion of the elbow, for example, meets immediate resistance as a result of the stretch reflex in the triceps muscle. Further stretch activates the inverse stretch reflex. The resistance to flexion suddenly collapses, and the arm flexes. Continued passive flexion stretches the muscle again, and the sequence may be repeated. This sequence of resistance followed by give when a limb is moved passively is known as the clasp-knife effect because of its resemblance to the closing of a pocket knife. It is also known as the lengthening reactionbecause it is the response of a spastic muscle to lengthening.
POLYSYNAPTIC REFLEXES: THE WITHDRAWAL REFLEX
Polysynaptic reflex paths branch in a complex fashion (Figure 12–6). The number of synapses in each of their branches varies. Because of the synaptic delay at each synapse, activity in the branches with fewer synapses reaches the motor neurons first, followed by activity in the longer pathways. This causes prolonged bombardment of the motor neurons from a single stimulus and consequently prolonged responses. Furthermore, some of the branch pathways turn back on themselves, permitting activity to reverberate until it becomes unable to cause a propagated transsynaptic response and dies out. Such reverberating circuits are common in the brain and spinal cord.
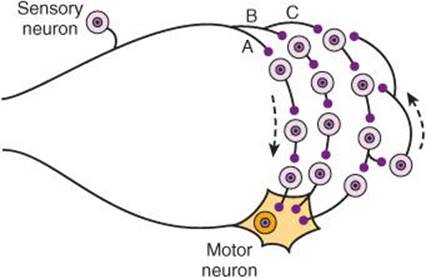
FIGURE 12–6 Diagram of polysynaptic connections between afferent and efferent neurons in the spinal cord. The dorsal root fiber activates pathway A with three interneurons, pathway B with four interneurons, and pathway C with four interneurons. Note that one of the interneurons in pathway C connects to a neuron that doubles back to other interneurons, forming reverberating circuits.
The withdrawal reflex is a typical polysynaptic reflex that occurs in response to a noxious stimulus to the skin or subcutaneous tissues and muscle. The response is flexor muscle contraction and inhibition of extensor muscles, so that the body part stimulated is flexed and withdrawn from the stimulus. When a strong stimulus is applied to a limb, the response includes not only flexion and withdrawal of that limb but also extension of the opposite limb. This crossed extensor response is properly part of the withdrawal reflex. Strong stimuli can generate activity in the interneuron pool that spreads to all four extremities. This spread of excitatory impulses up and down the spinal cord to more and more motor neurons is called irradiation of the stimulus, and the increase in the number of active motor units is called recruitment of motor units.
IMPORTANCE OF THE WITHDRAWAL REFLEX
Flexor responses can be produced by innocuous stimulation of the skin or by stretch of the muscle, but strong flexor responses with withdrawal are initiated only by stimuli that are noxious or at least potentially harmful (ie, nociceptive stimuli). The withdrawal reflex serves a protective function as flexion of the stimulated limb gets it away from the source of irritation, and extension of the other limb supports the body. The pattern assumed by all four extremities puts one in position to escape from the offending stimulus. Withdrawal reflexes are prepotent; that is, they preempt the spinal pathways from any other reflex activity taking place at the moment.
Many of the characteristics of polysynaptic reflexes can be demonstrated by studying the withdrawal reflex. A weak noxious stimulus to one foot evokes a minimal flexion response; stronger stimuli produce greater and greater flexion as the stimulus irradiates to more and more of the motor neuron pool supplying the muscles of the limb. Stronger stimuli also cause a more prolonged response. A weak stimulus causes one quick flexion movement; a strong stimulus causes prolonged flexion and sometimes a series of flexion movements. This prolonged response is due to prolonged, repeated firing of the motor neurons. The repeated firing is called after-discharge and is due to continued bombardment of motor neurons by impulses arriving by complicated and circuitous polysynaptic paths.
As the strength of a noxious stimulus is increased, the reaction time is shortened. Spatial and temporal facilitation occurs at synapses in the polysynaptic pathway. Stronger stimuli produce more action potentials per second in the active branches and cause more branches to become active; summation of the EPSPs to the threshold level for action potential generation occurs more rapidly.
FRACTIONATION & OCCLUSION
Another characteristic of the withdrawal response is the fact that supramaximal stimulation of any of the sensory nerves from a limb never produces as strong a contraction of the flexor muscles as that elicited by direct electrical stimulation of the muscles themselves. This indicates that the afferent inputs fractionate the motor neuron pool; that is, each input goes to only part of the motor neuron pool for the flexors of that particular extremity. On the other hand, if all the sensory inputs are dissected out and stimulated one after the other, the sum of the tension developed by stimulation of each is greater than that produced by direct electrical stimulation of the muscle or stimulation of all inputs at once. This indicates that the various afferent inputs share some of the motor neurons and that occlusion occurs when all inputs are stimulated at once.
SPINAL INTEGRATION OF REFLEXES
The responses of animals and humans to spinal cord injury (SCI) illustrate the integration of reflexes at the spinal level. The deficits seen after SCI vary, of course, depending on the level of the injury. Clinical Box 12–3 provides information on long-term problems related to SCI and recent advancements in treatment options.
CLINICAL BOX 12–3
Spinal Cord Injury
It has been estimated that the worldwide annual incidence of sustaining spinal cord injury (SCI) is between 10 and 83 per million of the population. Leading causes are vehicle accidents, violence, and sports injuries. The mean age of patients who sustain an SCI is 33 years old, and men outnumber women with a nearly 4:1 ratio. Approximately 52% of SCI cases result in quadriplegia and about 42% lead to paraplegia. In quadriplegic humans, the threshold of the withdrawal reflex is very low; even minor noxious stimuli may cause not only prolonged withdrawal of one extremity but marked flexion–extension patterns in the other three limbs. Stretch reflexes are also hyperactive. Afferent stimuli irradiate from one reflex center to another after SCI. When even a relatively minor noxious stimulus is applied to the skin, it may activate autonomic neurons and produce evacuation of the bladder and rectum, sweating, pallor, and blood pressure swings in addition to the withdrawal response. This distressing mass reflex can however sometimes be used to give paraplegic patients a degree of bladder and bowel control. They can be trained to initiate urination and defecation by stroking or pinching their thighs, thus producing an intentional mass reflex. If the cord section is incomplete, the flexor spasms initiated by noxious stimuli can be associated with bursts of pain that are particularly bothersome. They can be treated with considerable success with baclofen, a GABAB receptor agonist that crosses the blood–brain barrier and facilitates inhibition.
THERAPEUTIC HIGHLIGHTS
Treatment of SCI patients presents complex problems. Administration of large doses of glucocorticoids has been shown to foster recovery and minimize loss of function after SCI. They need to be given soon after the injury and then discontinued because of the well-established deleterious effects of long-term steroid treatment. Their immediate value is likely due to reduction of the inflammatory response in the damaged tissue. Due to immobilization, SCI patients develop a negative nitrogen balance and catabolize large amounts of body protein. Their body weight compresses the circulation to the skin over bony prominences, causing decubitus ulcers to form. The ulcers heal poorly and are prone to infection because of body protein depletion. The tissues that are broken down include the protein matrix of bone and this, plus the immobilization, cause Ca2+ to be released in large amounts, leading to hypercalcemia, hypercalciuria, and formation of calcium stones in the urinary tract. The combination of stones and bladder paralysis cause urinary stasis, which predisposes to urinary tract infection, the most common complication of SCI. The search continues for ways to get axons of neurons in the spinal cord to regenerate. Administration of neurotrophins shows some promise in experimental animals, and so does implantation of embryonic stem cells at the site of injury. Another possibility being explored is bypassing the site of SCI with brain-computer interface devices. However, these novel approaches are a long way from routine clinical use.
In all vertebrates, transection of the spinal cord is followed by a period of spinal shock during which all spinal reflex responses are profoundly depressed. Subsequently, reflex responses return and become hyperactive. The duration of spinal shock is proportional to the degree of encephalization of motor function in the various species. In frogs and rats it lasts for minutes; in dogs and cats it lasts for 1–2 h; in monkeys it lasts for days; and in humans it usually lasts for a minimum of 2 weeks.
Cessation of tonic bombardment of spinal neurons by excitatory impulses in descending pathways (see below) undoubtedly plays a role in development of spinal shock. In addition, spinal inhibitory interneurons that normally are themselves inhibited may be released from this descending inhibition to become disinhibited. This, in turn, would inhibit motor neurons. The recovery of reflex excitability may be due to the development of denervation hypersensitivity to the mediators released by the remaining spinal excitatory endings. Another contributing factor may be sprouting of collaterals from existing neurons, with the formation of additional excitatory endings on interneurons and motor neurons.
The first reflex response to appear as spinal shock wears off in humans is often a slight contraction of the leg flexors and adductors in response to a noxious stimulus (ie, the withdrawal reflex). In some patients, the knee jerk reflex recovers first. The interval between cord transection and the return of reflex activity is about 2 weeks in the absence of any complications, but if complications are present it is much longer. Once the spinal reflexes begin to reappear after spinal shock, their threshold steadily drops.
Circuits intrinsic to the spinal cord can produce walking movements when stimulated in a suitable fashion even after spinal cord transection in cats and dogs. There are two locomotor pattern generators in the spinal cord: one in the cervical region and one in the lumbar region. However, this does not mean that spinal animals or humans can walk without stimulation; the pattern generator has to be turned on by tonic discharge of a discrete area in the midbrain, the mesencephalic locomotor region, and, of course, this is only possible in patients with incomplete spinal cord transection. Interestingly, the generators can also be turned on in experimental animals by administration of the norepinephrine precursor L-dopa (levodopa) after complete section of the spinal cord. Progress is being made in teaching humans with SCI to take a few steps by placing them, with support, on a treadmill.
GENERAL PRINCIPLES OF CENTRAL ORGANIZATION OF MOTOR PATHWAYS
To voluntarily move a limb, the brain must plan a movement, arrange appropriate motion at many different joints at the same time, and adjust the motion by comparing plan with performance. The motor system “learns by doing” and performance improves with repetition. This involves synaptic plasticity. Damage to the cerebral cortex before or during childbirth or during the first 2–3 years of development can lead to cerebral palsy, a disorder that affects muscle tone, movement, and coordination (Clinical Box 12–4).
CLINICAL BOX 12–4
Cerebral Palsy
Cerebral palsy (CP) is a term used to describe any one of several nonprogressive neurological disorders that occur before or during childbirth or during early childhood. Prenatal factors, including exposure of the developing brain to hypoxia, infections, or toxins, may account for 70–80% of cases of CP. Typical symptoms of the disorder include spasticity, ataxia, deficits in fine motor control, and abnormal gait (crouched or “scissored gait”). Sensory deficits including loss of vision and hearing as well as learning difficulties and seizures often occur in children with CP. In developed countries, the prevalence of CP is 2–2.5 cases per 1000 live births; however, the incidence of CP in children who are born prematurely is much higher compared with children born at term. Based on differences in the resting tone in muscles and the limbs involved, CP is classified into different groups. The most prevalent type is spastic CP, which is characterized by spasticity, hyperreflexia, clonus, and a positive Babinski sign. These are all signs of damage to the corticospinal tract (see Clinical Box 12–5). Dyskinetic CP is characterized by abnormal involuntary movements (chorea and athetosis) and is thought to reflect damage to extrapyramidal motor areas. It is not uncommon to have signs of both types of CP to coexist. The rarest type is hypotonic CP, which presents with truncal and extremity hypotonia, hyperreflexia, and persistent primitive reflexes.
THERAPEUTIC HIGHLIGHTS
There is no cure for CP. Treatment often includes physical and occupational therapy. Botulinum toxin injections into affected muscles have been used to reduce muscle spasticity, especially in the gastrocnemius muscle. Other drugs used to treat muscle spasticity in patients with CP include diazepam (a benzodiazepine that binds to the GABAA receptor), baclofen (an agonist at presynaptic GABAB receptors in the spinal cord), and dantrolene (a direct muscle relaxant). Various surgeries have been used to treat CP, including selective dorsal rhizotomy (section of the dorsal roots) and tenotomy (severing the tendon in the gastrocnemius muscles).
There is considerable evidence for the general motor control scheme shown in Figure 12–7. Commands for voluntary movement originate in cortical association areas. The movements are planned in the cortex as well as in the basal ganglia and the lateral portions of the cerebellar hemispheres, as indicated by increased electrical activity before the movement. The basal ganglia and cerebellum funnel information to the premotor and motor cortex by way of the thalamus. Motor commands from the motor cortex are relayed in large part via the corticospinal tracts to the spinal cord and the corresponding corticobulbar tracts to motor neurons in the brain stem. However, collaterals from these pathways and a few direct connections from the motor cortex end on brain stem nuclei, which also project to motor neurons in the brain stem and spinal cord. These pathways can also mediate voluntary movement. Movement sets up alterations in sensory input from the special senses and from muscles, tendons, joints, and the skin. This feedback information, which adjusts and smoothes movement, is relayed directly to the motor cortex and to the spinocerebellum. The spinocerebellum projects in turn to the brain stem. The main brain stem pathways that are concerned with posture and coordination are the rubrospinal, reticulospinal, tectospinal, and vestibulospinal tracts.
