Sender Herschorn1 and Blayne K. Welk1
(1)
Division of Urology, Sunnybrook Health Sciences Centre, University of Toronto, 2075 Bayview Avenue, Suite MG408, Toronto, ON, Canada, M4N 3M5
Sender Herschorn
Email: s.herschorn@utoronto.ca
Abstract
Bladder augmentation with an ileal patch was first described by Von Mickulicz [1]. Different gastrointestinal segments were subsequently reported, colon by Lemoine in 1912 [2], sigmoid by Bisgard [3], cecum by Couvelaire [4], and stomach by Leong [5]. In 1950, Couvelaire began augmentation cystoplasty to treat contracted bladders as a result of tuberculosis, and the technique started to gain acceptance [4]. Other attempts using organic tissues such as peritoneum, omentum, human dura, skin, pericardium, placenta, gallbladder, free fascial grafts, and preserved bladder tissue were unsuccessful as were efforts using synthetic materials [6]. In 1959, Goodwin described the modern operative technique of using a detubularized ileal patch [7].
Introduction
Bladder augmentation with an ileal patch was first described by Von Mickulicz [1]. Different gastrointestinal segments were subsequently reported, colon by Lemoine in 1912 [2], sigmoid by Bisgard [3], cecum by Couvelaire [4], and stomach by Leong [5]. In 1950, Couvelaire began augmentation cystoplasty to treat contracted bladders as a result of tuberculosis, and the technique started to gain acceptance [4]. Other attempts using organic tissues such as peritoneum, omentum, human dura, skin, pericardium, placenta, gallbladder, free fascial grafts, and preserved bladder tissue were unsuccessful as were efforts using synthetic materials [6]. In 1959, Goodwin described the modern operative technique of using a detubularized ileal patch [7].
Bladder augmentation is often done in conjunction with other surgical procedures, such as creation of a continent stoma, or bladder outlet procedures to reduce urinary incontinence. This chapter will outline the indications and techniques of bladder augmentation and focus on short- and long-term complications and their management.
Indications
In 1977, Smith et al. [8] reviewed augmentation cystoplasty and suggested that the procedure was “a successful long-term solution for patients with small contracted bladders of almost any etiology.” Table 16.1 lists the current indications.
Table 16.1
Indications for augmentation cystoplasty (usually with associated symptoms of urinary incontinence, high detrusor pressures, or renal dysfunction refractory to other management options)
|
Indication |
|
|
Congenital |
Myelodysplasia |
|
Posterior urethral valves |
|
|
Exstrophy/epispadias complex |
|
|
Acquired neurogenic bladder |
Spinal cord injury |
|
Multiple sclerosis |
|
|
Acquired non-neurogenic bladder |
Overactive bladder |
|
Infectious |
Tuberculosis |
|
Schistosomiasis |
|
|
Inflammatory |
Radiation cystitis (interstitial cystitis) |
|
Iatrogenic |
Intraoperative loss of bladder wall |
|
Urinary undiversion |
Congenital Conditions
Myelodysplasia, a form of spinal dysraphism, may lead to neurogenic bladder dysfunction. Approximately 1/3 of patients have sphincter dyssyngeria, and the urodynamic pattern often changes as the child ages [9]. The failure of conservative or medical therapy to adequately treat urinary incontinence, high detrusor leak point pressures, and renal dysfunction are indications for bladder augmentation. It has been estimated with data approximately 5% [10] to 30% [11] of patients with spina bifida may undergo an augmentation cystoplasty. Augmentation is often combined with other procedures such as a catheterizable abdominal stoma and bladder neck procedures or slings to increase urinary outlet resistance.
Posterior urethral valves in males can lead to bladder dysfunction and renal failure. Augmentation cystoplasty may be required prior to renal transplantation [12–15]. Patients with exstrophy/epispadias complex also require bladder augmentation when staged functional reconstruction is unsuccessful [16–19].
Other congenital anomalies include sacral agenesis, cloacal exstrophy, imperforate anus, and persistent urogenital sinus [20].
Acquired Neurogenic Bladder
Spinal cord injury can lead to severe detrusor overactivity, poor bladder compliance, and decreased capacity over time. The changes are frequently related to the level of injury. Suprasacral spinal cord lesions often lead to detrusor overactivity with sphincter dyssynergia. This antagonistic dysfunction of the bladder and the outlet can impair detrusor compliance, and over time lead to reduced bladder capacity [21]. Sacral spinal cord lesions often lead to detrusor areflexia with a fixed, nonrelaxing sphincter. Generally the bladder has normal compliance; however over time decreased compliance and reduced capacity can develop [21].
Bladder augmentation may be indicated if incontinence, high detrusor leak point pressures, severe autonomic dysreflexia, or renal dysfunction occur due to failure of the bladder to store urine at a low pressure. Usually augmentation is considered when other measures such as behavioral modifications, anticholinergics, intravesical botulinum toxin, or rarely anterior nerve root stimulation are ineffective [22–24].
Multiple sclerosis is another cause of neurogenic bladder dysfunction that may result in detrusor overactivity with sphincter dyssynergia [25]. Bladder dysfunction can worsen over time, and progressive neuromuscular deterioration can make intermittent self-catheterization difficult [26]. Medical therapy with anticholinergics and intravesical botulinum toxin is usually the preferred treatment. However, occasional cases may be amenable to augmentation cystoplasty.
Overactive Bladder
Overactive bladder is a syndrome or symptom complex of urinary urgency, with or without urgency incontinence, urinary frequency, and nocturia [27]. Bladder augmentation is a treatment of last resort for refractory symptoms associated with detrusor overactivity that cannot be controlled with behavioral therapy, anticholinergics, intravesical botulinum toxin, or sacral/peripheral neuromodulation [28].
Infection
Genitourinary tuberculosis occurs in 10–20% of patients with pulmonary tuberculosis [29]. Tuberculous cystitis causes velvety granulations, bladder ulceration, and bladder wall thickening and can progress to severely reduced bladder capacity [26]. Tuberculosis, once a common indication for augmentation [30], is now a rarity due to better therapies and decreased incidence in the developed world [31, 32].
Schistosomiasis, an endemic parasitic infection found primarily in the Middle East and Africa, may cause bladder wall fibrosis due to granulomatous inflammation [33]. Reduced bladder capacity may be improved by augmentation [34].
Inflammatory Causes
Radiation changes may follow external beam radiation therapy for treatment of pelvic malignancy. Acute cystitis symptoms usually resolve within a few months, however occasionally seen bladder wall fibrosis may reduce bladder capacity and impair function [35]. Patient comorbidities and further oncologic treatment may limit augmentation in this group [36].
Bladder augmentation has been used as treatment for interstitial cystitis in patients with contracted, small capacity bladders [37]. However, augmentation has shown only modest success as treatment for pain associated with interstitial cystitis [26, 38]. Its use in this population is controversial [26, 39–41].
Iatrogenic
Augmentation cystoplasty may be necessary in patients with significant loss of the bladder wall due to surgical resection. This may be from the resection of locally advanced nonurologic cancer, or benign bladder resections. For patients with previous urinary diversion who did not undergo a cystectomy, redirecting the ureters to an augmentation cystoplasty may be a reasonable method of undiversion in some patients [42].
Contraindications
Serious bowel dysfunction, such as inflammatory bowel disease or after radiotherapy, in which removal of a segment will compromise absorption, is a contraindication. In patients with short gut syndrome, ileum and colon should not be used, although stomach may be an alternative. Another contraindication is when a patient is unwilling or unable to do clean intermittent catheterization (CIC), performed either by himself/herself or a caregiver [43].
Poor baseline renal function may predispose patients to severe electrolyte abnormalities and worsening renal function, and is a relative contraindication [43, 44] (although in patients with continuing renal dysfunction as a direct result of bladder dysfunction, augmentation may still be appropriate, and can slow the decrease in renal function [43]).
Surgical Considerations
Preoperative workup usually involves renal and bladder imaging (to assess renal anatomy, obstruction, and presence of stone disease), video-urodynamics (with special attention to the appearance of the bladder neck in order to assess the need for concomitant bladder neck or incontinence surgery), cystoscopy (to assess lower urinary tract anatomy), urine culture, complete blood count, renal function, and electrolyte levels. A history of bowel disease or surgery may require preoperative bowel imaging studies or colonoscopy. A full bowel preparation is generally used for these patients preoperatively however questions have been raised recently regarding its safety and need [45, 46].
