Asher Hirshberg
INTRODUCTION
Laparotomy is the most common operation performed for truncal trauma. It consists of a methodical sequence of steps that enable the surgeon to gain access to abdominal injuries, and identify and address them. These steps are guided by a series of priority-driven decisions that shape the operation.1
There are two modes of laparotomy for trauma, corresponding to the two major indications for the procedure: peritonitis and bleeding.2 The first mode is abdominal exploration in a hemodynamically stable patient with a tender abdomen. In these cases, the operation proceeds along the lines of an explorative laparotomy for an acute abdominal condition such as a hollow organ perforation: it is urgent but not hectic since there is no danger of imminent death. A less common but more dramatic mode is a crash laparotomy in a patient in shock with intra-abdominal hemorrhage.1 Here the patient’s life is in immediate jeopardy because on-table exsanguination is a real threat. Despite its hectic pace, a crash laparotomy is not merely an accelerated version of the first mode. Instead, it is a multidimensional effort that combines technical and team leadership skills. In a crash laparotomy, the surgeon has to calibrate the operative effort not only to the patient’s clinical condition but also to the capabilities of the surgical team and the available resources.
A crash laparotomy requires therefore not only a more expedient technical approach to the task but also a different frame of mind. When operating for peritonitis in the stable patient, the focus is on reconstructing the anatomy. In a crash laparotomy, the focus is on rapid control of hemorrhage and preservation of the patient’s physiology. The anatomical integrity of the repair is less important, and is sometimes temporarily sacrificed to prevent an irreversible physiological insult. It is in these adverse circumstances that the special expertise of the trauma surgeon can make a difference.
This chapter provides an overview of laparotomy for trauma, with an emphasis on crash laparotomy in an unstable patient. The first part of the chapter describes the guiding principles of the procedure from the perspective of the trauma surgeon. This is followed by a detailed description of the technical steps and key maneuvers of a trauma laparotomy against the background of ongoing decision making. The final part of the chapter addresses special types of abdominal exploration in the injured patient such as urgent or planned reoperation after damage control surgery as well as bedside laparotomy and the current role of laparoscopy.
PRINCIPLES
![]() The Core Mission
The Core Mission
In a trauma laparotomy, the core mission of the surgeon is to stop the bleeding. The success of the procedure hinges on the surgeon’s ability to rapidly reach the source of intra-abdominal hemorrhage and control it effectively. All else is of secondary importance simply because there is no alternative to achieving hemostasis. If the patient is not bleeding significantly, the mission then becomes to identify and repair other injuries (typically holes in the gut).
The importance of remaining focused on the core mission cannot be overemphasized. The multitrauma patient often presents with a bewildering array of injuries surrounded by clouds of clinical, administrative, and medicolegal issues that easily divert attention from the uncontrolled bleeding in the abdomen. It is therefore crucial to keep in mind that once the indication for laparotomy has been established in a trauma patient in shock, nothing should stand in the way of a rapid organized effort to put the patient in the operating room (OR) and bring the surgeon face-to-face with the bleeder as quickly as possible.
![]() The Surgeon’s Support Envelope
The Surgeon’s Support Envelope
In the exsanguinating patient with a hole in the inferior vena cava, the goal of the entire team effort is to insert a vascular suture line into the wall of the injured vessel. The circulating nurse supplies the suture, the scrub nurse loads it on a needle driver, and the anesthesiologist transfuses and ventilates the patient—but it is the surgeon who will accomplish the goal on behalf of the entire team.
The surgeon’s core mission cannot be accomplished without effective specialized support. Every trauma center therefore maintains a support envelope around the operating surgeon. The elements of this envelope are typically arranged in three tiers (Table 27-1). The inner tier is located inside the sterile field around the open abdomen and is immediately accessible to the surgeon. The middle tier is inside the OR but outside the sterile field, and the outer tier is outside the OR and consists of a wide array of assets and resources located at various distances from it.
TABLE 27-1 Anatomy of the Surgeon’s Support Envelope

Some elements of the support envelope (especially in the inner and middle tiers) participate in the entire procedure, whereas others come into play at various stages. The single most important element in the support envelope and the engine that keeps it up and running is the circulating nurse.
There is often a time lag between the decision to employ a specific element of the support envelope and its availability to the surgeon. This delay is inversely related to the tier in which the element is located. For example, there is no delay in getting hold of a Deaver retractor, but it takes at least a few minutes to bring a Rummel tourniquet into the operative field—and much longer than that to obtain a portable x-ray.
It is sometimes difficult—but nevertheless essential—to think of a crash laparotomy in terms of resource allocation. Many elements in the surgeon’s support envelope are finite and precious resources that must not be wasted. Since the envelope responds to the surgeon’s requests, it becomes the surgeon’s responsibility to use those resources judiciously. A case in point is the circulating nurse, the most important resource not only in the middle tier but also in the entire support envelope. The circulating nurse can propel the operation forward or slow it down to a crawl. Requesting a specific instrument from the OR storage area means that the circulating nurse will be temporarily unavailable for other tasks, some of which may be more important for the conduct of the operation. Similarly, there is little point in doing a 30-minute damage control laparotomy only to find on completion that mobilizing an SICU bed from the outer tier takes more time than the procedure itself.