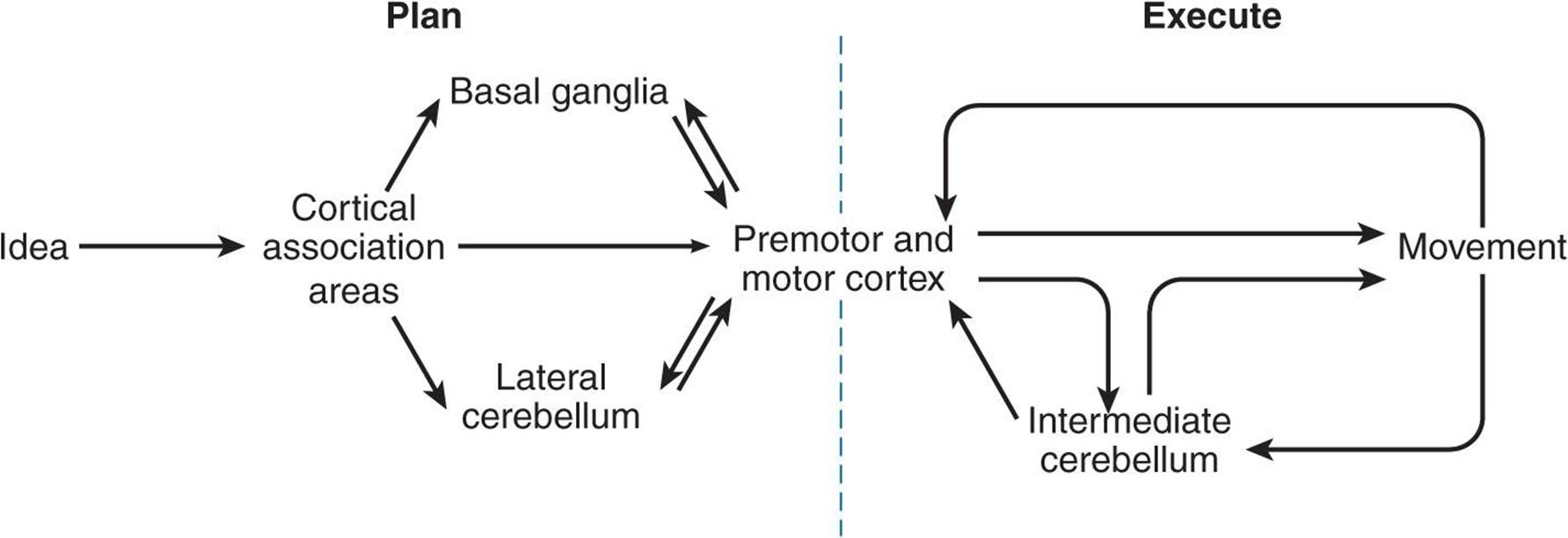
FIGURE 12–7 Control of voluntary movement. Commands for voluntary movement originate in cortical association areas. The cortex, basal ganglia, and cerebellum work cooperatively to plan movements. Movement executed by the cortex is relayed via the corticospinal tracts and corticobulbar tracts to motor neurons. The cerebellum provides feedback to adjust and smooth movement.
MOTOR CORTEX & VOLUNTARY MOVEMENT
PRIMARY MOTOR CORTEX
The reader can refer to Figure 8–8 for the locations of the major cortical regions involved in motor control. The primary motor cortex (M1) is in the precentral gyrus of the frontal lobe, extending into the central sulcus. By means of stimulation experiments in patients undergoing craniotomy under local anesthesia, this region was mapped to show where various parts of the body are represented in the precentral gyrus. Figure 12–8 shows the motor homunculuswith the feet at the top of the gyrus and the face at the bottom. The facial area is represented bilaterally, but the rest of the representation is generally unilateral, with the cortical motor area controlling the musculature on the opposite side of the body. The cortical representation of each body part is proportional in size to the skill with which the part is used in fine, voluntary movement. The areas involved in speech and hand movements are especially large in the cortex; use of the pharynx, lips, and tongue to form words and of the fingers and opposable thumbs to manipulate the environment are activities in which humans are especially skilled.
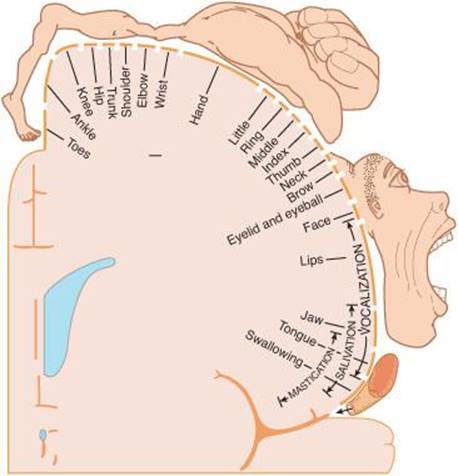
FIGURE 12–8 Motor homunculus. The figure represents, on a coronal section of the precentral gyrus, the location of the cortical representation of the various parts. The size of the various parts is proportional to the cortical area devoted to them. Compare with Figure 8–9 which shows the sensory homunculus. (Reproduced with permission from Penfield W, Rasmussen G: The Cerebral Cortex of Man. Macmillan, 1950.)
Modern brain imaging techniques such as positive emission tomography (PET) and functional magnetic resonance imaging (fMRI) have been used to map the cortex to identify motor areas. Figure 12–9 shows activation of the hand area of the motor cortex while repetitively squeezing a ball with either the right or left hand.
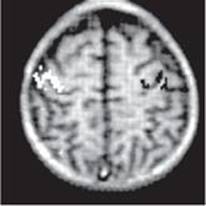
FIGURE 12–9 Hand area of motor cortex demonstrated by functional magnetic resonance imaging (fMRI) in a 7-year-old boy. Changes in signal intensity, measured using a method called echoplanar magnetic resonance imaging, result from changes in the flow, volume, and oxygenation of the blood. The child was instructed to repetitively squeeze a foam-rubber ball at the rate of two to four squeezes per second with the right or left hand. Changes in cortical activity with the ball in the right hand are shown in black. Changes in cortical activity with the ball in the left hand are shown in white. (Data from Novotny EJ, et al: Functional magnetic resonance imaging (fMRI) in pediatric epilepsy. Epilepsia 1994;35(Supp 8):36.)
The cells in the cortical motor areas are arranged in a columnar organization. The ability to elicit discrete movements of a single muscle by electrical stimulation of a column within M1 led to the view that this area was responsible for control of individual muscles. More recent work has shown that neurons in several cortical columns project to the same muscle; in fact, most stimuli activate more than one muscle. Moreover, the cells in each column receive fairly extensive sensory input from the peripheral area in which they produce movement, providing the basis for feedback control of movement. Some of this input may be direct and some is relayed from other parts of the cortex. The current view is that M1 neurons represent movements of groups of muscles for different tasks.
SUPPLEMENTARY MOTOR AREA
The supplementary motor area is on and above the superior bank of the cingulate sulcus on the medial side of the hemisphere. It projects to the primary motor cortex and also contains a map of the body; but it is less precise than in M1. The supplementary motor area may be involved primarily in organizing or planning motor sequences, while M1 executes the movements. Lesions of this area in monkeys produce awkwardness in performing complex activities and difficulty with bimanual coordination.
When human subjects count to themselves without speaking, the motor cortex is quiescent, but when they speak the numbers aloud as they count, blood flow increases in M1 and the supplementary motor area. Thus, both regions are involved in voluntary movement when the movements being performed are complex and involve planning.
PREMOTOR CORTEX
The premotor cortex is located anterior to the precentral gyrus, on the lateral and medial cortical surface; it also contains a somatotopic map. This region receives input from sensory regions of the parietal cortex and projects to M1, the spinal cord, and the brain stem reticular formation. This region may be concerned with setting posture at the start of a planned movement and with getting the individual prepared to move. It is most involved in control of proximal limb muscles needed to orient the body for movement.
POSTERIOR PARIETAL CORTEX
The somatic sensory area and related portions of the posterior parietal lobe project to the premotor cortex. Lesions of the somatic sensory area cause defects in motor performance that are characterized by inability to execute learned sequences of movements such as eating with a knife and fork. Some of the neurons are concerned with aiming the hands toward an object and manipulating it, whereas other neurons are concerned with hand–eye coordination. As described below, neurons in this posterior parietal cortex contribute to the descending pathways involved in motor control.
PLASTICITY
A striking discovery made possible by PET and fMRI is that the motor cortex shows the same kind of plasticity as already described for the sensory cortex in Chapter 8. For example, the finger areas of the contralateral motor cortex enlarge as a pattern of rapid finger movement is learned with the fingers of one hand; this change is detectable at 1 week and maximal at 4 weeks. Cortical areas of output to other muscles also increase in size when motor learning involves these muscles. When a small focal ischemic lesion is produced in the hand area of the motor cortex of monkeys, the hand area may reappear, with return of motor function, in an adjacent undamaged part of the cortex. Thus, the maps of the motor cortex are not immutable, and they change with experience.
CONTROL OF AXIAL & DISTAL MUSCLES
Within the brain stem and spinal cord, pathways and neurons that are concerned with the control of skeletal muscles of the trunk (axial) and proximal portions of the limbs are located medially or ventrally, whereas pathways and neurons that are concerned with the control of skeletal muscles in the distal portions of the limbs are located laterally. The axial muscles are concerned with postural adjustments and gross movements, whereas the distal limb muscles mediate fine, skilled movements. Thus, for example, neurons in the medial portion of the ventral horn innervate proximal limb muscles, particularly the flexors, whereas lateral ventral horn neurons innervate distal limb muscles. Similarly, the ventral corticospinal tract and medial descending brain stem pathways (tectospinal, reticulospinal, and vestibulospinal tracts) are concerned with adjustments of proximal muscles and posture, whereas the lateral corticospinal and rubrospinal tracts are concerned with distal limb muscles and, particularly in the case of the lateral corticospinal tract, with skilled voluntary movements. Phylogenetically, the lateral pathways are newer.
CORTICOSPINAL & CORTICOBULBAR TRACTS
The somatotopic organization just described for the motor cortex continues throughout the pathways from the cortex to the motor neurons. The axons of neurons from the motor cortex that project to spinal motor neurons form the corticospinal tracts, a large bundle of about 1 million fibers. About 80% of these fibers cross the midline in the medullary pyramids to form the lateral corticospinal tract (Figure 12–10). The remaining 20% make up the ventral corticospinal tract, which does not cross the midline until it reaches the level of the spinal cord at which it terminates. Lateral corticospinal tract neurons make monosynaptic connections to motor neurons, especially those concerned with skilled movements. Many corticospinal tract neurons also synapse on spinal interneurons antecedent to motor neurons; this indirect pathway is important in coordinating groups of muscles.
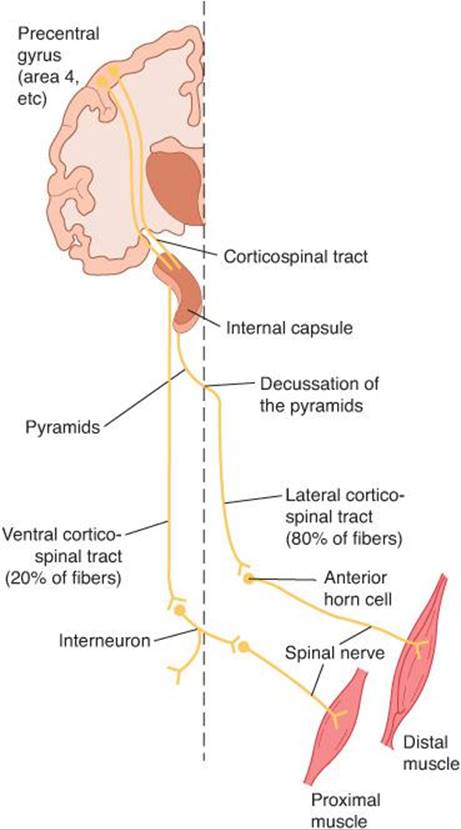
FIGURE 12–10 The corticospinal tracts. This tract originates in the precentral gyrus and passes through the internal capsule. Most fibers decussate in the pyramids and descend in the lateral white matter of the spinal cord to form the lateral division of the tract which can make monosynaptic connections with spinal motor neurons. The ventral division of the tract remains uncrossed until reaching the spinal cord where axons terminate on spinal interneurons antecedent to motor neurons.
The trajectory from the cortex to the spinal cord passes through the corona radiata to the posterior limb of the internal capsule. Within the midbrain they traverse the cerebral peduncle and the basilar pons until they reach the medullary pyramids on their way to the spinal cord.
The corticobulbar tract is composed of the fibers that pass from the motor cortex to motor neurons in the trigeminal, facial, and hypoglossal nuclei. Corticobulbar neurons end either directly on the cranial nerve nuclei or on their antecedent interneurons within the brain stem. Their axons traverse through the genu of the internal capsule, the cerebral peduncle (medial to corticospinal tract neurons), to descend with corticospinal tract fibers in the pons and medulla.
The motor system can be divided into lower and upper motor neurons. Lower motor neurons refer to the spinal and cranial motor neurons that directly innervate skeletal muscles. Upper motor neurons are those in the cortex and brain stem that activate the lower motor neurons. The pathophysiological responses to damage to lower and upper motor neurons are very distinctive (see Clinical Box 12–5).