The bladder is usually exposed through a midline lower abdominal incision, and the bowel segment is assessed for its suitability for use. The surgeon assesses the ease of moving the segment down to the bladder combined with the possible nutritional and metabolic consequences that will be discussed below. The bowel segment is usually detubularized to maximize the surface area (and therefore the resulting bladder volume), and reduces bowel contractions and postoperative detrusor pressure [47].
Ileum is often the preferred segment due to its familiarity among urologists, low complication rate, and tolerable metabolic profile [26, 44]. It may result in lower postoperative maximal detrusor pressures, and may reduce uninhibited contractions more effectively than sigmoid [48]. A 20–40 cm segment is selected (depending on the need), at least 20 cm proximal to the ileocecal valve. It is detubularized and used in various configurations for augmentation (Figs. 16.1 [49] and 16.2).
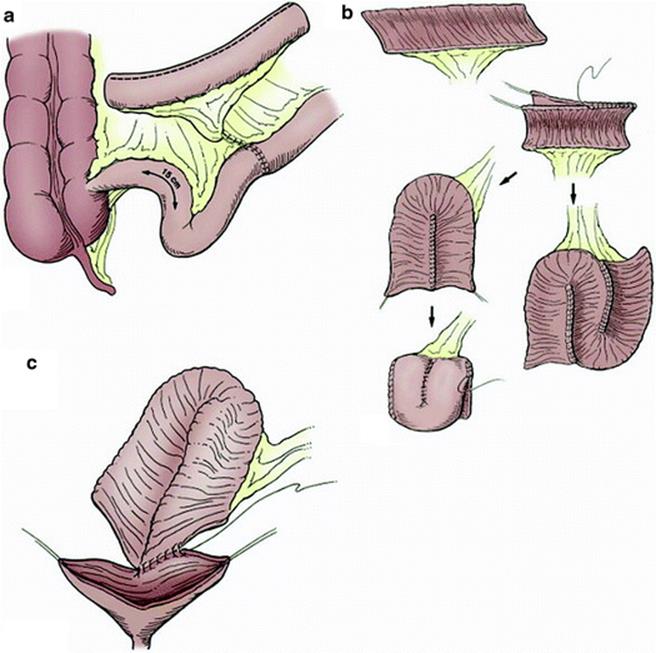
Fig. 16.1
(a) Ileocystoplasty. A 20–40-cm segment of ileum at least 15 cm from the ileocecal valve is removed and opened on its antimesenteric border. Ileoileostomy reconstitutes the bowel. (b) The opened ileal segment should be reconfigured. This can be done in a U, S, or W configuration. It can be further folded as a cup patch. (c) The reconfigured ileal segment is anastomosed widely to the native bladder (from Adams and Joseph in Campbell-Walsh urology [49])
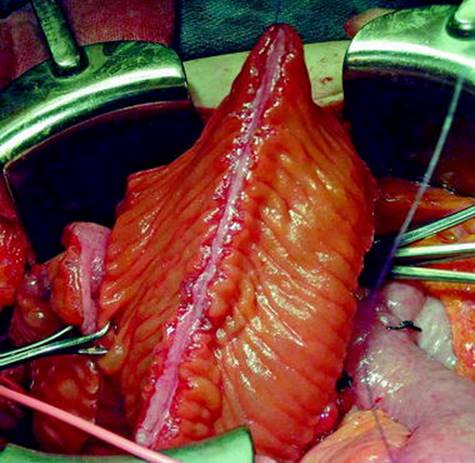
Fig. 16.2
A 40 cm length of ileum is shown. The segment has been isolated from the GI tract and reconfigured. The antimesenteric border was incised and the bowel segment was detubularized into an inverted U-shaped. It will be anastomosed to the bladder
Sigmoid is an alternative and has been reported to have a lower rate of bowel obstruction [50, 51]. A 15–20 cm detubularized segment can be used.
Another alternative is cecum and ascending colon that can be mobilized up to the hepatic flexure. Cecum can be detubularized and used alone or in conjunction with a 15–30 cm segment of detubularized ileum to form the augment. Ileum or appendix can be used as a continent catheterizing channel with the ileocecal valve (or intravesical tunneling of the appendix) providing the continence mechanism. The ileal segment can also be used as a bladder “chimney” to reach resected or obstructed ureters for reimplantation if necessary.
Stomach is rarely used and jejunum should be avoided because of associated metabolic complications.
Alternative procedures for bladder augmentation include ureterocystoplasty (which is an option in patients with megaureter and an ipsilateral nonfunctional kidney [52, 53]) and autoaugmentation. Autoaugmentation involves performing a detrusor myectomy to create a large, low-pressure bladder diverticulum. Autoaugmentation avoids the complications associated with bowel, however it has poor reported long-term efficacy [54–58].
Once the bowel segment has been selected, the bladder is usually opened with a sagittal incision to bivalve it (“clam” cystoplasty [59]). An alternative is a wide U-shaped anterior or posterior incision that effectively creates a large flap for a wide anastomosis [60]. Supratrigonal bladder excision [61] can also be done. The ureteric orifices are identified to avoid damage. The bowel segment is sutured to the bladder with a wide anastomosis to ensure good drainage of the augmentation. A pelvic drain, suprapubic tube, and foley catheter may be placed for the postoperative period.
Reports of completely intraperitoneal laparoscopic, robotic-assisted, and single port augmentation cystoplasties in both adults and children have been published. These procedures require advanced laparoscopic skills and are not yet widely used [62–64].
Follow-Up
Close follow-up is necessary in the immediate postoperative period until indwelling catheters are removed, and the patient adjusts to CIC and bladder irrigations. The augmentation usually enlarges with time. Long-term follow-up consists of renal imaging, renal function tests, electrolyte measurements (to test for metabolic derangements), and complete blood count (to detect pernicious anemia). Some authors have advocated screening cystoscopy 5–10 years after augmentation to assess for bladder cancer; however this is controversial [65, 66]. Urodynamics may be done if there is a change in symptoms, onset of new hydronephrosis, or worsening renal function.
The overall complication rates in various series range from 3 to 41% depending on the duration of follow-up and completeness of reporting [67, 68].
Early Postoperative Complications
With any major abdominal surgery, there are associated cardiovascular, respiratory, and gastrointestinal complications. Postoperative mortality rates have been reported between 0 and 3.2% [43, 67, 69–76], and were generally the result of postoperative myocardial infarction (0–2.7%) and pulmonary embolus/deep vein thrombosis (0–7%) [39]. There have been a small number of reports of other severe complications, such as major bleeding requiring reoperation [39] and necrosis of the bowel segment [8, 76].
Small bowel obstruction requiring operative intervention may occur in 3–6% of patients, and approximately 5–6% of patients may develop a wound infection or dehiscence [43]. Anastomotic leak from the bladder occurs in 2–4% of patients. Postoperative ileus is common, and prolonged ileus occurs in approximately 5% of patients [43]. Severe postoperative complications are less frequent in contemporary case series [43].
Continence and Urodynamic Outcomes
Several groups have reported long-term functional outcomes in adult and pediatric populations. Blaivas et al. [60] reported on 65 adult patients who underwent augmentation cystoplasty (primarily with an ileocecal segment) with or without creation of an abdominal stoma (and included an additional 11 patients who had a continent diversion). At a mean follow-up of 5 years, 70% considered themselves cured, and 18% considered themselves improved. Failures consisted almost exclusively of interstitial cystitis patients. Mean bladder capacity increased from 166 to 572 mL, and mean maximal detrusor pressure fell from 53 to 14 cm H2O. Flood et al. [36] reported on 122 augmentation cystoplasties (67% ileocystoplasty, 30% ileocecocystoplasty) with a mean follow-up of 3 years. They had a primarily adult population. They reported similar urodynamic improvements, a 75% cure rate, and a 20% improvement rate in incontinence.
Quek and Ginsberg [77] reported durability of the urodynamic improvements and 96% patient satisfaction among 24 patients with a mean follow-up of 8 years (range 4–13).
Herschorn and Hewitt [67] preformed a cross-sectional survey of 59 adults who underwent augmentation cystoplasty (usually with additional simultaneous reconstructive procedures) at a median follow-up of 6 years. Sixty-seven percent of patients reported complete continence, and 30% reported only mild incontinence (requiring on average 1–2 pads per day). Almost all patients were very satisfied with their urologic management.
Results in the pediatric populations are similar, although the majority of patients require additional reconstructive procedures such as ureteral reimplantation, bladder neck procedures, and creation of catheterizable channels. Lopez Pereira et al. reported on 29 children with a mean follow-up of 11 years [78]. Mean postoperative bladder capacity increased from 90 to 521 mL, and mean maximal detrusor pressure fell from 45 to 10 cm H2O. Shekarriz et al. reported a 95% continence rate among 133 pediatric patients at a mean follow-up of 5 years [50].