In using the resources of the support envelope, the surgeon must learn to be flexible and rapidly consider the cost of every request in terms of the precious time and attention of team members. The temptation to request “nice to have” items must be resisted for the sake of feasible solutions that move the operation forward. Close acquaintance with each of the three tiers and knowledge of the realistic capabilities of the key assets around the operative field are crucial to the smooth conduct of a trauma laparotomy.
![]() Preparation
Preparation
The less stable the patient, the less time should be spent on preoperative preparations. In the trauma resuscitation bay, in addition to the primary and secondary surveys and corresponding resuscitative maneuvers, it is very helpful (but not essential) to obtain chest and abdominal x-rays in patients with abdominal gunshot wounds. These x-rays help delineate the bullet trajectory and avoid missing injuries. This is especially important with multiple gunshot wounds. Some bullet trajectories carry special significance.3 A thoracoabdominal trajectory is associated with higher morbidity, and a trajectory across the abdominal midline (transabdominal) in a hypotensive patient is an early predictor of the need for damage control and a marker of high mortality.
The arrival of the hemodynamically unstable patient to the OR is often hectic and disorganized. The surgeon and the OR team must then convert the scramble into a methodical effort and rapidly deploy the surgeon’s support envelope.
This obligatory logistical interval from the patient’s entry into the OR to skin incision is known in the trauma surgery jargon as “the black hole.” During this interval, the patient is moved to the operating table, positioned, and prepared, but nothing is done to address the bleeding. The ability to minimize the duration of this interval is a measure of the efficiency of the OR team.
Effective surgeons recognize the need to minimize the “black hole” interval and to start cutting as quickly as possible, but at the same time they are also aware of the importance of proper positioning and preparation. It is crucial that the surgeon share a brief outline of the operative plan with the OR team (e.g., “We are going to insert a right chest tube, and then do a laparotomy and possibly also a right thoracotomy”). Rather than delegate the preparatory steps to junior members of the team, effective surgeons minimize or eliminate their time at the scrub sink and remain with the patient throughout the “black hole” interval, orienting the team and coordinating the effort.