CLINICAL BOX 12–5
Lower versus Upper Motor Neuron Damage
Lower motor neurons are those whose axons terminate on skeletal muscles. Damage to these neurons is associated with flaccid paralysis, muscular atrophy, fasciculations (visible muscle twitches that appear as flickers under the skin), hypotonia (decreased muscle tone), and hyporeflexia or areflexia. An example of a disease that leads to lower motor neuron damage is amyotrophic lateral sclerosis (ALS). “Amyotrophic” means “no muscle nourishment” and describes the atrophy that muscles undergo because of disuse. “Sclerosis” refers to the hardness felt when a pathologist examines the spinal cord on autopsy; the hardness is due to proliferation of astrocytes and scarring of the lateral columns of the spinal cord. ALS is a selective, progressive degeneration of α-motor neurons. This fatal disease is also known as Lou Gehrig disease in remembrance of a famous American baseball player who died of ALS. The worldwide annual incidence of ALS has been estimated to be 0.5–3 cases per 100,000 people. The disease has no racial, socioeconomic, or ethnic boundaries. The life expectancy of ALS patients is usually 3–5 years after diagnosis. ALS is most commonly diagnosed in middle age and affects men more often than women. Most cases of ALS are sporadic in origin; but 5–10% of the cases have a familial link. Possible causes include viruses, neurotoxins, heavy metals, DNA defects (especially in familial ALS), immune system abnormalities, and enzyme abnormalities. About 40% of the familial cases have a mutation in the gene for Cu/Zn superoxide dismutase (SOD-1)on chromosome 21. SOD is a free radical scavenger that reduces oxidative stress. A defective SOD-1 gene permits free radicals to accumulate and kill neurons. Some evidence suggests an increase in the excitability of deep cerebellar nuclei due to the inhibition of small-conductance calcium-activated potassium (SK) channels contributes to the development of cerebellar ataxia.
Upper motor neurons typically refer to corticospinal tract neurons that innervate spinal motor neurons, but they can also include brain stem neurons that control spinal motor neurons. Damage to these neurons initially causes muscles to become weak and flaccid but eventually leads to spasticity, hypertonia (increased resistance to passive movement), hyperactive stretch reflexes, and abnormal plantar extensor reflex (positive Babinski sign). The Babinski sign is dorsiflexion of the great toe and fanning of the other toes when the lateral aspect of the sole of the foot is scratched. In adults, the normal response to this stimulation is plantar flexion in all the toes. The Babinski sign is believed to be a flexor withdrawal reflex that is normally held in check by the lateral corticospinal system. It is of value in the localization of disease processes, but its physiologic significance is unknown. However, in infants whose corticospinal tracts are not well developed, dorsiflexion of the great toe and fanning of the other toes is the natural response to stimuli applied to the sole of the foot.
THERAPEUTIC HIGHLIGHTS
One of the few drugs shown to modestly slow the progression of ALS is riluzole, a drug that opens the SK channels. Spasticity associated with motor neuron disease can be reduced by the muscle relaxant baclofen (a derivative of GABA); in some cases a subarachnoid infusion of baclofen is given via an implanted lumbar pump. Spasticity can also be treated with tizanidine, a centrally acting α2-adrenoceptor agonist; its effectiveness is thought to be due to increasing presynaptic inhibition of spinal motor neurons. Botulinum toxin is also approved for the treatment of spasticity; this toxin acts by binding to receptors on the cholinergic nerve terminals to decrease the release of acetylcholine, causing neuromuscular blockade.
ORIGINS OF CORTICOSPINAL & CORTICOBULBAR TRACTS
Corticospinal and corticobulbar tract neurons are pyramidal shaped and located in layer V of the cerebral cortex (see Chapter 14). The cortical areas from which these tracts originate were identified on the basis of electrical stimulation that produced prompt discrete movement. About 31% of the corticospinal tract neurons are from the primary motor cortex. The premotor cortex and supplementary motor cortex account for 29% of the corticospinal tract neurons. The other 40% of corticospinal tract neurons originate in the parietal lobe and primary somatosensory area in the postcentral gyrus.
ROLE IN MOVEMENT
The corticospinal and corticobulbar system is the primary pathway for the initiation of skilled voluntary movement. This does not mean that movement—even skilled movement—is impossible without it. Nonmammalian vertebrates have essentially no corticospinal and corticobulbar system, but they move with great agility. Cats and dogs stand, walk, and run after complete destruction of this system. Only in primates are relatively marked deficits produced.
Careful section of the pyramids producing highly selective destruction of the lateral corticospinal tract in laboratory primates produces prompt and sustained loss of the ability to grasp small objects between two fingers and to make isolated movements of the wrists. However, the animal can still use the hand in a gross fashion and can stand and walk. These deficits are consistent with loss of control of the distal musculature of the limbs, which is concerned with fine-skilled movements. On the other hand, lesions of the ventral corticospinal tract produce axial muscle deficits that cause difficulty with balance, walking, and climbing.
BRAIN STEM PATHWAYS INVOLVED IN POSTURE AND VOLUNTARY MOVEMENT
As mentioned above, spinal motor neurons are organized such that those innervating the most proximal muscles are located most medially and those innervating the more distal muscles are located more laterally. This organization is also reflected in descending brain stem pathways (Figure 12–11).
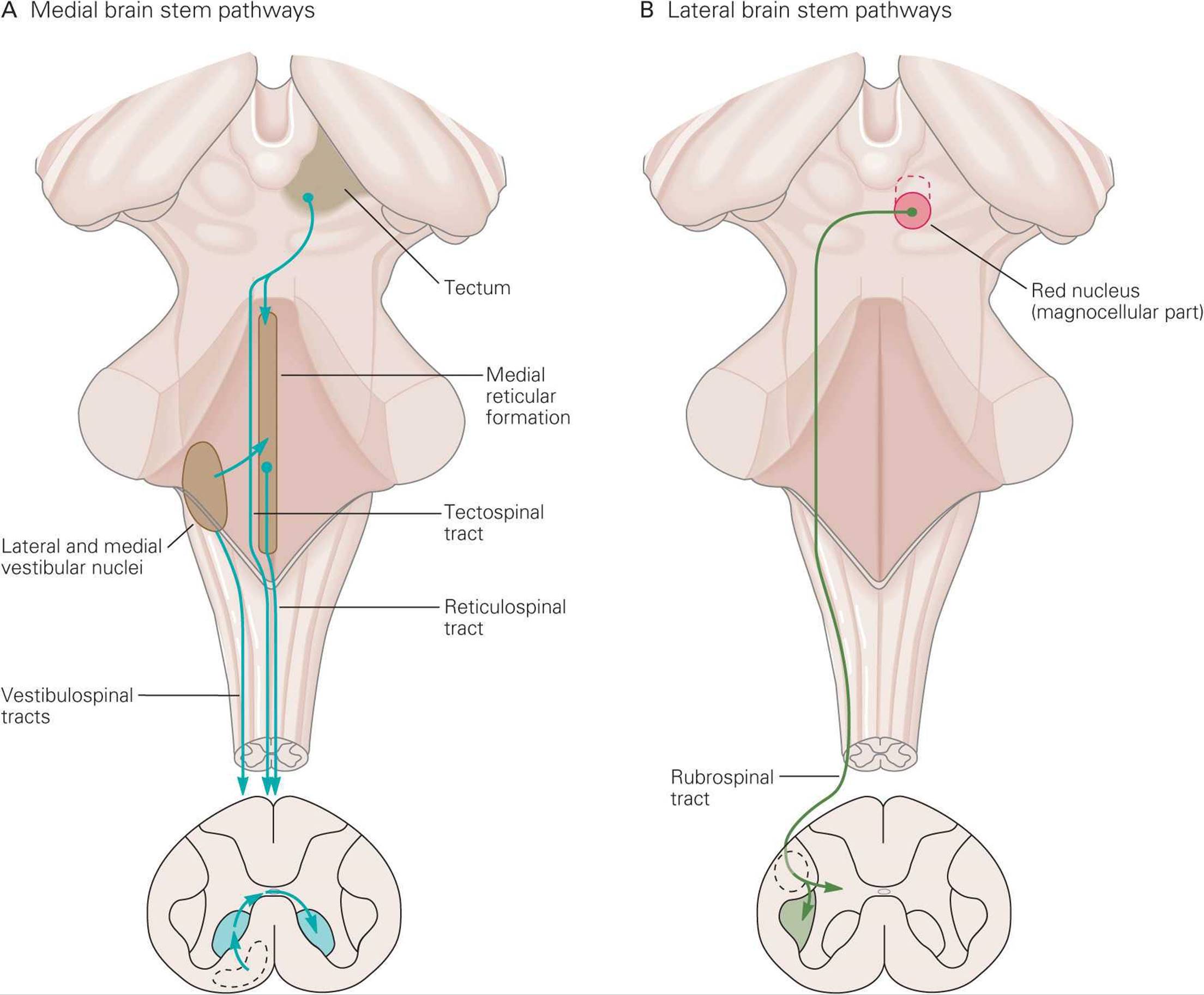
FIGURE 12–11 Medial and lateral descending brain stem pathways involved in motor control. A) Medial pathways (reticulospinal, vestibulospinal, and tectospinal) terminate in ventromedial area of spinal gray matter and control axial and proximal muscles. B) Lateral pathway (rubrospinal) terminates in dorsolateral area of spinal gray matter and controls distal muscles. (From Kandel ER, Schwartz JH, Jessell TM [editors]: Principles of Neural Science, 4th ed. McGraw-Hill, 2000.)
MEDIAL BRAIN STEM PATHWAYS
The medial brain stem pathways, which work in concert with the ventral corticospinal tract, are the pontine and medullary reticulospinal, vestibulospinal, and tectospinal tracts. These pathways descend in the ipsilateral ventral columns of the spinal cord and terminate predominantly on interneurons and long propriospinal neurons in the ventromedial part of the ventral horn to control axial and proximal muscles. A few medial pathway neurons synapse directly on motor neurons controlling axial muscles.
The medial and lateral vestibulospinal tracts are involved in vestibular function and are briefly described in Chapter 10. The medial tract originates in the medial and inferior vestibular nuclei and projects bilaterally to cervical spinal motor neurons that control neck musculature. The lateral tract originates in the lateral vestibular nuclei and projects ipsilaterally to neurons at all spinal levels. It activates motor neurons to antigravity muscles (eg, proximal limb extensors) to control posture and balance.
The pontine and medullary reticulospinal tracts project to all spinal levels. They are involved in the maintenance of posture and in modulating muscle tone, especially via an input to γ-motor neurons. Pontine reticulospinal neurons are primarily excitatory and medullary reticulospinal neurons are primarily inhibitory.
The tectospinal tract originates in the superior colliculus of the midbrain. It projects to the contralateral cervical spinal cord to control head and eye movements.
LATERAL BRAIN STEM PATHWAY
The main control of distal muscles arise from the lateral corticospinal tract, but neurons within the red nucleus of the midbrain cross the midline and project to interneurons in the dorsolateral part of the spinal ventral horn to also influence motor neurons that control distal limb muscles. This rubrospinal tract excites flexor motor neurons and inhibits extensor motor neurons. This pathway is not very prominent in humans, but it may play a role in the posture typical of decorticate rigidity (see below).
POSTURE-REGULATING SYSTEMS
In the intact animal, individual motor responses are submerged in the total pattern of motor activity. When the neural axis is transected, the activities integrated below the section are cut off, or released, from the control of higher brain centers and often appear to be accentuated. Release of this type, long a cardinal principle in neurology, may be due in some situations to removal of an inhibitory control by higher neural centers. A more important cause of the apparent hyperactivity is loss of differentiation of the reaction so that it no longer fits into the broader pattern of motor activity. Research using animal models has led to information on the role of cortical and brain stem mechanisms involved in control of voluntary movement and posture. The deficits in motor control seen after various lesions mimic those seen in humans with damage in the same structures.
DECEREBRATION
A complete transection of the brain stem between the superior and inferior colliculi permits the brain stem pathways to function independent of their input from higher brain structures. This is called a midcollicular decerebrationand is diagramed in Figure 12–12 by the dashed line labeled A. This lesion interrupts all input from the cortex (corticospinal and corticobulbar tracts) and red nucleus (rubrospinal tract), primarily to distal muscles of the extremities. The excitatory and inhibitory reticulospinal pathways (primarily to postural extensor muscles) remain intact. The dominance of drive from ascending sensory pathways to the excitatory reticulospinal pathway leads to hyperactivity in extensor muscles in all four extremities which is called decerebrate rigidity. This resembles what ensues after uncal herniation due to a supratentorial lesion. Uncal herniation can occur in patients with large tumors or a hemorrhage in the cerebral hemisphere. Figure 12–13A shows the posture typical of such a patient. Clinical Box 12–6 describes complications related to uncal herniation.