A number of authors have compared the outcomes of ileum, ileocecal, and sigmoid segments, and have not shown any consistent advantages of any segment in terms of urinary continence or renal function [76, 79–81]. Urodynamically demonstrated contractions might persist postoperatively with colonic segments [48, 82].
Long-Term Consequences
The possible long-term consequences of augmentation are listed in Table 16.2 and discussed below. Complications requiring intervention may occur years after the original surgery [67, 68]. This underscores the necessity of long-term follow-up.
Table 16.2
Long-term consequences of augmentation cystoplasty and potential management strategies
|
Description |
Management |
|
|
Growth retardation and osteopenia |
Conflicting evidence on presence of linear growth reduction |
Consider monitoring bone mineral density |
|
Chronic acidosis may lead to osteopenia |
Treat acidosis |
|
|
Electrolyte abnormalities |
||
|
Ileum/colon |
Hyperchloremic, metabolic acidosis ± hypokalemia |
Chloride restriction, bicarbonate, niacin, chlorpromazine |
|
Stomach |
Hypochloremic, hypokalemia, metabolic alkalosis ± hematuria-dysuria syndrome |
IV fluids, potassium supplementation, histamine antagonists, proton pump inhibitors |
|
Renal insufficiency |
May occur as a result of complications associated with augmentation cystoplasty, especially in patients with poor preoperative renal function |
Postoperative monitoring of renal function |
|
Vitamin B12 deficiency |
Due to ileal resection |
Postoperative monitoring of complete blood count |
|
B12 supplementation |
||
|
Bladder cancer |
Increased risk of aggressive bladder cancer among patients with neurogenic bladder; controversial if the augmentation is an independent risk factor |
Aggressive investigation of hematuria, frequent urinary infections, or penile/scrotal discharge |
|
Bladder perforation |
Consider if any patient with peritonitis, septic shock, abdominal pain and distension, nausea and vomiting, fever, referred shoulder pain, or intraperitoneal fluid |
In stable patients, a trial of conservative therapy may be attempted. |
|
Standard treatment is laparotomy for surgical repair |
||
|
Stone disease |
Due to metabolic alterations, poor bladder emptying, mucus, and chronic infection |
Endoscopic, percutaneous, or open surgical procedure |
|
Increased fluid intake and dietary modifications |
||
|
Mucus |
Produced by the bowel segment |
Bladder irrigations |
|
Acetylcysteine/urea irrigations |
||
|
Urinary tract infection |
Asymptomatic bacteriuria is common |
Antibiotic therapy for symptomatic infections |
|
Symptomatic urinary infection require treatment |
Antibiotic prophylaxis or intravesical irrigations for frequent symptomatic infections |
|
|
Bowel dysfunction |
Due to alterations to bile acid metabolism; often exacerbates underlying neurogenic bowel or irritable bowel syndrome |
Low fat diet |
|
Antidiarrheal medication |
||
|
Bile acid binders (cholestyramine) |
||
|
Voiding dysfunction |
Incomplete emptying or inability to void |
CIC is commonly required postoperatively |
|
Incontinence may occur due to an incompetent outlet |
Surgical treatment of incontinence is common |
|
|
Bowel dysfunction |
Due to alterations to bile acid metabolism; often exacerbates underlying neurogenic bowel or irritable bowel syndrome |
Low fat diet |
|
Antidiarrheal medication |
||
|
Bile acid binders (cholestyramine) |
||
|
Pregnancy |
Vaginal delivery preferable |
|
|
Urologic assistance is helpful during elective cesarean sections |
||
Growth Retardation and Decreased Bone Mineral Density
Small case series by Mundy and Nurse [83] and Wagstaff et al. [84] were the first to suggest there is a decrease in linear growth in children after augmentation cystoplasty. Since then, several additional studies have been published, of which two suggested there is approximately a 15% decrease in linear growth after augmentation, and six which did not demonstrate a significant change to linear growth [85, 86]. There is also contradictory evidence as to whether decreased bone mineral density or osteopenia is a result of the augmentation [86]. In a case series of 24 children followed for an average of 9 years after augmentation, Hafez et al. reported a 20% incidence of significant osteopenia [87]. The osteopenia is likely a result of buffering of the acidosis by the skeletal system, which leads to changes in bone mineralization [88]. Correction of this acidosis may improve bone density [89]. Other mechanisms of osteopenia include reduced renal tubular reabsorption of calcium and intestinal malabsorption of calcium [90]. The long-term impact of the osteopenia and how it affects children as adults is still unknown [86].
Management includes appropriate screening and treatment of postoperative metabolic acidosis. Patients with renal failure are more likely to have uncompensated acidosis and should be followed closely and treated for this complication. Some authors have advocated bone mineral density measurements after augmentation [87].
Electrolyte Abnormalities
The expected pattern of metabolic abnormality is dependent on the segment of bowel used in the augmentation cystoplasty. Other factors that influence the severity of the electrolyte imbalance include the surface area of the augmentation, urine pH, and the urine contact time [90].
Ileum and Colon
The classic electrolyte pattern is hyperchloremic metabolic acidosis. The symptoms associated with metabolic acidosis are fatigue, anorexia, weight loss, and polydipsia. There are several possible mechanisms: frequent pyelonephritis may lead to distal tubular acidification defect, urea in the urine may be metabolized by intestinal flora to ammonium which is then absorbed by the bowel, loss of bicarbonate from the bowel that can lead to metabolic acidosis, or chloride that is actively transported from the bowel into the urine leads to reabsorption of ammonium or hydrogen ions [91]. The most likely mechanism is ammonium substituting for sodium in a sodium-hydrogen ion antiport; this antiport is coupled with a bicarbonate-chloride exchanger, leading to a net reabsorption of hydrogen ion, ammonium, and chloride [92]. Hypokalemia can occur during treatment of an acidosis, which unmasks low total body potassium, or as a result of renal potassium wasting (seen more frequently with colonic segments) [92, 93]. Associated hypocalcemia and hypomagnesemia (usually restricted to patients with renal insufficiency, and more commonly seen in colonic augmentations) may be due to reduced renal reabsorption due to a high level of sulfate that is reabsorbed from the bowel, or due to chronic acidosis causing calcium mobilization and subsequent activation of parathyroid hormone [93, 94].
Normal renal function can often compensate for this acidosis; the majority of patients will have a measurable abnormality [95], however it will only be clinically relevant in approximately 10–20% of patients [43, 96]. The absorptive properties of the bowel may be attenuated with time due to mucosal atrophy [97, 98]. Treatment of the acidosis is usually considered once the base excess falls below −2.5 mmol/L [93, 96]. Therapy consists of dietary chloride restriction, bicarbonate supplementation (sodium bicarbonate, potassium citrate), and maximal urinary drainage [94]. Niacin or chlorpromazine inhibits active chloride transportation in the intestine, and may be useful, especially when the solute load of bicarbonate therapy is undesirable [98].
Stomach
The classic electrolyte pattern is hypochloremic, hypokalemic, metabolic alkalosis. Clinical symptoms associated include pelvic pain, fatigue, mental status changes, seizures, or cardiac arrhythmias [93]. Treatment of the electrolyte disturbance involves maximal bladder drainage, normal saline fluid resuscitation, and potassium replacement when necessary [93, 99]. Long-term therapy with potassium chloride may be necessary [93]. Acid secretion can be suppressed with histamine antagonists, or proton pump inhibitors [93].
Hematuria-dysuria syndrome is characterized by excess acid secretion causing peptic ulcer disease, hematuria and dysuria; it occurs in up to 25% of patients, and treatment with a proton pump inhibitor is required intermittently or continuously in a small proportion of patients [100].
Hyperammonemia
The liver is responsible for metabolizing ammonium (absorbed from an augmentation cystoplasty) into urea. Impaired hepatic function or sepsis can lead to the inability of the liver to cope with the hyperammonemia; symptomatically this presents as ammoniagenic encephalopathy [94]. Treatment is maximal urinary drainage, low protein diet, ammonium binders (such as lactulose or neomycin), and in severe cases, intravenous arginine glutamate [93].
