Federica Cadeddu, Franco Salis and Giovanni Milito1
(1)
Department of Experimental Medicine and Surgery, Tor Vergata University, Rome, Italy
Giovanni Milito
Email: giovanni.milito@virgilio.it
Abstract
Chronic constipation is a common self-reported gastrointestinal problem that affects between 2% and 34% of adults in various populations studied [1].
12.1 Biofeedback Therapy for Dyssynergic Defecation
12.1.1 Pelvic Floor Dyssynergia
Chronic constipation is a common self-reported gastrointestinal problem that affects between 2% and 34% of adults in various populations studied [1].
Approximately half of constipated patients suffer from obstructed defecation. Obstructed defecation is a broad term of the pathophysiologic condition describing the inability to evacuate contents from the rectum. Obstructed defecation is often a functional disorder, associated with psychoneurosis, rectal hyposensation, and anismus. It may result from functional, metabolic, mechanical, and anatomical derangements involving a rectoanal evacuatory mechanism. Failure to release and paradoxical contraction of puborectalis muscle and anal sphincters during straining are thought to be the main functional causes of obstructed defecation [2, 3].
Pelvic floor dyssynergia is usually defined on the basis of symptoms and physiologic and radiologic studies. Symptoms include a feeling of incomplete evacuation and rectal obstruction, passage of hard stools, rectal or vaginal digitations, and excessive straining in the constipated patient (stool frequency of fewer than three times per week).
Diagnostic criteria for pelvic floor dyssynergia include those for functional constipation, namely two or more of six symptoms present for the last 3 months with an onset more than 6 months in the past; the symptoms are straining, lumpy or hard stools, sensation of incomplete evacuation, sensation of anorectal obstruction/blockage, or manual maneuvers to facilitate defecation for more than one-quarter of bowel movements, or less than three bowel movements per week [4, 5].
To meet the criteria for a diagnosis of pelvic floor dyssynergia, the patient must also undergo objective diagnostic testing and demonstrate at least two of three abnormalities: impaired evacuation of the rectum, inappropriate contraction or less than 20% relaxation of the pelvic floor muscles (Figs. 12.1 and 12.2) and inadequate propulsive forces during defecation [4].
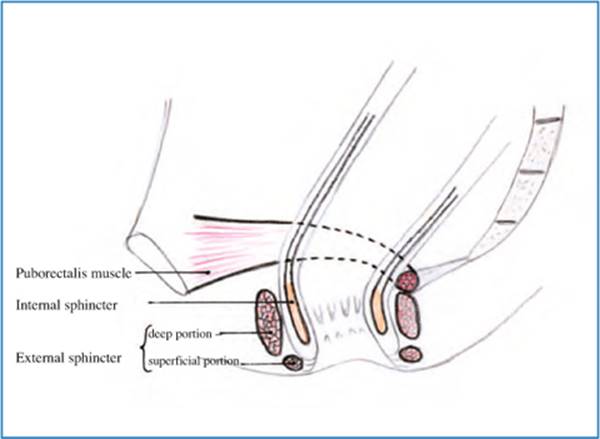
Fig. 12.1
Anatomy of anal sphincters and puborectalis muscle. Red puborectalis muscle, yellow external sphincter deep portion, pale yellow external sphincter superficial portion, orange internal sphincter
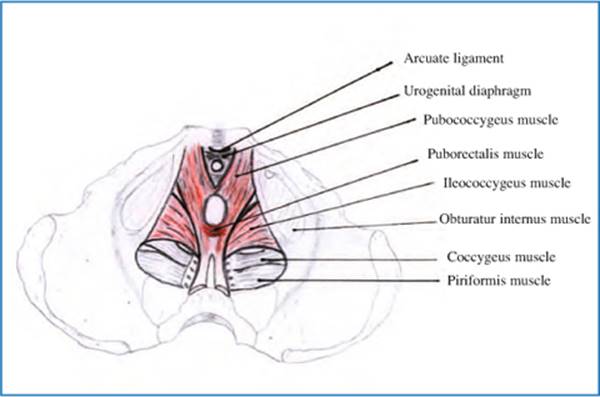
Fig. 12.2
Anatomy of levator ani complex
Concerning the diagnosis, careful perineal and digital rectal examination, colonic transit time study, anorectal manometry, anal electromyography (EMG) defecography, or dynamic magnetic resonance imaging (MRI) of the pelvic floor usually help to assess a correct diagnosis.
Anorectal manometry provides a comprehensive assessment of anal pressures, rectoanal reflexes, rectal pressures, sensation, and compliance. Several types of recording devices are available, but perfused catheters and balloon probes are among the most commonly used. A paradoxical increment in anal pressure on straining efforts is a distinctive feature of dyssynergic defecation [4] (Figs. 12.3 and 12.4). An increment in muscle motor activity on straining may be demonstrated by means of EMG either by intra-anal electrodes or by electrodes taped to the perianal skin.