The operative field for torso trauma extends from the chin to above the knees, between the posterior axillary lines and with both arms fully abducted (Fig. 27-1). This wide sterile field provides free access to the abdomen and chest as well as both groins, while giving the anesthesia team access to both upper extremities and the head and neck. It reflects the underlying principle of preparing the operative field for a “worst case scenario” where the surgeon may have to enter other visceral compartments in mid-operation.4 As the surgical team defines, drapes, and prepares to work in the operative field, the inner and middle tiers of the surgeon’s support envelope organize and deploy. Before making the incision, the surgeon should take a moment to pause, look up from the operative field, take mental stock of the overall situation in the OR, and ascertain that everyone is ready.
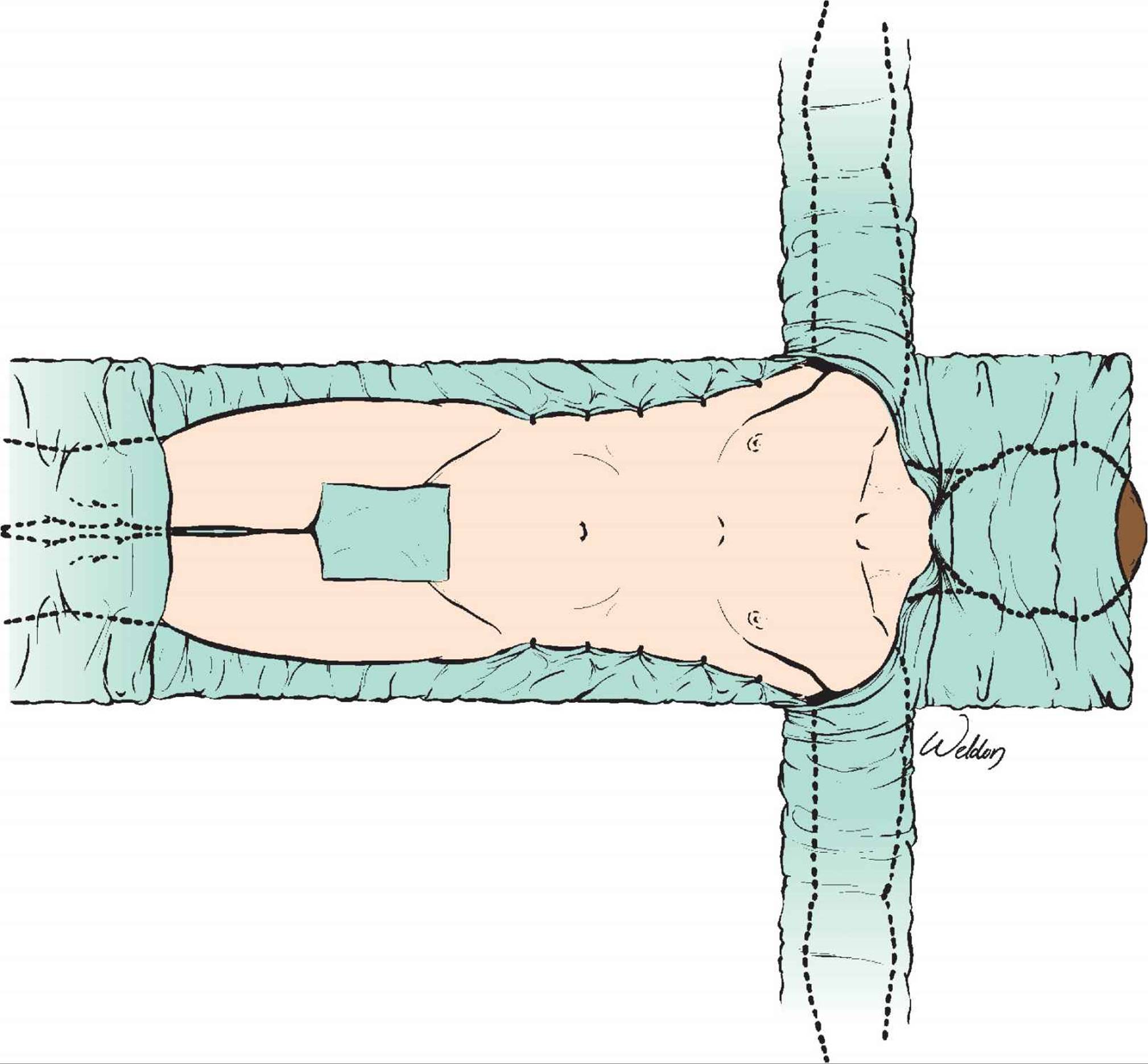
FIGURE 27-1 The wide operative field for torso trauma provides free access to the abdomen, chest, and groins. It reflects the principle of planning for unexpected obstacles during the procedure such as the need to extend the operation into another cavity to access the groins.
![]() The Operative Sequence
The Operative Sequence
Every trauma laparotomy follows a generic sequence of operative steps (Fig. 27-2).1 This sequence reflects the underlying logic and priorities of the procedure, but is often modified according to the clinical circumstances and operative findings. For example, in the absence of significant bleeding there is no need to pack the abdomen for temporary hemostasis and the surgeon proceeds directly to exploration. If a major obvious bleeder (such as injury to an abdominal great vessel) is encountered shortly after entering the abdomen, temporary hemostasis often merges with a definitive vascular repair, and full exploration of the peritoneal cavity takes place later.
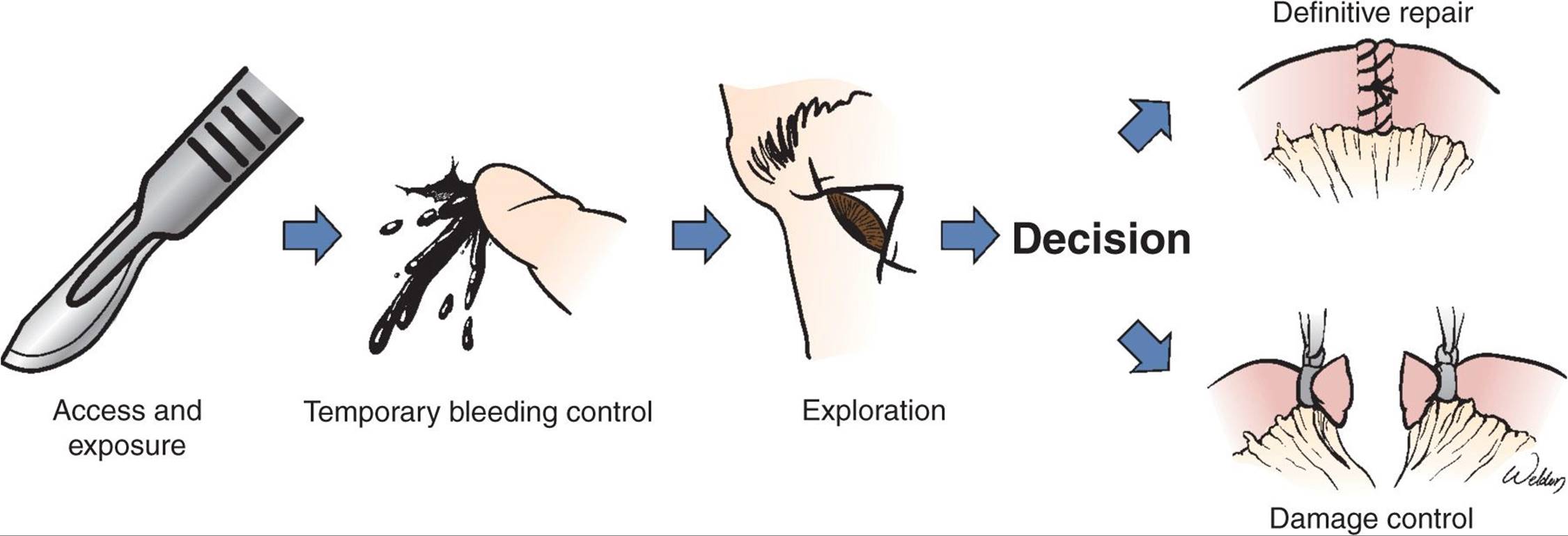
FIGURE 27-2 A schematic depiction of the generic sequence of steps in a trauma laparotomy. This sequence reflects the global priorities but is frequently modified as needed. The key decision point is the choice of operative profile that shapes the rest of the procedure as well as the patient’s postoperative course.