CLINICAL BOX 12–6
Uncal Herniation
Space-occupying lesions from large tumors, hemorrhages, strokes, or abscesses in the cerebral hemisphere can drive the uncus of the temporal lobe over the edge of the cerebellar tentorium, compressing the ipsilateral cranial nerve III (uncal herniation). Before the herniation these patients experience a decreased level of consciousness, lethargy, poorly reactive pupils, deviation of the eye to a “down and out” position, hyperactive reflexes, and a bilateral Babinski sign (due to compression of the ipsilateral corticospinal tract). After the brain herniates, the patients are decerebrate and comatose, have fixed and dilated pupils, and eye movements are absent. Once damage extends to the midbrain, a Cheyne–Stokes respiratory pattern develops, characterized by a pattern of waxing-and-waning depth of respiration with interposed periods of apnea. Eventually, medullary function is lost, breathing ceases, and recovery is unlikely. Hemispheric masses closer to the midline compress the thalamic reticular formation and can cause coma before eye findings develop (central herniation). As the mass enlarges, midbrain function is affected, the pupils dilate, and a decerebrate posture ensues. With progressive herniation, pontine vestibular and then medullary respiratory functions are lost.
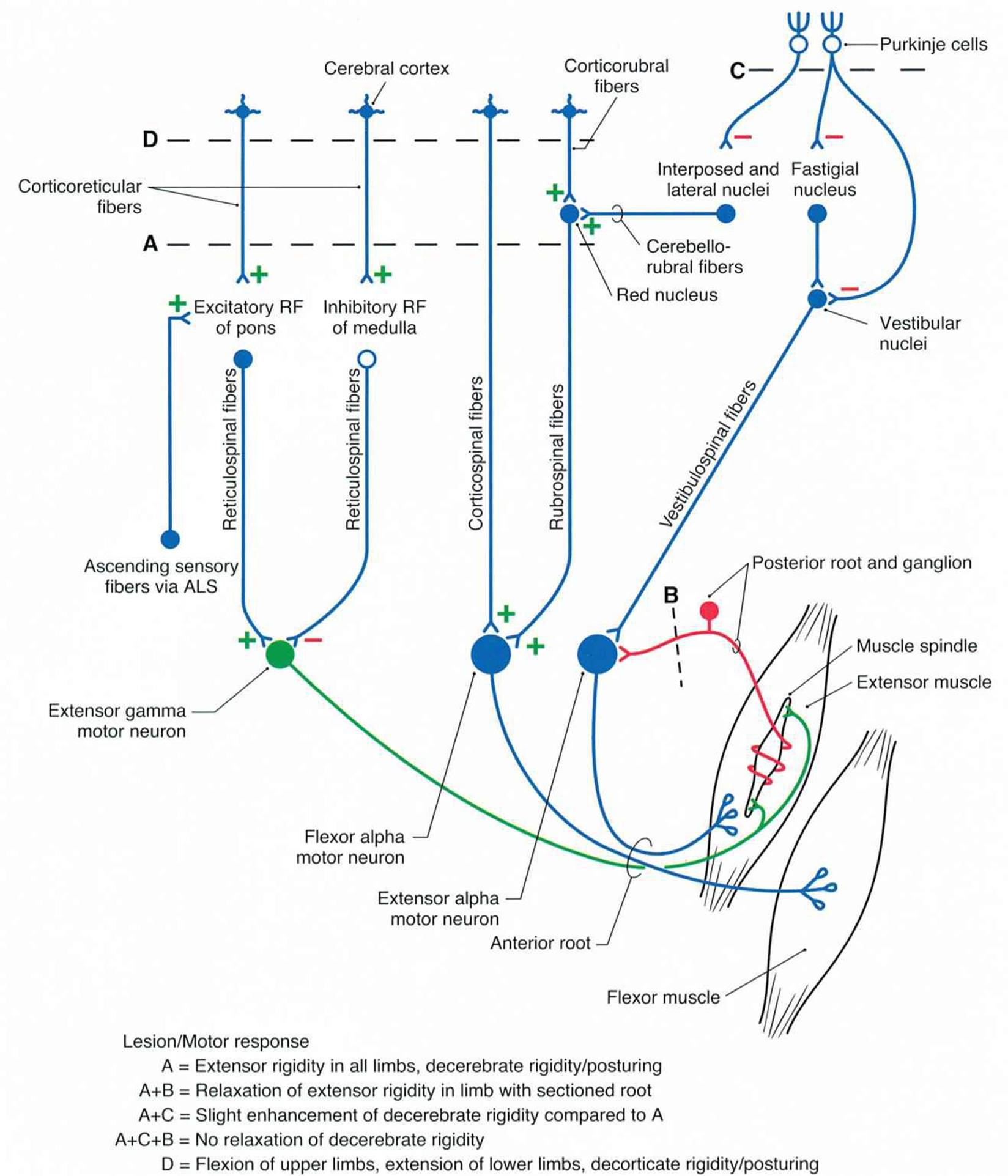
FIGURE 12–12 A circuit drawing representing lesions produced in experimental animals to replicate decerebrate and decorticate deficits seen in humans. Bilateral transections are indicated by dashed lines A, B, C, and D. Decerebration is at a midcollicular level (A), decortication is rostral to the superior colliculus, dorsal roots sectioned for one extremity (B), and removal of anterior lobe of cerebellum (C). The objective was to identify anatomic substrates responsible for decerebrate or decorticate rigidity/posturing seen in humans with lesions that either isolate the forebrain from the brain stem or separate rostral from caudal brain stem and spinal cord. (Reproduced with permission from Haines DE [editor]: Fundamental Neuroscience for Basic and Clinical Applications, 3rd ed. Elsevier, 2006.)
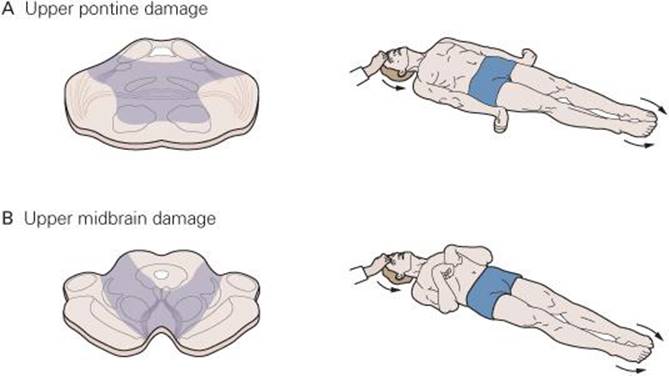
FIGURE 12–13 Decerebrate and decorticate postures. A) Damage to lower midbrain and upper pons causes decerebrate posturing in which lower extremities are extended with toes pointed inward and upper extremities extended with fingers flexed and forearms pronate. Neck and head are extended. B) Damage to upper midbrain may cause decorticate posturing in which upper limbs are flexed, lower limbs are extended with toes pointed slightly inward, and head is extended. (Modified from Kandel ER, Schwartz JH, Jessell TM [editors]: Principles of Neural Science, 4th ed. McGraw-Hill, 2000.)
In midcollicular decerebrate cats, section of dorsal roots to a limb (dashed line labeled B in Figure 12–12) immediately eliminates the hyperactivity of extensor muscles. This suggests that decerebrate rigidity is spasticity due to facilitation of the myotatic stretch reflex. That is, the excitatory input from the reticulospinal pathway activates γ-motor neurons which indirectly activate α-motor neurons (via Ia spindle afferent activity). This is called the gamma loop.
The exact site of origin within the cerebral cortex of the fibers that inhibit stretch reflexes is unknown. Under certain conditions, stimulation of the anterior edge of the precentral gyrus can cause inhibition of stretch reflexes and cortically evoked movements. This region, which also projects to the basal ganglia, has been named the suppressor strip.
There is also evidence that decerebrate rigidity leads to direct activation of α-motor neurons. If the anterior lobe of the cerebellum is removed in a decerebrate animal (dashed line labeled C in Figure 12–12), extensor muscle hyperactivity is exaggerated (decerebellate rigidity). This cut eliminates cortical inhibition of the cerebellar fastigial nucleus and secondarily increases excitation to vestibular nuclei. Subsequent dorsal root section does not reverse the rigidity, thus it was due to activation of α-motor neurons independent of the gamma loop.
DECORTICATION
Removal of the cerebral cortex (decortication; dashed line labeled D in Figure 12–12) produces decorticate rigidity which is characterized by flexion of the upper extremities at the elbow and extensor hyperactivity in the lower extremities (Figure 12–13B). The flexion can be explained by rubrospinal excitation of flexor muscles in the upper extremities; the hyperextension of lower extremities is due to the same changes that occur after midcollicular decerebration.
Decorticate rigidity is seen on the hemiplegic side after hemorrhages or thromboses in the internal capsule. Probably because of their anatomy, the small arteries in the internal capsule are especially prone to rupture or thrombotic obstruction, so this type of decorticate rigidity is fairly common. Sixty percent of intracerebral hemorrhages occur in the internal capsule, as opposed to 10% in the cerebral cortex, 10% in the pons, 10% in the thalamus, and 10% in the cerebellum.
BASAL GANGLIA
ORGANIZATION OF THE BASAL GANGLIA
The term basal ganglia (or basal nuclei) is applied to five interactive structures on each side of the brain (Figure 12–14). These are the caudate nucleus, putamen, and globus pallidus (three large nuclear masses underlying the cortical mantle), the subthalamic nucleus, and substantia nigra. The caudate nucleus and putamen collectively form the striatum; the putamen and globus pallidus collectively form the lenticular nucleus.
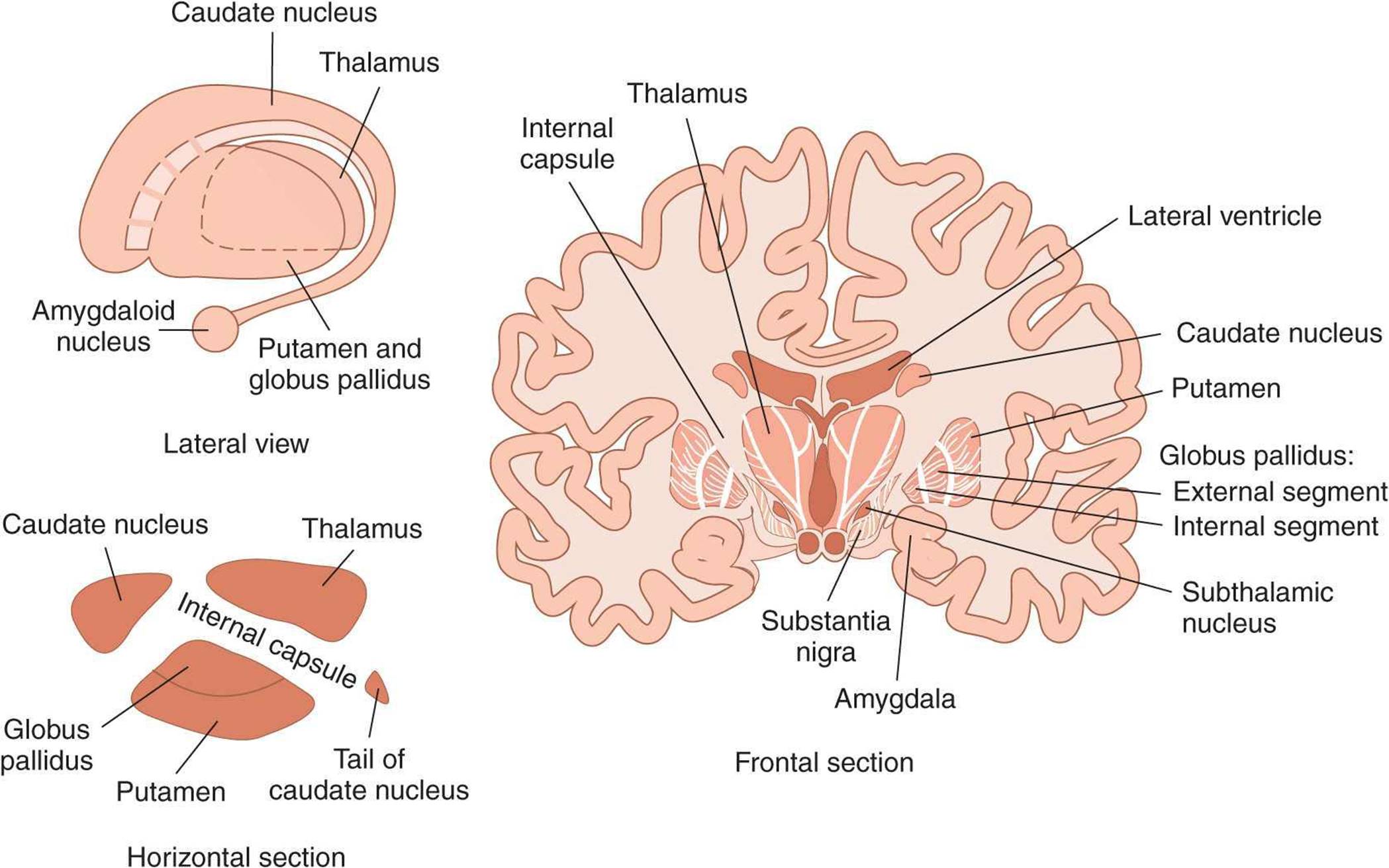
FIGURE 12–14 The basal ganglia. The basal ganglia are composed of the caudate nucleus, putamen, and globus pallidus and the functionally related subthalamic nucleus and substantia nigra. The frontal (coronal) section shows the location of the basal ganglia in relation to surrounding structures.