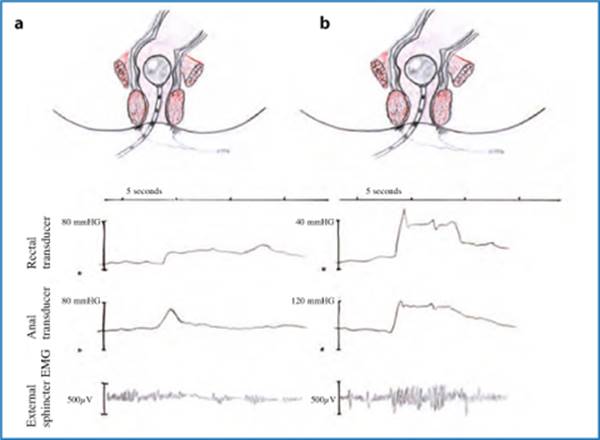
Fig. 12.3
Anal manometry and electromyography during defecation in a normal person (a) and in presence of pelvic floor dyssynergia (b)
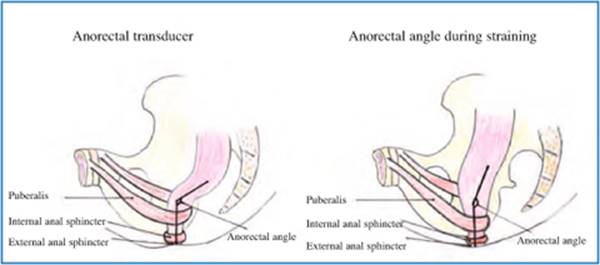
Fig. 12.4
Anorectal angle at rest and during straining in absence of pelvic floor dyssynergia
Defecography is a radiographic test providing morphological and functional information on the anorectum. Several parameters may be assessed, such as pelvic floor descent, anorectal angle, rectocele, and rectal prolapse. Failure of the anorectal angle to become more oblique on straining provides indirect evidence of defective pelvic floor relaxation, and impaired evacuation of contrast material is also suggestive of dyssynergia [4].
Anal EMG may be recorded either by intra-anal probes or by perianal EMG electrodes attached to the skin [6, 7]. The EMG activity used in biofeedback training is the averaged activity of large numbers of muscle cells rather than the activity of small groups of muscle cells innervated by a single axon. This averaged EMG activity is recorded with large electrodes on the skin or the mucosa of the anal canal rather than with needle electrodes. Averaged EMG recorded in this way is proportional to the strength of contraction of the underlying muscles (Fig. 12.4).
Defective expulsion is commonly investigated by asking the patient to defecate a 50 ml water-filled rectal balloon; patients with functional defecation disorders usually fail this test [7]. Some patients also have a higher threshold for perceiving the urge to defecate [6], but the clinical significance of this sensory dysfunction is ill-defined, in contrast to the relevance of rectal sensory impairment in fecal incontinence [8].
Patients with functional defecation disorders are often unresponsive to conservative medical management, and the surgical division of the puborectalis muscle (which has been proposed for the treatment of dyssynergic defecation) has resulted in poor benefit and an unacceptable risk of anal incontinence [1, 3]. Treatment with botulinum toxin injection may provide temporary improvement, but it remains an investigational treatment. Therefore, the gold standard of therapy of pelvic floor dyssynergia is conservative and is based on a high-fiber diet, physical activity, and biofeedback training [2].
12.1.2 Biofeedback Techniques for Pelvic Floor Dyssynergia
Biofeedback is a conditioning treatment where information about a physiologic process (contraction and relaxation of a muscle) is converted to a simple visual or auditory signal to enable the patient to learn to control the disordered function. Biofeedback is considered appropriate when specific pathophysiological mechanisms are known and the voluntary control of responses can be learned with the aid of systematic information about functions not usually monitored at a conscious level [6]. Biofeedback involves the use of pressure measurements (manometry) or averaged EMG activity within the anal canal to teach patients how to relax pelvic floor muscles when straining to defecate.
Various biofeedback techniques (intra-anal and perianal EMG monitoring, manometric anal probe biofeedback, intra-rectal balloon expulsion biofeedback, ultrasound biofeedback) have been investigated, but none has a superior success rate, which ranges from 30% to over 90% [9–11].
Despite controversy about the method of biofeedback and the number of sessions needed, it seems reasonable to use this generally safe technique as the initial treatment for pelvic floor dyssynergia.