Gaining access and exposure (a long midline incision and evisceration of the bowel) and gaining temporary hemostasis (by packing or manual pressure) are routine tasks that are rapidly accomplished with minimal support. Effective temporary hemostasis is a crucial early success because it gives the surgeon a first rough impression of the injuries, and provides a time window to pause, think, and plan ahead. The anesthesia team now has time to regroup and catch up on previous blood loss. Additional instruments (e.g., vascular tray), help (such as an experienced assistant), and technology (autotransfusion device) can be mobilized from the middle and outer support tiers. Failure to achieve early temporary hemostasis in the face of severe hemorrhage means that the surgeon and the team are denied all these advantages: the scramble continues (as does the bleeding) and can fatally derail the entire operation.
Methodical exploration of the peritoneal cavity allows the surgeon to discover and define the injuries. However, in real life this methodical exploration may be interrupted by the urgent need to address another injury or enter another anatomical compartment (e.g., the chest). In other cases, the necessity to bail out to prevent an irreversible physiological insult (see below) may force the surgeon to abort the systematic exploration altogether, deferring it until reoperation.
The most pivotal decision in the operative sequence is between definitive repair and damage control. By making this decision, the surgeon chooses an operative profile and shapes the remainder of the procedure. Definitive repair means repair or resection of the injured organs, reconstruction of the anatomy, and formal abdominal closure. This is the traditional trauma laparotomy and it applies to the great majority of cases. Damage control (see Chapter 38) means a rapid bailout using temporary measures to control bleeding and spillage and temporary abdominal closure with the intention to return to abdomen in the next few days. Since damage control is associated with increased morbidity, it is used selectively in the most severely injured patients.
A “forced bailout” is a situation where the patient’s physiology is so precarious that damage control remains the surgeon’s only feasible option. This occurs when the surgeon erroneously decides to proceed with definitive repair, having either underestimated the injuries or overestimated the capabilities of the surgical team. As the patient continues to bleed and becomes hypothermic, edematous, and coagulopathic, the surgeon is forced to pack the abdomen and bail out to avert an irreversible physiological insult. Similarly, ominous injury patterns such as multifocal or multicavitary exsanguination5 push the patient to the brink of death so quickly that a forced bailout is the only option if the surgeon managed to control the bleeding.
![]() The Surgeon
The Surgeon
Effective surgeons always plan ahead. They conduct the operation with a clear sense of the core mission and how they plan to accomplish it. They anticipate difficulties and plan to avert or meet them. It is commonly said that an effective scrub tech must stay at least one step “ahead of the operation,” always preparing the instruments for the next step. In fact, this is only one example of dealing with the delays and time lags inherent in the structure of the surgeon’s support envelope. Obviously the elements of the middle and outer tiers must be even further ahead of the operation, with the circulator obtaining a device that will be needed in 5–10 minutes and the anesthesia team ordering blood products that will be used in half an hour or later. To confidently steer the operation in the right direction, the surgeon must therefore formulate a plan that extends well beyond the next few steps and has a clear end point (e.g., “We must pack the liver, tie off the bowel injuries and be in the SICU in less than an hour”).
Maintaining a continuous ongoing dialogue with the OR team is another hallmark of an effective surgeon. Amidst the drama of operating on a massively bleeding patient, it is sometimes easy to forget that team members outside the operative field cannot do their jobs intelligently without understanding at least the broad picture of what is being done and where the operation is heading. It is a sad fact that surgeons, anesthesiologists, and OR nurses spend their entire careers working as part of OR teams—yet train as individuals. Surgeons are taught to think of themselves as captains of the operative ship, yet are rarely, if ever, trained in collaborative teamwork.6,7
Team leadership in the OR is more about carefully listening to input from others and interacting with them than about making an endless stream of requests for esoteric instruments. A good example is communication with the anesthesia team who has crucial information that the surgeon simply cannot do without. Assessing the patient’s physiological reserves hinges on a set of variables such as core temperature and total blood loss, all of which are monitored by the anesthesia team but are also needed by the surgeon for an intelligent decision to bail out. The surgeon must also be acutely aware that the patient may be bleeding outside the operative field (e.g., into the pleural space) and the only clues that something is amiss in another visceral compartment will be noticed by the anesthesia team. Therefore, open communication lines and constant interaction between surgeon and anesthesiologist across the sterile drapes are absolutely essential for a safe laparotomy. Such a dialogue ensures that the anesthesiologist knows what the injuries are, understands the surgeon’s intentions, and tailors the anesthesia plan accordingly.