The globus pallidus is divided into external and internal segments (GPe and GPi); both regions contain inhibitory GABAergic neurons. The substantia nigra is divided into a pars compacta which uses dopamine as a neurotransmitter and a pars reticulata which uses GABA as a neurotransmitter. There are at least four neuronal types within the striatum. About 95% of striatal neurons are medium spiny neurons that use GABA as a neurotransmitter. The remaining striatal neurons are all aspiny interneurons that differ in terms of size and neurotransmitters: large (acetylcholine), medium (somatostatin), and small (GABA).
Figure 12–15 shows the major connections to and from and within the basal ganglia along with the neurotransmitters within these pathways. There are two main inputs to the basal ganglia; they are both excitatory (glutamate), and they both terminate in the striatum. They are from a wide region of the cerebral cortex (corticostriate pathway) and from intralaminar nuclei of the thalamus (thalamostriatal pathway). The two major outputs of the basal ganglia are from GPi and substantia nigra pars reticulata. Both are inhibitory (GABAergic) and both project to the thalamus. From the thalamus, there is an excitatory (presumably glutamate) projection to the prefrontal and premotor cortex. This completes a full cortical-basal ganglia-thalamic-cortical loop.
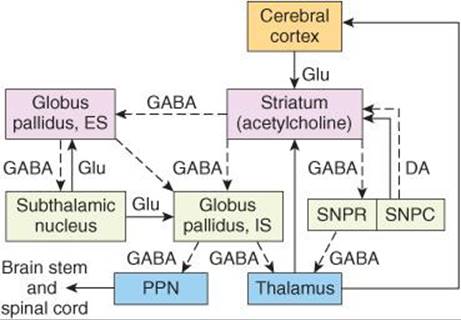
FIGURE 12–15 Diagrammatic representation of the principal connections of the basal ganglia. Solid lines indicate excitatory pathways, dashed lines inhibitory pathways. The transmitters are indicated in the pathways, where they are known. DA, dopamine; Glu, glutamate. Acetylcholine is the transmitter produced by interneurons in the striatum. ES, external segment; IS, internal segment; PPN, pedunculopontine nuclei; SNPC, substantia nigra, pars compacta; SNPR, substantia nigra, pars reticulata. The subthalamic nucleus also projects to the pars compacta of the substantia nigra; this pathway has been omitted for clarity.
The connections within the basal ganglia include a dopaminergic nigrostriatal projection from the substantia nigra pars compacta to the striatum and a GABAergic projection from the striatum to substantia nigra pars reticulata. There is an inhibitory projection from the striatum to both GPe and GPi. The subthalamic nucleus receives an inhibitory input from GPe, and in turn the subthalamic nucleus has an excitatory (glutamate) projection to both GPe and GPi.
FUNCTION
Neurons in the basal ganglia, like those in the lateral portions of the cerebellar hemispheres, discharge before movements begin. This observation, plus careful analysis of the effects of diseases of the basal ganglion in humans and the effects of drugs that destroy dopaminergic neurons in animals, have led to the idea that the basal ganglia are involved in the planning and programming of movement or, more broadly, in the processes by which an abstract thought is converted into voluntary action (Figure 12–7). They influence the motor cortex via the thalamus, and the corticospinal pathways provide the final common pathway to motor neurons. In addition, GPi projects to nuclei in the brain stem, and from there to motor neurons in the brain stem and spinal cord. The basal ganglia, particularly the caudate nuclei, also play a role in some cognitive processes. Possibly because of the interconnections of this nucleus with the frontal portions of the neocortex, lesions of the caudate nuclei disrupt performance on tests involving object reversal and delayed alternation. In addition, lesions of the head of the left but not the right caudate nucleus and nearby white matter in humans are associated with a dysarthric form of aphasia that resembles Wernicke aphasia (see Chapter 15).
DISEASES OF THE BASAL GANGLIA IN HUMANS
Three distinct biochemical pathways in the basal ganglia normally operate in a balanced fashion: (1) the nigrostriatal dopaminergic system, (2) the intrastriatal cholinergic system, and (3) the GABAergic system, which projects from the striatum to the globus pallidus and substantia nigra. When one or more of these pathways become dysfunctional, characteristic motor abnormalities occur. Diseases of the basal ganglia lead to two general types of disorders: hyperkinetic and hypokinetic. The hyperkinetic conditions are those in which movement is excessive and abnormal, including chorea, athetosis, and ballism. Hypokinetic abnormalities include akinesia and bradykinesia.
Chorea is characterized by rapid, involuntary “dancing” movements. Athetosis is characterized by continuous, slow writhing movements. Choreiform and athetotic movements have been likened to the start of voluntary movements occurring in an involuntary, disorganized way. In ballism, involuntary flailing, intense, and violent movements occur. Akinesia is difficulty in initiating movement and decreased spontaneous movement. Bradykinesia is slowness of movement.
In addition to Parkinson disease, which is described below, there are several other disorders known to involve a malfunction within the basal ganglia. A few of these are described in Clinical Box 12–7. Huntington disease is one of an increasing number of human genetic diseases affecting the nervous system that are characterized by trinucleotide repeat expansion. Most of these involve cytosine-adenine-guanine (CAG) repeats (Table 12–1), but one involves CGG repeats and another involves CTG repeats (T refers to thymine). All of these are in exons; however, a GAA repeat in an intron is associated with Friedreich’s ataxia. There is also preliminary evidence that increased numbers of a 12-nucleotide repeat are associated with a rare form of epilepsy.
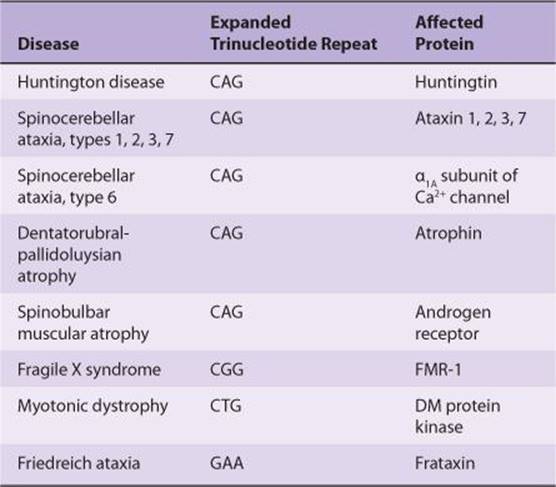
TABLE 12–1. Examples of trinucleotide repeat diseases.
CLINICAL BOX 12–7
Basal Ganglia Diseases
The initial detectable damage in Huntington disease is to medium spiny neurons in the striatum. This loss of this GABAergic pathway to the globus pallidus external segment releases inhibition, permitting the hyperkinetic features of the disease to develop. An early sign is a jerky trajectory of the hand when reaching to touch a spot, especially toward the end of the reach. Later, hyperkinetic choreiform movements appear and gradually increase until they incapacitate the patient. Speech becomes slurred and then incomprehensible, and a progressive dementia is followed by death, usually within 10–15 years after the onset of symptoms. Huntington disease affects 5 out of 100,000 people worldwide. It is inherited as an autosomal dominant disorder, and its onset is usually between the ages of 30 and 50. The abnormal gene responsible for the disease is located near the end of the short arm of chromosome 4. It normally contains 11–34 cytosine-adenine-guanine (CAG) repeats, each coding for glutamine. In patients with Huntington disease, this number is increased to 42–86 or more copies, and the greater the number of repeats, the earlier the age of onset and the more rapid the progression of the disease. The gene codes for huntingtin, a protein of unknown function. Poorly soluble protein aggregates, which are toxic, form in cell nuclei and elsewhere. However, the correlation between aggregates and symptoms is less than perfect. It appears that a loss of the function of huntingtin occurs that is proportional to the size of the CAG insert. In animal models of the disease, intrastriatal grafting of fetal striatal tissue improves cognitive performance. In addition, tissue caspase-1 activity is increased in the brains of humans and animals with the disease, and in mice in which the gene for this apoptosis-regulating enzyme has been knocked out, progression of the disease is slowed.
Another basal ganglia disorder is Wilson disease (or hepatolenticular degeneration), which is a rare disorder of copper metabolism which has an onset between 6 and 25 years of age, affecting about four times as many females as males. Wilson disease affects about 30,000 people worldwide. It is a genetic autosomal recessive disorder due to a mutation on the long arm of chromosome 13q. It affects the copper-transporting ATPase gene (ATP7B) in the liver, leading to an accumulation of copper in the liver and resultant progressive liver damage. About 1% of the population carries a single abnormal copy of this gene but does not develop any symptoms. A child who inherits the gene from both parents may develop the disease. In affected individuals, copper accumulates in the periphery of the cornea in the eye accounting for the characteristic yellow Kayser–Fleischer rings. The dominant neuronal pathology is degeneration of the putamen, a part of the lenticular nucleus. Motor disturbances include “wing-beating” tremor or asterixis, dysarthria, unsteady gait, and rigidity.
Another disease commonly referred to as a disease of the basal ganglia is tardive dyskinesia. This disease indeed involves the basal ganglia, but it is caused by medical treatment of another disorder with neuroleptic drugs such as phenothiazides or haloperidol. Therefore, tardive dyskinesia is iatrogenic in origin. Long-term use of these drugs may produce biochemical abnormalities in the striatum. The motor disturbances include either temporary or permanent uncontrolled involuntary movements of the face and tongue and cogwheel rigidity. The neuroleptic drugs act via blockade of dopaminergic transmission. Prolonged drug use leads to hypersensitivity of D3 dopaminergic receptors and an imbalance in nigrostriatal influences on motor control.
THERAPEUTIC HIGHLIGHTS
Treatment for Huntington disease is directed at treating the symptoms and maintaining quality of life as there is no cure. In general, drugs used to treat the symptoms of this disease have side effects such as fatigue, nausea, and restlessness. In August 2008, the U.S. Food and Drug Administration approved the use of tetrabenazine to reduce choreiform movements that characterize the disease. This drug binds reversibly to vesicular monoamine transporters (VMAT) and thus inhibits the uptake of monoamines into synaptic vesicles. It also acts as a dopamine receptor antagonist. Tetrabenazine is the first drug to receive approval for individuals with Huntington disease. It is also used to treat other hyperkinetic movement disorders such as tardive dyskinesia. Chelating agents (eg, penicillamine, trienthine) are used to reduce the copper in the body in individuals with Wilson disease. Tardive dyskinesia has proven to be difficult to treat. Treatment in patients with psychiatric disorders is often directed at prescribing a neuroleptic with less likelihood of causing the disorder. Clozapine is an example of an atypical neuroleptic drug that has been an effective substitute for traditional neuroleptic drugs but with less risk for development of tardive dyskinesia.
PARKINSON DISEASE
Parkinson disease has both hypokinetic and hyperkinetic features. It was originally described in 1817 by James Parkinson and is named for him. Parkinson disease is the first disease identified as being due to a deficiency in a specific neurotransmitter (see Clinical Box 12–8). In the 1960s, Parkinson disease was shown to result from the degeneration of dopaminergic neurons in the substantia nigra pars compacta. The fibers to the putamen (part of the striatum) are most severely affected.
CLINICAL BOX 12–8
Parkinson Disease
There are 7–10 million people worldwide who have been diagnosed with Parkinson disease. Men are 1.5 times more likely than women to develop the disease. In the US, nearly 60,000 individuals are newly diagnosed each year. Parkinsonism occurs in sporadic idiopathic form in many middle-aged and elderly individuals and is one of the most common neurodegenerative diseases. It is estimated to occur in 1–2% of individuals over age 65. Dopaminergic neurons and dopamine receptors are steadily lost with age in the basal ganglia in normal individuals, and an acceleration of these losses apparently precipitates parkinsonism. Symptoms appear when 60–80% of the nigrostriatal dopaminergic neurons degenerate.
Parkinsonism is also seen as a complication of treatment with the phenothiazine group of tranquilizer drugs and other drugs that block dopaminergic D2 receptors. It can be produced in rapid and dramatic form by injection of 1-methyl-4-phenyl-1,2,5,6-tetrahydropyridine (MPTP). This effect was discovered by chance when a drug dealer in northern California supplied some of his clients with a homemade preparation of synthetic heroin that contained MPTP. MPTP is a prodrug that is metabolized in astrocytes by the enzyme monoamine oxidase (MOA-B) to produce a potent oxidant, 1-methyl-4-phenylpyridinium (MPP+). In rodents, MPP+ is rapidly removed from the brain, but in primates it is removed more slowly and is taken up by the dopamine transporter into dopaminergic neurons in the substantia nigra, which it destroys without affecting other dopaminergic neurons to any appreciable degree. Consequently, MPTP can be used to produce parkinsonism in monkeys, and its availability has accelerated research on the function of the basal ganglia.
THERAPEUTIC HIGHLIGHTS
There is no cure for Parkinson disease, and drug therapies are designed to treat the symptoms. Sinemet, which is a combination of levodopa (L-dopa) and carbidopa, is the most commonly used drug for the treatment of Parkinson disease. The addition of carbidopa to L-dopa increases its effectiveness and prevents the conversion of L-DOPA to dopamine in the periphery and thus reduces some of the adverse side effects of L-dopa (nausea, vomiting, and cardiac rhythm disturbances). Dopamine agonists have also proven effective in some patients with Parkinson disease; these include apomorphine, bromocriptine, pramipexole, and ropinirole. Taken in combination with levodopa, cathechol-O-methyltransferase (COMT) inhibitors (eg, entacapone) are another class of drugs used to treat this disease. They act by blocking the breakdown of L-dopa, allowing more of it to reach the brain to increase the level of dopamine. MAO-B inhibitors (eg, selegiline) also prevent the breakdown of dopamine. They can be given soon after diagnosis and delay the need for levodopa.