Renal Insufficiency
Deterioration of renal function may occur in 0–15% of patients after augmentation [43]. It is unknown whether this is a direct result of the augmentation or due to associated complications [101]. Renal insufficiency occurs, independent of the bowel segment selected [102, 103]. The etiology of renal dysfunction may be urinary stone disease, bacteriuria, high detrusor pressures, vesicoureteral reflux, unrecognized obstruction, and lack of compliance with catheterization [102]. One study suggests approximately 5% of patients will have renal dysfunction after augmentation without a clear etiology [102]. Some authors have demonstrated that baseline renal function is a significant predictor of renal deterioration after augmentation cystoplasty, with increased risk when creatinine clearance is <40 mL/min [8, 43, 104, 105]. Other studies in children and adults with baseline renal dysfunction did not appear to have accelerated renal failure after augmentation cystoplasty [67, 106]. There is no consensus on the order of a staged augmentation cystoplasty and a renal transplant [106].
Postoperatively, patients should have renal imaging and serum creatinine measurements to screen for renal insufficiency [94]. Serum creatinine can be difficult to interpret in this population, due to a low muscle mass in neurogenic patients, and increased reabsorption of urine creatinine by the ileum. Nuclear renograms may be better for definitive measurement.
Vitamin B12 Deficiency
Vitamin B12 is bound to intrinsic factor in the duodenum, which allows is to be absorbed in the terminal ileum. With ileocystoplasty, the most distal 15 cm of the ileum should be preserved to prevent this complication [94]. Vitamin B12 deficiency may cause megaloblastic anemia and neurologic changes [94]. In nutritionally normal individuals, it takes up to 3 years for the livers store of B12 to be depleted, and the resulting deficiency to manifest. The incidence of B12 deficiency related to ileal resection is 3–20% [94, 107].
This complication may be treated prophylactically with B12 supplementation if more than 50 cm of ileum is used for the bladder augmentation [108]. Otherwise, patients should have complete blood counts in follow-up to screen for pernicious anemia.
Bladder Cancer
Bladder cancer has been reported in young patients after augmentation [68, 109, 110]. It has also been reported that spinal cord injury patients and spina bifida patients develop bladder cancer at a young age (40–50 years), they have an increased risk of locally advanced disease, an increased number of adenocarcinomas and squamous cell carcinomas, and a short median survival after diagnosis [66, 111]. In a matched cohort study from a registry of patients with bladder dysfunction due to neurologic abnormalities, exstrophy, and posterior urethral valves, Higuchi et al. did not find a significant difference in the incidence of bladder cancer among patients with augmentation cystoplasty (using ileum or colon) compared to patients managed with intermittent catheterization [65]. The authors did demonstrate that the incidence of bladder cancer was higher in both groups with congenital bladder anomalies independent of augmentation status when compared to the SEER database. Possible reasons for a higher rate of bladder cancer in patients with neurogenic bladder may be reduced intracellular antioxidant activity (leading to increased rates of DNA mutation) [112], impaired DNA repair in the bowel due to the hyperosmolar urine [113], and immunosuppressant use in patients after renal transplantation [65]. However, patients who have undergone a gastric augmentation may have a higher cancer risk compared to other bowel segments [65].
Urologists should have a particular awareness of the potential for aggressive bladder cancer in this population, whether or not they have had an augmentation cystoplasty. Symptoms such as hematuria, frequent urinary infections, or penile/scrotal discharge need to be aggressively investigated; visual changes in the bladder due to the augmentation, recent infections, or catheterization can make cystoscopy challenging, and biopsy or CT should be considered if there is any uncertainty [111].
Bladder Perforation
This is a potentially life-threatening complication that occurs in approximately 6–13% of patients [20, 114–118]. Patients with neurogenic bladders, those with competent bladder necks, those without a catheterizable channel, and those who abuse alcohol appear to be at an increased risk [20, 43, 119, 120]. Perforation can occur at any time postoperatively, even years later. It can present with fever, abdominal pain and distension with intraperitoneal fluid, nausea and vomiting, referred shoulder pain, peritonitis, and septic shock [50, 116]; because of neurological abnormalities of these patients, the presenting symptoms are often nonspecific. Diagnosis can be made with a CT cystogram; standard fluoroscopic cystography has a 10–20% false negative rate [50, 115, 121]. CT or US can demonstrate intraperitoneal fluid, which is an important sign that bladder perforation has occurred [122]. Due to the augmentation, extraperitoneal ruptures are rare [123]. The area of perforation is usually at the bowel-bladder anastomosis, or within the weaker bowel wall [115]. The etiology of bladder perforation is thought to be from traumatic catheterization, acute over distension, or increased intravesical pressure chronic over distension (from CIC noncompliance) or infection leading to localized areas of ischemia and necrosis [121, 124].
The treatment of patients with large perforations and clinical instability usually is laparotomy for surgical repair. In patients that are stable, (usually with a small perforation), a trial of conservative therapy (foley catheter and antibiotics) may be considered [124, 125]. Mortality is high in patients with clinical instability on presentation, and those with a delayed diagnosis; overall mortality has been estimated at up to 25% [114, 126, 127]. If clinical suspicion is high, and imaging is negative, the patient should still be treated as a possible bladder perforation [43]. There is a 25% rate of recurrence of bladder perforation after the initial episode [20, 121, 128].
Stone Disease
Patients are at increased risk for bladder and upper tract calculi. Urinary calculi have been reported in 9–15% of patients after augmentation [43, 67, 129–131], and in some series as high as 50% [132]. Many of the risk factors for stones are present in patients that undergo augmentation, and may not be directly related to the surgical procedure [133]. Patients with a continent catheterizable channel (which may not drain the bladder completely), those using urethral CIC (compared to those voiding spontaneous), and patients with urease splitting bacteriuria are at increased risk [43, 130]. Possible reasons for stone formation include chronic bacteriuria (a significant risk factor in multivariable analysis [134]), intravesical foreign bodies, elevated postvoid residuals, and mucus secretion from the bowel segment [135]. Similar to regular stone forming population, dietary choices, and inadequate fluid intake increase the risk of stone disease [136]. Metabolic changes, such as hypercalciuria and hypocitraturia secondary to metabolic acidosis, water loss through the cystoplasty bowel segment, and mild enteric hyperoxaluria (from the bowel resection or antibiotic-related deficiency of oxalobacter formigenes) can predispose these patients to stone formation [132, 136, 137]. Most stones are struvite due to frequent bacteriuria, or calcium oxalate; they are usually mixed with calcium phosphate due to the alkalotic urine [132, 136, 138].
Treatment of stones includes endoscopic, percutaneous, or open surgical procedures depending on the stone size, location, and patient factors [43, 129]. Prevention of stones consists of bladder irrigation, which may [139] or may not [140] be preventive role, increased fluid intake, decreased salt, purine and oxalate intake, and medical therapy directed by 24 h urine and stone analysis.
Mucus
Ileal and colonic segments used in augmentations continue to produce mucus. Up to 40 g of mucus can be produced daily and continues over time despite villous atrophy [141]. Colonic bowel segments produce more mucus than ileal segments [143]. The mucus is thought to help reduce malignant changes [142], however it has been implicated as a causative factor in urinary tract infections, stone formation, poor bladder emptying, and bladder perforation [43].
Problematic mucus secretion can be treated with daily bladder irrigations. These can be augmented with acetylcysteine or urea irrigations which help dissolve mucus [143], or oral ranitidine which may help to reduce mucus production [144].
Urinary Tract Infection
Asymptomatic bacteriuria is nearly universal among augmentation enterocystoplasty patients and usually does not require treatment except in cases of urease splitting organisms (such as Proteus and Klebsiella) [145]. Studies in ileal conduits have shown that bacteria freely adhere to bowel mucosa, and do not incite an inflammatory reaction [146]. This chronic bacteriuria has been cited as a risk factor for stone disease, incontinence, and bladder cancer [43, 147]. The most common organism is Escherichia coli [148].
Symptomatic urinary tract infection, which occurs in 5–40% of patients [43, 76, 80], requires antibiotic treatment. Risk factors are similar to asymptomatic bacteriuria and include urinary stasis, mucus production, and intermittent catheterization [39]. Symptoms may be nonspecific if bladder sensation is absent and include incontinence, abdominal pain, hematuria, new onset foul urine, and lethargy.
Management of urinary tract infection consists of appropriate antibiotic therapy. In patients with frequent symptomatic infections despite oral antibiotic prophylaxis, intravesical irrigation with antibiotics may reduce symptomatic infections [149]. In a small pilot study of 15 patients after ileocystoplasty, cranberry extract reduced asymptomatic bacteriuria [150].