One of the hallmarks of a crash laparotomy as compared to the more leisurely pace of laparotomy for peritonitis is the frequency of sudden changes in the operative plan. It is essential that the surgeon update the OR team of these changes directly and without delay.
Trauma laparotomy is performed by general and trauma surgeons who vary widely in their trauma experience. From time to time, surgeons may face a technical challenge that they feel either is outside their comfort zone or requires more competent assistance. The correct thing to do is to get experienced help. There are several good “pause points” along the operative sequence where the surgeon can stop operating, assess the situation, and decide to summon help. For example, after obtaining effective temporary hemostasis, the surgeon may decide to pause the operation for a short time and recruit a better assistant from the outer support tier. Another valid “pause point” is at the end of exploration, where the surgeon may decide not to open a central retroperitoneal hematoma and instead summon a colleague with more vascular experience. This is certainly a wiser decision than plunging into the hematoma and losing control (and the patient). Pausing the operation with the situation temporarily at hand and seeking help is never a sign of weakness but rather a marker of sound judgment and of personal and professional maturity.
Most trauma centers are also academic institutions where resident training closely follows patient care as a high priority. Doing a crash laparotomy in a teaching setup is a special challenge because the surgeon must ensure that resident teaching does not interfere with the smooth and timely flow of the procedure. In other words, the teaching experience must be carefully tailored both to the specific situation in the operative field and to the resident’s capabilities. During a teaching-oriented crash laparotomy, the teacher does not actually hold the knife or needle driver but nevertheless maintains full control of all aspects of the operation, interacts with the team, and coordinates the support while the trainee focuses on the technical aspects of the operation. Making a crash laparotomy an educational experience for the resident without compromising patient safety is the hallmark of a true surgical educator.
TECHNIQUES AND MANEUVERS
![]() Gaining Access and Exposure
Gaining Access and Exposure
The most expedient access to the peritoneal cavity is through a long midline incision. Speed is especially important during a crash laparotomy when the patient is in shock. A bold xiphopubic skin incision skirting the umbilicus is rapidly followed by sharp division of the subcutaneous fat down to the fascia. The surgeon then “gains the midline” by identifying the decussating fibers of the anterior rectus sheath.8Sharply dividing the linea alba with the scalpel exposes the preperitoneal fat throughout the entire length of the incision. In hypotensive patients, bleeding from the divided skin and subcutaneous fat is minimal due to peripheral vasoconstriction, and the entire incision can be completed with three long precise strokes of the scalpel.
In most patients, the peritoneum just cranial to the umbilicus is very thin or has a defect, and is covered with scant preperitoneal fat. Poking a finger through this area (Fig. 27-3) is the quickest way to enter the peritoneal cavity. The hole is then enlarged by incising both the peritoneum and overlaying preperitoneal fat together to the full extent of the incision using Mayo scissors. Dividing the falciform ligament between clamps provides access to the right upper quadrant and completes the incision.
