Deanna L. Kelly, Elaine Weiner, and Heidi J. Wehring
LEARNING OBJECTIVES
Upon completion of the chapter, the reader will be able to:
1. Explain the pathophysiologic mechanisms that are thought to underlie schizophrenia.
2. Recognize the signs and symptoms of schizophrenia and be able to distinguish among positive, negative, and cognitive symptoms of the illness.
3. Identify the treatment goals for a patient with schizophrenia.
4. Recommend appropriate antipsychotic medications based on patient-specific data.
5. Compare the side-effect profiles of individual antipsychotics.
6. Describe the components of a monitoring plan to assess the effectiveness and safety of antipsychotic medications.
7. Educate patients and families about schizophrenia, treatments, and the importance of adherence to antipsychotic treatment.
KEY CONCEPTS
![]() A diagnosis of schizophrenia is made clinically, as there are no psychological assessments, brain imaging, or laboratory examinations that confirm the diagnosis.
A diagnosis of schizophrenia is made clinically, as there are no psychological assessments, brain imaging, or laboratory examinations that confirm the diagnosis.
![]() The goals of treatment are to reduce symptomatology, decrease psychotic relapses, and improve patient functioning and social outcomes.
The goals of treatment are to reduce symptomatology, decrease psychotic relapses, and improve patient functioning and social outcomes.
![]() Patients presenting with odd behaviors, illogical thought processes, bizarre beliefs, and hallucinations should be assessed for schizophrenia.
Patients presenting with odd behaviors, illogical thought processes, bizarre beliefs, and hallucinations should be assessed for schizophrenia.
![]() The cornerstone of treatment is antipsychotic medications. Because most patients with schizophrenia relapse when not medicated, long-term treatment is usually necessary.
The cornerstone of treatment is antipsychotic medications. Because most patients with schizophrenia relapse when not medicated, long-term treatment is usually necessary.
![]() Psychosocial support is needed to help improve functional outcomes.
Psychosocial support is needed to help improve functional outcomes.
![]() Compared to the older antipsychotics (first-generation antipsychotics [FGAs]), the more recently developed second-generation antipsychotics (SGAs) are associated with a lower risk of motor side effects (tremor, stiffness, restlessness, and dyskinesia); may offer greater benefits for affective, negative, and cognitive symptoms; and may prolong the time to psychotic relapse.
Compared to the older antipsychotics (first-generation antipsychotics [FGAs]), the more recently developed second-generation antipsychotics (SGAs) are associated with a lower risk of motor side effects (tremor, stiffness, restlessness, and dyskinesia); may offer greater benefits for affective, negative, and cognitive symptoms; and may prolong the time to psychotic relapse.
![]() SGAs as a class are heterogeneous with regard to side-effect profiles. Many SGAs carry an increased risk for weight gain and for the development of glucose and lipid abnormalities; therefore careful monitoring is essential.
SGAs as a class are heterogeneous with regard to side-effect profiles. Many SGAs carry an increased risk for weight gain and for the development of glucose and lipid abnormalities; therefore careful monitoring is essential.
![]() Education of the patient and family regarding the benefits and risks of antipsychotic medications and the importance of adherence to their therapeutic regimens must be integrated into pharmacologic management.
Education of the patient and family regarding the benefits and risks of antipsychotic medications and the importance of adherence to their therapeutic regimens must be integrated into pharmacologic management.
In most cases schizophrenia is a chronically debilitating disorder and is likely one of the most devastating of chronic medical illnesses. Conceptually, schizophrenia might better be thought of as a clinical syndrome, comprising several disease entities that manifest with psychotic symptoms, including hallucinations, delusions, and disordered thinking. Commonly, these more flagrant symptoms are accompanied by more insidious ones, including cognitive impairment (abnormalities in thinking, reasoning, attention, memory, and perception), impaired insight and judgment, loss of motivation (avolition), loss of emotional range (restricted affect), and a decrease in spontaneous speech (poverty of speech). The latter three symptoms are termed negative symptoms, and when taken together, are frequently called the deficit syndrome. Cognitive impairments and negative symptoms account for much of the poor social and functional outcomes observed in schizophrenia. Schizophrenia is the fourth leading cause of disability among adults and is associated with substantially lower rates of employment, marriage, and independent living compared to population norms. Approximately 10% of people with schizophrenia die by suicide.1 However, earlier diagnosis and treatment, as well as advances in research and newer treatment developments, have led to better outcomes for people who suffer from this complex and challenging illness.
Patient Encounter, Part 1
AC is a 28-year-old, single African American male with an approximately 3-year history of paranoia, increasing use of marijuana and cocaine, and poor work performance. His symptoms seemed to intensify around his being caught taking money from his girlfriend’s bank account to buy cocaine. Since then he has become suspicious that the police were watching his movements and that people on the street knew personal information about him. He has left several jobs one after another due to his belief that other employees were sabotaging him and that people were talking behind his back. He occasionally hears his father speaking to him but has not seen him in 10 years. He is sad and hopeless about the state of his life and very guilty about his substance abuse and stealing from his girlfriend. Though supportive of him, the patient’s girlfriend is feeling somewhat frustrated by the patient’s withdrawal from her and reluctance to socialize to the extent that they had in the past.
What diagnoses are suggested by this presentation?
What additional information would help to clarify the diagnosis?
EPIDEMIOLOGY AND ETIOLOGY
One percent of the world’s population suffers from schizophrenia, and symptoms usually first present in late adolescence or early adulthood.1 While equally prevalent between genders, symptoms generally appear earlier in males, and males have a younger age at first hospitalization (15–24 years) compared to females (25–34 years).
The etiology of schizophrenia remains largely unknown, though the evidence strongly supports a genetic basis for the disorder. First-degree relatives of patients with schizophrenia carry a 10% risk of developing the disorder. When both parents have the diagnosis, the risk to their offspring is 40%. For monozygotic twins, the likelihood of one twin developing the illness if the other twin has schizophrenia is about 50%. Many genes have been weakly associated with the development of schizophrenia; however, no clear association exists for any one gene. There is probably no single “schizophrenic gene,” but research continues in the hopes of more fully exploring candidate genes.2 Environmental stimuli or triggers along with genetic liability may contribute to the expression of the illness. Some data suggest that intrauterine exposure to viral or bacterial infections may be a risk factor; however, more research is needed in this area.
PATHOPHYSIOLOGY
The oldest theory associated with the pathophysiology of schizophrenia is the dopamine hypothesis, which proposes that psychosis is due to excessive dopamine in the brain. This hypothesis was formed in the late 1950s, following the discovery that chlorpromazine, the first antipsychotic drug, acted as a postsynaptic dopamine antagonist. Studies have also shown that drugs which cause an increase in dopamine (e.g., cocaine and amphetamines) increase psychotic symptoms, while drugs that decrease dopamine (as do all current antipsychotic medications) decrease psychotic symptoms. A wide array of scientific work over the last several decades has revealed a more complicated picture with both hyperdopaminergic, as well as hypodopaminergic brain regions. Hypodopaminergic activity observed in the prefrontal lobe is thought to relate to the core negative symptoms associated with schizophrenia. Thus, a more modern reworking of the dopamine hypothesis is the “dysregulation hypothesis” which takes these findings into account.3 It is possible, however, that the dopamine abnormalities hypothesized to underlie the etiology of schizophrenia may represent compensatory changes that occur secondary to other pathophysiologic abnormalities intrinsic to the illness. Other neurotransmitter systems have also been implicated in schizophrenia. Some investigators have suggested that a combined dysfunction of the dopamine and glutamate transmitter systems may better explain the disorder.4 In particular, it is hypothesized that glutamate, possibly through malfunctioning N-methyl-D-aspartate (NMDA) receptors, interacts to impact dopaminergic activity in areas of the brain such as the mesolimbic and mesocortical pathways. NMDA antagonists such as phencyclidine (PCP) and ketamine can elicit a state resembling the psychotic symptoms of schizophrenia, including positive, negative, and cognitive symptoms. There has also been a great deal of speculation regarding a role for serotonin receptor antagonism in antipsychotic efficacy,5 as many second-generation antipsychotics (SGAs) are active at serotonin receptors. Serotonin receptor binding may be important to drug action, possibly by modulating dopamine activity in mesocortical pathways. However, a compelling pathophysiologic theory relating to dopamine and serotonin receptor affinities does not yet exist. It is important to note that to date, antipsychotics without any primary or secondary dopamine-modulating properties have been ineffective for the treatment of positive symptoms of schizophrenia.
CLINICAL PRESENTATION AND DIAGNOSIS
People with schizophrenia may appear uncooperative, suspicious, hostile, anxious, or aggressive due to their misinterpretation of reality. They may have poor hygiene and appear unkempt, as psychosis, as well as depressive symptoms, may lead to impaired self-care. Sleep and appetite are often disturbed. People with schizophrenia often have difficulty living independently in the community and have difficulty forming close relationships with others. Additionally, they have problems with initiating or maintaining employment. Comorbid medical disorders, such as type 2 diabetes and chronic obstructive pulmonary disease, are prevalent in schizophrenia due to sedentary lifestyles, poor dietary habits leading to obesity, and/or heavy cigarette smoking. Approximately 85% of people with schizophrenia smoke, and approximately 50% use drugs and alcohol, rates that are much higher than in the general population.6
Clinical Presentation of Schizophrenia
General
![]() Schizophrenia is a chronic disorder of thought and affect, causing a significant disturbance in the individual’s ability to function vocationally and interpersonally. The onset of symptoms in most cases is insidious, usually preceded by a prodromal phase characterized by gradual social withdrawal, diminished interests, changes in appearance and hygiene, changes in cognition, and bizarre or odd behaviors. Despite the tendency of the media to portray a stereotype, the clinical presentation of a person with schizophrenia is extremely varied. Hallmark symptoms include psychotic symptoms, negative symptoms, and cognitive impairments that last for at least 6 months.
Schizophrenia is a chronic disorder of thought and affect, causing a significant disturbance in the individual’s ability to function vocationally and interpersonally. The onset of symptoms in most cases is insidious, usually preceded by a prodromal phase characterized by gradual social withdrawal, diminished interests, changes in appearance and hygiene, changes in cognition, and bizarre or odd behaviors. Despite the tendency of the media to portray a stereotype, the clinical presentation of a person with schizophrenia is extremely varied. Hallmark symptoms include psychotic symptoms, negative symptoms, and cognitive impairments that last for at least 6 months.
Symptoms
Psychotic symptoms: These symptoms are sometimes called positive symptoms, as they are “added on to” a person’s normal experience. They may include hallucinations (distortions or exaggeration of perception), delusions (fixed false beliefs), and thought disorder (illogical thought and speech). Hallucinations, most frequently auditory, can also be visual, olfactory, gustatory, and tactile. Auditory hallucinations may be experienced as voices or as thoughts that feel distinct from the person’s own thoughts. The content of the hallucinations is variable but often they are threatening or commanding (i.e., commanding the person to perform a particular action). Patients may feel compelled to perform the commanded task or may experience much anxiety when they do not. Delusions frequently involve fixed false beliefs despite invalidating evidence, and may be bizarre in nature. Often they have paranoid themes, which may make the patient suspicious of others. The characteristic thought disorder of schizophrenia includes loosening of associations, tangentiality, thought blocking, concreteness, circumstantiality, and perseveration. Thinking and speech may be incomprehensible and illogical. Subtle disturbances in associative thinking may develop years before disorganized thinking (formal thought disorder).
