Akitsugu Kawashima
Abstract
Some reports have stated that surgical treatment is needed for patients with moyamoya disease involving hypoperfusion in the anterior cerebral artery (ACA) territory. We introduced a new direct bypass technique using the long superficial temporal artery (STA) graft for such cases in 2010. Moyamoya patients who need reconstruction in the ACA territory also frequently need reconstruction in the middle cerebral artery (MCA) territory. In this chapter, we describe STA-ACA/MCA double bypasses with long grafts. This powerful revascularization technique can supply much blood flow in the greater part of the frontal lobe. It is essential to the procedure that the 10-cm-long STA graft passes intradurally under the residual bone bridge between the two separate craniotomies for the ACA and the MCA recipient arteries to prevent kinking of the long STA graft. Difficulties of this procedure are long STA graft preparation/set-up and anastomosis to the ACA cortical arteries, which are smaller in size than the MCA cortical arteries and lie in the sulcus in many cases. It is very important to prepare for the graft to avoid damage to the graft, which can include hypertextention, heat form coagulator, and insufficient dissection layer. Also, close attention should be paid to a natural STA graft course. Key to successful anastomosis is (1) having a good view of suturing, (2) avoiding suturing the contralateral wall, and (3) suturing suitable margin and interval of stitches, specifically making as small number of stitches as possible to expand the orifice.
Keywords: moyamoya disease, STA-MCA bypass, STA- ACA bypass, long graft, anastomosis
There have been some reports about surgical treatment for patients with moyamoya disease involving hypoperfusion in the anterior cerebral artery (ACA) territory. Some procedures of indirect revascularization in the ACA territory were reported to help cases of moyamoya disease to prevent ischemic stroke. However, effectiveness of indirect revascularization has some limitations, taking several months to obtain its effect and having potentially insufficient effect compared to direct bypass. Superficial temporal artery-middle cerebral artery (STA-MCA) bypass may improve ischemia in both the ACA and the MCA territories through leptomeningeal anastomosis between capillary vessels of the ACA and the MCA. However, it’s unclear before operation whether blood supply in the ACA territory from STA-MCA bypass through leptomeningeal anastomosis is sufficient.
Previously, direct bypass in the ACA territory using the peripheral branch of the STA for the graft was conducted. We introduced new techniques using the long STA graft, which can allow wide blood supply from the bypass in 2010. Moyamoya patients who need reconstruction in the ACA territory also require frequently reconstruction in the MCA territory. In this chapter, we describe STA- ACA/MCA double bypasses with a long graft. This procedure has the potential for powerful revascularization that can provide blood supply in the whole frontal lobe.
The indication for STA-ACA/MCA bypasses with long grafts in cases of moyamoya disease are as follows: (1) hypoperfusion with poor vasoreactivity in the ACA and the MCA territories based on cerebral blood flow (CBF) study and angiographic study, and (2) ischemic symptoms including the lower extremities. A typical angiographic finding is severe internal carotid artery terminal stenosis/occlusion with poor collateral blood flow from ipsilateral posterior cerebral artery, contralateral ACA, or ipsilateral MCA.
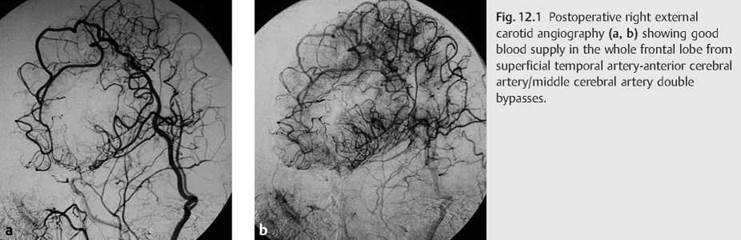
This procedure requires much time (about 5 hours) and takes a lot of effort, specifically a large skin incision with two separate craniotomies, long STA graft dissection, and anastomosis for small-size recipient artery. However, this procedure can achieve immediate blood supply from the bypasses in both ACA and MCA territories. Moreover, this powerful revascularization has the possibility to provide blood flow in the greater part of the frontal lobe (Fig. 12.1a, b).
12.4.1 Strength
• This procedure can provide immediate extensive blood supply in the whole frontal lobe in many cases.
12.4.2 Weaknesses
• Large skin incision and two separate craniotomies are needed in this procedure.
• Long graft dissection, usually frontal branch preparation from the galeal side, should occur, with care not to damage the graft.
• The condition of anastomosis to the ACA cortical arteries is more difficult than the condition of anastomosis to the MCA cortical arteries because size of recipient arteries in the ACA territory are smaller than in the MCA territory and recipient arteries in the ACA territory usually lie in the sulcus, thus allowing only a narrow working space.