In December 2010, investigators at Southern Methodist University and The University of Texas at Dallas reported that they have identified a family of small molecules that shows promise in protecting brain cell damage in diseases such as Parkinson, Alzheimer, and Huntington. This would be a major step forward as the first drug that would act as a neuroprotective agent to stop brain cell death as opposed to merely treating symptoms of the neurological disease.
The U.S. Food and Drug Administration has approved the use of deep brain stimulation (DBS) as a method for treating Parkinson disease. DBS reduces the amount of L-dopa patients need and thus reduce its adverse side effects (eg, involuntary movements called dyskinesias). DBS has been associated with reducing tremors, slowness of movements, and gait problems in some patients. Surgical treatments in general are reserved for those who have exhausted drug therapies or who have not responded favorably to them. Lesions in GPi (pallidotomy) or in the subthalamic nucleus (thalamotomy) have been performed to help restore the output balance of the basal ganglia toward normal (see Figure 12–17). Another surgical approach is to implant dopamine-secreting tissue in or near the basal ganglia. Transplants of the patient’s own adrenal medullary tissue or carotid body works for a while, apparently by functioning as a sort of dopamine minipump, but long-term results have been disappointing. Results with transplantation of fetal striatal tissue have been better, and there is evidence that the transplanted cells not only survive but make appropriate connections in the host’s basal ganglia. However, some patients with transplants develop dyskinesias due to excessive levels of dopamine.
The hypokinetic features of Parkinson disease are akinesia and bradykinesia, and the hyperkinetic features are cogwheel rigidity and tremor at rest. The absence of motor activity and the difficulty in initiating voluntary movements are striking. There is a decrease in the normal, unconscious movements such as swinging of the arms during walking, the panorama of facial expressions related to the emotional content of thought and speech, and the multiple “fidgety” actions and gestures that occur in all of us. The rigidity is different from spasticity because motor neuron discharge increases to both the agonist and antagonist muscles. Passive motion of an extremity meets with a plastic, dead-feeling resistance that has been likened to bending a lead pipe and is therefore called lead pipe rigidity. Sometimes a series of “catches” takes place during passive motion (cogwheel rigidity), but the sudden loss of resistance seen in a spastic extremity is absent. The tremor, which is present at rest and disappears with activity, is due to regular, alternating 8-Hz contractions of antagonistic muscles.
A current view of the pathogenesis of the movement disorders in Parkinson disease is shown in Figure 12–16. In normal individuals, basal ganglia output is inhibitory via GABAergic nerve fibers. The dopaminergic neurons that project from the substantia nigra to the putamen normally have two effects: they stimulate the D1 dopamine receptors, which inhibit GPi via direct GABAergic receptors, and they inhibit D2 receptors, which also inhibit the GPi. In addition, the inhibition reduces the excitatory discharge from the subthalamic nucleus to the GPi. This balance between inhibition and excitation somehow maintains normal motor function. In Parkinson disease, the dopaminergic input to the putamen is lost. This results in decreased inhibition and increased excitation from the subthalamic nucleus to the GPi. The overall increase in inhibitory output to the thalamus and brain stem disorganizes movement.
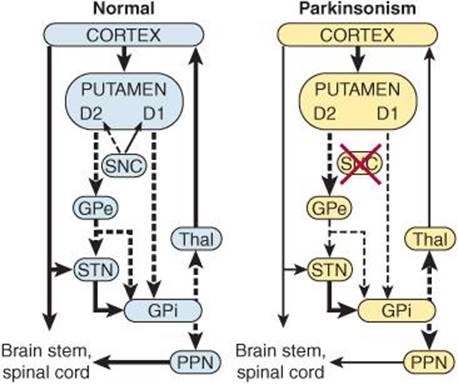
FIGURE 12–16 Probable basal ganglia-thalamocortical circuitry in Parkinson disease. Solid arrows indicate excitatory outputs and dashed arrows inhibitory outputs. The strength of each output is indicated by the width of the arrow. GPe, external segment of the globus pallidus; GPi, internal segment of the globus pallidus; PPN, pedunculopontine nuclei; SNC, pars compacta of the substantia nigra; STN, subthalamic nucleus; Thal, thalamus. See text for details. (Modified from Grafton SC, DeLong M: Tracing the brain circuitry with functional imaging. Nat Med 1997;3:602.)
Familial cases of Parkinson disease occur, but these are uncommon. The genes for at least five proteins can be mutated. These proteins appear to be involved in ubiquitination. Two of the proteins, α-synuclein and barkin, interact and are found in Lewy bodies. The Lewy bodies are inclusion bodies in neurons that occur in all forms of Parkinson disease. However, the significance of these findings is still unsettled.
An important consideration in Parkinson disease is the balance between the excitatory discharge of cholinergic interneurons and the inhibitory dopaminergic input in the striatum. Some improvement is produced by decreasing the cholinergic influence with anticholinergic drugs. More dramatic improvement is produced by administration of L-dopa (levodopa). Unlike dopamine, this dopamine precursor crosses the blood–brain barrier and helps repair the dopamine deficiency. However, the degeneration of these neurons continues and in 5–7 years the beneficial effects of L-dopa disappear.
CEREBELLUM
The cerebellum sits astride the main sensory and motor systems in the brain stem (Figure 12–17). The medial vermis and lateral cerebellar hemispheres are more extensively folded and fissured than the cerebral cortex. The cerebellum weighs only 10% as much as the cerebral cortex, but its surface area is about 75% of that of the cerebral cortex. Anatomically, the cerebellum is divided into three parts by two transverse fissures. The posterolateral fissure separates the medial nodulus and the lateral flocculus on either side from the rest of the cerebellum, and the primary fissure divides the remainder into an anterior and a posterior lobe. Lesser fissures divide the vermis into smaller sections, so that it contains 10 primary lobules numbered I–X from superior to inferior.
FIGURE 12–17 A midsagittal section through the cerebellum. The medial vermis and lateral cerebellar hemispheres have many narrow, ridge-like folds called folia. Although not shown, the cerebellum is connected to the brain stem by three pairs of peduncles (superior, middle, and inferior). (Reproduced with permission, from Waxman SG: Clinical Neuroanatomy, 26th ed. McGraw-Hill, 2010.)
The cerebellum is connected to the brain stem by three pairs of peduncles that are located above and around the fourth ventricle. The superior cerebellar peduncle includes fibers from deep cerebellar nuclei that project to the brain stem, red nucleus, and thalamus. The middle cerebellar peduncle contains only afferent fibers from the contralateral pontine nuclei, and the inferior cerebellar peduncle a mixture of afferent fibers from the brain stem and spinal cord and efferent fibers to the vestibular nuclei.
The cerebellum has an external cerebellar cortex separated by white matter from the deep cerebellar nuclei. The middle and inferior cerebellar peduncles carry afferent fibers into the cerebellum where they are called mossy and climbing fibers. These fibers emit collaterals to the deep nuclei and pass to the cortex. There are four deep nuclei: the dentate, the globose, the emboliform, and the fastigial nuclei. The globose and the emboliform nuclei are sometimes lumped together as the interpositus nucleus.
ORGANIZATION OF THE CEREBELLUM
The cerebellar cortex has three layers: an external molecular layer, a Purkinje cell layer that is only one cell thick, and an internal granular layer. There are five types of neurons in the cortex: Purkinje, granule, basket, stellate, and Golgi cells (Figure 12–18). The Purkinje cells are among the biggest neurons in the CNS. They have very extensive dendritic arbors that extend throughout the molecular layer. Their axons, which are the only output from the cerebellar cortex, project to the deep cerebellar nuclei, especially the dentate nucleus, where they form inhibitory synapses. They also make inhibitory connections with neurons in the vestibular nuclei.
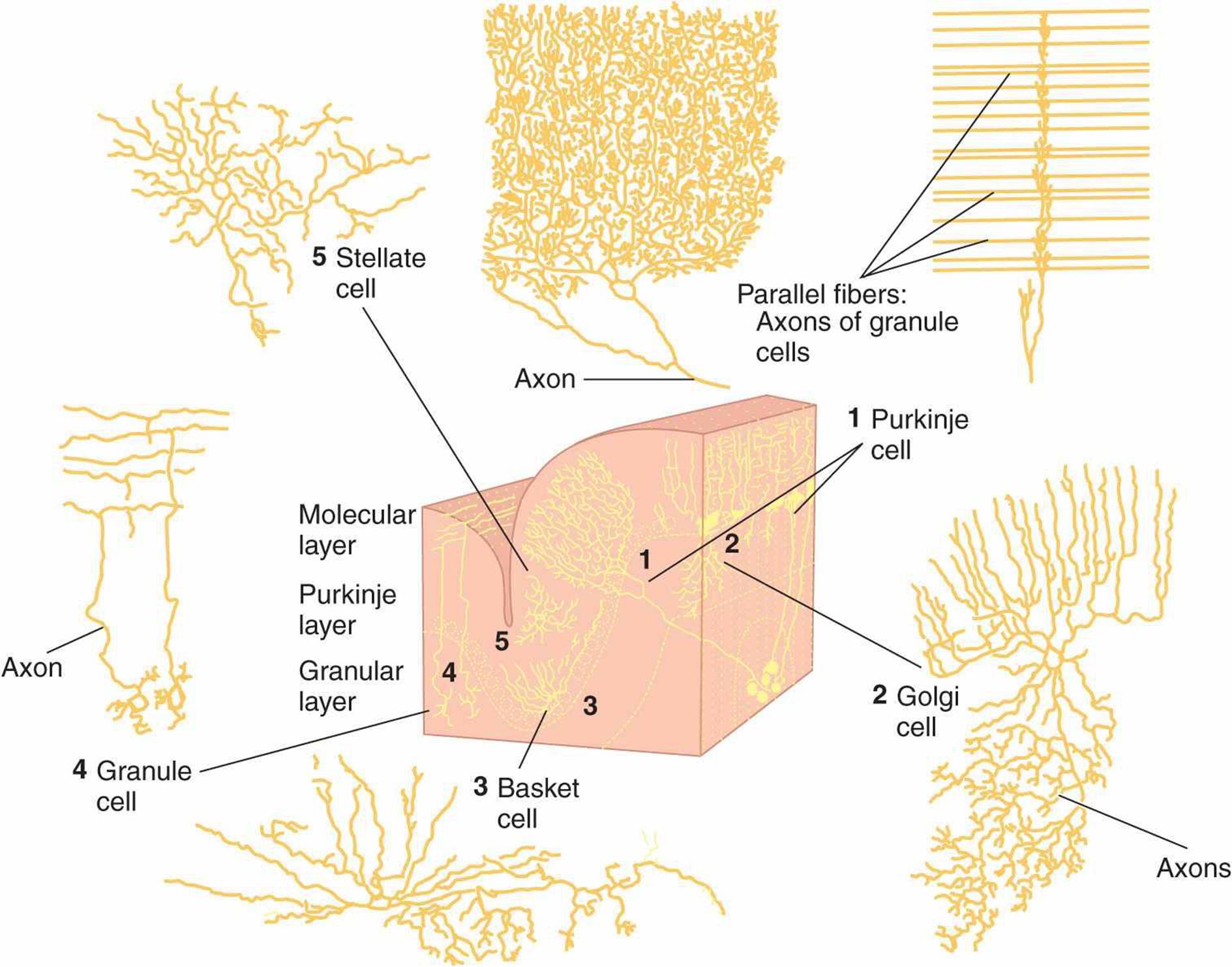
FIGURE 12–18 Location and structure of five neuronal types in the cerebellar cortex. Drawings are based on Golgi-stained preparations. Purkinje cells (1) have processes aligned in one plane; their axons are the only output from the cerebellum. Axons of granule cells (4) traverse and make connections with Purkinje cell processes in molecular layer. Golgi (2), basket (3), and stellate (5) cells have characteristic positions, shapes, branching patterns, and synaptic connections. (For 1 and 2, After Ramon y Cajal S: Histologie du Systeme Nerveux II., C.S.I.C. Madrid; For 3-5, After Palay SL, Chan-Palay V: Cerebellar Cortex. Berlin: Springer-Verlag, 1974.)
The cerebellar granule cells, whose cell bodies are in the granular layer, receive excitatory input from the mossy fibers and innervate the Purkinje cells (Figure 12–19). Each sends an axon to the molecular layer, where the axon bifurcates to form a T. The branches of the T are straight and run long distances; thus, they are called parallel fibers. The dendritic trees of the Purkinje cells are markedly flattened and oriented at right angles to the parallel fibers. The parallel fibers form excitatory synapses on the dendrites of many Purkinje cells, and the parallel fibers and Purkinje dendritic trees form a grid of remarkably regular proportions.