Negative symptoms: So called because they are qualities “taken away” from the personality, include impoverished speech and thinking, lack of social drive, flatness of emotional expression, and apathy. Though quite ubiquitous, these symptoms are difficult to evaluate because they occur in a continuum with normality, and can be due to secondary causes, including medication side effects, mood disorder, environmental understimulation, or demoralization. When due to schizophrenia itself, they are termed primary negative symptoms or deficit symptoms. The best strategy for differentiating primary from secondary negative symptoms is to observe for their persistence over time, despite efforts at resolving the other causes. Approximately 10% to 15% of people with schizophrenia may present primarily with negative symptoms; these people may be referred to as having a deficit syndrome.
Cognitive symptoms: Neuropsychological research shows that patients with schizophrenia show abnormalities in the areas of attention, processing speed, verbal and visual memory, working memory, and problem solving. There is a loss of, on average, one standard deviation of preillness IQ, with the average IQ between 80 and 84.
![]() A diagnosis of schizophrenia is made clinically, as there are no psychological assessments, brain imaging, or laboratory examinations that confirm the diagnosis.
A diagnosis of schizophrenia is made clinically, as there are no psychological assessments, brain imaging, or laboratory examinations that confirm the diagnosis.
Biological markers are being investigated, but currently, the diagnosis is made by ruling out other causes of psychosis and meeting specified diagnostic criteria that are based on symptoms and functioning. Family history of psychiatric disorders is helpful in supporting the diagnosis. The commonly accepted diagnostic criteria for schizophrenia are from the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision (DSM-IV-TR)7 (Table 37–1).
COURSE AND PROGNOSIS
The onset of psychosis, whether insidious or acute, is marked by difficulties for patients, families, and clinicians. The severity of symptoms may be denied, and the nature of behavioral disturbances may be misunderstood. Patients may try to keep symptoms hidden from family and friends; they may also isolate themselves from social support networks. The gradual development of psychosis, combined with the frequent misunderstanding of symptoms, generally leads to a substantial time period between symptom onset and diagnosis and treatment. While not unequivocal, recent data suggest that people with fewer episodes of acute psychosis and those treated early on in their illness may have a better prognosis. Therefore, the first challenge of optimal therapy is to move treatment initiation closer to the onset of psychosis.
Table 37–1 Diagnostic Criteria for Schizophrenia
|
A. Characteristic symptoms: Two (or more) of the following, each present for a significant portion of time during a 1-month period (or less if successfully treated):a |
|
• Delusions |
|
• Hallucinations |
|
• Disorganized speech (e.g., frequent derailment/incoherence) |
|
• Grossly disorganized/catatonic behavior |
|
• Negative symptoms (e.g., flat affect, alogia, avolition) |
|
B. Social/occupational dysfunction: For a significant portion of the time since the onset of the disturbance, one or more major areas of functioning such as work, interpersonal relations, or self-care are markedly below the level achieved prior to onset (or when onset is in childhood or adolescence, failure to achieve the expected level of interpersonal, academic, or occupational achievement) |
|
C. Duration: Continuous signs of disturbance persist for at least 6 months. This 6-month period must include at least 1 month of symptoms (or less if successfully treated) that meet criterion A (i.e., active-phase symptoms), and may include periods of prodromal or residual symptoms. During these prodromal or residual periods, the signs of disturbance may be manifested by only negative symptoms, or by two or more symptoms listed in category A present in an attenuated form (e.g., odd beliefs, unusual perceptual experiences) |
|
D. Ruling out other disorders: |
|
• Schizoaffective and mood disorder exclusion: Schizoaffective disorder and mood disorder with psychotic features have been ruled out because either (a) no major depressive, manic, or mixed episodes have occurred concurrently with the active-phase symptoms; or (b) if mood episodes have occurred during active-phase symptoms, their total duration has been brief relative to the duration of the active and residual periods |
|
• Substance/general medical condition exclusion: The disturbance is not due to the direct physiologic effects of a substance (e.g., drug of abuse or medication) or a general medical condition |
|
• Pervasive developmental disorder: If there is a history of autistic disorder or another pervasive developmental disorder, the additional diagnosis of schizophrenia is made only if prominent delusions or hallucinations are also present for at least a month (or less if successfully treated) |
a Only one symptom from category A is required if delusions are bizarre or hallucinations consist of a voice maintaining a running commentary on the person’s behavior or thoughts, or two or more voices conversing. Adapted from Ref. 7.
Although the course of schizophrenia is variable, the long-term prognosis for independent function is often poor. The course of illness is marked by intermittent acute psychotic episodes with a downward decline in psychosocial functioning. Over time, a patient may become more withdrawn, bizarre, and nonfunctional. Complete return to full premorbid functioning is uncommon. Many of the more dramatic and acute symptoms fade with time, but severe residual symptoms may persist. Family and friends often find this illness difficult to interpret and understand. Involvement with the law is fairly common for misdemeanors such as vagrancy, loitering, and disturbing the peace. The overall life expectancy is shortened primarily due to suicide, accidents, and the inability of self-care. The lifetime risk of suicide for people with schizophrenia is about 10%.8 Persistent compliance with a tolerable drug regimen improves prognosis, though relapse without medication exceeds 50% annually.
TREATMENT
Desired Outcomes
![]() The goal is for people with schizophrenia to receive, as early in their course as possible, comprehensive treatment designed to achieve functional outcomes. While in the past, the primary treatment goal was to decrease positive symptoms and the associated hostile and aggressive behaviors, newer approaches to treatment have a wider focus, including positive, negative, depressive, and anxious symptoms, as well as preserving cognition. The aim is to not only reduce symptomatology and psychotic relapses, but also to improve functional and social outcomes.9 It is important to keep in mind that some symptoms respond earlier than others. Combativeness, hostility, sleep disturbances, appetite, and hallucinations may be some of the first symptoms to improve. Improvements in negative symptoms, cognitive functioning, social skills, and judgment generally require a longer period to improve.
The goal is for people with schizophrenia to receive, as early in their course as possible, comprehensive treatment designed to achieve functional outcomes. While in the past, the primary treatment goal was to decrease positive symptoms and the associated hostile and aggressive behaviors, newer approaches to treatment have a wider focus, including positive, negative, depressive, and anxious symptoms, as well as preserving cognition. The aim is to not only reduce symptomatology and psychotic relapses, but also to improve functional and social outcomes.9 It is important to keep in mind that some symptoms respond earlier than others. Combativeness, hostility, sleep disturbances, appetite, and hallucinations may be some of the first symptoms to improve. Improvements in negative symptoms, cognitive functioning, social skills, and judgment generally require a longer period to improve.
Over the past few years, partial recovery and remission have become an increasingly prominent paradigm for treatment of schizophrenia. A range of interventions must be incorporated into long-term treatment strategies, including pharmacologic interventions and psychosocial therapies. Current treatment planning is increasingly focused on functional outcomes by providing treatment and recovery-oriented services to people with schizophrenia. Also, implementation of evidence-based practice, whereby new information from the published literature gets incorporated into clinicians’ prescribing behavior, has led to the use of interventions that promote a remission or recovery attitude.10 Moreover, recent attempts have been made to address and measure patient satisfaction with treatments and instill hope and optimism in order to attempt to empower patients. Unfortunately, this new paradigm is in the earliest stages of implementation, and despite these advances and recent treatment improvements, long-term outcomes currently remain poor, and many patients fail to receive comprehensive care.
Patient Encounter, Part 2
Past Psychiatric History: Though AC describes himself as being depressed “all my life” and remembers that he felt able to read peoples’ minds in high school, he denied prior psychiatric treatment until his first hospitalization 2 years ago at the height of his paranoia. At that time, he believed there was a conspiracy against him. He lived in constant fear that his phone was tapped and his home was bugged. He felt he was being watched in public places, and therefore he began avoiding going out. He experienced voices commenting on his behavior and believed the television was talking to him and that shows were about him. At times he felt his “brain was being squeezed” for information, so that his mind was being read. He believed his girlfriend was having affairs, and he could not be reassured. He reported decreased appetite, difficulty sleeping, and suicidal ideation. His toxicology screen and blood alcohol test in the emergency department were negative. In the hospital he was given a diagnosis of major depressive disorder with psychotic features. He was started on olanzapine 10 mg/day for psychosis and fluoxetine 20 mg/day for depression, with some improvement.
Past Medical History: He has no history of medical illness, head trauma, or seizure disorder.
Social History: He grew up in an upper middle class family, completed high school, and entered the Navy where he was discharged dishonorably due to not following rules. He began using alcohol in high school and continued to use other substances intermittently since then. He has held several jobs, the longest for 6 months. He thinks he might apply for disability.
Family Psychiatric History: His father had an alcohol problem.
Mental Status Exam
Appearance: Nicely dressed and groomed. No abnormal movements. Poor eye contact
Speech: Quiet and somewhat monotonous
Mood: Nervous
Affect: Guarded and mildly anxious with restricted range
Thought content: Adequate historian but with a tendency to leave out detail. Experiences hearing others call his name, and interpreting the car lights coming down his street as meaning people are out to get him. He denies suicidal or homicidal thoughts
Thought processes: Logical but vague
Cognition: Grossly intact
Insight and judgment: Mixed, as he can at times question his thinking, but at other times has full conviction of his beliefs. He is currently taking his medication and cooperating with the evaluation appointments
Given this additional information, how has your differential diagnosis changed?
What are the goals of outpatient treatment?
What pitfalls are likely to occur during outpatient treatment?
General Approach to Treatment
![]() Patients presenting with odd behaviors, illogical thought processes, bizarre beliefs, and hallucinations should be assessed for schizophrenia.
Patients presenting with odd behaviors, illogical thought processes, bizarre beliefs, and hallucinations should be assessed for schizophrenia.
A person presenting with the initial episode of psychosis requires a comprehensive assessment and careful diagnosis, as psychosis is not pathognomonic for schizophrenia. The differential diagnosis includes a wide array of neurologic, psychiatric, and general medical conditions (Table 37–2). Often, people present with a short history of psychopathology and cannot recall historical information accurately. This lack of information, along with frequent comorbid substance abuse, medical illnesses, and psychosocial stressors often confound the case. Patients presenting with psychotic symptoms, particularly those in their initial episode, should have a thorough medical and laboratory (electrolytes, blood urea nitrogen, serum creatinine, urinalysis, liver and thyroid function profile, syphilis serology, serum pregnancy test, and urine toxicology) evaluation, including a careful review of systems and a physical exam that includes a neurologic evaluation. At the minimum, in accordance with the American Psychiatric Association Practice Guidelines11 and the Expert Consensus Guideline Series for Schizophrenia,12 psychotic patients should have a medical workup at the time of admission to rule out other diagnoses or contributing factors.