Biofeedback training protocols vary among different centers [6, 7]. A mainstay of behavior therapy is to first explain the anorectal dysfunction and discuss its relevance with the patient before approaching the treatment [3, 7]. Biofeedback interventions for pelvic floor disorders are directed at teaching patients to relax their pelvic floor muscles while simultaneously applying a downward intra-abdominal pressure to generate propulsive force (Valsalva maneuver). Patients are shown anal manometry or EMG recordings displaying their anal function and are taught through trial and error to relax the pelvic floor and anal muscles during straining [6–8]. This objective is first pursued with the help of visual feedback on pelvic floor muscle contraction, accompanied by continuous encouragement from the therapist. When the patient has learned to relax the pelvic floor muscles during straining, the visual and auditory help are gradually withdrawn [6]. Another retraining option is to simulate defecation by means of an air-filled balloon attached to a catheter, which is slowly withdrawn from the rectum while the patient concentrates on the evoked sensation and tries to facilitate its passage [3, 7]. In the next phase of training, the patient is taught to defecate the balloon by bearing down, without the assistance of the therapist. Some centers also add a balloon sensory retraining to lower the urge perception threshold [12]. The number of training sessions is not standardized, but 4–6 sessions are frequently provided. Individual training sessions last 30–60 min.
Therapeutic sessions are professionally demanding, and a highly trained and motivated therapist is essential. No study has addressed the necessary training required for an individual to administer biofeedback therapy. In particular, it is unclear whether the adequate provider should be a physician, psychologist, or nurse. Experience varies among centers, but the low cost reimbursement provided for behavior therapy is likely to influence future choices.
12.1.3 Biofeedback for Pelvic Floor Dyssynergia: Literature Results
Controlled studies systematically comparing different biofeedback protocols with each other are lacking. In the literature, the symptomatic improvement rate has been shown to vary widely between 44% and 100% in several uncontrolled clinical trials. Subsequent randomized trials have confirmed the higher efficacy of biofeedback compared with standard therapy and laxatives.
Chiarioni et al. [13] randomized 109 dyssynergic patients to EMG biofeedback training or to polyethylene glycol and assessed outcomes at 6 and 12 months. Biofeedback patients were more satisfied than the control group (80% vs. 22%, p < 0.001) and reported greater reductions in blocked or incomplete bowel movements, straining, abdominal pain, and use of enemas and suppositories. Stool frequency increased in both groups.
Rao et al. [14] compared anal pressure biofeedback to two control conditions - sham biofeedback and standard care - in 77 patients. Standard care subjects received diet and life style advice, laxatives, and scheduled evacuations. At 3 months follow-up, the biofeedback reported significantly more complete spontaneous bowel movements, defecation improvement, and higher satisfaction than the sham-treated group.
Heymen et al. [15] compared EMG biofeedback to two control conditions: diazepam (5 mg) (a skeletal muscle relaxant) or placebo 1 h before attempted defecation. Prior to randomization, all 117 patients were provided with enhanced standard care that included diet, lifestyle measures, stool softeners, and scheduled evacuations during a 4-week run-in and those who reported adequate relief at the end of run-in (n = 18, 15%) were excluded. Eighty-four patients were randomized. At 3 months follow-up, biofeedback-treated patients reported significantly more unassisted bowel movements than placebo-treated patients (p = 0.005) In the intent-to-treat analysis, 70% of the biofeedback group reported adequate relief compared with 23% of diazepam-treated patients (p < 0.001) and 38% of placebo-treated patients (p = 0.017).
Although it is encouraging that more controlled studies have been carried out in recent years, these trials were heterogeneous with regard to inclusion criteria, treatment protocols, and end points.
The mechanism of action of biofeedback therapy is also not known. Although various parameters of colonic and anorectal function show improvement, and one study showed improvement in distal colonic blood flow [16], the precise alterations are unclear. Recent studies using bidirectional cortical-evoked potentials and transcranial magnetic stimulations suggest significant bidirectional brain-gut dysfunction in patients with dyssynergic defecation [16]. Whether an improvement in bowel function correlates with an improvement in brain-gut dysfunction remains to be explored.
Finally, concerning the technique of choice, a recent meta-analysis showed that in open-label studies, the mean success rate with pressure biofeedback was slightly greater than with EMG biofeedback (78% vs. 70%) [17]. No differences were found between anal versus perianal EMG recording. In addition, adding balloon feedback did not seem to influence the therapeutic outcome [17]. However, the majority of studies in the last 10 years have utilized EMG biofeedback rather than pressure feedback, even in the absence of scientific evidence [17]. There are no standardized protocols, and centers use different combinations of laboratory EMG training, home EMG training, and balloon feedback, depending on the researchers’ experience.