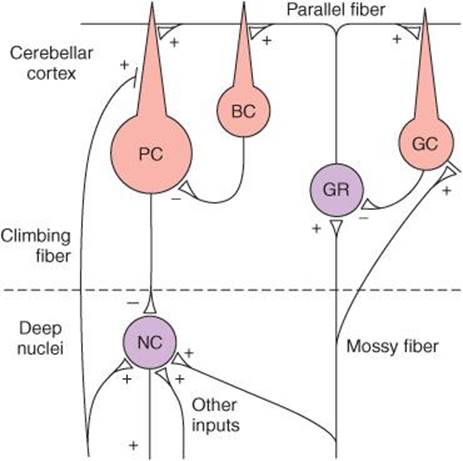
FIGURE 12–19 Diagram of neural connections in the cerebellum. Plus (+) and minus (–) signs indicate whether endings are excitatory or inhibitory. BC, basket cell; GC, Golgi cell; GR, granule cell; NC, cell in deep nucleus; PC, Purkinje cell. Note that PCs and BCs are inhibitory. The connections of the stellate cells, which are not shown, are similar to those of the basket cells, except that they end for the most part on Purkinje cell dendrites.
The other three types of neurons in the cerebellar cortex are inhibitory interneurons. Basket cells (Figure 12–18) are located in the molecular layer. They receive excitatory input from the parallel fibers and each projects to many Purkinje cells (Figure 12–19). Their axons form a basket around the cell body and axon hillock of each Purkinje cell they innervate. Stellate cells are similar to the basket cells but are located in the more superficial molecular layer. Golgi cells are located in the granular layer. Their dendrites, which project into the molecular layer, receive excitatory input from the parallel fibers (Figure 12–19). Their cell bodies receive excitatory input via collaterals from the incoming mossy fibers. Their axons project to the dendrites of the granule cells where they form an inhibitory synapse.
As already mentioned, the two main inputs to the cerebellar cortex are climbing fibers and mossy fibers. Both are excitatory (Figure 12–19). The climbing fibers come from a single source, the inferior olivary nuclei. Each projects to the primary dendrites of a Purkinje cell, around which it entwines like a climbing plant. Proprioceptive input to the inferior olivary nuclei comes from all over the body. On the other hand, the mossy fibers provide direct proprioceptive input from all parts of the body plus input from the cerebral cortex via the pontine nuclei to the cerebellar cortex. They end on the dendrites of granule cells in complex synaptic groupings called glomeruli. The glomeruli also contain the inhibitory endings of the Golgi cells mentioned above.
The fundamental circuits of the cerebellar cortex are thus relatively simple (Figure 12–19). Climbing fiber inputs exert a strong excitatory effect on single Purkinje cells, whereas mossy fiber inputs exert a weak excitatory effect on many Purkinje cells via the granule cells. The basket and stellate cells are also excited by granule cells via their parallel fibers; and the basket and stellate cells, in turn, inhibit the Purkinje cells (feedforward inhibition). Golgi cells are excited by the mossy fiber collaterals and parallel fibers, and they inhibit transmission from mossy fibers to granule cells. The neurotransmitter released by the stellate, basket, Golgi, and Purkinje cells is GABA, whereas the granule cells release glutamate. GABA acts via GABAA receptors, but the combinations of subunits in these receptors vary from one cell type to the next. The granule cell is unique in that it appears to be the only type of neuron in the CNS that has a GABAA receptor containing the α6 subunit.
The output of the Purkinje cells is in turn inhibitory to the deep cerebellar nuclei. As noted above, these nuclei also receive excitatory inputs via collaterals from the mossy and climbing fibers. It is interesting, in view of their inhibitory Purkinje cell input, that the output of the deep cerebellar nuclei to the brain stem and thalamus is always excitatory. Thus, almost all the cerebellar circuitry seems to be concerned solely with modulating or timing the excitatory output of the deep cerebellar nuclei to the brain stem and thalamus. The primary afferent systems that converge to form the mossy fiber or climbing fiber input to the cerebellum are summarized in Table 12–2.
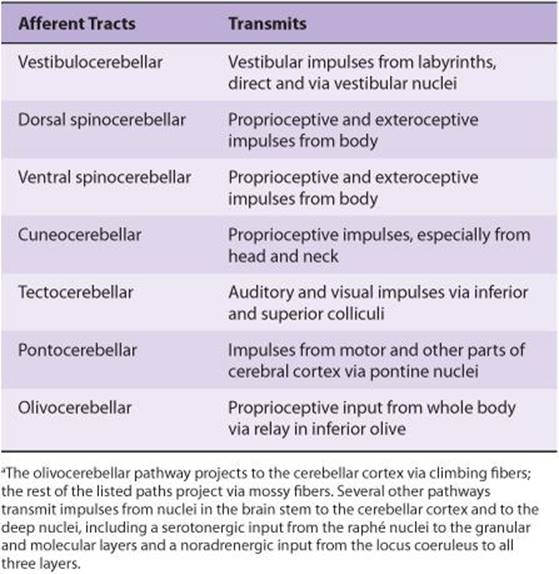
TABLE 12–2. Function of principal afferent systems to the cerebellum.a
FUNCTIONAL DIVISIONS
From a functional point of view, the cerebellum is divided into three parts (Figure 12–20). The nodulus in the vermis and the flanking flocculus in the hemisphere on each side form the vestibulocerebellum (or flocculonodular lobe). This lobe, which is phylogenetically the oldest part of the cerebellum, has vestibular connections and is concerned with equilibrium and eye movements. The rest of the vermis and the adjacent medial portions of the hemispheres form the spinocerebellum, the region that receives proprioceptive input from the body as well as a copy of the “motor plan” from the motor cortex. By comparing plan with performance, it smoothes and coordinates movements that are ongoing. The vermis projects to the brain stem area concerned with control of axial and proximal limb muscles (medial brain stem pathways), whereas the hemispheres of the spinocerebellum project to the brain stem areas concerned with control of distal limb muscles (lateral brain stem pathways). The lateral portions of the cerebellar hemispheres are called the cerebrocerebellum. They are the newest from a phylogenetic point of view, reaching their greatest development in humans. They interact with the motor cortex in planning and programming movements.
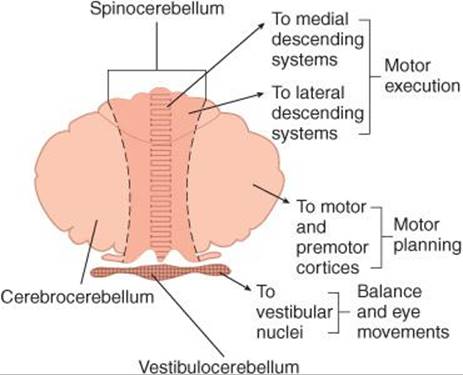
FIGURE 12–20 Three functional divisions of the cerebellum. The nodulus in the vermis and the flanking flocculus in the hemisphere on each side form the vestibulocerebellum which has vestibular connections and is concerned with equilibrium and eye movements. The rest of the vermis and the adjacent medial portions of the hemispheres form the spinocerebellum, the region that receives proprioceptive input from the body as well as a copy of the “motor plan” from the motor cortex. The lateral portions of the cerebellar hemispheres are called the cerebrocerebellum which interacts with the motor cortex in planning and programming movements. (Modified from Kandel ER, Schwartz JH, Jessell TM [editors]: Principles of Neural Science, 4th ed. McGraw-Hill, 2000.)
Most of the vestibulocerebellar output passes directly to the brain stem, but the rest of the cerebellar cortex projects to the deep nuclei, which in turn project to the brain stem. The deep nuclei provide the only output for the spinocerebellum and the cerebrocerebellum. The medial portion of the spinocerebellum projects to the fastigial nuclei and from there to the brain stem. The adjacent hemispheric portions of the spinocerebellum project to the emboliform and globose nuclei and from there to the brain stem. The cerebrocerebellum projects to the dentate nucleus and from there either directly or indirectly to the ventrolateral nucleus of the thalamus.
CEREBELLAR DISEASE
Damage to the cerebellum leads to several characteristic abnormalities, including hypotonia, ataxia, and intention tremor. Motor abnormalities associated with cerebellar damage vary depending on the region involved. Figure 12–21 illustrates some of these abnormalities. Additional information is provided in Clinical Box 12–9.
CLINICAL BOX 12–9
Cerebellar Disease
Most abnormalities associated with damage to the cerebellum are apparent during movement. The marked ataxia is characterized as incoordination due to errors in the rate, range, force, and direction of movement. Ataxia is manifest not only in the wide-based, unsteady, “drunken” gait of patients, but also in defects of the skilled movements involved in the production of speech, so that slurred, scanning speech results. Many types of ataxia are hereditary, including Friedreich’s ataxia and Machado-Joseph disease. There is no cure for hereditary ataxias. Voluntary movements are also highly abnormal when the cerebellum is damaged. For example, attempting to touch an object with a finger results in overshooting. This dysmetria or past-pointing promptly initiates a gross corrective action, but the correction overshoots to the other side, and the finger oscillates back and forth. This oscillation is called an intention tremor. Another characteristic of cerebellar disease is inability to “put on the brakes” to stop movement promptly. Normally, for example, flexion of the forearm against resistance is quickly checked when the resistance force is suddenly broken off. The patient with cerebellar disease cannot stop the movement of the limb, and the forearm flies backward in a wide arc. This abnormal response is known as the rebound phenomenon. This is one of the important reasons these patients show dysdiadochokinesia, the inability to perform rapidly alternating opposite movements such as repeated pronation and supination of the hands. Finally, patients with cerebellar disease have difficulty performing actions that involve simultaneous motion at more than one joint. They dissect such movements and carry them out one joint at a time, a phenomenon known as decomposition of movement. Other signs of cerebellar deficit in humans point to the importance of the cerebellum in the control of movement.
Motor abnormalities associated with cerebellar damage vary depending on the region involved. The major dysfunction seen after damage to the vestibulocerebellum is ataxia, dysequilibrium, and nystagmus. Damage to the vermis and fastigial nucleus (part of the spinocerebellum) leads to disturbances in control of axial and trunk muscles during attempted antigravity postures and scanning speech. Degeneration of this portion of the cerebellum can result from thiamine deficiency in alcoholics or malnourished individuals. The major dysfunction seen after damage to the cerebrocerebellum is delays in initiating movements and decomposition of movement.
THERAPEUTIC HIGHLIGHTS
Management of ataxia is primarily supportive; it often includes physical, occupational, and speech therapy. Attempts to identify effective drug therapies have met with little success. Deep brain stimulation of the ventral intermediate nucleus of the thalamus may reduce cerebellar tremor, but it is less effective in reducing ataxia. There is some evidence that a deficiency in coenzyme Q10 (CoQ10) contributes to the abnormalities seen in some forms of familial ataxia. If low levels of CoQ10 are detected, treatment to replace the missing CoQ10 has been shown to be beneficial.
FIGURE 12–21 Typical defects associated with cerebellar disease. A) Lesion of the right cerebellar hemisphere delays initiation of movement. The patient is told to clench both hands simultaneously; right hand clenches later than left (shown by recordings from a pressure bulb transducer squeezed by the patient). B) Dysmetria and decomposition of movement shown by patient moving his arm from a raised position to his nose. Tremor increases on approaching the nose. C) Dysdiadochokinesia occurs in the abnormal position trace of hand and forearm as a cerebellar subject tries alternately to pronate and supinate forearm while flexing and extending elbow as rapidly as possible. (From Kandel ER, Schwartz JH, Jessell TM [editors]: Principles of Neural Science, 4th ed. McGraw-Hill, 2000.)
THE CEREBELLUM & LEARNING
The cerebellum is concerned with learned adjustments that make coordination easier when a given task is performed over and over. As a motor task is learned, activity in the brain shifts from the prefrontal areas to the parietal and motor cortex and the cerebellum. The basis of the learning in the cerebellum is probably the input via the olivary nuclei. The mossy fiber–granule cell–Purkinje cell pathway is highly divergent, allowing an individual Purkinje cell to receive input from many mossy fibers arising from different regions. In contrast, a Purkinje cell receives input from a single climbing fiber but it makes 2000–3000 synapses on it. Climbing fiber activation produces a large, complex spike in the Purkinje cell, and this spike produces long-term modification of the pattern of mossy fiber input to that particular Purkinje cell. Climbing fiber activity is increased when a new movement is being learned, and selective lesions of the olivary complex abolish the ability to produce long-term adjustments in certain motor responses.
CHAPTER SUMMARY
![]() A muscle spindle is a group of specialized intrafusal muscle fibers with contractile polar ends and a noncontractile center that is located in parallel to the extrafusal muscle fibers and is innervated by types Ia and II afferent fibers and efferent γ-motor neurons. Muscle stretch activates the muscle spindle to initiate reflex contraction of the extrafusal muscle fibers in the same muscle (stretch reflex).
A muscle spindle is a group of specialized intrafusal muscle fibers with contractile polar ends and a noncontractile center that is located in parallel to the extrafusal muscle fibers and is innervated by types Ia and II afferent fibers and efferent γ-motor neurons. Muscle stretch activates the muscle spindle to initiate reflex contraction of the extrafusal muscle fibers in the same muscle (stretch reflex).
![]() A Golgi tendon organ is a netlike collection of knobby nerve endings among the fascicles of a tendon that is located in series with extrafusal muscle fibers and innervated by type Ib afferents. They are stimulated by both passive stretch and active contraction of the muscle to relax the muscle (inverse stretch reflex) and function as a transducer to regulate muscle force.