Table 37–2 Disorders That May Present With Psychotic Symptoms

![]() Because early detection and intervention in schizophrenia is important for maximizing outcomes, treatment with antipsychotic medications should begin as soon as psychotic symptoms are recognized. Antipsychotic medications are the cornerstone of therapy for people with schizophrenia, and most patients are on lifelong therapy as nonadherence and discontinuation of antipsychotics are associated with high relapse rates. If other symptoms are present such as depression and anxiety, these symptoms should also be aggressively treated. Additionally, psychosocial treatments should be used concomitantly to improve patient outcomes.
Because early detection and intervention in schizophrenia is important for maximizing outcomes, treatment with antipsychotic medications should begin as soon as psychotic symptoms are recognized. Antipsychotic medications are the cornerstone of therapy for people with schizophrenia, and most patients are on lifelong therapy as nonadherence and discontinuation of antipsychotics are associated with high relapse rates. If other symptoms are present such as depression and anxiety, these symptoms should also be aggressively treated. Additionally, psychosocial treatments should be used concomitantly to improve patient outcomes.
Nonpharmacologic Therapy
![]() Psychosocial support is needed to help improve functional outcomes. Only 30% of patients respond robustly to antipsychotics, another 30% respond partially, and another 30% have a minimal response. Patients who do respond often continue to have residual symptoms such as amotivation, isolation, and impaired social functioning, thus limiting their participation in social, vocational, and educational endeavors. Psychosocial interventions are based on the premise that increased psychosocial functioning will lead to improvements in subjective feelings of self-esteem and life satisfaction. A few of the best-supported and most promising approaches to psychosocial rehabilitation are social skills training (SST), cognitive-behavioral therapy (CBT), and cognitive remediation (CR). These treatments are mainly utilized as targeted treatments for social and cognitive impairments and as adjuncts to pharmacotherapy for psychotic symptoms.13 Most psychosocial studies have been carried out in the United Kingdom, and findings are not yet widely utilized in the United States. Nonetheless, there are substantial data to support that family education and vocational support help to improve long-term functional outcomes.
Psychosocial support is needed to help improve functional outcomes. Only 30% of patients respond robustly to antipsychotics, another 30% respond partially, and another 30% have a minimal response. Patients who do respond often continue to have residual symptoms such as amotivation, isolation, and impaired social functioning, thus limiting their participation in social, vocational, and educational endeavors. Psychosocial interventions are based on the premise that increased psychosocial functioning will lead to improvements in subjective feelings of self-esteem and life satisfaction. A few of the best-supported and most promising approaches to psychosocial rehabilitation are social skills training (SST), cognitive-behavioral therapy (CBT), and cognitive remediation (CR). These treatments are mainly utilized as targeted treatments for social and cognitive impairments and as adjuncts to pharmacotherapy for psychotic symptoms.13 Most psychosocial studies have been carried out in the United Kingdom, and findings are not yet widely utilized in the United States. Nonetheless, there are substantial data to support that family education and vocational support help to improve long-term functional outcomes.
Pharmacologic Therapy
Generally, all people with schizophrenia should be treated with antipsychotics, and adjunctive medications should be used when necessary to treat specific symptoms or comorbid diagnoses. Medications specifically targeting negative symptoms and cognitive deficits are actively being investigated. Because each schizophrenic patient presents differently, each treatment regimen should be individually tailored. After a period of nearly 20 years during which only first-generation antipsychotics (FGAs; typical antipsychotics) were available, nine new antipsychotics have been marketed in the United States since 1990. These agents, known as SGAs (atypical antipsychotics), include risperidone (Risperdal), olanzapine (Zyprexa), quetiapine (Seroquel), ziprasidone (Geodon; Zeldox), aripiprazole (Abilify), paliperidone (Invega), iloperidone (Fanapt), asenapine (Saphris), and clozapine (Clozaril). Clozapine, the prototype of this class of medications, is reserved as second-line therapy due to its unusual side-effect profile (see below). ![]() Compared to the older FGAs, the more recently developed SGAs are associated with a lower risk of motor side effects (tremor, stiffness, restlessness, and dyskinesia); may offer greater benefits for affective, negative, and cognitive symptoms; and may prolong the time to psychotic relapse.
Compared to the older FGAs, the more recently developed SGAs are associated with a lower risk of motor side effects (tremor, stiffness, restlessness, and dyskinesia); may offer greater benefits for affective, negative, and cognitive symptoms; and may prolong the time to psychotic relapse.
Since the introduction of SGAs the use of FGAs has been progressively decreasing, and the current market share for these agents in treatment of schizophrenia is less than 10%. This occurred due to the touted lower side-effect profile and other benefits of SGAs in several domains of the illness. Early comparative studies used high dose, high-potency FGAs such as haloperidol and concluded that SGAs were more effective in some domains. More recently, a large multisite clinical trial (N = greater than 1,400 patients) was designed to examine the effectiveness of SGAs relative to a midpotency FGA, perphenazine (the Clinical Antipsychotics Trials of Intervention Effectiveness; CATIE trial). This study demonstrated that FGAs may be as effective as SGAs when using the primary endpoint of time to discontinuation of medication.14 Thus, while these results suggest that midpotency FGAs may be similar in efficacy to SGAs, the CATIE trial had several limitations, and other evidence suggests SGAs may be more effective in some illness domains, such as relapse prevention, affective and negative symptoms, and cognitive function. The SGAs have historically been much more expensive than the FGAs, however risperidone and clozapine are now available in lower cost generic formulations, and other SGAs will lose patent protection in the next 5 years. In conclusion, when selecting among the various agents, the risk benefit profile becomes fundamental, as all the antipsychotics are associated with somewhat different side-effect profiles.
Second-Generation (Atypical) Antipsychotics
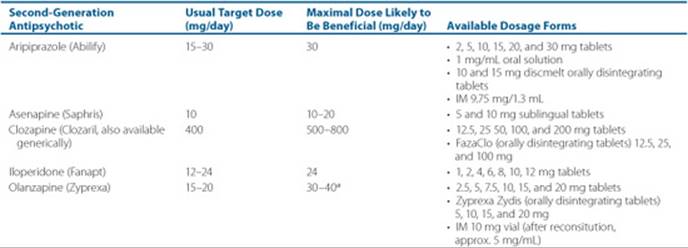
While FGAs exert most of their effect through dopamine-receptor blockade at the dopamine2 (D2) receptor, the SGAs may work through a more complicated mechanism, as SGAs have greater affinity for serotonin receptors than for dopamine receptors (Fig. 37–1). Despite being very heterogeneous with regard to receptor binding, the overall efficacy among the SGAs is similar.14 Recently new comparative data among SGAs and FGAs (lower doses) have reported that overall efficacy is similar between groups, which suggests a reevaluation of the place of FGAs in therapy is needed.14 Only clozapine, however, has demonstrated superior efficacy for some patients (see the section on treatment-resistant patients). An important distinction of the SGAs as a class is their lower propensity to cause extrapyramidal symptoms(EPS) and tardive dyskinesia (TD). The annual risk of TD is considered to be less than 1.5% per year in adults (less than 54 years old) taking SGAs compared to approximately a 5% per year risk in those taking FGAs.15 Pharmacologic profiles and side-effect profiles, however, are very different among the agents. ![]() SGAs as a class are heterogeneous with regard to side-effect profiles. Many SGAs carry an increased risk for weight gain and for the development of glucose and lipid abnormalities; therefore, careful monitoring is essential.Table 37–3 lists the SGA agents, recommended dosing, and dosage forms available. Table 37–4 lists the side-effect profiles of the SGAs and haloperidol. Clozapine is discussed in the section on treatment-resistant patients.
SGAs as a class are heterogeneous with regard to side-effect profiles. Many SGAs carry an increased risk for weight gain and for the development of glucose and lipid abnormalities; therefore, careful monitoring is essential.Table 37–3 lists the SGA agents, recommended dosing, and dosage forms available. Table 37–4 lists the side-effect profiles of the SGAs and haloperidol. Clozapine is discussed in the section on treatment-resistant patients.
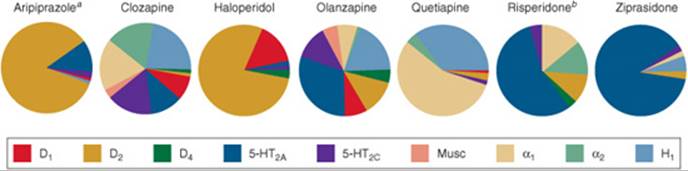
FIGURE 37–1. Profiles for receptor binding of second-generation antipsychotics and haloperidol.a Partial agonist activity at D2 receptors, whereas all others are D2 antagonists.bPaliperidone data are unavailable, but would be expected to be similar to risperidone. Iloperidone and asenapine are not shown here due to recency of approval. (D, dopamine receptor; H1, histamine receptor; 5-HT, serotonin receptor; Musc, muscarinic receptor; α1 and α2, receptors.) Adapted from The Journal of Clinical Psychiatry and Physicians Post Graduate Press. Collaborative Working Group on Clinical Trial Evaluations. J Clin Psychiatry 1998; 59[Suppl 12]:7, and Pharmacologic Treatment of Schizophrenia. Caddo, OK. Professional Communications, Inc, 2003:52, with permission.