Bowel Dysfunction
Bowel dysfunction after bowel resection for augmentation or diversion occurs in approximately 20–50% of patients [67, 151, 152]. The most common symptom is diarrhea seen in about 25% of patients, however potentially more distressing symptoms of fecal urgency and incontinence and nocturnal bowel movements are also common [151]. Bowel dysfunction is more common among patients with a neurologic diagnosis as a result of associated neurogenic bowel dysfunction and among patients with previous radiation or bowel resections [151, 152]. Approximately 30% of patients with irritable bowel syndrome have detrusor overactivity; this may be due to an intrinsic disorder of smooth muscle calcium metabolism [152].
Specific surgical factors may contribute to postoperative changes in bowel function that lead to diarrhea. Bile acids, generated in the liver and secreted into the small intestine, are necessary for fat absorption. Bile acids are reabsorbed in the distal ileum, enter the liver, and participate in the feedback mechanism for regeneration. Resection of long sections of the terminal ileum can lead to bile acid malabsorption. Bile acids entering the colon may cause diarrhea by inducing water and salt secretion and by promoting motility [153]. Ileal resection of more than 100 cm results in severe bile acid malabsorption that cannot be compensated for by increased hepatic synthesis. In such cases, steatorrhea results from impaired micelle formation due to decreased luminal concentrations of conjugated bile acids. In shorter ileal resections, bile acid malabsorption can usually be compensated for by an increase in hepatic synthesis; and malabsorbed bile acids cause the diarrhea rather than steatorrhea [154, 155]. Resection of the ileocecal valve leads to bacterial colonization of the distal ileum that destroys the bile acids. The lack of bile acids, which leads to unabsorbed fatty acids in the large bowel, stimulates the colon to secrete more water and mucus, increase motility, and prompt defecation [156].
Treatment of this complication involves a low fat diet and antidiarrheal medications. Bile acid-related diarrhea can be diagnosed with a selenium homocholic acid taurine test, or a therapeutic trial of bile acid binders such as cholestyramine [156] may be helpful.
Voiding Dysfunction and Incontinence
The interposition of bowel into the bladder usually prevents the efficient detrusor contractions that are necessary for voiding [157]. The urethral outlet resistance may be high due to neurologic disease, or concomitant surgery to treat incontinence. Some patients are able to void spontaneously with abdominal straining.
If the patient is unable to void, or has complications from incomplete emptying, he/she will need to use CIC to empty their bladder. This is necessary in 25–100% of neurogenic patients, and a lower proportion of neurologically intact patients [43].
Continence rates range from 60 to 100% [67, 77]. Nocturnal incontinence can occur due to failure of the urethral sphincter to respond to contractions of the augmented bowel, and increased urine output due to water loss from the augmented bowel segment. Daytime incontinence can be due to stress incontinence, detrusor overactivity, or from phasic contractions of the augmented bowel segment [158, 159]. These phasic contractions are usually <40 cm H2O, and occur at higher volumes [77].
Treatment of incontinence in these patients includes behavioral modification (such as more frequent CIC), anticholinergics, and surgical procedures such as midurethral slings, bladder neck slings or bladder neck reconstruction, and artificial urinary sphincters [43, 160]. Occasionally repeat augmentation is necessary [131].
Pregnancy
While not a postoperative complication, pregnancy after augmentation cystoplasty is becoming more common [121]. Complications such as premature labor, urinary tract infection, renal dysfunction, and urinary tract obstruction are more prevalent [161]. Patients usually require antibiotic treatment of bacteriuria; screening urinalysis for infection or proteinuria is not accurate due to mucus from the augmentation cystoplasty [162].
Vaginal delivery is preferable [162, 163], however there is controversy as to whether cesarean section is necessary for patients with artificial sphincters and bladder neck procedures [43, 162]. If an elective cesarean section is scheduled for other reasons, urologic assistance during the surgery and a high segment section may help avoid damage to the bladder augmentation [43, 162]. The bowel segment can survive inadvertent damage to the vascular pedicle, however this may lead to eventual contraction of the bowel segment [164].
Conclusion
Bladder augmentation with intestine has been successfully used to treat various conditions that results in small capacity bladders. The surgical technique involves detubularization and reconfiguration of a segment of bowel (usually the ileum or colon) to create a patch. A successful clinical outcome is dependent upon creating a large capacity, low-pressure reservoir to store urine; additional procedures to aid in catheterization or continence are often necessary. Potential complications have been well described and are usually reported in case series. Medical and surgical treatments of complications are similarly well elucidated although some are still controversial. Since complications may occur at any time after surgery prolonged follow-up and monitoring are essential.
References
1.
Mikulicz J. Zur Operation der angeborenen Blasenspalte. Zentralbl Chir. 1899;26:641–3.
2.
Charghi A, Charbonneau J, Gauthier GE. Colocystoplasty for bladder enlargement and bladder substitution: a study of late results in 31 cases. J Urol. 1967;97:849–56.PubMed
3.
Bisgard JD. Substitution of the urinary bladder with a segment of sigmoid: an experimental study. Ann Surg. 1943;117:106–9.PubMed
4.
Couvelaire R. [The “little bladder” of genito-urinary tuberculosis; classification, site and variants of bladder-intestine transplants]. J Urol Medicale Chir. 1950;56:381–434.PubMed
5.
Leong CH. Use of the stomach for bladder replacement and urinary diversion. Ann R Coll Surg Engl. 1978;60:283–9.PubMed
6.
Elbahnasy AM, Shalhav A, Hoenig DM, Figenshau R, Clayman RV. Bladder wall substitution with synthetic and non-intestinal organic materials. J Urol. 1998;159:628–37.PubMed
7.
Goodwin WE, Winter CC, Barker WF. Cup-patch technique of ileocystoplasty for bladder enlargement or partial substitution. Surg Gynecol Obstet. 1959;108:240–4.PubMed
8.
Smith RB, van Cangh P, Skinner DG, Kaufman JJ, Goodwin WE. Augmentation enterocystoplasty: a critical review. J Urol. 1977;118:35–9.PubMed
9.
Sidi AA, Dykstra DD, Gonzalez R. The value of urodynamic testing in the management of neonates with myelodysplasia: a prospective study. J Urol. 1986;135:90–3.PubMed
10.
Lendvay TS, Cowan CA, Mitchell MM, Joyner BD, Grady RW. Augmentation cystoplasty rates at children’s hospitals in the United States: a pediatric health information system database study. J Urol. 2006;176:1716–20.PubMed
11.
Kaefer M, Pabby A, Kelly M, Darbey M, Bauer SB. Improved bladder function after prophylactic treatment of the high risk neurogenic bladder in newborns with myelomentingocele. J Urol. 1999;162: 1068–71.PubMed
12.
Aki FT, et al. Renal transplantation in children with augmentation enterocystoplasty. Transplant Proc. 2006;38:554–5.PubMed
13.
Fisang C, Hauser S, Muller SC. Ureterocystoplasty: an ideal method for vesical augmentation in children. Aktuelle Urol. 2010;41 Suppl 1:S50–2.PubMed
14.
Kajbafzadeh AM, Quinn FM, Duffy PG, Ransley PG. Augmentation cystoplasty in boys with posterior urethral valves. J Urol. 1995;154:874–7.PubMed
15.
Peters CA, et al. The urodynamic consequences of posterior urethral valves. J Urol. 1990;144:122–6.PubMed
16.
Amirzargar MA, et al. Reconstruction of bladder and urethra using ileocecal segment and appendix in patients with exstrophy-epispadias complex: the first report of a new surgical approach. Int Urol Nephrol. 2007;39:779–85.PubMed
17.
De Castro R, Pavanello P, Domini R. Indications for bladder augmentation in the exstrophy-epispadias complex. Br J Urol. 1994;73:303–7.PubMed
18.
Gearhart JP, Peppas DS, Jeffs RD. The failed exstrophy closure: strategy for management. Br J Urol. 1993;71:217–20.PubMed
19.
Lund DP, Hendren WH. Cloacal exstrophy: a 25-year experience with 50 cases. J Pediatr Surg. 2001;36: 68–75.PubMed
20.
Metcalfe PD, et al. Spontaneous bladder perforations: a report of 500 augmentations in children and analysis of risk. J Urol. 2006;175:1466–70; discussion 1461–70.
21.
Jeong SJ, Cho SY, Oh SJ. Spinal cord/brain injury and the neurogenic bladder. Urol Clin North Am. 2010;37:537–46.PubMed
22.
Sidi A, Becher E, Reddy P, Dykstra D. Augmentation enterocystoplasty for the management of voiding dysfunction in spinal cord injury patients. J Urol. 1990;143:83–5.PubMed
23.
Van Rey F, Heesakkers J. Applications of neurostimulation for urinary storage and voiding dysfunction in neurological patients [review] [33 refs]. Urol Int. 2008;81:373–8.PubMed
24.