12.2 Biofeedback Therapy for Fecal Incontinence
12.2.1 Fecal Incontinence
Fecal incontinence (FI) is a debilitating and embarrassing problem facing approximately 2.2% of the US general population over 65 years old. The etiology of FI is multifactorial and can be due to several factors including neuropathic, traumatic, congenital, and obstetric trauma, as well as iatrogenic injuries caused by injudicious fistula surgery, hemorrhoidectomy, and lateral internal sphincterotomy, among others [18]. FI symptoms can range from mild to severe and the work-up and treatments of this disorder are just as varied. Patients may complain of incontinence to flatus, or liquid or solid stools. In some patients, just the concern that an accident may happen adversely affects their daily quality of life and limits their ability to interact socially due to fear and embarrassment. Several scoring systems have been created and validated to help patients and their medical practitioners quantify the severity of symptoms and the effects of FI on their daily life. These scores are used by physicians to plan treatment strategies and by researchers to study the outcomes of FI treatments [18].
The mechanism of fecal continence is extremely complex despite the simplicity that physicians often ascribe to it. The sphincter mechanism requires the ability to discriminate between solid, liquid, and gas; voluntarily allowing for the passage of one while holding the other components [19]. Treating FI requires an understanding of this complex pelvic floor musculature, innervation, and function, as well as the mechanisms that must be present to ensure continence. The internal and external sphincters and the puborectalis muscle comprise the sphincter mechanism. The internal anal sphincter (IAS) is a continuation of the circular, smooth, involuntary muscle of the rectum that accounts for the resting tone of the anus. The rectoanal inhibitory reflex allows the internal sphincter to relax in response to rectal distension, preparing the anal canal for defecation [18, 19]. The external anal sphincter (EAS) provides voluntary control over defecation and provides the squeezing pressure measured by anal manometry. The puborectalis is a U-shaped muscle that controls the rectoanal angle that increases during defecation. Both parasympathetic and sympathetic nerves provide the innervation of this sphincter complex. The pudendal nerve innervates both the puborectalis and EAS and when neurogenic incontinence is present, latency of this nerve can be detected [19]. Because of the embarrassing nature of FI, symptoms are often hidden by patients and thus are underreported and undertreated. Once these symptoms are voiced, it is important to obtain a detailed account of the incontinence. Descriptions of partial incontinence to only gas or liquid stools, and occasional or complete involuntary passage of solid stools should be provided to assess severity. Episodes of soiling or leakage and use of protective pads for undergarments are important, as well as the thorough assessment of general bowel habits. A careful history of anorectal surgery, colorectal disease, anal intercourse, obstetric trauma, rectal prolapse, and neurologic disorders should be taken.
Details of the patient’s stool frequency, consistency, or frequency of incontinent episodes should be obtained to assess the severity FI symptoms. There have been several score indices created to quantify symptoms: the Fecal Incontinence Severity Index (FISI), or the Cleveland Clinic Incontinence Score that combines the loss of flatus, liquid, and solid stools as well as impact of quality of life to assess the severity of FI. Other scoring systems specifically address the effects of FI on quality of life, as in Fecal Incontinence Quality of Life Questionnaire (FIQL) published by the American Society of Colon and Rectal Surgeons. The clinician can use these tools to assess the severity of symptoms and thus recommend a strategy for evaluation and treatment [19].
Regarding the diagnostic tests [20], the first step is to identify whether the incontinence is secondary to diarrhea. If so, endoscopic mucosal evaluation, stool tests, and breath tests may be useful. Anorectal manometry quantifies IAS and EAS function, rectal sensation, rectoanal reflexes, and rectal compliance. Anal endosonography provides an assessment of the thickness and structural integrity of the EAS and IAS and can detect scarring, loss of muscle tissue, and other local pathology. It is performed by using a 7–12 mHz rotating transducer with a focal length of 1–4 cm. More recently, three-dimensional ultrasound imaging has become available, which provides better delineation of anal sphincters and puborectalis and surrounding structures Rectal balloon distention with incremental volumes of air can be used for the assessment of both sensory responses and compliance. Incontinent patients may exhibit rectal hyposensitivity or hypersensitivity. MRI provides superior imaging with better spatial resolution of the EAS. A major contribution of anal MRI has been the recognition of external sphincter atrophy, and sometimes without pudendal neuropathy. The addition of dynamic pelvic MRI by using fast imaging sequences or MRI colpocystography, which involves rectal filling with ultrasound gel and having the patient evacuate while lying inside the magnet, may each provide better delineation of pelvic anatomy. The use of an endoanal coil significantly enhances the resolution and allows more precise definition of sphincter muscles. However, comparative studies with other technology, and costs and clinical utility have not been assessed. EMG of the anal sphincter identifies sphincter injury as well as denervation-reinnervation potentials that indicate neuropathy. Finally, the integrity of the entire spino-anorectal pathways that control anorectal function can be assessed by magnetic stimulation and recording of motor-evoked potentials [19, 20].