Table 37–3 Second-Generation (Atypical) Antipsychotics

Risperidone
Risperidone, a benzisoxazole derivative, was the first SGA to be marketed following the release of clozapine. Risperidone is the only SGA available for administration as a long-acting injection in the United States, and is also the only first-line oral SGA to become available generically. It has high binding affinity to both serotonin 2A (5-HT2A) and D2 receptors and binds to α1 and α2 receptors, with very little blockade of cholinergic receptors.16 Multicenter registry trials found 6 to 16 mg of risperidone at least as equally efficacious as haloperidol (20 mg), while 6 mg of risperidone produced EPS at a rate no different than placebo.17 Risperidone is also approved for relapse prevention and is associated with significantly lower relapse rates than long-term haloperidol treatment.18 At clinically effective doses (less than or equal to 6 mg/day), EPSs are low, although higher doses are clearly associated with a greater incidence of EPS. Risperidone is associated with serum prolactin elevations that are similar to or greater than those seen with the FGAs. Elevated prolactin levels can, but do not always, lead to clinical symptoms such as hormonal problems (e.g., amenorrhea, galactorrhea, and gynecomastia) or sexual dysfunction. Risperidone is associated with mild to moderate weight gain, and mild elevations in lipid and glucose may occur. However, patients chronically treated with other antipsychotics such as olanzapine may experience a decline in cholesterol and triglyceride levels when changed to risperidone monotherapy.14
Table 37–4 Comparative Side Effects Among the SGAs and Haloperidola

Olanzapine
Olanzapine has greater affinity for 5-HT2A than for D2 receptors. In addition, the compound has affinity at the binding sites of D4, D3, 5-HT3, 5-HT6, α1-adrenergic, muscarinic1–5 (M1–5), and histamine1 (H1) receptors.19 Multicenter clinical trials have reported that the effectiveness of olanzapine is at least equal to that of haloperidol for the treatment of positive symptoms20,21 and equal or superior to haloperidol for the treatment of negative symptoms. The premarketing clinical trials reported no significant differences in efficacy among dosage groups. However, the higher dose range appeared to offer the greatest benefits for both positive and negative symptoms when compared to haloperidol (20 mg). In one study, olanzapine (16.3 mg/day mean modal dose) was superior to haloperidol (16.4 mg/day) for the treatment of negative symptoms,20 while another study found that 13.2 mg olanzapine was superior for both positive and negative symptoms compared to 11.8 mg haloperidol.21 Among the first-line antipsychotic agents, olanzapine is associated with the longest time to treatment discontinuation,14 a finding that suggests that olanzapine may somewhat differ from the other SGAs in effectiveness. Olanzapine has a low rate of EPS and causes slight, transient prolactin elevations. However, clinically significant weight gain occurs with olanzapine across the dosage range. The degree of weight gain is similar to that seen with clozapine and greater than that observed with the other SGAs. Olanzapine is also associated with hypertriglyceridemia, increased fasting glucose, and new-onset type 2 diabetes (i.e., metabolic syndrome), and among the first-line SGAs, it is associated with the greatest elevations in these metabolic parameters.14
Quetiapine
Structurally quetiapine is related to clozapine and olanzapine. Quetiapine has high affinity for 5-HT2A receptors and lower affinity for D2 and D1 receptors. This drug has some affinity for α1, α2, and H1receptors, and very little for muscarinic receptors. The efficacy of quetiapine for psychosis was established in two controlled trials, which found that maximum benefits occurred at 300 mg per day or greater.22,23 The most effective doses of quetiapine may be higher than 500 mg/day. Quetiapine may have beneficial effects for anxious and depressive symptoms. Because of its low D2 occupancy, motor side effects and prolactin elevations are usually not seen with quetiapine. Orthostasis occurred in 4% of subjects in clinical trials. Sedation may occur but is generally transient. Mild weight gain and minor elevations in triglycerides can occur.
Ziprasidone
Ziprasidone was developed specifically to be a compound that blocks D2 receptors, but also binds with even greater affinity to central 5-HT2A receptors. As a result, ziprasidone has a binding affinity ratio of 11:1 for 5-HT2A:D2receptors. Ziprasidone also has a relatively high affinity for 5-HT2C, 5-HT, oj1-adrenergic, and D1 receptors.24 Several short-term, placebo-controlled, premarketing clinical trials led to the recommended dose range of 40 to 160 mg daily with food.25,26 Current dosing suggests that efficacy may be greater at doses over 200 mg/day. Liability for EPS, weight gain, and lipid elevations were very low in the clinical trials. Ziprasidone is associated with some prolongation of the QT interval corrected for heartrate (QTc) interval in adults. However, drug overdose data and studies of pharmacokinetic interactions have thus far shown little evidence that significant QTc prolongation may occur. It is advised to utilize caution, however, when using medications that may affect ziprasidone metabolism and when it is prescribed along with other medications known to prolong QTc. Please see the Pharmacokinetics section of this chapter for further discussion of this topic. Caution should also be used when prescribing ziprasidone in situations that have increased risk for prolongation of QTc including comorbid diabetes, electrolyte disturbances (e.g., low serum sodium and potassium concentrations), heavy alcohol consumption, female gender, and congenital QTc disorder.
Aripiprazole
Aripiprazole was formulated in the early 1980s to function as a potential dopamine modulator, with both antagonist and agonist activity at the D2 receptor. It is the first D2 partial agonist available for the treatment of schizophrenia and is sometimes referred to as a third-generation antipsychotic. This novel mechanism is termed a “dopamine system stabilizer,” functioning as an antagonist in a hyperdopaminergic state and as an agonist when in a hypodopaminergic state. Aripiprazole is also a partial agonist at 5-HT1A receptors, an antagonist at 5-HT2A receptors, and also has affinity for D3receptors. Additionally, it has a moderate affinity for α1 and H1 receptors with no appreciable affinity for the M1 receptor.27 The recommended starting dose is 10 to 15 mg daily given without regard to meals. No titration is required, as the starting dose is an effective dose. The recommended dosing range is 10 to 30 mg/day; doses greater than 30 mg have not been systematically evaluated. Side effects are low with sedation and nausea and vomiting occurring most frequently. Elevations in weight, lipids, and glucose are generally negligible, and due to its partial dopamine receptor agonism, it does not usually cause elevations in prolactin. In fact, patients switched to aripiprazole from other antipsychotic agents may experience decreases in prolactin levels.
Paliperidone
Paliperidone is the separately marketed 9-hydroxy metabolite of risperidone. The effects of risperidone and paliperidone in the body are expected to be similar, as the effects of risperidone are thought to be a result of both the parent drug and the 9-hydroxy metabolite. The neurotransmitter receptor binding affinity is also similar between the two agents, with paliperidone having a greater affinity at 5-HT2Acompared to D2 receptors. Unlike many other antipsychotic medications, the majority of paliperidone is excreted unchanged. Paliperidone is available as an extended release tablet, given once daily. Patients may be told to expect to see the shell of the tablet in the stool, as it does not dissolve in the digestive tract. For adults with schizophrenia, the recommended dose is 6 mg every morning. No dose titration is required; however if needed, the dose can be adjusted at five day intervals to doses of 3 to 12 mg once daily. Side effects of paliperidone are expected to be similar to those of risperidone, including the potential for dose related EPS and prolactin elevation.28 A long-acting injectible form of paliperidone has been recently approved.
Patient Encounter, Part 3
Soon after his discharge from the hospital, AC stopped taking his olanzapine. He had gained a lot of weight, and he didn’t believe the medication was helping. He continued to have difficulties maintaining employment and being involved socially. With encouragement from his girlfriend, AC sought treatment again and was started on risperidone 2.5 mg a day. He continued to experience paranoia, still feeling unable to function up to his potential at work, and still tending to be reclusive. A new therapist supported his involvement in Alcoholics Anonymous, and AC began to question the depression diagnosis. She referred him to a center specializing in schizophrenia where the diagnosis was established to be schizophrenia, paranoid type, continuous with depressive episode, single episode, alcohol dependence, early full remission; and polysubstance abuse, early full remission.
Why was risperidone selected as the next antipsychotic choice for AC?
Describe nonpharmacologic treatment strategies that may be appropriate for AC.
Discuss a risperidone dosing plan, and list side effects AC may experience on this medication.
Iloperidone (Fenapt)
Iloperidone was approved in the United States in 2009 and is indicated for acute treatment of adults with schizophenia. Iloperidone exhibits high affinity for 5HT2a and dopamine D2 and D3 receptors and acts as an antagonist at these receptors, as well as the 5HT1a and norepinephrine α1/α2c receptors. Doses must be titrated due to the risk of orthostatic hypotension and starts at 1 mg twice daily, titrating daily up to a close of 24 mg total daily dose (12 mg twice daily). Iloperidone has the potential to prolong the QT interval, so clinicians should consider using other drugs first. Common adverse reactions include dizziness, dry mouth, fatigue, orthostatic hypotension, tachy cardia, and weight gain.
Table 37–5 First-Generation (Typical) Antipsychotics
Asenapine (Saphris)
Asenapine was approved in the United States in 2009 and is indicated for the acute treatment of schizophrenia in adults. Asenapine’s mechanism of action is thought to be associated with antagonistic activity at 5HT2a and D2receptors. Asenapine also exhibits a high affinity for other serotonerigic and dopaminergic receptors, as well as α1 and α2 adrenergic receptors and H1 receptors. The recommended starting and target dose for the treatment of symptoms of schizophrenia is 5 mg sublingually twice a day. Tablets must be placed under the tongue and allowed to be dissolved completely; the tablets should not be chewed or swallowed. Patients should refrain from drinking or eating for 10 minutes post administration. In controlled trials no added benefit was seen with doses above 10 mg twice daily, but there was an increase in adverse effects. Adverse effects commonly seen with asenapine may include somnolence, dizziness, and akathisia.
First-Generation (Typical) Antipsychotics
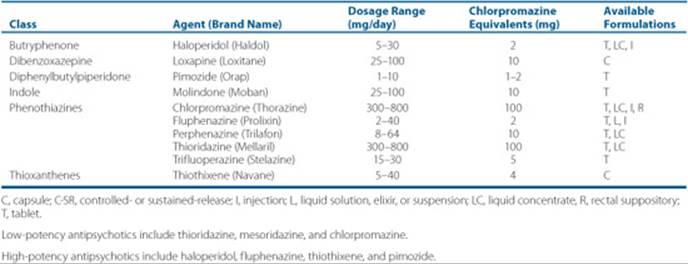
FGAs are high-affinity D2 receptor antagonists. During chronic treatment, these agents block 65% to 80% of D2 receptors in the striatum and block dopamine receptors in the other dopamine tracts in the brain as well.29 Clinical response is generally associated with 60% D2-receptor blockade, while 70% and 80% are associated with hyperprolactinemia and EPS, respectively. This group of antipsychotics was widely used from the 1950s to the 1990s, when the SGAs began to replace these agents as first-line therapy.
Dosages for these agents are frequently given as chlorpromazine equivalent dosages, which are defined as the dosage of any of the FGAs equipotent with 100 mg of chlorpromazine. The target dose recommendation for acute psychosis is 400 to 600 chlorpromazine equivalents, unless the patient’s history indicates that this dose may result in intolerable side effects. Generally maintenance therapy should provide a dose of 300 to 600 chlorpromazine equivalents for maximum efficacy. Information on dosing and available dosage forms is provided in Table 37–5. All FGAs are equally efficacious in groups of patients when used in equipotent doses. However, individual variation does occur, such that a patient may not respond equally to each antipsychotic. Selection of a particular antipsychotic should be based on patient variables, such as the need to avoid certain side effects or interactions with concomitant medications. Previous patient or family history of response is also helpful in the selection of a particular agent.
Decanoates
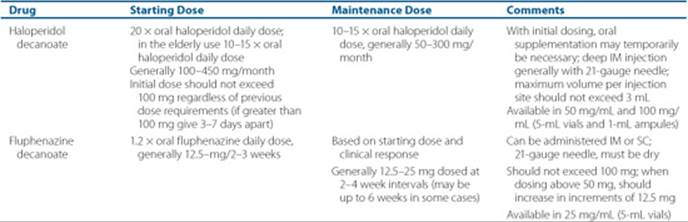
Long-acting, depot preparations are available for two FGAs (fluphenazine decanoate and haloperidol decanoate) in the United States. These compounds are esterified antipsychotics formulated in sesame seed oil for deep intramuscular (IM) injection. Because these are long-acting preparations, patients should be exposed to the oral form of the drug first to ensure tolerability. With initial dosing of haloperidol decanoate, oral supplementation may temporarily be necessary, as the drug accumulates over many weeks, not reaching steady state until 4 to 5 dosing intervals have elapsed. The pharmacokinetic profiles of the depot agents are useful parameters for strategic dosing. Patients may be dosed with fluphenazine decanoate on a 1- to 3-week interval, while haloperidol decanoate is usually dosed once a month. Conversion from oral to depot and maintenance dosing recommendations are shown in Table 37–6. Based on these recommendations, a reasonable estimate is that 12.5 mg (0.5 mL) of fluphenazine decanoate given every 2 weeks is approximately equivalent to 10 mg/day of fluphenazine orally. A maintenance haloperidol decanoate dose of 150 mg every 4 weeks is approximately equivalent to 10 mg/day of oral haloperidol. Initial decanoate injections should be preceded by a small test dose.