Lewis J, Cheng E. Non-traditional management of the neurogenic bladder: tissue engineering and neuromodulation [review] [77 refs]. Scientific World Journal. 2007;7:1230–41.PubMed
25.
Litwiller S, Frohman E, Zimmern P. Multiple sclerosis and the urologist. J Urol. 1999;161:743–57.PubMed
26.
Reyblat P, Ginsberg D. Augmentation cystoplasty: what are the indications? [review] [41 refs]. Curr Urol Rep. 2008;9:452–8.PubMed
27.
Abrams P, et al. The standardisation of terminology in lower urinary tract function: report from the standardisation sub-committee of the International Continence Society. Urology. 2003;61:37–49.PubMed
28.
Reyblat P, Ginsberg D. Augmentation enterocystoplasty in overactive bladder: is there still a role? Curr Urol Rep. 2010;11:432–9.PubMed
29.
Wise G, Shteynshlyuger A. An update on lower urinary tract tuberculosis. Curr Urol Rep. 2008;9:305–13.PubMed
30.
Abel B, Gow J. Results of caecocystoplasty for tuberculous bladder contracture. Br J Urol. 1978;50: 511–6.PubMed
31.
Wesolowski S. Late resuls of cystoplasty in chronic tuberculous cystitis. Br J Urol. 1970;42:697–703.PubMed
32.
de Figueiredo A, Lucon A, Srougi M. Bladder augmentation for the treatment of chronic tuberculous cystitis. Clinical and urodynamic evaluation of 25 patients after long term follow-up. Neurourol Urodyn. 2006;25:433–40.PubMed
33.
Kehinde E, Anim J, Hira P. Parasites of urological importance. Urol Int. 2008;81:1–13.PubMed
34.
Badr M, Zaher M. Ileocystoplasty in the treatment of bilharzial contracted bladder. J Egypt Med Assoc. 1959;42:33–49.PubMed
35.
Smit S, Heyns C. Management of radiation cystitis. Nat Rev Urol. 2010;7:206–14.PubMed
36.
Flood H, et al. Long-term results and complications using augmentation cystoplasty in reconstructive urology. Neurourol Urodyn. 1995;14:297–309.PubMed
37.
Webster G, Maggio M. The management of chronic interstitial cystitis by substitution cystoplasty. J Urol. 1989;141:287–91.PubMed
38.
Nielsen K, Kromann-Andersen B, Steven K, Hald T. Failure of combined supratrigonal cystectomy and Mainz ileocecocystoplasty in intractable interstitial cystitis: is histology and mast cell count a reliable predictor for the outcome of surgery? J Urol. 1990;144:255–8; discussion 258–9.
39.
Worth P. The treatment of interstitial cystitis by cystolysis with observations on cystoplasty. A review after 7 years. Br J Urol. 1980;52:232.PubMed
40.
Nurse D, Parry J, Mundy A. Problems in the surgical treatment of interstitial cystitis. Br J Urol. 1991;68:153–4.PubMed
41.
MacDermott J, Charpied G, Tesluk H, Stone A. Recurrent interstitial cystitis following cystoplasty: fact or fiction? J Urol. 1990;144:37–40.PubMed
42.
Herschorn S, Rangaswamy S, Radomski SB. Urinary undiversion in adults with myelodysplasia: long-term followup. J Urol. 1994;152:329–33.PubMed
43.
Greenwell T, Venn S, Mundy A. Augmentation cystoplasty [review] [166 refs]. BJU Int. 2001;88:511–25.PubMed
44.
Kilic N, et al. Bladder augmentation: urodynamic findings and clinical outcome in different augmentation techniques. Eur J Pediatr Surg. 1999;9:29–32.PubMed
45.
Matsou A, et al. Mechanical bowel preparation before elective colorectal surgery: is it necessary? Tech Coloproctol. 2011;15 Suppl 1:59–62.
46.
Wells T, Plante M, McAlpine JN. Preoperative bowel preparation in gynecologic oncology: a review of practice patterns and an impetus to change. Int J Gynecol Cancer. 2011;21:1135–42.PubMed
47.
Hinman FJ. Selection of intestinal segments for bladder substitution: physical and physiological characteristics. J Urol. 1988;139:519–23.PubMed
48.
Radomski S, Herschorn S, Stone A. Urodynamic comparison of ileum vs. sigmoid in augmentation cystoplasty for neurogenic bladder dysfunction. Neurourol Urodyn. 1995;14:231–7.PubMed
49.
Adams MC, Joseph DB. Urinary tract reconstruction in children. In: Wein A, Kavoussi LR, Novick AC, Partin AW, Peters CA, editors. Campbell-Walsh urology, vol. 4. Philadelphia, PA: Saunders Elsevier; 2007. p. 3656–702.
50.
Shekarriz B, Upadhyay J, Demirbilek S, Barthold J, Gonzalez R. Surgical complications of bladder augmentation: comparison between various enterocystoplasties in 133 patients. Urology. 2000;55:123–8.PubMed
51.
Gough D. Enterocystoplasty [review] [40 refs]. BJU Int. 2001;88:739–43.PubMed
52.
Hitchcock R, Duffy P, Malone P. Ureterocystoplasty: the ‘bladder’ augmentation of choice. Br J Urol. 1994;73:575–9.PubMed
53.
Anderson P, Dewan P. Ureterocystoplasty: an alternative reconstructive procedure to enterocystoplasty in suitable cases. J Pediatr Surg. 2001;36:962.PubMed
54.
MacNeily A, Afshar K, Coleman G, Johnson H. Autoaugmentation by detrusor myotomy: its lack of effectiveness in the management of congenital neuropathic bladder. J Urol. 2003;170:1643–6; discussion 1646.
55.
Stohrer M, et al. Bladder autoaugmentation in adult patients with neurogenic voiding dysfunction. Spinal Cord. 1997;35:456–62.PubMed
56.
Skobejko-Wlodarska L, Strulak K, Nachulewicz P, Szymkiewicz C. Bladder autoaugmentation in myelodysplastic children. Br J Urol. 1998;81 Suppl 3:114–6.PubMed
57.
Duel B, Gonzalez R, Barthold J. Alternative techniques for augmentation cystoplasty [review] [134 refs]. J Urol. 1998;159:998–1005.PubMed
58.
Cartwright P, Snow B. Bladder autoaugmentation: early clinical experience. J Urol. 1989;142:505–8.PubMed
59.
Bramble F. The clam cystoplasty [review] [21 refs]. Br J Urol. 1990;66:337–41.PubMed
60.
Blaivas J, et al. Long-term followup of augmentation enterocystoplasty and continent diversion in patients with benign disease. J Urol. 2005;173:1631–4.PubMed
61.
Gil Vernet SG. [Neurogenic bladder, neuromuscular bladder and intestinal bladder]. Acta Urol Belg. 1962;30:405–20.PubMed
62.
Traxel E, Minevich E, Noh P. A review: the application of minimally invasive surgery to pediatric urology: lower urinary tract reconstructive procedures [review] [38 refs]. Urology. 2010;76:115–20.PubMed
63.
Noguera R, et al. Laparoscopic augmentation enterocystoplasty through a single trocar. Urology. 2009;73:1371–4.PubMed
64.
Challacombe B, Dasgupta P. Reconstruction of the lower urinary tract by laparoscopic and robotic surgery. Curr Opin Urol. 2007;17:390–5.PubMed
65.
Higuchi T, Granberg C, Fox J, Husmann D. Augmentation cystoplasty and risk of neoplasia: fact, fiction and controversy. J Urol. 2010;184: 2492–6.PubMed
66.
Austin J. Long-term risks of bladder augmentation in pediatric patients. Curr Opin Urol. 2008;18:408–12.PubMed
67.
Herschorn S, Hewitt R. Patient perspective of long-term outcome of augmentation cystoplasty for neurogenic bladder. Urology. 1998;52:672–8.PubMed
68.
Metcalfe PD, Rink RC. Bladder augmentation: complications in the pediatric population. Curr Urol Rep. 2007;8:152–6.PubMed
69.
Fontaine E, et al. Combined modified rectus fascial sling and augmentation ileocystoplasty for neurogenic incontinence in women. J Urol. 1997;157:109–12.PubMed
70.
Cheng C, Hendry W, Kirby R, Whitfield H. Detubularisation in cystoplasty: clinical review. Br J Urol. 1991;67:303–7.PubMed
71.
Sethia KK, Webb RJ, Neal DE. Urodynamic study of ileocystoplasty in the treatment of idiopathic detrusor instability. Br J Urol. 1991;67:286–90.PubMed
72.