12.2.2 Biofeedback Techniques for Fecal Incontinence
Biofeedback treatment of FI was proposed by Engel and coworkers, 30 years ago [21]. Patients were taught to improve their ability to voluntarily contract the external anal sphincter during rectal filling, by improving the strength of the sphincter (motor skills training), or by increasing the ability to perceive weak rectal distention (discrimination training), or by combining the previous two mechanisms (training in the coordination of sphincter contractions with rectal sensation). No side-effects were reported and the treatment was generally well accepted. Further trials showed that therapeutic goals can be achieved through training that employs measurements of pressures (manometry) or electrical activity (EMG) in the anal canal [22].
12.2.2.1 Manometric Biofeedback
Biofeedback training aimed at increasing the strength of the EAS has usually been carried out by recording anal canal pressures, coupled with visual/auditory signals proportional to the pressures themselves. Anal pressure may be recorded by balloon probes or by perfused catheters [23]. During manometric recording, the patient is required to squeeze as in preventing defecation while being given visual feedback and verbal guidance on how to reach this goal. The patients may also be taught to inhibit wrong responses such as contraction of the abdominal muscles. Asking the patient to squeeze may be obtained in response to balloon distention of the rectum or without rectal distention [24]. Some authors have suggested that improving squeeze duration is more important than maximizing anal strength. Therefore, patients are taught to pursue this therapeutic goal as a part of the biofeedback protocol [24].
12.2.2.2 Electromyography Biofeedback
Strengthening the pelvic floor muscle may also be achieved by showing the patient a recording of the integrated (average) EMG activity from the striated muscles that surround the anal canal. In EMG training, the patient is asked to squeeze and relax without rectal distention, and home exercises in which the patient is required to repeatedly squeeze the pelvic floor muscles (Kegel exercises) are usually added to the training to further strengthen these muscles. Other methods of EMG recording of the pelvic floor employ an anal plug with surface electrodes; this is very easy to use and requires no preparation [25].
12.2.2.3 Sensory Discrimination Training
This is aimed at increasing the patient’s ability to perceive and respond to rectal distention [26]. After inserting a catheter-mounted balloon into the recrum, the latter is inflated with different air volumes; the patient is then asked to signal when the feeling of distention is perceived, or to contract the pelvic floor muscles in response to the distention. For these purposes, easily perceived distention with large volumes of air is given at first, and the volumes of distention are gradually decreased until the patient is able to perceive them with difficulty. Repeated distention slightly above and below the sensory threshold of the patient, coupled with the investigators’ feedback on the accuracy of detection, teach the patient to recognize distention of even weaker intensity [23].This type of sensory training is often coupled to sphincter strength training, asking the patient always to contract (as strongly as possible) in response to rectal distention and providing feedback on the strength of contraction and accuracy of detection [23]. Several pieces of evidence suggest that sensory discrimination training (aimed at reducing the threshold for perception of rectal distention) is very important for an effective biofeedback procedure [21]. We have recently evaluated 24 patients with severe, solid-stool FI [27] by teaching them to squeeze in response to rectal distention; the patients were evaluated 3 months after biofeedback training, and were classified as responders (< 75% decrease of incontinence episodes) or nonresponders. Comparison of the two groups showed that responders displayed significantly lower sensory thresholds after training with respect to nonresponders, but squeeze pressures were not significantly different between groups. Sensory thresholds measured before biofeedback training were good predictors of which patients would respond to it; in fact, patients with more severe sensory impairment had poor response to biofeedback training [27]. Sphincter strength and severity of FI before biofeedback training were not useful as predictors of outcome.
12.2.3 Biofeedback and Fecal Incontinence: Literature Results
Most of the available studies concerning the use of biofeedback to treat FI have been carried out by manometric means; however, a clear superiority of pressure versus EMG feedback has not been reported [28].
A review of all the studies reported in the literature, regardless of etiology, shows that about two-thirds of patients display at least a 75% decrease in episodes of FI [29], although only about 50% develop complete continence. However, it must be stressed that: (1) no uniform criteria for defining improvement or assessing outcome have been adopted; (2) inclusion criteria differed; (3) treatment protocols varied; and (4) only few prospective, randomized, parallelgroup studies have been published, not enough to draw conclusions on the overall efficacy of biofeedback training. In addition, recent randomized studies have not confirmed the optimistic outcome of previous open studies.