Table 37–6 Antipsychotic Dosing of Long-Acting Preparations

Side Effects of FGAs
The FGAs are associated with a host of side effects that largely differ between high- and low-potency agents. In general, the low-potency agents are less likely to cause EPS than the high-potency agents. Of note, lower doses and midpotency agents may be associated with less EPS than once believed and similar to SGAs in clinical trials.14
FGAs are associated with EPS, which includes akathisia (motor and/or subjective restlessness), dystonia (muscle spasm), and pseudoparkinsonism (akinesia, tremor, and rigidity). These fairly common motor side effects are caused by dopamine antagonism in the nigrostriatal pathways. Akathisia is the most frequently occurring motor side effect, with approximately 20% to 40% of people treated with FGA drugs experiencing an objective or subjective feeling of restlessness. Roughly half of the cases of akathisia present within one month of antipsychotic initiation although the onset of akathisia many times may be within 5 to 10 days after the first dose or increase in dosage. Younger people and those taking high doses of high-potency antipsychotics are at greater risk for the development of akathisia. Acute dystonic reactions are abrupt in onset and are usually seen within 24 to 96 hours after a first dose or increase in dosage. Characteristic signs and symptoms include abnormal positioning or spasm of the muscles of the head, neck, limbs, or trunk. Dystonia may occur in 10% to 20% of patients. There is higher risk for dystonia in young male patients and those taking high-potency FGAs. Pseudoparkinsonism resembles idiopathic Parkinson’s disease, and features may be present in up to 30% to 60% of people treated with FGAs. The onset of symptoms is usually seen within 1 to 2 weeks following initiation of dosing or a dose increase. Risk factors include older age, female gender, high doses, and possibly those with depressive symptoms.30
TD is a movement disorder characterized by abnormal choreiform (rapid, objectively purposeless, irregular, and spontaneous movement) and athetoid (slow and irregular) movements occurring late in onset in relation to initiation of antipsychotic therapy. This adverse effect usually develops over several months or after at least 3 months of cumulative exposure to neuroleptic medications. Severity of TD can range from mild and barely noticeable to severe. In some cases TD may be severe enough to interfere with ambulation, and may stigmatize persons taking antipsychotics if symptoms are visible to others. The estimated average prevalence of TD is 20% with a range of 13% to 36%. The incidence of new cases per treatment year with FGAs is approximately 5%.31 TD is reversible in one-third to one-half of cases with the cessation of the antipsychotic.32 When the antipsychotic is tapered or discontinued, there is typically initial worsening of abnormal movements. Risk factors for TD include older age, longer duration of antipsychotic treatment, and presence of EPS, substance abuse, and mood disorders. SGAs carry a lower risk of TD than FGAs. A pooled analysis of studies indicated that new onset TD incidence was 0.8% with SGA treatment versus 5.4% for FGAs.15 A more recent review of studies published from 2004 to 2008 reported a smaller difference in incidence of TD between agents, at 3.9% for SGAs and 5.5% for FGAs.33
Table 37–7 Side Effects of FGAs
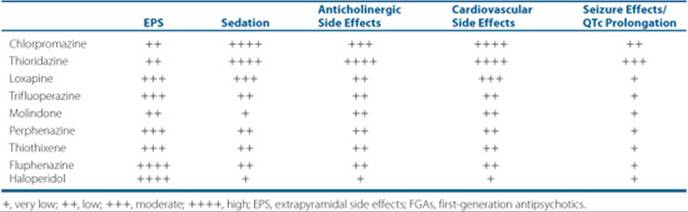
Neuroleptic malignant syndrome (NMS), a life-threatening emergency characterized by severe muscular rigidity, autonomic instability, and altered consciousness, can occur uncommonly with all the FGAs, and may also occur with SGAs. Rapid dose escalation, the use of high-potency FGAs at higher doses, and younger patients are associated with a higher risk of NMS. When NMS is diagnosed or suspected, antipsychotics should be discontinued and supportive and symptomatic treatment begun (e.g., antipyretics, cooling blanket, IV fluids, oxygen, monitoring of liver enzymes, and complete blood cell count). Dopamine agonists (e.g., bromocriptine) should be considered in moderate to severe cases.
Additionally, dermatologic side effects, photosensitivity, and cataracts may occur with the phenothiazine agents. Sedation is caused from H1-receptor antagonism; anticholinergic side effects (constipation, blurred vision, dry mouth, and urinary retention) are caused from M1-receptor antagonism; and α1-receptor blockade is associated with orthostatic hypotension and tachycardia (Table 37–7). QTc prolongation may occur with lower-potency FGAs, and thioridazine has a black box warning for QTc prolongation.
Treatment Guidelines and Algorithms
Due to the plethora of choices and the rising costs of SGAs, algorithms and treatment/consensus guidelines have been developed for the treatment of schizophrenia. The most widely accepted algorithm in the United States was developed as part of the Texas Implementation of Medication Algorithms (TIMA). A national panel of experts developed this algorithm, most recently updated in 2006.34 Algorithms go beyond guidelines, providing a framework for clinical decision making at critical decision points. According to the TIMA schizophrenia algorithm, SGAs (except clozapine) should be utilized as first-line treatment. The choice of SGA is guided by consideration of the side-effect profiles and the clinical characteristics of the patient. Treatment with a given drug should be continued for 4 to 6 weeks in order to assess response. If only partial response or nonresponse is noted, a trial of a second SGA should be initiated (Fig. 37–2). Other similar guidelines include the American Psychiatric Association Practice Guidelines for schizophrenia,11 The Expert Consensus Guideline Series,12 and the Schizophrenia Patient Outcomes Research Team (PORT) Treatment Recommendations.35 In contrast to the TIMA guidelines, the PORT recommends either the use of FGAs or SGAs as first-line therapy.
Treatment Adherence
Medication nonadherence in schizophrenia is common and often associated with symptom relapse.36 Estimates of nonadherence to antipsychotics range from approximately 24% to 88% with a mean of approximately 50%. Subjects who are nonadherent have approximately a fourfold greater risk of a relapse than those who are adherent. Adherence to antipsychotic regimens is problematic for many reasons. Neurocognitive deficits and paranoid symptoms may hamper adherence to the therapeutic regimen, and identification of nonadherence by caretakers and providers can be challenging. Frequently, there is no obvious connection between nonadherence and symptom exacerbation, as there may be no immediate consequences of missing a dose, and patients may relapse suddenly after only several weeks or months of nonadherence. Antipsychotic side effects such as EPS, weight gain, and sexual dysfunction are also a major contributing factor to treatment nonadherence, with 25% to 66% of subjects citing adverse effects as the primary reason for nonadherence.37 Several other factors are associated with nonadherence, including younger age, delusions, substance abuse, and deficit symptoms. For patients who have relapsed several times due to nonadherence, have a history of dangerous behavior, or risk a significant loss of social/vocational gains when relapsed, treatment with long-acting formulations should be encouraged. Risperidone is currently the only SGA with a long-acting formulation available. It uses a microsphere technology that maintains stable blood levels for about 2 weeks. Dosing is generally 25 to 50 mg every 2 weeks with oral supplementation in the first 3 weeks.
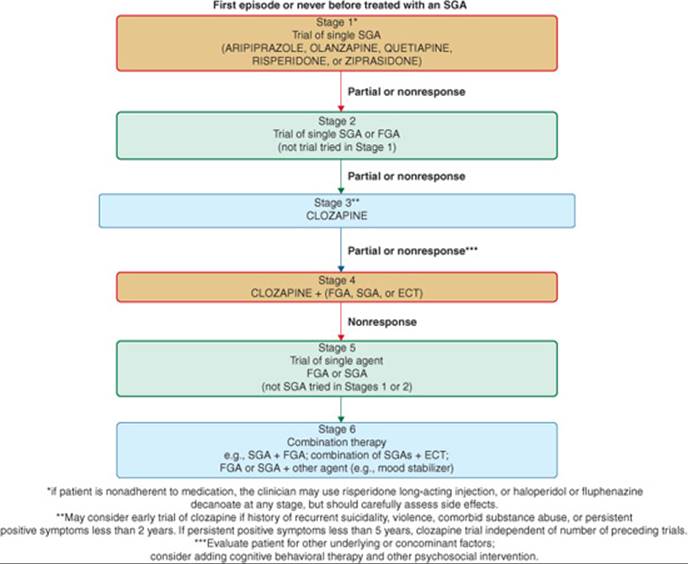
FIGURE 37–2. TIMA algorithm for antipsychotic treatment in schizophrenia. Choice of antipsychotic should be guided by considering the clinical characteristics of the patient and the efficacy and side-effect profiles of the medication. Any stage may be skipped depending on the clinical picture or history of antipsychotic failures. (ECT, electroconvulsive therapy; FGA, first-generation antipsychotic; SGA, second-generation antipsychotic.) (Adapted from the Texas Department of State Health Services.)
Table 37–8 Suggestions for First-Line Antipsychotic Therapy in Specific Patients
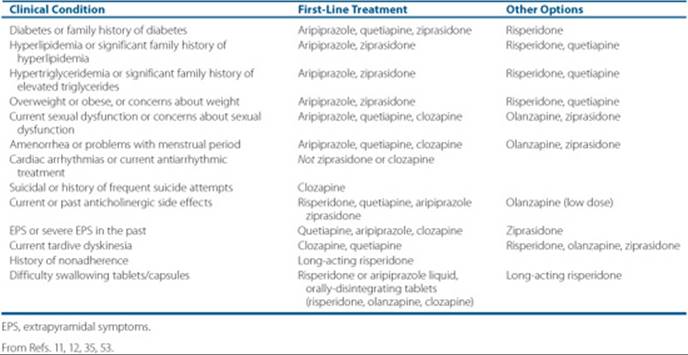
Patient-Specific Antipsychotic Selection
Patient-specific characteristics may help guide the selection of antipsychotic treatment. Based on expert consensus guidelines, Table 37–8 provides evidence-based suggestions for SGA selection to aid the clinician in individualizing treatment. FGAs continue to have a place in therapy for patients who cannot afford the SGAs or for those who have responded favorably to these agents in the past. However, in the next few years several of the SGAs will be available generically, and cost may become less of an advantage for FGAs. New data suggest that the efficacy of FGAs is comparable to SGAs,14 however clinicians must evaluate the risk to benefit profile on an individualized basis.