Kockelbergh R, et al. Clam enterocystoplasty in general urological practice. Br J Urol. 1991;68:38–41.PubMed
73.
George V, Russell G, Shutt A, Gaches C, Ashken M. Clam ileocystoplasty. Br J Urol. 1991;68:487–9.PubMed
74.
Gonzalez R, Sidi A, Zhang G. Urinary undiversion: indications, technique and results in 50 cases. J Urol. 1986;136:13–6.PubMed
75.
Gearhart J, Albertsen P, Marshall F, Jeffs R. Pediatric applications of augmentation cystoplasty: the Johns Hopkins experience. J Urol. 1986;136:430–2.PubMed
76.
Mitchell M, Kulb T, Backes D. Intestinocystoplasty in combination with clean intermittent catheterization in the management of vesical dysfunction. J Urol. 1986;136:288–91.PubMed
77.
Quek M, Ginsberg D. Long-term urodynamics followup of bladder augmentation for neurogenic bladder. J Urol. 2003;169:195–8.PubMed
78.
Lopez Pereira P, et al. Enterocystoplasty in children with neuropathic bladders: long-term follow-up. J Pediatr Urol. 2008;4:27–31.PubMed
79.
Kreder K, Webster G. Management of the bladder outlet in patients requiring enterocystoplasty. J Urol. 1992;147:38–41.PubMed
80.
Khoury J, Timmons S, Corbel L, Webster G. Complications of enterocystoplasty. Urology. 1992;40:9–14.PubMed
81.
Lockhart J, Bejany D, Politano V. Augmentation cystoplasty in the management of neurogenic bladder disease and urinary incontinence. J Urol. 1986;135:969–71.PubMed
82.
Goldwasser B, Barrett DM, Webster GD, Kramer SA. Cystometric properties of ileum and right colon after bladder augmentation, substitution or replacement. J Urol. 1987;138:1007–8.PubMed
83.
Mundy A, Nurse D. Calcium balance, growth and skeletal mineralisation in patients with cystoplasties. Br J Urol. 1992;69:257–9.PubMed
84.
Wagstaff K, Woodhouse C, Duffy P, Ransley P. Delayed linear growth in children with enterocystoplasties. Br J Urol. 1992;69:314–7.PubMed
85.
Taskinen S, Makitie O, Fagerholm R. Intestinal bladder augmentation at school age has no adverse effects on growth. J Pediatr Urol. 2008;4:40–2.PubMed
86.
Mingin G, Maroni P, Gerharz E, Woodhouse C, Baskin L. Linear growth after enterocystoplasty in children and adolescents: a review [review] [14 refs]. World J Urol. 2004;22:196–9.PubMed
87.
Hafez A, et al. Long-term evaluation of metabolic profile and bone mineral density after ileocystoplasty in children. J Urol. 2003;170:1639–41; discussion 1632–41.
88.
Vajda P, et al. Metabolic findings after colocystoplasty in children. Urology. 2003;62:542–6; discussion 546.
89.
Abes M, Sarihan H, Madenci E. Evaluation of bone mineral density with dual x-ray absorptiometry for osteoporosis in children with bladder augmentation. J Pediatr Surg. 2003;38:230–2.PubMed
90.
McDougal W. Metabolic complications of urinary intestinal diversion. J Urol. 1992;147:1199–208.PubMed
91.
Koch M, McDougal W, Reddy P, Lange P. Metabolic alterations following continent urinary diversion through colonic segments. J Urol. 1991;145:270–3.PubMed
92.
Stein R, Schroder A, Thuroff J. Bladder augmentation and urinary diversion in patients with neurogenic bladder: non-surgical considerations. J Pediatr Urol. 2011;8(2):145–52.PubMed
93.
Tanrikut C, McDougal W. Acid-base and electrolyte disorders after urinary diversion. World J Urol. 2004;22:168–71.PubMed
94.
Gilbert S, Hensle T. Metabolic consequences and long-term complications of enterocystoplasty in children: a review [review] [71 refs]. J Urol. 2005;173:1080–6.PubMed
95.
Mitchell M, Piser J. Intestinocystoplasty and total bladder replacement in children and young adults: followup in 129 cases. J Urol. 1987;138:579–84.PubMed
96.
Poulsen A, Steven K. Acid-base metabolism after bladder substitution with the ileal urethral Kock reservoir. Br J Urol. 1996;78:47–53.PubMed
97.
Hall M, Koch M, Halter S, Dahlstedt S. Morphologic and functional alterations of intestinal segments following urinary diversion. J Urol. 1993;149:664–6.PubMed
98.
Akerlund S, Forssell-Aronsson E, Jonsson O, Kock N. Decreased absorption of 22Na and 36Cl in ileal reservoirs after exposure to urine. An experimental study in patients with continent ileal reservoirs for urinary or fecal diversion. Urol Res. 1991;19:249–52.PubMed
99.
Plawker M, Rabinowitz S, Etwaru D, Glassberg K. Hypergastrinemia, dysuria-hematuria and metabolic alkalosis: complications associated with gastrocystoplasty. J Urol. 1995;154:546–9.PubMed
100.
Kurzrock E, Baskin L, Kogan B. Gastrocystoplasty: long-term followup [review] [32 refs]. J Urol. 1998;160:2182–6.PubMed
101.
Jonsson O, Olofsson G, Lindholm E, Tornqvist H. Long-time experience with the Kock ileal reservoir for continent urinary diversion. Eur Urol. 2001;40:632–40.PubMed
102.
Fontaine E, Leaver R, Woodhouse C. The effect of intestinal urinary reservoirs on renal function: a 10-year follow-up. BJU Int. 2000;86:195–8.PubMed
103.
Whitmore III WF, Gittes RF. Reconstruction of the urinary tract by cecal and ileocecal cystoplasty: review of a 15-year experience. J Urol. 1983;129:494–8.PubMed
104.
Decter R, Bauer S, Mandell J, Colodny A, Retik A. Small bowel augmentation in children with neurogenic bladder: an initial report of urodynamic findings. J Urol. 1987;138:1014–6.PubMed
105.
Kuss R, Bitker M, Camey M, Chatelain C, Lassau J. Indications and early and late results of intestino-cystoplasty: a review of 185 cases. J Urol. 1970;103:53–63.PubMed
106.
Ivancic V, et al. Progression of renal insufficiency in children and adolescents with neuropathic bladder is not accelerated by lower urinary tract reconstruction. J Urol. 2010;184:1768–74.PubMed
107.
Matsui U, Topoll B, Miller K, Hautmann R. Metabolic long-term follow-up of the ileal neobladder. Eur Urol. 1993;24:197–200.PubMed
108.
Pannek J, Haupt G, Schulze H, Senge T. Influence of continent ileal urinary diversion on vitamin B12 absorption. J Urol. 1996;155:1206–8.PubMed
109.
Shaw J, Lewis MA. Bladder augmentation surgery—what about the malignant risk? Eur J Pediatr Surg. 1999;9 Suppl 1:39–40.PubMed
110.
Carr LK, Herschorn S. Early development of adenocarcinoma in a young woman following augmentation cystoplasty for undiversion. J Urol. 1997;157:2255–6.PubMed
111.
Kalisvaart J, Katsumi H, Ronningen L, Hovey R. Bladder cancer in spinal cord injury patients. Spinal Cord. 2010;48:257–61.PubMed
112.
Barrington J, Jones A, James D, Smith S, Stephenson T. Antioxidant deficiency following clam enterocystoplasty. Br J Urol. 1997;80:238–42.PubMed
113.
Dixon B, Chu A, Henry J, Kim R, Bissler J. Increased cancer risk of augmentation cystoplasty: possible role for hyperosmolal microenvironment on DNA damage recognition. Mutat Res. 2009;670:88–95.PubMed
114.
Couillard D, Vapnek J, Rentzepis M, Stone A. Fatal perforation of augmentation cystoplasty in an adult [review] [22 refs]. Urology. 1993;42:585–8.PubMed
115.
Braverman R, Lebowitz R. Perforation of the augmented urinary bladder in nine children and adolescents: importance of cystography. AJR Am J Roentgenol. 1991;157:1059–63.PubMed
116.
Fontaine E, Leaver R, Woodhouse C. Diagnosis of perforated enterocystoplasty. J R Soc Med. 2003;96:393–4.PubMed
117.
DeFoor W, Tackett L, Minevich E, Wacksman J, Sheldon C. Risk factors for spontaneous bladder perforation after augmentation cystoplasty [review] [15 refs]. Urology. 2003;62:737–41.PubMed
118.