In a first randomized controlled study, Whitehead et al. compared biofeedback plus behavioral management with behavioral management alone in children with FI due to myelomeningocele [30]; both groups displayed significant improvement, suggesting that biofeedback has the same effects as behavioral management for most children with myelomeningocele. In a second controlled study, van der Plas and coworkers [31] studied 71 children with FI without constipation and randomized them to standard care and laxatives or standard care and laxatives plus biofeedback. At 12–18 months follow-up, approximately 50% of children in both groups showed significant improvement in symptoms. A trend toward better outcome was shown in the biofeedback group, but statistical significance was not reached. In the first randomized study of biofeedback in adults with FI, Miner et al. [32] randomized 25 patients to three sessions of either sensory discrimination training without biofeedback on sphincter strength, or equivalent distention without feedback on the accuracy of their detection of the strength of contractions. Patients in the sensory training group had significant decrease of frequency of episodes of incontinence with respect to controls, but between-group differences did not reach statistical significance (probably due to small sample size). Control patients were then given sensory training, and displayed improvement in continence. Thereafter, all patients were randomized again to sphincter-strengthening exercises without biofeedback or to squeezing in response to rectal distention with feedback. Overall, the patients had further improvement of continence in this second step of the study, but no significant differences were observed between groups, suggesting that sensory training is important for the treatment of incontinence, although the results are not definitive due to the small sample size. Recently, the St Mark group reported a large, randomized, controlled study on 171 adults with FI [33]. Patients were randomized to four groups: (1) standard care with advice; (2) standard care with advice, plus anal sphincter exercises taught verbally and via digital examinations; (3) group 2 protocol plus biofeedback therapy run at the clinic; (4) group 3 plus home biofeedback device. Approximately half of the patients in all groups reported improvement of symptoms at 1 year follow-up. Interestingly, quality-of-life measurements, bowel symptoms, and anal sphincter pressures were improved by similar percentages in all groups. Biofeedback therapy yielded no greater benefit than did standard care with advice on an intention-to-treat analysis.
Moreover, another prospective, randomized, controlled study comparing pelvic floor exercises plus anal exercises taught via digital examination with either manometry or anal ultrasound-guided biofeedback in 120 adults with FI failed to show any additional benefit of behavior therapy over Kegel exercises in terms of clinical outcome, quality-of-life measurements, and anal pressures [34]. In that trial, a clinical benefit was evident in the short term in approximately 70% of all patients. The same group then reported this clinical benefit as substantially preserved in the long-term follow-up. Interestingly, quality-of-life measurements and subjective perception of catching up with incontinence improved even in patients whose incontinence scores worsened. Therefore, intervention per se seems to improve subjective symptoms perception in FI.
12.3 Anorectal Pain
Two types of functional anorectal pain are recognized, which may overlap but have different presentation with regard to symptom duration, frequency, and intensity. Levator ani syndrome generally presents as a dull or indistinct feeling of pressure, ache, or pain in the upper rectum. Rome II diagnostic criteria require this to have been experienced 12 weeks out of the last 12 months and for the duration of the episodes to be at least 20 min [26]. It has been reported to affect 6.6% of the general population [35], is more common in women than men, and most commonly affects individuals between the ages of 30 and 60 years. Proctalgia fugax presents as sudden, severe anal, or lower rectal pain that lasts from a few seconds to a few minutes. The pain is often intense enough to disrupt normal life activities [36], but rarely occurs more often than five times a year. Population prevalence estimates for proctalgia fugax vary from 8% to 14% [35], but because of the brief and infrequent nature of the episodes, few sufferers seek medical attention for the condition.
12.3.1 Biofeedback Techniques for Anorectal Pain
All three of the prospective studies of biofeedback for anorectal pain have used pressure feedback from the EAS.
Regardless of the specific biofeedback protocols used to treat anorectal disorders, the in clinic biofeedback training is almost invariably supplemented with other potentially therapeutic components. These often include advice, reassurance and patient education, prescribed pelvic floor home exercises, practice with balloon defecation, laxatives, enemas, fiber supplements, or the use of EMG biofeedback home trainer devices.
The number of biofeedback sessions used in published studies to treat patients for functional anorectal disorders varies greatly, and ranges from 1–12 sessions. Many studies reported high success rates with as few as three or four sessions per patient.
12.3.2 Biofeedback and Anorectal Pain: Literature Results
Three clinical trials of biofeedback for functional anorectal pain were found in the literature, none of which were randomized.