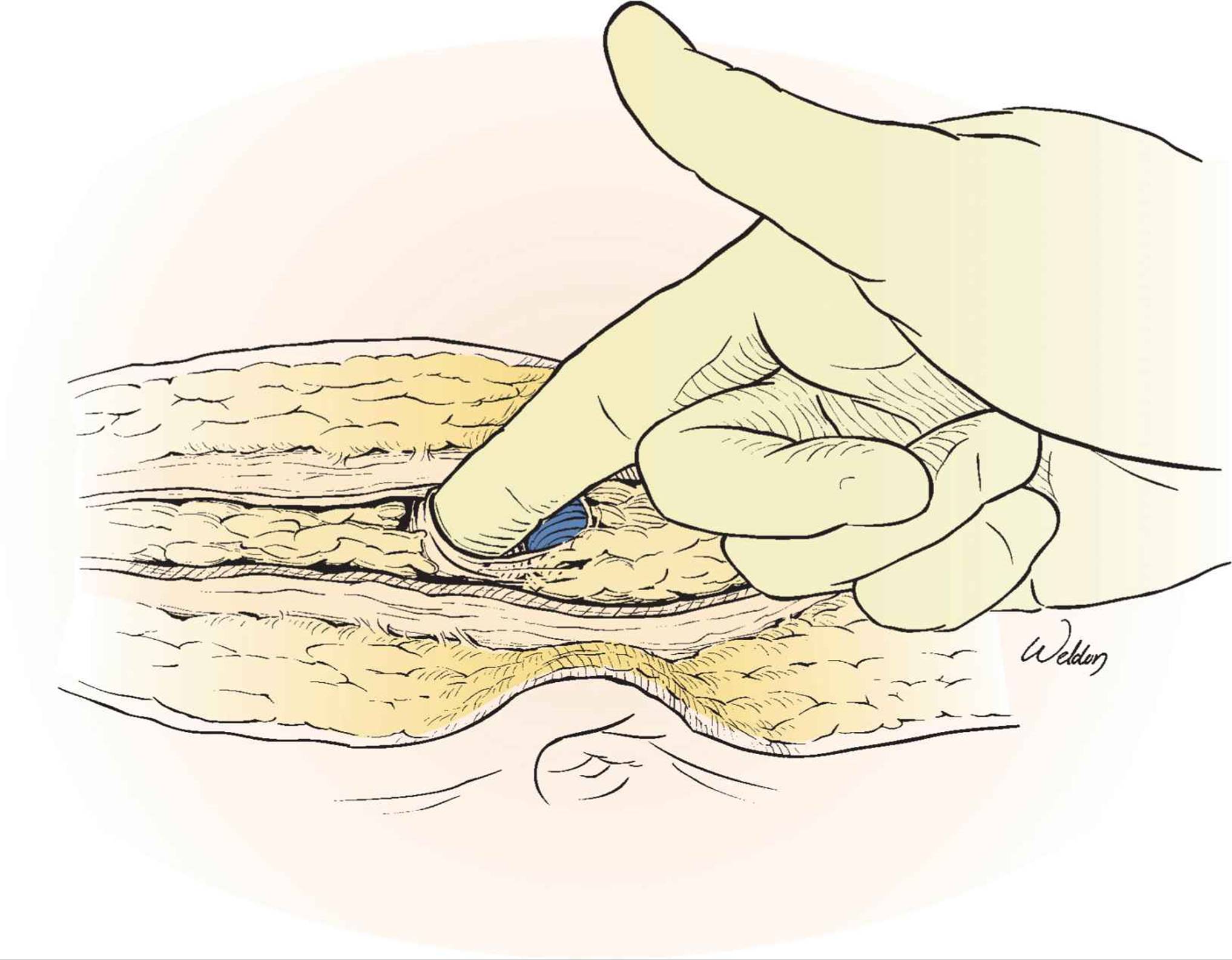
FIGURE 27-3 The quickest way to enter the peritoneal cavity is through the very thin (or absent) peritoneum immediately cranial to the umbilicus. After having divided the linea alba throughout the entire length of the incision, the surgeon can easily locate this area, bluntly poke a finger through it, and then incise the peritoneum and preperitoneal fat with heavy scissors.
When gaining rapid access to the peritoneal cavity, the major pitfall is iatrogenic injury. The left lateral lobe of the liver, transverse colon, and bladder are at risk in the upper, middle, and lower parts of the incision, respectively. In patients with a pelvic fracture, limiting the incision to the upper abdomen avoids entering a retroperitoneal hematoma that may extend into the preperitoneal space.
In patients with previous laparotomy scars, the surgeon can either carefully reenter through the old scar or try to skirt around it. Most surgeons choose the former option, which entails carrying the skin incision beyond the previous scar into “virgin” territory. The peritoneal cavity is thus entered in an area where adhesions to the anterior abdominal wall are less likely, and the incision is gradually extended into the previous scar, carefully avoiding bowel loops that are often adherent to the undersurface of the scar. This process can be time consuming and frustrating in a hypotensive patient. The other option is to bypass the troublesome area by making a completely new incision in a different orientation, such as a bilateral subcostal incision.
The final and often neglected key maneuver that provides access to the abdominal cavity is complete evisceration of the small bowel (Fig. 27-4): gathering all the loops into a surgical towel outside the abdomen and to the patient’s right. This pivotal step converts the bloody mess inside the injured abdomen into an organized workspace.1

FIGURE 27-4 Complete evisceration of the small bowel immediately on entering the peritoneal cavity is a key maneuver that is often forgotten by the inexperienced. Gathering all bowel loops outside the abdomen and always to the patient’s right is a pivotal step in organizing the peritoneal cavity into a convenient workspace.
![]() Achieving Temporary Hemostasis
Achieving Temporary Hemostasis
In blunt abdominal trauma, temporary bleeding control is typically achieved with empirical packing. Blindly shoving laparotomy pads into the abdomen without evisceration is not an effective way to achieve temporary hemostasis. After rapidly eviscerating the bowel and evacuating the blood from the peritoneal cavity using open laparotomy pads and suction, the surgeon proceeds to swiftly pack the abdomen without attempting to precisely identify the injuries (hence the term “empirical” packing).
With the assistant elevating the corresponding portion of the abdominal wall using a large retractor, the surgeon places a hand over the dome of the liver, gently pulling it toward the midline. Several laparotomy pads are then placed above and then below the liver, creating a hemostatic “sandwich” that approximates disrupted tissue planes. The same technique of packing over the surgeon’s hand and creating a “sandwich” is repeated above and below the spleen (Fig. 27-5). The paracolic gutters and pelvis are packed next. Empirical packing is especially useful in blunt trauma, since the most common sources of bleeding are the spleen, liver, and small bowel mesentery. Bleeding from the solid organs can often be temporarily controlled by packing (unless it is arterial), while mesenteric bleeders are immediately apparent in the eviscerated bowel and are easily accessible.