Special Populations
Adolescents
Epidemiologic data show that 10% to 30% of patients with schizophrenia develop their first psychotic symptoms prior to their 18th birthday. The diagnosis of schizophrenia in children and adolescents is often difficult to make, and the differential diagnosis includes pervasive developmental disorders, attention-deficit/hyperactivity disorder, and language or communication disorders. The existence of prominent hallucinations or delusions, however, helps make the diagnosis, as they are not a prominent part of the other disorders. Fifty-four percent to 90% of patients developing schizophrenia before age 18 have premorbid abnormalities such as withdrawal, odd traits, and isolation.38
Treatment for psychotic children and adolescents ideally involves an intensive and comprehensive program with a highly structured environment that includes special education and psychoeducation. Day treatment, hospitalization, or long-term residential treatment may be necessary. Pharmacologic treatment is indicated if psychotic symptoms cause significant impairments or interfere with other interventions. Children and adolescents are more vulnerable to EPS, particularly dystonias, than are adults. Due to concerns about EPS and TD, it has been recommended that pharmacotherapy in children and adolescents should be initiated with SGAs. Aripiprazole and risperidone are both FDA-indicated for the treatment of schizophrenia in adolescents aged 13 to 17. Recommended initiation and target dosing is lower for adolescents than adults.
Agents with significant sedation and anticholinergic side effects are not preferred, as they can cause attention difficulties and cognitive dulling that may interfere with optimal school performance. Compared to adults, children and adolescents tend to gain more weight on these agents. Young patients should be started on lower doses than adults and should be titrated at a slower rate. Side effects should be monitored closely during initiation and throughout maintenance therapy. Informed consent, addressing the rationale for treatment, and potential risks and benefits of therapy, should be obtained from the parents or guardians prior to treatment with any antipsychotic medication, and assent should be obtained from the child as well.
Elderly
Psychotic symptoms in late life (greater than 65 years of age) are generally a result of an ongoing chronic illness carried over from younger life; however, a small percentage of patients develop psychotic symptoms de novo, defined as late-life schizophrenia. However, other illnesses presenting with psychotic symptoms are common in this population, as approximately one-third of patients with Alzheimer’s disease, Parkinson’s disease, and vascular dementia experience psychotic symptoms. The majority of data for antipsychotic use in the elderly comes from experience treating these other disease states.
Antipsychotics can be safe and effective for the treatment of psychosis in the elderly, if used at lower doses than those commonly used in younger adults. Older adults are particularly vulnerable to the side effects of FGAs. Parkinsonian symptoms reportedly occur in over 50% of all elderly patients receiving these agents, and the cumulative annual incidence of TD in middle-aged and elderly patients is over 25%. With the SGAs, the risk appears to be approximately 4%. However, these data are based on a limited number of studies, and future investigations are likely to yield more specific information regarding TD risk with the SGAs.39 The likelihood of reversing this potentially debilitating condition diminishes with age. Orthostasis, estimated to occur in 5% to 30% of geriatric patients, is a major contributing factor to the occurrence of falls that often leads to fractures, injuries, and loss of independence. Low-potency antipsychotics and clozapine are more likely to cause significant drops in orthostatic blood pressure. Antipsychotics may cause or worsen anticholinergic effects including constipation, dry mouth, urinary retention, and cognitive impairment. Cognitive impairment may lead to decreased independence, and a more rapid decline in cognitive functioning may occur in the elderly treated with antipsychotics than the younger adult population. As a result of data showing a statistically significant increase in mortality rate in elderly dementia patients who are treated with SGAs, all SGAs now carry a warning, and patients and families should be informed of this risk prior to treatment with these agents. Dosing in the elderly is initiated lower and titration is slower than in adults. Maximum doses are often one-half of adult doses.
Dually Diagnosed
The prevalence of substance dependence and abuse among persons with schizophrenia is significantly higher than in the general population. Conservative estimates of the proportion of schizophrenic patients abusing alcohol and/or illicit drugs range from one-third to as many as one-half.40 The most common drugs of abuse are cannabis and cocaine and alcohol abuse. Unfortunately, substance use often worsens the course and complicates the treatment of schizophrenia. Dually-diagnosed patients are more likely to be nonadherent with treatment. Characteristically, they have a poorer response rate to FGAs, have more severe psychosis, and have higher rates of relapse and rehospitalization compared to patients who are not abusing substances. Some studies have found that EPS may occur more frequently in substance-abusing patients, and alcohol use is a risk factor for developing TD. There is a growing body of literature indicating that SGAs are effective in this population, and some studies have shown a reduction in the use of drugs and alcohol with SGAs.41
Treatment-Resistant Patients
For approximately 20% to 30% of people with schizophrenia, drug treatment is ineffective. A standard definition of treatment resistance is persistent positive symptoms despite treatment with at least two different antipsychotics given at adequate doses (at least 600 chlorpromazine equivalents) for an adequate duration (4–6 weeks). In addition, patients must have a moderately severe illness as defined by rating instruments, and have a persistence of illness for at least 5 years.42 These patients are often highly symptomatic and require extensive periods of hospital care.
Clozapine
To date, clozapine remains the only drug with proven and superior efficacy in treatment-resistant patients, and it is currently the only drug approved for the treatment-resistant schizophrenic. Studies have shown a response of approximately 30% to 50% in these well-defined treatment-resistant patients. Clinical trials have consistently found clozapine to be superior to traditional antipsychotics for treatment-refractory patients, and it is efficacious even after nonresponse to other SGAs and in partially responsive patients. It is often effective even in those who have had a poor response to other medication for years. Recent studies have demonstrated that it has a beneficial effect for aggression and suicidality, which led to the FDA approval for the treatment of suicidal behavior in people with psychosis.43
A great deal of interest has been generated in understanding what pharmacologic properties of clozapine contribute to its superior efficacy, but to date this has not been clearly elucidated. Clozapine has a low affinity for D2receptors, also blocks D1 receptors, and is a 5-HT2A antagonist. Its unsurpassed efficacy suggests that there is a neuropharmacologic effect associated with clozapine that is to date unique to this agent.
Clozapine’s use is limited by its association with the rare but life-threatening risk of agranulocytosis. Other side effects include seizures, as well as common unpleasant side effects including sedation, enuresis, anticholinergic effects, weight gain, and hypersalivation. The long-term hematologic monitoring (see monitoring section) required for prevention of agranulocytosis can represent a barrier to both patients and care providers. The optimal plasma level of clozapine is a minimum trough level of 300 to 350 ng/mL (300–350 mcg/L or 0.92 to 1.07 μmol/L or 918–1,071 nmol/L), usually corresponding to a daily dose of 200 to 400 mg, although dosage must be individualized. According to published guidelines and recommendations, clozapine should be considered after two failed antipsychotic trials, but may be considered sooner if the individual patient situation warrants.34
Other Strategies
Approximately 30% of patients treated with clozapine will not respond, and another 30% will have only a partial response to the drug. Treatment options with other medications are limited after clozapine nonresponse. For these patients, the best evidence points to augmentation using an FGA, SGA, or electroconvulsive therapy (ECT). The only available data support the combination of clozapine with risperidone; however, controlled trial results are mixed. Other potential strategies include combining antipsychotics with mood stabilizers (e.g., lithium, lamotrigine, valproate, and topiramate).34
Acutely Psychotic Patients
Psychiatric emergencies occur in a variety of settings, including emergency departments, psychiatric units, medical facilities, and outpatient settings. Although verbal interventions are recommended as initial interventions, most psychiatric emergencies require both pharmacologic and psychological interventions. Often, the psychiatrist must make decisions based on limited information and history. IM formulations are available for a number of FGAs and three SGAs (aripipra-zole, ziprasidone, and olanzapine). These formulations are safe and effective in treating acute psychosis. These SGAs are now recommended as first-line therapy in agitated schizophrenia patients; however, IM lorazepam with or without concomitant oral (tablets, liquid, or disintegrating tablets) SGAs are also used. Concomitant olanzapine and benzodiazepines may cause orthostasis. High dosing of FGAs, termed rapid neuroleptization, is no longer recommended.
Pregnancy and Lactation
When to use antipsychotics in pregnancy and during lactation remains a complicated decision based on a careful analysis of risks and benefits. Women with schizophrenia, even those who are unmedicated have a significantly greater risk for stillbirth, infant death, preterm delivery, low infant birth weight, and infants that are small for gestational age. Furthermore, women who experience a relapse in psychotic symptoms during pregnancy are at the greatest risk for birth complications.44 Because risks related to psychotic relapse may be more detrimental than antipsychotic treatment to both mother and baby, antipsychotics are often continued during this period.
Essentially all antipsychotic medications pass through the placenta. High-potency FGAs are associated with a low risk for congenital abnormalities; however, limb defects and dyskinesias have been reported.45 Low-potency phenothiazine antipsychotics may increase the risk of congenital abnormalities when used in the first trimester. According to the relevant literature published to date, there appears to be little risk associated with the first-line SGAs.46Long-term neurobehavioral studies of children exposed to SGA in utero have not yet been done, however. On the basis of available data, generalization is impossible, and each case and specific antipsychotic should be weighed on an individual basis. While SGAs are excreted in breast milk, most case reports have reported a low frequency of deleterious effects on the infant. Women taking SGAs may have enhanced fertility compared to women taking FGAs, as SGAs (except risperidone and paliperidone) are less likely to cause prolactin elevations leading to anovulation. Therefore, careful discussion with patients, including education about birth control, must occur.
Pharmacokinetics
Pharmacokinetic and pharmacodynamic issues are not generally a major concern with antipsychotic treatment; however, additive side effects may occur with combined treatment, and a few clinically significant drug interactions are notable (Table 37–9). All of the antipsychotics are highly protein-bound; however, protein-binding interactions are generally not clinically significant. Absorption of most antipsychotics is not affected by food, with the exception of ziprasidone, the absorption of which is increased by 60% to 70% when given with meals.