Bauer S, et al. Perforation of the augmented bladder. J Urol. 1992;148:699–703.PubMed
119.
Novak T, Salmasi A, Mathews R, Lakshmanan Y, Gearhart J. Complications of complex lower urinary tract reconstruction in patients with neurogenic versus nonneurogenic bladder—is there a difference? J Urol. 2008;180:2629–34.PubMed
120.
Fox J, Husmann D. Continent urinary diversion in childhood: complications of alcohol abuse developing in adulthood. J Urol. 2010;183:2342–6.PubMed
121.
Kurzrock E. Pediatric enterocystoplasty: long-term complications and controversies. World J Urol. 2009;27:69–73.PubMed
122.
Malone T, Lisle D, McNabb C, Ryan G. Traumatic rupture of an augmented bladder: computed tomography appearances. J Trauma. 2009;67:E85–7.PubMed
123.
Glass R, Rushton H. Delayed spontaneous rupture of augmented bladder in children: diagnosis with sonography and CT. AJR Am J Roentgenol. 1992;158:833–5.PubMed
124.
Slaton J, Kropp K. Conservative management of suspected bladder rupture after augmentation enterocystoplasty. J Urol. 1994;152:713–5.PubMed
125.
Wolff J, Boeckmann W, Jakse G. Spontaneous bladder rupture following enterocystoplasty can be treated conservatively. Urol Int. 1994;52:113–4.PubMed
126.
Rushton H, Woodard J, Parrott T, Jeffs R, Gearhart J. Delayed bladder rupture after augmentation enterocystoplasty. J Urol. 1988;140:344–6.PubMed
127.
Elder J, Snyder H, Hulbert W, Duckett J. Perforation of the augmented bladder in patients undergoing clean intermittent catheterization. J Urol. 1988;140:1159–62.PubMed
128.
Rink R. Bladder augmentation. Options, outcomes, future. Urol Clin North Am. 1999;26:111–23.PubMed
129.
Kronner K, et al. Bladder calculi in the pediatric augmented bladder. J Urol. 1998;160:1096–8.PubMed
130.
Nurse D, McInerney P, Thomas P, Mundy A. Stones in enterocystoplasties. Br J Urol. 1996;77:684–7.PubMed
131.
Metcalfe P, et al. What is the need for additional bladder surgery after bladder augmentation in childhood? J Urol. 2006;176:1801–5.PubMed
132.
Palmer L, et al. Urolithiasis in children following augmentation cystoplasty. J Urol. 1993;150:726–9.PubMed
133.
Barroso U, Jednak R, Fleming P, Barthold J, Gonzalez R. Bladder calculi in children who perform clean intermittent catheterization. BJU Int. 2000;85:879–84.PubMed
134.
DeFoor W, et al. Bladder calculi after augmentation cystoplasty: risk factors and prevention strategies. J Urol. 2004;172:1964–6.PubMed
135.
Khoury A, et al. Stone formation after augmentation cystoplasty: the role of intestinal mucus. J Urol. 1997;158:1133–7.PubMed
136.
Hamid R, Robertson W, Woodhouse C. Comparison of biochemistry and diet in patients with enterocystoplasty who do and do not form stones. BJU Int. 2008;101:1427–32.PubMed
137.
Woodhouse C, Robertson W. Urolithiasis in enterocystoplasties [review] [29 refs]. World J Urol. 2004;22:215–21.PubMed
138.
Robertson W, Woodhouse C. Metabolic factors in the causation of urinary tract stones in patients with enterocystoplasties. Urol Res. 2006;34:231–8.PubMed
139.
Hensle T, Bingham J, Lam J, Shabsigh A. Preventing reservoir calculi after augmentation cystoplasty and continent urinary diversion: the influence of an irrigation protocol. BJU Int. 2004;93:585–7.PubMed
140.
Brough R, O’Flynn K, Fishwick J, Gough D. Bladder washout and stone formation in paediatric enterocystoplasty. Eur Urol. 1998;33:500–2.PubMed
141.
Murray K, Nurse D, Mundy A. Secreto-motor function of intestinal segments used in lower urinary tract reconstruction. Br J Urol. 1987;60:532–5.PubMed
142.
Iannoni C, et al. Abnormal patterns of colorectal mucin secretion after urinary diversion of different types: histochemical and lectin binding studies. Hum Pathol. 1986;17:834–40.PubMed
143.
Gillon G, Mundy A. The dissolution of urinary mucus after cystoplasty. Br J Urol. 1989;63:372–4.PubMed
144.
George V, et al. The effect of ranitidine on urine mucus concentration in patients with enterocystoplasty. Br J Urol. 1992;70:30–2.PubMed
145.
Akerlund S, Campanello M, Kaijser B, Jonsson O. Bacteriuria in patients with a continent ileal reservoir for urinary diversion does not regularly require antibiotic treatment. Br J Urol. 1994;74:177–81.PubMed
146.
Bruce A, Reid G, Chan R, Costerton J. Bacterial adherence in the human ileal conduit: a morphological and bacteriological study. J Urol. 1984;132: 184–8.PubMed
147.
Greenwell T, Woodhams S, Smalley T, Mundy A. Effect of antibiotics on enterocystoplasty urinary nitrosamine levels. Urology. 2001;58:660–4.PubMed
148.
Wagstaff K, Woodhouse C, Rose G, Duffy P, Ransley P. Blood and urine analysis in patients with intestinal bladders. Br J Urol. 1991;68:311–6.PubMed
149.
van Nieuwkoop C, den Exter P, Elzevier H, den Hartigh J, van Dissel J. Intravesical gentamicin for recurrent urinary tract infection in patients with intermittent bladder catheterisation. Int J Antimicrob Agents. 2010;36:485–90.PubMed
150.
Botto H, Neuzillet Y. Effectiveness of a cranberry (Vaccinium macrocarpon) preparation in reducing asymptomatic bacteriuria in patients with an ileal enterocystoplasty. Scand J Urol Nephrol. 2010;44:165–8.PubMed
151.
Singh G, Thomas D. Bowel problems after enterocystoplasty. Br J Urol. 1997;79:328–32.PubMed
152.
N’Dow J, Leung H, Marshall C, Neal D. Bowel dysfunction after bladder reconstruction. J Urol. 1998;159:1470–4; discussion 1474–5.
153.
Jacobsen O, et al. Effect of enterocoated cholestyramine on bowel habit after ileal resection: a double blind crossover study. Br Med J (Clin Res Ed). 1985;290:1315–8.
154.
Hofmann AF, Poley JR. Role of bile acid malabsorption in pathogenesis of diarrhea and steatorrhea in patients with ileal resection. I. Response to cholestyramine or replacement of dietary long chain triglyceride by medium chain triglyceride. Gastroenterology. 1972;62:918–34.PubMed
155.
Fromm H, Malavolti M. Bile acid-induced diarrhoea. Clin Gastroenterol. 1986;15:567–82.PubMed
156.
Pattni S, Walters J. Recent advances in the understanding of bile acid malabsorption. Br Med Bull. 2009;92:79–93.PubMed
157.
Strawbridge L, Kramer S, Castillo O, Barrett D. Augmentation cystoplasty and the artificial genitourinary sphincter. J Urol. 1989;142:297–301.PubMed
158.
Robertson A, Davies J, Webb R, Neal D. Bladder augmentation and replacement. Urodynamic and clinical review of 25 patients. Br J Urol. 1991;68:590–7.PubMed
159.
McInerney PD, DeSouza N, Thomas PJ, Mundy AR. The role of urodynamic studies in the evaluation of patients with augmentation cystoplasties. Br J Urol. 1995;76:475–8.PubMed
160.
Lopez Pereira P, Somoza Ariba I, Martinez Urrutia M, Lobato Romero R, Monroe E. Artificial urinary sphincter: 11-year experience in adolescents with congenital neuropathic bladder. Eur Urol. 2006;50:1096–101.PubMed
161.
Hill D, Kramer S. Management of pregnancy after augmentation cystoplasty. J Urol. 1990;144:457–9.PubMed
162.
Niknejad K, Atala A. Bladder augmentation techniques in women. Int Urogynecol J Pelvic Floor Dysfunct. 2000;11:156–69.PubMed
163.
Fenn N, Barrington J, Stephenson T. Clam enterocystoplasty and pregnancy. Br J Urol. 1995;75: 85–6.PubMed
164.
Kearse WJ, St Clair S, Hixson C, Ritchey M. Functional characteristics of enterocystoplasty after interruption of the mesenteric blood supply. J Urol. 1993;150:593–6.PubMed