A Golgi tendon organ is a netlike collection of knobby nerve endings among the fascicles of a tendon that is located in series with extrafusal muscle fibers and innervated by type Ib afferents. They are stimulated by both passive stretch and active contraction of the muscle to relax the muscle (inverse stretch reflex) and function as a transducer to regulate muscle force.
![]() The flexor withdrawal reflex is a polysynaptic reflex that is initiated by nociceptive stimuli; it can serve as a protective mechanism to present further injury.
The flexor withdrawal reflex is a polysynaptic reflex that is initiated by nociceptive stimuli; it can serve as a protective mechanism to present further injury.
![]() Spinal cord transection is followed by a period of spinal shock during which all spinal reflex responses are profoundly depressed. In humans, recovery begins about 2 weeks after the injury.
Spinal cord transection is followed by a period of spinal shock during which all spinal reflex responses are profoundly depressed. In humans, recovery begins about 2 weeks after the injury.
![]() The supplementary cortex, basal ganglia, and cerebellum participate in the planning of skilled movements. Commands from the primary motor cortex and other cortical regions are relayed via the corticospinal and corticobulbar tracts to spinal and brainstem motor neurons. Cortical areas and motor pathways descending from the cortex are somatotopically organized.
The supplementary cortex, basal ganglia, and cerebellum participate in the planning of skilled movements. Commands from the primary motor cortex and other cortical regions are relayed via the corticospinal and corticobulbar tracts to spinal and brainstem motor neurons. Cortical areas and motor pathways descending from the cortex are somatotopically organized.
![]() The ventral corticospinal tract and medial descending brain stem pathways (tectospinal, reticulospinal, and vestibulospinal tracts) regulate proximal muscles and posture. The lateral corticospinal and rubrospinal tracts control distal limb muscles for fine motor control and skilled voluntary movements.
The ventral corticospinal tract and medial descending brain stem pathways (tectospinal, reticulospinal, and vestibulospinal tracts) regulate proximal muscles and posture. The lateral corticospinal and rubrospinal tracts control distal limb muscles for fine motor control and skilled voluntary movements.
![]() Decerebrate rigidity leads to hyperactivity in extensor muscles in all four extremities; it is actually spasticity due to facilitation of the stretch reflex. It resembles what is seen with uncal herniation due to a supratentorial lesion. Decorticate rigidity is flexion of the upper extremities at the elbow and extensor hyperactivity in the lower extremities. It occurs on the hemiplegic side after hemorrhage or thrombosis in the internal capsule.
Decerebrate rigidity leads to hyperactivity in extensor muscles in all four extremities; it is actually spasticity due to facilitation of the stretch reflex. It resembles what is seen with uncal herniation due to a supratentorial lesion. Decorticate rigidity is flexion of the upper extremities at the elbow and extensor hyperactivity in the lower extremities. It occurs on the hemiplegic side after hemorrhage or thrombosis in the internal capsule.
![]() The basal ganglia include the caudate nucleus, putamen, globus pallidus, subthalamic nucleus, and substantia nigra. The connections between the parts of the basal ganglia include a dopaminergic nigrostriatal projection from the substantia nigra to the striatum and a GABAergic projection from the striatum to substantia nigra.
The basal ganglia include the caudate nucleus, putamen, globus pallidus, subthalamic nucleus, and substantia nigra. The connections between the parts of the basal ganglia include a dopaminergic nigrostriatal projection from the substantia nigra to the striatum and a GABAergic projection from the striatum to substantia nigra.
![]() Parkinson disease is due to degeneration of the nigrostriatal dopaminergic neurons and is characterized by akinesia, bradykinesia, cogwheel rigidity, and tremor at rest. Huntington disease is characterized by choreiform movements due to the loss of the GABAergic inhibitory pathway to the globus pallidus.
Parkinson disease is due to degeneration of the nigrostriatal dopaminergic neurons and is characterized by akinesia, bradykinesia, cogwheel rigidity, and tremor at rest. Huntington disease is characterized by choreiform movements due to the loss of the GABAergic inhibitory pathway to the globus pallidus.
![]() The cerebellar cortex contains five types of neurons: Purkinje, granule, basket, stellate, and Golgi cells. The two main inputs to the cerebellar cortex are climbing fibers and mossy fibers. Purkinje cells are the only output from the cerebellar cortex, and they generally project to the deep nuclei. Damage to the cerebellum leads to several characteristic abnormalities, including hypotonia, ataxia, and intention tremor.
The cerebellar cortex contains five types of neurons: Purkinje, granule, basket, stellate, and Golgi cells. The two main inputs to the cerebellar cortex are climbing fibers and mossy fibers. Purkinje cells are the only output from the cerebellar cortex, and they generally project to the deep nuclei. Damage to the cerebellum leads to several characteristic abnormalities, including hypotonia, ataxia, and intention tremor.
MULTIPLE-CHOICE QUESTIONS
For all questions, select the single best answer unless otherwise directed.
1. When dynamic γ-motor neurons are activated at the same time as α-motor neurons to muscle,
A. prompt inhibition of discharge in spindle Ia afferents takes place.
B. clonus is likely to occur.
C. the muscle will not contract.
D. the number of impulses in spindle Ia afferents is smaller than when α discharge alone is increased.
E. the number of impulses in spindle Ia afferents is greater than when α discharge alone is increased.
2. The inverse stretch reflex
A. occurs when Ia spindle afferents are inhibited.
B. is a monosynaptic reflex initiated by activation of the Golgi tendon organ.
C. is a disynaptic reflex with a single interneuron inserted between the afferent and efferent limbs.
D. is a polysynaptic reflex with many interneurons inserted between the afferent and efferent limbs.
E. uses type II afferent fibers from the Golgi tendon organ.
3. Withdrawal reflexes are not
A. initiated by nociceptive stimuli.
B. prepotent.
C. prolonged if the stimulus is strong.
D. an example of a flexor reflex.
E. accompanied by the same response on both sides of the body.
4. While exercising, a 42-year-old female developed sudden onset of tingling in her right leg and an inability to control movement in that limb. A neurological exam showed a hyperactive knee jerk reflex and a positive Babinski sign. Which of the following is not characteristic of a reflex?
A. Reflexes can be modified by impulses from various parts of the CNS.
B. Reflexes may involve simultaneous contraction of some muscles and relaxation of others.
C. Reflexes are chronically suppressed after spinal cord transection.
D. Reflexes involve transmission across at least one synapse.
E. Reflexes often occur without conscious perception.
5. Increased neural activity before a skilled voluntary movement is first seen in the
A. spinal motor neurons.
B. precentral motor cortex.
C. midbrain.
D. cerebellum.
E. cortical association areas.
6. A 58-year-old woman was brought to the emergency room of her local hospital because of a sudden change of consciousness. All four limbs were extended, suggestive of decerebrate rigidity. A brain CT showed a rostral pontine hemorrhage. Which of the following describes components of the central pathway responsible for control of posture?
A. The tectospinal pathway terminates on neurons in the dorsolateral area of the spinal ventral horn that innervate limb muscles.
B. The medullary reticulospinal pathway terminates on neurons in the ventromedial area of the spinal ventral horn that innervate axial and proximal muscles.
C. The pontine reticulospinal pathway terminates on neurons in the dorsomedial area of the spinal ventral horn that innervate limb muscles.
D. The medial vestibular pathway terminates on neurons in the dorsomedial area of the spinal ventral horn that innervate axial and proximal muscles.
E. The lateral vestibular pathway terminates on neurons in the dorsolateral area of the spinal ventral horn that innervate axial and proximal muscles.
7. A 38-year-old female had been diagnosed with a metastatic brain tumor. She was brought to the emergency room of her local hospital because of irregular breathing and progressive loss of consciousness. She also showed signs of decerebrate posturing. Which of the following is not true about decerebrate rigidity?
A. It involves hyperactivity in extensor muscles of all four limbs.
B. The excitatory input from the reticulospinal pathway activates γ-motor neurons which indirectly activate α-motor neurons.
C. It is actually a type of spasticity due to inhibition of the stretch reflex.
D. It resembles what ensues after uncal herniation.
E. Lower extremities are extended with toes pointed inward.
8. Which of the following describes a connection between components of the basal ganglia?
A. The subthalamic nucleus releases glutamate to excite the globus pallidus, internal segment.
B. The substantia nigra pars reticulata releases dopamine to inhibit the striatum.
C. The substantia nigra pars compacta releases dopamine to excite the globus pallidus, external segment.
D. The striatum releases acetylcholine to excite the substantia nigra pars reticulata.
E. The globus pallidus, external segment releases glutamate to excite the striatum.
9. A 60-year old male was diagnosed 15 years ago with Parkinson disease. He has been taking Sinemet and, until recently, has been able to continue to work and help with routine jobs around the house. Now his tremor and rigidity interfere with these activities. His physician has suggested that he undergo deep brain stimulation therapy. The therapeutic effect of L-dopa in patients with Parkinson disease eventually wears off because
A. antibodies to dopamine receptors develop.
B. inhibitory pathways grow into the basal ganglia from the frontal lobe.
C. there is an increase in circulating α-synuclein.
D. the normal action of nerve growth factor (NGF) is disrupted.
E. the dopaminergic neurons in the substantia nigra continue to degenerate.
10. An 8-year-old girl was brought to her pediatrician because her parents noted frequent episodes of gait unsteadiness and speech difficulties. Her mother was concerned because of a family history of Friedreich’s ataxia. Which of the following is a correct description of connections involving cerebellar neurons?
A. Basket cells release glutamate to activate Purkinje cells.
B. Climbing fiber inputs exert a strong excitatory effect on Purkinje cells, and mossy fiber inputs exert a strong inhibitory effect on Purkinje cells.
C. Granule cells release glutamate to excite basket cells and stellate cells.
D. The axons of Purkinje cells are the sole output of the cerebellar cortex, and they release glutamate to excite the deep cerebellar nuclei.
E. Golgi cells are inhibited by mossy fiber collaterals.
11. After falling down a flight of stairs, a young woman is found to have partial loss of voluntary movement on the right side of her body and loss of pain and temperature sensation on the left side below the midthoracic region. It is probable that she has a lesion
A. transecting the left half of the spinal cord in the lumbar region.
B. transecting the left half of the spinal cord in the upper thoracic region.
C. transecting sensory and motor pathways on the right side of the pons.
D. transecting the right half of the spinal cord in the upper thoracic region.
E. transecting the dorsal half of the spinal cord in the upper thoracic region.
12. At the age of 30, a male postal worker reported weakness in his right leg. Within a year the weakness had spread to his entire right side. A neurological examination revealed flaccid paralysis, muscular atrophy, fasciculations, hypotonia, and hyporeflexia of muscles in the right arm and leg. Sensory and cognitive function tests were normal. Which of the following diagnosis is likely?
A. A large tumor in the left primary motor cortex.
B. A cerebral infarct in the region of the corona radiate.
C. A vestibulocerebellar tumor.
D. Damage to the basal ganglia.
E. Amyotrophic lateral sclerosis.
CHAPTER RESOURCES
Alexi T, Liu X-Z, Qu Y, et al: Neuroprotective strategies for basal ganglia degeneration: Parkinson’s and Huntington’s diseases. Prog Neurbiol 2000;60:409.
De Zeeuw CI, Strata P, Voogd J: The Cerebellum: From Structure to Control. Elsevier, 1997.
Ditunno JF Jr, Formal CF: Chronic spinal cord injury. N Engl J Med 1994; 330:550.
Graybiel AM, Delong MR, Kitai ST: The Basal Ganglia VI. Springer, 2003.
Hunt CC: Mammalian muscle spindle: Peripheral mechanisms. Physiol Rev 1990;70: 643.
Jankowska E: Interneuronal relay in spinal pathways from proprioceptors. Prog Neurobiol 1992;38:335.
Jueptner M, Weiller C: A review of differences between basal ganglia and cerebellar control of movements as revealed by functional imaging studies. Brain 1998;121:1437.
Latash ML: Neurophysiological Basis of Movement, 2nd ed. Human Kinetics, 2008.
Lemon RN: Descending pathways in motor control. Annu Rev Neurosci 2008;31:195.
Lundberg A: Multisensory control of spinal reflex pathways. Prog Brain Res 1979;50:11.
Manto MU, Pandolfo M: The Cerebellum and its Disorders. Cambridge University Press, 2001.
Matyas F, Sreenivasan V, Marbach F, Wacongne C, Barsy B, Mateo C, Aronoff R, Petersen CCH: Motor control of sensory cortex. Science 2010;26:1240.
McDonald JW, Liu X-Z, Qu Y, et al: Transplanted embryonic stem cells survive, differentiate and promote recovery in injured rat spinal cord. Nature Med 1999;5:1410.
Nudo RJ: Postinfarct cortical plasticity and behavioral recovery. Stroke 2007;38:840.
Ramer LM, Ramer MS, Steeves JD: Setting the stage for functional repair of spinal cord injuries: a cast of thousands. Spinal Cord 2005;43:134.
Stein RB, Thompson AK: Muscle reflexes and motion: How, what, and why? Exerc Sport Sci Rev 2006;34:145.