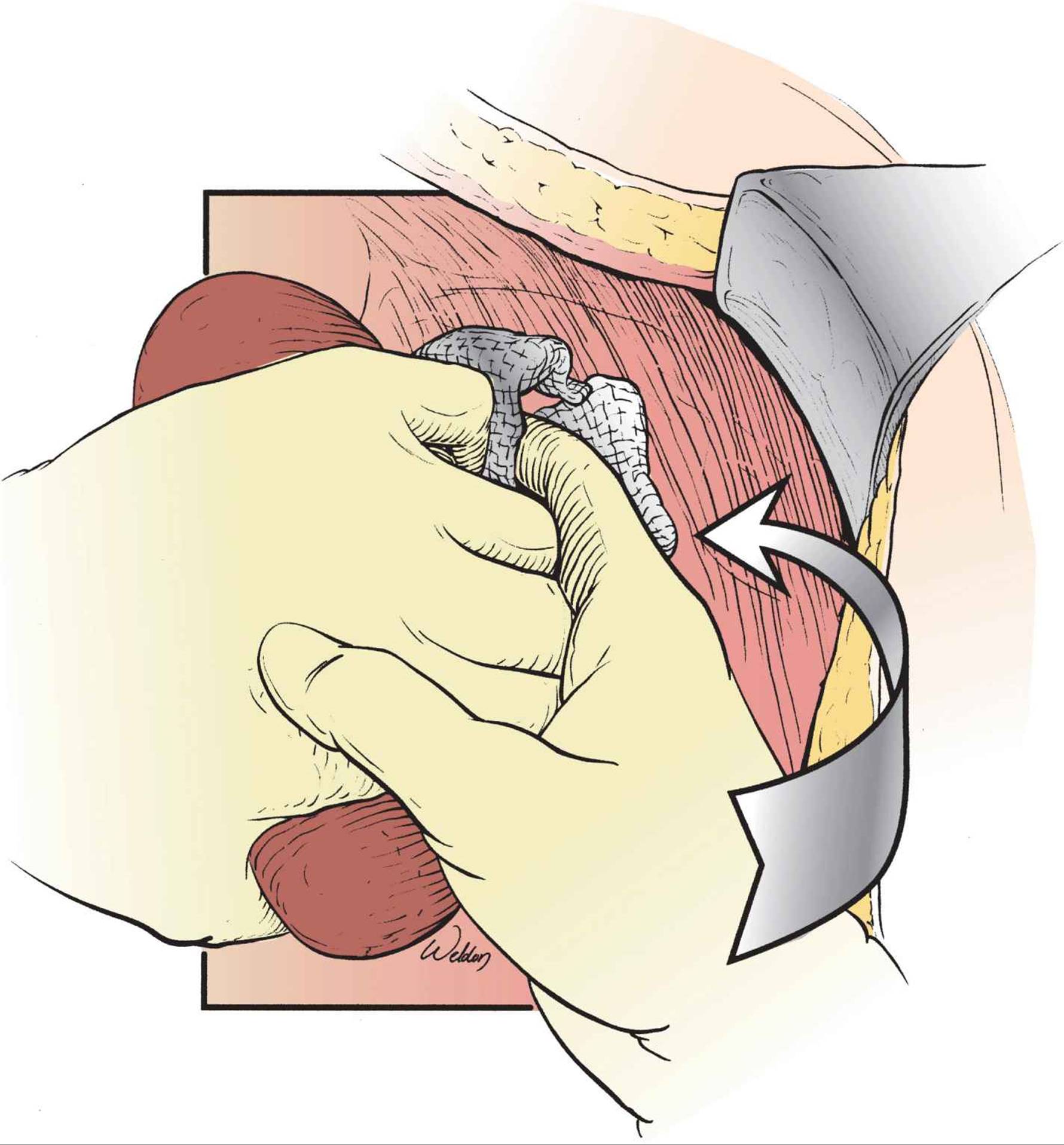
FIGURE 27-5 Empirical packing of the left upper quadrant to achieve temporary hemostasis. The surgeon uses one hand to pull the spleen medially and places laparotomy pads over that hand above and below the spleen to create a hemostatic “sandwich.” A similar technique is used in the right upper quadrant, both paracolic gutters, and the pelvis. Empirical packing is especially useful in blunt trauma.
In penetrating trauma, empirical packing is less useful. Knowing the presumed trajectory of the wounding agent allows the surgeon to focus on a specific quadrant or area of the abdomen. After evisceration, it is often possible to address the bleeder directly using atraumatic measures such as manual or digital pressure. Blind clamping in a pool of blood should be avoided because it is ineffective and dangerous. Packing can be used to temporarily control bleeding from an injured solid organ or a retroperitoneal hematoma.