Table 37–9 Metabolism and Drug Interactions With Antipsychotics
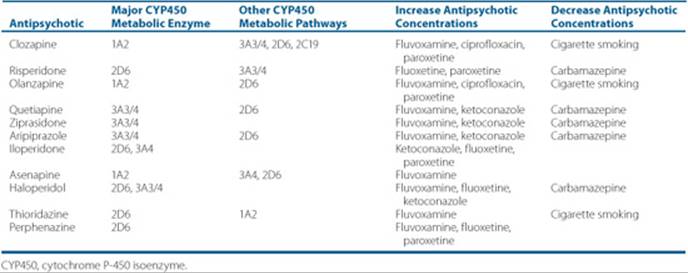
All antipsychotics are, at least to some extent, metabolized by hepatic microsomal enzymes to water-soluble compounds that are excreted by the kidneys. Table 37–9 lists the primary metabolic enzymes and some potential drug interactions for the antipsychotic medications. Clinicians should assess the clinical impact of the addition or discontinuation of these drugs in individual patients and should make appropriate adjustments when necessary. Unlike the other antipsychotics, ziprasidone is mostly metabolized by aldehyde oxidase, a metabolic system independent of the CYP450 system. Paliperidone, the 9-hydroxy metabolite of risperidone, is mostly excreted unchanged in the urine, although up to one-third may be metabolized. Another recent discovery in metabolism of antipsychotics and their distribution involves the transmembrane energy-dependent efflux transporter, P-glycoprotein. This may limit the ability of a number of drugs to penetrate the blood–brain barrier and therefore impact pharmacologic activity in the brain.47 Antipsychotics currently known to use this pathway include perphenazine, haloperidol, fluphenazine, quetiapine, risperidone, and olanzapine.48
Due to its propensity to cause prolongation of the QTc interval, the use of ziprasidone with other agents that prolong the QTc interval should be avoided. These other agents include, but are not limited to, antiarrhythmic medications such as quinidine and sotalol, certain FGAs (chlorpromazine, droperidol, mesoridazine, pimozide, and thioridazine), and certain antibiotics (e.g., gatifloxacin, halofantrine, mefloquine, moxifloxacin, and pentamidine). Ziprasidone has not been shown to have clinically significant drug interactions with the CYP4503A3/4 inhibitors regarding increasing the QTc; however, higher doses, especially given with inhibitors of CYP4503A3/4 (e.g., ketoconazole and erythromycin), should be used cautiously. Clinicians should also be vigilant to avoid pharmacodynamic interactions with any of the antipsychotics involving additive side effects from combination therapies. Side effects that may be worsened with combination therapies include sedation, hypotension, anticholinergic symptoms, and weight gain or metabolic abnormalities.
Adjunct Pharmacologic Treatments
The judicious use of pharmacologic therapies other than antipsychotics is often necessary in the treatment of patients with schizophrenia. Concomitant medications may be indicated for the treatment of motor side effects, anxiety, depression, mood elevation, and possibly refractory psychotic symptoms. Anticholinergic medications (e.g., benztropine, 1–2 mg two times daily; trihexyphenidyl, 1–3 mg three times daily; and diphenhydramine, 25–50 mg two times daily) are used to effectively treat EPS, thereby improving the tolerability of these medications. They may be prescribed prophylactically with high-D2-binding agents or in patients at risk for EPS, or for treatment of EPS. β-Blockers (e.g., propranolol in doses of 30–120 mg/day) are sometimes effective for patients who develop akathisia. In some situations, such as on an inpatient unit, the concomitant use of benzodiazepines (e.g., lorazepam in doses of 1–3 mg/day) with the SGAs may be necessary for agitation and insomnia.
Antidepressants may be useful for patients with depressive symptoms that are not due to negative symptomatology or emotional blunting secondary to parkinsonian-type side effects. Since suicide and depression are linked, aggressive treatment is necessary when depression is present. Selective serotonin reuptake inhibitors (SSRIs) are the preferred agents, but may inhibit the CYP450 enzymes, thus raising plasma concentrations of clozapine, olanzapine, and haloperidol. Mood stabilizers, such as lithium and the anticonvulsants, have long been used adjunctively with antipsychotics to treat the affective component of schizoaffective disorder. Lastly, much research is currently underway to develop better treatments for primary negative symptoms and cognitive impairment; however, no approved treatments are yet available.
OUTCOME EVALUATION
Patient Education
![]() Education of the patient and family regarding the benefits and risks of antipsychotic medications and the importance of adherence to their therapeutic regimens must be integrated into pharmacologic management. Too often, patients receive little education about schizophrenia and may not have a good understanding of their diagnosis. Meetings for purposes of medication management offer an opportunity to provide some education on the general illness. Discuss the nature and course of schizophrenia and be prepared to accept that the patient might have a different understanding of the nature of his or her illness. Key points to cover include:
Education of the patient and family regarding the benefits and risks of antipsychotic medications and the importance of adherence to their therapeutic regimens must be integrated into pharmacologic management. Too often, patients receive little education about schizophrenia and may not have a good understanding of their diagnosis. Meetings for purposes of medication management offer an opportunity to provide some education on the general illness. Discuss the nature and course of schizophrenia and be prepared to accept that the patient might have a different understanding of the nature of his or her illness. Key points to cover include:
• Involve families in the education and treatment plans, since family psychoeducation may decrease relapse, improve symptomatology, and enhance psychosocial and family outcomes.49
• Be clear that there is no cure for schizophrenia and that medications only help to decrease the symptoms.
• Explain common side effects of medications.
• Discuss rare but dangerous side effects that may also occur.
• Stress the importance of medication and treatment adherence for improving long-term outcomes in schizophrenia.
Symptom Monitoring
Develop a good working alliance with the patient. When a solid therapeutic foundation is not formed, patients are frequently reluctant to share their psychotic experiences. Once a solid working relationship and some knowledge of the patient’s psychotic experiences exist, perform a more structured interview. Many assessments are available to more objectively rate positive and negative symptoms, level of function, and life satisfaction. The most commonly used scales include:
Table 37–10 Monitoring Protocol for Patients on SGAs
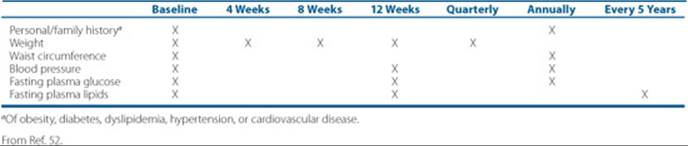
Table 37–11 Monitoring of WBC and Absolute Neutrophil Count During Clozapine Treatment
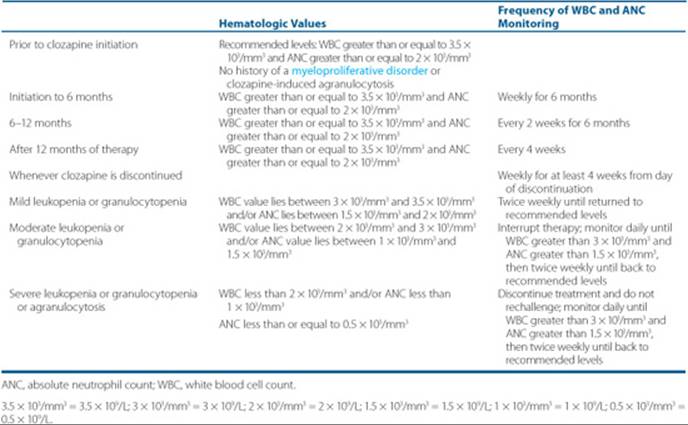
• Positive and Negative Symptom Scale (PANSS)
• Brief Psychiatric Rating Scale (BPRS)
• Clinical Global Impression (CGI) Scale
Using these scales on a regular basis, particularly when switching medications or changing doses, is a more reliable means of monitoring for improvement. Certainly symptom assessments cannot capture the full range of possible improvements a patient may experience, but they can be useful in deciding whether a medication is having substantial benefit.
Patient Encounter, Part 4
With his care providers, AC discussed several possible medication choices. Because there had been improvement of auditory hallucinations on risperidone, the initial choice was made to increase the dose to target the ongoing paranoid thinking. AC was seen weekly to monitor for emergence of EPS or akathisia. He was able to tolerate an increase to 4 mg of risperidone daily with improvement in his paranoid thinking. He went on several job interviews and accepted part-time employment at a coffee shop. When he complained of emergence of sexual dysfunction, his prolactin level was determined to be 42 ng/mL (42 mcg/L) (normal prolactin levels are less than or equal to 18–20 ng/mL [18-20 mcg/L] in men and less than or equal to 24 ng/mL [24 mcg/L] in women). He discussed risks and benefits of therapy with his clinicians and decided to continue risperidone for now, as he had improved in his ability to work and get out more in the community. He remains interested in having an aripiprazole trial in the future, as he hopes he might more easily lose the weight he gained while taking olanzapine and resume normal sexual functioning. He is no longer taking an antidepressant, and though he has some low days, he does not meet the criteria for diagnosis of depressive episode. He continues to be free of substance abuse, and his girlfriend continues to support his treatment. He still hopes to return to work in his field of expertise someday.
Based on the information above, create a care plan for this patient’s treatment. Your plan should include: (a) a statement of the drug-related concerns and/or problems, (b) the goals of therapy, and (c) a plan for monitoring and follow-up to determine whether the goals have been achieved and adverse effects minimized.
Side Effect Monitoring
![]() Regularly monitor patients for side effects and overall health status while taking antipsychotic medications.50,51 Perform orthostatic blood pressure measurements before initiating antipsychotics and throughout treatment. Ask about impaired menstruation, libido, and sexual performance regularly. Encourage patients to have annual eye exams, as several of the antipsychotic medications have been associated with the premature development of cataracts. Check body weight, fasting glucose, glycosylated hemoglobin, and lipid profile at baseline, at 4 months after initiation of medication, and then yearly.52For patients who are at higher risk of developing diabetes and those who gain weight, check body weight more often (Table 37–10). Encourage patients to act proactively against weight gain through healthier eating and exercise. Perform a baseline ECG for patients with pre-existing cardiovascular disease and risk for arrhythmia. With clozapine therapy there is a risk for the development of agranulocytosis, which is greatest in the first 6 months of treatment. Current guidelines require the monitoring of white blood cell counts (WBCs) prior to drug dispensing (Table 37–11).
Regularly monitor patients for side effects and overall health status while taking antipsychotic medications.50,51 Perform orthostatic blood pressure measurements before initiating antipsychotics and throughout treatment. Ask about impaired menstruation, libido, and sexual performance regularly. Encourage patients to have annual eye exams, as several of the antipsychotic medications have been associated with the premature development of cataracts. Check body weight, fasting glucose, glycosylated hemoglobin, and lipid profile at baseline, at 4 months after initiation of medication, and then yearly.52For patients who are at higher risk of developing diabetes and those who gain weight, check body weight more often (Table 37–10). Encourage patients to act proactively against weight gain through healthier eating and exercise. Perform a baseline ECG for patients with pre-existing cardiovascular disease and risk for arrhythmia. With clozapine therapy there is a risk for the development of agranulocytosis, which is greatest in the first 6 months of treatment. Current guidelines require the monitoring of white blood cell counts (WBCs) prior to drug dispensing (Table 37–11).
Patient Care and Monitoring
1. Assess the patient’s symptoms, review patient and family history, and obtain initial medical evaluation to rule out other causes of psychosis.
2. Periodically review patient data for consistency with diagnostic criteria, and regularly monitor changes in symptomatology.
3. Obtain a thorough history of prescription medication use, and determine what treatments have been helpful in the past, which treatments the patient is currently receiving, and previous side effects experienced.
4. Determine whether the patient is taking an appropriate antipsychotic drug and dose and whether the patient has other symptoms that may need to be treated.
5. Educate the patient, and the family if possible, about the disease state, medication treatments, possible side effects, and goals of treatment.
6. Develop a plan to assess the effectiveness of the current treatment regimen. Also, consider alternative treatments if current treatment is ineffective.
7. Encourage a healthy lifestyle, including eliminating or decreasing substance abuse and cigarette use, as well as appropriate nutritional counseling and exercise suggestions.