12.4.3 Opportunity
• This procedure can provide extensive blood supply in the greater part of the frontal lobe in many cases, so improvement of cognitive function may be established.
12.4.4 Threats
• Using a long STA graft may increase the risk of graft kink/rotation and graft damage to the vessel wall of the STA. This may cause an increase in the rate of graft occlusion in long-term follow-up.
• The risk of skin trouble after operation may increase caused by the long STA dissection.
Contraindications to STA-ACA/MCA bypasses with long grafts are as follows: cases with insufficient STA development and young pediatric cases. There are probably 10 to 20% of cases where STAs are too small in size or too many in branching to be prepared as the long graft. In our experience of 62 cases, blood supply from bypasses has been commonly developing in the greater part of the frontal lobe. However, blood supply from bypasses that gradually shifted to indirect collaterals was shown particularly in pediatric cases less than 10 years old.
To determine the suitable indication of this procedure, it is very important to evaluate preoperative angiographical findings and CBF study. To prepare the STA long graft, preoperative evaluation of the development of the STA is also needed. Taking 100 mg aspirin daily starting more than 1 week before operation is necessary to prevent intraoperative thrombosis formation at the orifice of the anastomosis. The patient should stop the use of aspirin on the day of the operation. Intravenous boluses of heparin are not needed, and aspirin is restarted 3 to 5 days after operation depending on presence of postoperative hemorrhage.
STA-ACA/MCA bypasses with long grafts may be challenging procedures because of the preparation of long graft of STA and the smaller size of the recipient artery with a narrow anastomosis field. However, we believe that bypass surgeons who have had certain experience with direct bypasses in cases of moyamoya disease can achieve successful bypasses in this procedure. Large skin incision and two separate craniotomies may lead to skin trouble or infection at a higher rate than the STA-MCA single bypass. In fact, rate of postoperative skin trouble and infection needing additional treatment are 0 and 1.5%, respectively, in our 68 procedures. Also, possible complications in terms of direct bypass are perioperative infarction and hyperperfusion syndrome, the same as standard direct bypass procedure.
The patient is placed in the supine position under general anesthesia. The head is elevated approximately 20 cm and fixed without pin fixation. The PaCO2 should remain at 35 to 42 mm Hg during the operation. Anesthesia is maintained with Propofol venous infusion (6-10 mg/kg/h). Mannitol is not used in this operation. After the operation, the patient is kept sedated for 2 to 6 hours postoperatively and his or her blood pressure is controlled to stay between 100 and 120 mm Hg to prevent postoperative hemorrhage caused by hyperperfusion.
The skin incision is made on the parietal branch of the STA and turned frontally to the midline (Fig. 12.2a). One of the two STA branches undergoes about a 10-cm dissection for the STA-ACA bypass. In most patients, the frontal branch of the STA is selected, and the other branch is undergoes about a 6-cm dissection for the STA-MCA bypass. The frontal branch of the STA is dissected from the galeal side. The temporal muscle is divided, and two separate craniotomies, a frontotemporal and a mediofrontal, each 5 cm in diameter, are performed. The craniotomy for the STA-ACA bypass is made around the coronal suture (Fig. 12.2b). Blue line is made on the long STA graft to avoid twisting the graft (Fig. 12.2c).
A long STA graft is passed intradurally under the residual bone bridge between the two craniotomies to prevent kinking (Fig. 12.2d). The recipient artery, which is a cortical branch from the ACA, is frequently located around the level of the coronal suture. However, there are some possibilities of facing difficulty in finding the proper size of the recipient artery. Some tips to detect the proper recipient artery are mentioned in Chapter 12.11. The long STA graft is anastomosed to the cortical artery of the ACA with 10-0 monofilament nylon using intermittent sutures. The short graft STA is anastomosed to the cortical artery of the MCA in the same manner (Fig. 12.2e).
The dura is closed, the bone flaps are returned, and the temporal muscle and the skin are closed while avoiding kinking of the STA graft during these procedures.

Intraoperative difficulties of STA-ACA/MCA bypasses with long grafts are divided into problems of the graft and location of the recipient arteries.
Regarding the graft, there are two difficult points. One is preparation of the graft. In many cases, the frontal branch of the STA is selected for the graft of the STA-ACA bypass. It is dissected from the galeal side as long as possible (approximately 10 cm). Especially dissection of the distal part of the frontal branch of the STA should receive close attention to prevent vessel wall damage. Careful dissection of the graft is one of the keys to achieving a good bypass. The other is prevention of kinking/rotating long graft including recipient artery around anastomosis. The long STA graft is easily kinked/rotated; however, it tends to be overlooked. The recipient artery around anastomosis is also easy to kink due to the unnatural graft course. Strongly taking care of the full-length long graft is very important.