Only one study was found in the literature that examined biofeedback treatment in any kind of controlled fashion. In that study, Ger and colleagues (2002) [37] provided three different treatments to adults who had anorectal pain without organic pathology and had failed conservative medical management: biofeedback (14 patients) electrogalvanic stimulation (29 patients), and steroid caudal block (11 patients). At follow-up 2–36 months after treatment completion, 43% of biofeedback patients versus 38% of those who had electrogalvanic stimulation and 18% of patients who had steroid block treatment reported successful pain relief. These results were not statistically different between groups. Apart from the lack of randomization into groups and the broad variability in follow-up time points across participants, the conclusions that may be made from this study are very limited due to the confounding factor that many participants received more than one of the test treatments, in varying order.
A couple of uncontrolled studies have suggested that biofeedback may be successfully used to treat anorectal pain. Grimaud and colleagues (1991) [38] treated 12 patients with functional anorectal pain with pressure biofeedback to enhance control of theEAS, and reported that all patients improved, with a mean of eight sessions needed to achieve benefit. The improvement was maintained in 11 of the 12 patients beyond a 16-month follow-up.
Heah et al. (1997) [39] treated 16 patients with balloon pressure biofeedback. The patients were significantly lower on pain scores as a group after treatment. Success rate was not stated in this report. However, it was noted that all but 2 of the 16 patients discontinued use of analgesics after treatment.
References
1.
Lembo A, Camilleri M (2003) Chronic constipation. N Engl J Med 349:1360–1368PubMedCrossRef
2.
Pescatori M, Milito G, Fiorino M, Cadeddu F (2009) Complications and reinterventions after surgery for obstructed defecation. Int J Colorectal Dis 24:951–959PubMedCrossRef
3.
Chiarioni G, Salandini L, Whitehead WE (2005) Biofeedback benefits only patients with outlet dysfunction, not patients with isolated slow transit constipation. Gastroenterology 129:86–97PubMedCrossRef
4.
Bharucha AE, Wald A, Enck P, Rao S (2006) Functional anorectal disorders. Gastroenterology 130:1510–1518PubMedCrossRef
5.
Longstreth GF, Thompson WG, Chey WD et al (2006) Functional bowel disorders. Gastroenterology 130:1480–1491PubMedCrossRef
6.
Scarlett YV (2005) Anorectal manometry and biofeedback. In: Drossman DA, Shaheen NJ, Grimm IS (eds) Handbook of gastroenterologic procedures. Lippincott Williams & Wilkins, Philadelphia, pp 341–348
7.
Bassotti G, Chistolini F, Sietchiping-Nzepa F (2004) Biofeedback for pelvic floor dysfunction in constipation. BMJ 328:393–396PubMedCrossRef
8.
Chiarioni G, Bassotti G, Stanganini S et al (2002) Sensory retraining is key to biofeedback therapy for formed stool fecal incontinence. Am J Gastroenterol 97:109–117PubMedCrossRef
9.
Fleshman JW, Dreznik Z, Meyer K et al (1992) Outpatient protocol for biofeedback therapy of pelvic floor outlet obstruction. Dis Colon Rectum 35:1–7PubMedCrossRef
10.
Palsson OS, Heymen S, Whitehead WE (2004) Biofeedback treatment for functional anorectal disorders: a comprehensive efficacy review. Appl Psychophysiol Biofeedback 29:153–174PubMedCrossRef
11.
Lau CW, Heymen S, Alabaz O et al (2004) Prognostic significance of rectocele, intussusception, and abnormal perineal descent in biofeedback treatment for constipated patients with paradoxical puborectalis contraction. Dis Colon Rectum 43:478–482CrossRef
12.
Rao SS, Welcher KD, Pelsang RE (1997) Effects of biofeedback therapy on anorectal function in obstructive defecation. Dig Dis Sci 42:2197–2205PubMedCrossRef
13.
Chiarioni G, Whitehead WE, Pezza V et al (2006) Biofeedback is superior to laxatives for normal transit constipation due to pelvic floor dyssynergia. Gastroenterology 130:657–664PubMedCrossRef
14.
Rao SS, Kinkade KJ, Schulze KS et al (2005) Biofeedback therapy (bt) for dyssynergic constipation-randomized controlled trial. Gastroenterology 128 Suppl 2:A269
15.
Heymen S, Scarlett Y, Jones K et al (2005) Randomized controlled trial shows biofeedback to be superior to alternative treatments for patients with pelvic floor dyssynergia-type constipation. Gastroenterology 128 Suppl 2:A266
16.
Emmanuel AV, Kamm MA (2001) Response to a behavioural treatment, biofeedback, in constipated patients is associated with improved gut transit and autonomic innervation. Gut 49:214–219PubMedCrossRef
17.
Heymen S, Jones KR, Scarlett Y, Whitehead WE (2003) Biofeedback treatment of constipation: a critical review. Dis Colon Rectum 46:1208–1217PubMedCrossRef
18.