8. Determine the role of psychosocial treatments.
9. Evaluate the patient for the presence of adverse drug reactions, drug interactions, and allergies.
10. Monitor the appropriate laboratory measures to prevent or minimize metabolic abnormalities and other side effects.
11. Stress the importance of adherence with the treatment regimen and maintain treatment even if the patient is feeling well.
Commonly used rating scales to monitor for EPS include the Simpson Angus Scale (SAS) and the Extrapyramidal Symptom Rating Scale (ESRS). Akathisia is commonly monitored by the Barnes Akathisia Scale (BAS). The emergence of dyskinesias (writhing or involuntary movements) could represent the emergence of TD. Monitor for TD at least annually, and if FGAs are used, patients should be evaluated at each visit. The most commonly used instrument to measure these symptoms is the Abnormal Involuntary Movement Scale (AIMS).
Abbreviations Introduced in This Chapter
 Self-assessment questions and answers are available at http://www.mhpharmacotherapy.com/pp.html.
Self-assessment questions and answers are available at http://www.mhpharmacotherapy.com/pp.html.
REFERENCES
1. Schultz SK, Andreasen NC. Schizophrenia. Lancet 1999;353(9162):1425–1430.
2. Abdolmaleky HM, Thiagalingam S, Wilcox M. Genetics and epigenetics in major psychiatric disorders: Dilemmas, achievements, applications, and future scope. Am J Pharmacogenomics 2005;5(3):149–160.
3. Abi-Dargham A. Do we still believe in the dopamine hypothesis? New data bring new evidence. Int J Neuropsychopharmacol 2004;7(Suppl 1):S1–S5.
4. Laruelle M, Kegeles LS, Abi-Dargham A. Glutamate, dopamine, and schizophrenia: From pathophysiology to treatment. Ann NY Acad Sci 2003;1003:138–158.
5. Meltzer HY, Li Z, Kaneda Y, Ichikawa J. Serotonin receptors: Their key role in drugs to treat schizophrenia. Prog Neuropsychopharmacol Biol Psychiatry 2003;27(7):1159–1172.
6. Goff DC, Cather C, Evins AE, et al. Medical morbidity and mortality in schizophrenia: guidelines for psychiatrists. J Clin Psychiatry 2005;66(2):183–194.
7. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th ed. Text rev. Washington, DC: American Psychiatric Association, 2000.
8. Pompili M, Girardi P, Ruberto A, Taterelli R. Toward a new prevention of suicide in schizophrenia. World J Biol Psychiatry 2004;5(4):201–210.
9. Casey DE. Long-term treatment goals: Enhancing healthy outcomes. CNS Spectr 2003;8(11 Suppl 2):26–28.
10. Resnick SG, Fontana A, Lehman AF, Rosenheck RA. An empirical conceptualization of the recovery orientation. Schizophr Res 2005;75(1):119–128.
11. American Psychiatric Association. Practice guidelines for the treatment of patients with schizophrenia. Am J Psychiatry 2004;161(2 Suppl):1–56.
12. The expert consensus guideline series: treatment of schizophrenia. J Clin Psychiatry 1996;57(Suppl 12B):31.
13. Bellack AS. Skills training for people with severe mental illness. Psychiatr Rehabil 2004;27(4):375–391.
14. Lieberman JA, Stroup TS, McEvoy JP, et al. For the Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) Investigators. Effectiveness of antipsychotic drugs in people with chronic schizophrenia. N Engl J Med 2005;353(12):1209–1223.
15. Correll CU, Leucht S, Kane JM. Lower risk for tardive dyskinesia associated with second-generation antipsychotics: A systematic review of 1-year studies. Am J Psychiatry 2004;161(3):414–425.
16. Schotte A, Janssen PFM, Gommeren W, et al. Risperidone compared with new and reference antipsychotic drugs: In vitro and in vivo receptor binding. Psychopharmacology 1996;124(1–2):57–73.
17. Marder SR, Meibach RC. Risperidone in the treatment of schizophrenia. Am J Psychiatry 1994;151(6):825–835.
18. Csernansky JG, Mahmoud R, Brenner R, Risperidone-USA-79 Study Group. A comparison of risperidone and haloperidol for the prevention of relapse in patients with schizophrenia. N Engl J Med 2002;346(1):16–22.
19. Richelson E. Receptor pharmacology of neuroleptics: Relation to clinical effects. J Clin Psychiatry 1999;60(Suppl 10):5–14.
20. Beasley CM, Tollefson G, Tran P, et al. Olanzapine versus placebo and haloperidol: Acute phase results of the North American double-blind olanzapine trial. Neuropsychopharmacology 1996;14(2):111–123.
21. Tollefson GD, Beasley CM, Tran PV, et al. Olanzapine versus haloperidol in the treatment of schizophrenia and schizoaffective and schizophreniform disorders: Results of an international collaborative trial. Am J Psychiatry 1997;154(4):457–465.
22. Arvanitis LA, Miller BG, the Seroquel Trial 13 Study Group. Multiple fixed doses of Seroquel (quetiapine) in patients with acute exacerbation of schizophrenia: A comparison with haloperidol and placebo. Biol Psychiatry 1997;42(4):233–246.
23. Small JG, Hirsch SR, Arvenitis LA, et al. Quetiapine in patients with schizophrenia: a high- and low-dose double-blind comparison with placebo. Arch Gen Psychiatry 1997;54(6):549–557.
24. Seeger TF, Seymour PA, Schmidt AW, et al. Ziprasidone (CP-88,059): A new antipsychotic with combined dopamine and serotonin receptor antagonist activity. J Pharmacol Exp Ther 1995;275(1):101–113.
25. Keck P, Buffenstein A, Ferguson J, et al. Ziprasidone 40 and 120 mg/day in the acute exacerbation of schizophrenia and schizoaffective disorder: A 4-week placebo-controlled trial. Psychopharmacology 1998;140(2):173–184.
26. Daniel DG, Zimbroff DL, Potkin SG, et al. Ziprasidone 80 mg/day and 160 mg/day in the acute exacerbation of schizophrenia and schizoaffective disorder: A 6-week placebo-controlled trial. Neuropsychopharmacology 1999;20(5):491–505.
27. Shapiro DA, Renock S, Arrington E, et al. Aripiprazole, a novel atypical antipsychotic drug with a unique and robust pharmacology. Neuropsychopharmacology 2003;28(8):1400–1411.
28. Dolder C, Nelson M, Deyo Z. Paliperidone for schizophrenia. Am J Health Syst Pharm 2008;65(5):403–413.
29. Kapur S, Seeman P. Does fast dissociation from the dopamine d(2) receptor explain the action of atypical antipsychotics? A new hypothesis. Am J Psychiatry 2001;158(3):360–369.
30. Wirshing WC. Movement disorders associated with neuroleptic treatment. J Clin Psychiatry 2001;62:Suppl 21:15–18.
31. Margolese HC, Chouinard G, Kolivakis TT, et al. Tardive dyskinesia in the era of typical and atypical antipsychotics. Part 2: Incidence and management strategies in patients with schizophrenia. Can J Psychiatry 2005;50(11):703–714.
32. Egan MF, Apud J, Wyatt RJ. Treatment of tardive dyskinesia. Schizophr Bull 1997;23(4):583–609.
33. Correll CU, Schenk EM. Tardive dyskinesia and new antipsychotics. Curr Opin Psychiatry 2008;21:151–156.
34. Moore TA, Buchanan RW, Buckley PF, et al. The Texas Medication Algorithm Project antipsychotic algorithm for schizophrenia: 2006 update. J Clin Psychiatry 2007;68(11):1751–1762.
35. Lehman AF, Kreyenbuhl J, Buchanan RW, et al. The schizophrenia patient outcomes research team (PORT): Updated treatment recommendations 2003. Schizophr Bull 2004;30(2):193–217.
36. Byerly MJ, Nakonezny PA, Lescouflair E. Antipsychotic medication adherence in schizophrenia. Psychiatr Clin N Am 2007;30(3):437–452.
37. Perkins DO. Predictors of noncompliance in patients with schizophrenia. J Clin Psychiatry 2002;63:1121–1128.
38. Remschmidt H. Early-onset schizophrenia as a progressive-deteriorating developmental disorder: Evidence from child psychiatry. J Neural Transm 2002;109:101–117.
39. Jeste DV, Caligiuri MP, Paulsen JS, et al. Risk of tardive dyskinesia in older patients. A prospective longitudinal study of 266 outpatients. Arch Gen Psychiatry 1995;52:756–765.
40. Tsuang J, Fong TW. Treatment of patients with schizophrenia and substance abuse disorders. Curr Pharm Des 2004;10(18):2249–2261.
41. Wobrock T, Soyka M. Wobrock T, Soyka M. Pharmacotherapy of schizophrenia with comorbid substance use disorder-reviewing the evidence and clinical research. Prog Neuropsychopharmacol Biol Psychiatry 2008;32(6):1375–1385.
42. Conley RR, Kelly DL. Management of treatment resistance in schizophrenia. Biol Psychiatry 2001;50(11):898–911.
43. Meltzer HY. Suicide in schizophrenia, clozapine and adoption of evidence-based medicine. J Clin Psychiatry 2005;60(Suppl 12):47–50.
44. Nilsson E, Lichtenstein P, Cnattingius S, et al. Women with schizophrenia: pregnancy outcome and infant death among their offspring. Schizophr Res 2002;58(2–3):221–229.
45. Diav-Citrin O, Schechtman S, Ornoy S, et al. Safety of haloperidol and penfluridol in pregnancy: a multicenter, prospective, controlled study. J Clin Psychiatry 2005;66(3):317–322.
46. McKenna K, Koren G, Tetelbaum M, et al. Pregnancy outcome of women using atypical antipsychotic drugs: a prospective comparative study. J Clin Psychiatry 2005;66(4):444–449.
47. Wang JS, Taylor R, Ruan Y, et al. Olanzapine penetration into brain is greater in transgenic Abcb1a P-glycoprotein-deficient mice and FVB1 (wild type) animals. Neuropsychopharmacology 2004;29(3):551–557.
48. Sandson NB, Armstrong SC, Cozza KL. An overview of psychotropic drug-drug interactions. Psychosomatics 2005;46(5):464–494.
49. Murray-Swank AB, Dixon L. Family psychoeducation as evidence-based practice. CNS Spect 2004;9(12):905–912.
50. Marder SR, Essock SM, Miller AL, et al. The Mount Sinai conference on the pharmacotherapy of schizophrenia. Schizophr Bull 2002;28(1): 5–16.
51. Marder SR, Essock SM, Miller AL, et al. Physical health monitoring of patients with schizophrenia. Am J Psychiatry 2004;161(8):1334–1349.
52. American Diabetes Association, American Psychiatric Association, American Association of Clinical Endocrinologists, North American Association for the Study of Obesity. Consensus development conference on antipsychotic drugs and obesity and diabetes. Diabetes Care 2004;27(2):596–601.
53. Love RC, Mackowick M, Carpenter D, Burks EJ. Expert consensus-based medication-use evaluation criteria for atypical antipsychotic drugs. Am J Health Syst Pharm 2003;60:2455–2470.