Size of the recipient artery in the ACA territory tends to be smaller than in the MCA territory. Suitable recipient arteries of the ACA could be detected in the 5 cm in diameter craniotomy around the coronal suture as mentioned above. However, the suitable recipient artery could not be found on the brain surface in some cases. Good size of the recipient artery can be detected in the sulcus in such a case. Recipient artery in the sulcus is a tough condition for anastomosis procedures in spite of the artery having less than 1 cm of depth and being very narrow and watery.
To prevent kinking/rotating, a blue line is drawn on the long graft (Fig. 12.2c) and the long STA graft is passed intradurally under the residual bone bridge between two separate craniotomies (Fig. 12.2d).
A relatively large size cortical ACA is located in the sulcus frequently. Indocyanine green fluorescence system helps us to find suitable recipient arteries, showing dull and dark but having a large size vessel (Fig. 12.3a-c). There are salvage maneuvers to make the more superficial and wider working space for the recipient artery in the sulcus, pulling the arachnoid at the margin of the sulcus laterally with 9-0 monofilament suturing, sheeting the suitable rubber under the recipient artery (Fig. 12.4a, b).
We use silicone rubber stent during anastomosis to make reliable anastomosis. It helps us to get a good visualization of vessel walls to keep up the threedimensional shape of the recipient artery after osteotomy and to avoid suturing the contralateral wall (Fig. 12.5a, b). The silicone rubber stent is strongly effective, especially for small-sized and thin-vessel-wall recipient arteries.



12.12.1 Graft Management
It’s very important to prepare for the graft procedure to avoid damaging it, which could include hypertextention, transfer of overheating from the coagulator to the graft, and insufficient dissection layer. Also, the natural graft course should be paid close attention (Fig. 12.2e).
12.12.2 Anastomosis
Key to successful anastomosis is (1) having a good view of the anastomotic field, (2) avoiding suturing the contralateral wall, and (3) suturing a suitable margin and interval of stitches, specifically making as small number of stitches as possible to expand the orifice (Fig. 12.5a, b).
12.12.3 Training
Training is indispensable because steady hands make good anastomosis. We have trained using a silicone microtube. One handled hours of training has prepared us for the bypass surgery. One thousand hours of training has helped us to achieve a mastery skill level.
Suggested Readings
Fujimura M, Shimizu H, Inoue T, Mugikura S, Saito A, Tominaga T. Significance of focal cerebral hyperperfusion as a cause of transient neurologic deterioration after extracranial-intracranial bypass for moyamoya disease: comparative study with non-moyamoya patients using N-isopropyl-p-[(123)I]iodoamphetamine single-photon emission computed tomography. Neurosurgery. 2011; 68(4):957-964, discussion 964-965
Iwama T, Hashimoto N, Miyake H, Yonekawa Y. Direct revascularization to the anterior cerebral artery territory in patients with moyamoya disease: report of five cases. Neurosurgery. 1998; 42(5):1157-1161, discussion 1161-1162
Karasawa J, Kikuchi H, Furuse S, Kawamura J, Sakaki T. Treatment of moyamoya disease with STA-MCA anastomosis. J Neurosurg. 1978; 49 (5):679-688
Kawashima A, Kawamata T, Yamaguchi K, Hori T, Okada Y. Successful superficial temporal artery-anterior cerebral artery direct bypass using a long graft for moyamoya disease: technical note. Neurosurgery. 2010; 67(3) Suppl Operative:ons145-ons149, discussion ons149
Khan N, Schuknecht B, Boltshauser E, et al. Moyamoya disease and moya- moya syndrome: experience in Europe; choice of revascularisation procedures. Acta Neurochir (Wien). 2003; 145(12):1061-1071, discussion 1071
Matsushima T, Inoue T, Suzuki SO, Fujii K, Fukui M, Hasuo K. Surgical treatment of moyamoya disease in pediatric patients—comparison between the results of indirect and direct revascularization procedures. Neurosurgery. 1992; 31(3):401 -405
Miyamoto S, Akiyama Y, Nagata I, et al. Long-term outcome after STA-MCA anastomosis for moyamoya disease. Neurosurg Focus. 1998; 5(5):e5
Okada Y, Shima T, Yamane K, Yamanaka C, Kagawa R. Cylindrical or T-shaped silicone rubber stents for microanastomosis—technical note. Neurol Med Chir (Tokyo). 1999; 39(1):55-57, discussion 57-58