Bharucha AE (2003) Fecal incontinence. Gastroenterology 124:1672–1685PubMedCrossRef
19.
Rudolph W, Galandiuk S (2002) A practical guide to the diagnosis and management of fecal incontinence. Mayo Clin Proc 77:271–275PubMedCrossRef
20.
Diamant NE, Kamm MA, Wald A, Whitehead WE (1999) AGA technical review on anorectal testing technique. Gastroenterology 116:735–760PubMedCrossRef
21.
Engel BT, Nikoomanesh P, Schuster MM (1974) Operant conditioning of rectosphincteric responses in the treatment of fecal incontinence. N Engl J Med 290:646–649PubMedCrossRef
22.
Bassotti G, Whitehead WE (1994) Biofeedback as a treatment approach to gastrointestinal tract disorders. Am J Gastroenterol 89:158–164PubMed
23.
Whitehead WE, Heymen S, Schuster MM (2002) Motility as a therapeutic modality: bio-feedback treatment of gastrointestinal disorders. In Schuster MM, Crowell MD, Koch KL (eds) Schuster atlas of gastrointestinal motility, 2nd edn. BC Decker, Hamilton, pp 381–397
24.
Patankar SK, Ferrara A, Larach SW et al (1997) Electromyographic assessment of bio-feedback training for fecal incontinence and chronic constipation. Dis Colon Rectum 40:907–911PubMedCrossRef
25.
Eisman E, Tries J (1993) A new probe for measuring electromyographic activity from multiple sites in the anal canal. Dis Colon Rectum 36:946–952PubMedCrossRef
26.
Whitehead WE, Wald A, Diamant NE (2000) Functional disorders of the anus and rectum. In Drossman DA, Corazziari E, Talley NJ, Thompson WG, Whitehead WE (eds) Rome II. The functional gastrointestinal disorders, 2nd edn. Degnon Associates, McLean, VA, pp 483–532
27.
Chiarioni G, Scattolini C, Bonfante F, Vantini I (1993) Liquid stool incontinence with severe urgency: anorectal function and effective bio-feedback therapy. Gut 34:1576–1580PubMedCrossRef
28.
Heymen S, Jones KR, Ringel Y et al (2001) Biofeedback treatment of fecal incontinence: a critical review. Dis Colon Rectum 44:728–736PubMedCrossRef
29.
Enck P (1993) Biofeedback training in disordered defecation: a critical review. Dig Dis Sci 38:1953–1960PubMedCrossRef
30.
Whitehead WE, Parker L, Bosmajian L et al (1986) Treatment of fecal incontinence in children with spina bifida: comparison of bio-feedback and behavioral modification. Arch Phys Med Rehabil 67:218–224PubMed
31.
van der Plas RN, Benninga MA, Redekop WK et al (1996) Randomised trial of bio-feedback training for encopresis. Arch Dis Child 75:367–374PubMedCrossRef
32.
Miner PB, Donnelly TC, Read NW (1990) Investigation of mode of action of bio-feedback in treatment of fecal incontinence. Dig Dis Sci 35:1291–1298PubMedCrossRef
33.
Norton C, Chelvanayagam S, Wilson-Barnett J et al (2003) Randomized controlled trial of biofeedback for fecal incontinence. Gastroenterology 125:1320–1329PubMedCrossRef
34.
Solomon MJ, Pager CK, Rex J, Roberts RA, Manning J (2003) Randomized, controlled trial of bio-feedback with anal manometry, transanal ultrasound, or pelvic floor retraining with digital guidance alone in the treatment of mild to moderate fecal incontinence. Dis Colon Rectum 46:703–710PubMedCrossRef
35.
Drossman DA, Andruzzi E, Temple RD et al (1993) U.S. householder survey of functional gastrointestinal disorders. Prevalence, sociodemography, and health impact. Dig Dis Sci 38:1569–1580PubMedCrossRef
36.
Thompson WG, Heaton KW (1980) Proctalgia fugax. J R Coll Physicians Lond 14:247–248PubMed
37.
Ger GC, Wexner S, Jorge JM et al (1993) Evaluation and treatment of chronic intractable rectal pain-A frustrating endeavor. Dis Colon Rectum 36:139–145PubMedCrossRef
38.
Grimaud JC, Bouvier M, Naudy B et al (1991) Manometric and radiologic investigations and biofeedback treatment of chronic idiopathic anal pain. Dis Colon Rectum 34:690–695PubMedCrossRef
39.
Heah SM, Ho YH, Tan M, Leong AF (1997) Biofeedback is effective treatment for levator ani syndrome. Dis Colon Rectum 40:187–189PubMedCrossRef