In a rapidly exsanguinating patient, most surgeons would consider supraceliac aortic clamping. This is a resuscitative maneuver, not a hemostatic technique. Instead of formally clamping the lowermost thoracic aorta through the crus of the diaphragm,9 which entails blind dissection through the crus and carries the risk of iatrogenic injury, it is safer to simply compress the supraceliac aorta manually through a hole in the lesser omentum. The aorta is identified by palpation as it emerges beneath the diaphragm immediately below and to the right of the abdominal esophagus, and is manually compressed against the spine.4
Successful temporary hemostasis is a pivotal moment in the operation because it has a significant psychological impact on the OR team. It lowers the sense of extreme urgency and turmoil that sometimes characterizes the previous steps. The unknowns in the abdomen are now at least partially known. Stress levels among team members are lower. The situation seems temporarily at hand. There is time to take a deep breath, think, and convert a frantic scramble into a disciplined procedure.
![]() Exploring the Peritoneal Cavity
Exploring the Peritoneal Cavity
The mesentery of the transverse colon divides the peritoneal cavity into a supramesocolic and an inframesocolic compartment, and each must be explored in turn using a consistent methodical sequence. Pulling the transverse colon cranially enables the surgeon and assistant to run the gut from the ligament of Treitz to the rectum, using two pairs of hands in a coordinated motion that flips each segment to inspect both sides of the bowel and mesentery. Spillage from holes in the gut is temporarily controlled using Babcock or Allis clamps. Colonic perforations can often be smelled before they are seen. Inspection of the bladder and the female reproductive organs concludes the exploration of the inframesocolic compartment.
Pulling the transverse colon caudad allows the surgeon to explore the supramesocolic compartment from right to left. The liver and gallbladder are inspected and palpated for injuries, followed by palpation of the right kidney. This is followed by inspection of the anterior stomach and proximal duodenum, and then palpation of the spleen and the left kidney. The surgeon then inspects each hemidiaphragm and notes if it is convex or is bulging into the abdomen. The posterior peritoneum is inspected for retroperitoneal hematoma.
Entering the lesser sac is easiest through the left side of the greater omentum (which is typically thinner and less vascular). This provides a reasonable view of the posterior gastric wall and the body and tail of the pancreas. This exposure is good enough to inspect the lesser sac, but not to work in it. A wider exposure can be achieved by taking down the entire length of the greater omentum from the transverse colon along the bloodless line.
In patients with penetrating abdominal trauma, injuries tend to be missed at several less accessible locations, especially in obese patients (Table 27-2).10 The key to avoiding missed injuries during laparotomy for penetrating trauma hinges on two technical principles:
TABLE 27-2 Common Locations of Missed Injuries in Penetrating Abdominal Trauma
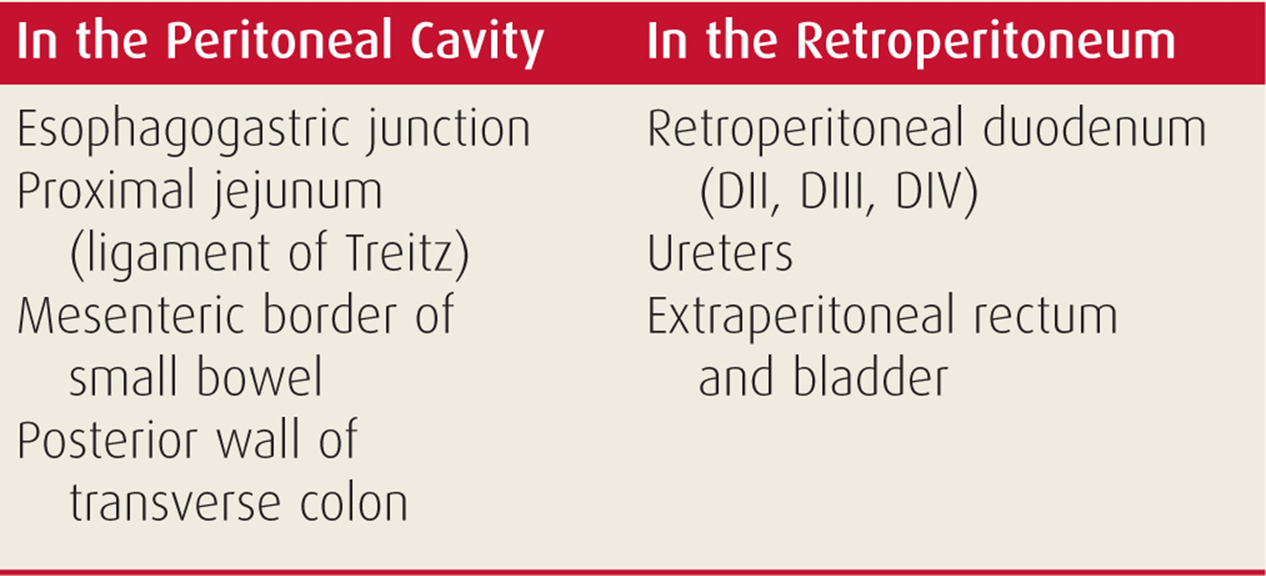
• The surgeon must be able to reconstruct the path of the wounding missile, and the trajectory must be linear and make sense. Inability to “connect the dots” between different injuries is often a clue to a missed injury.
• Finding an odd number of perforations in a hollow organ is cause for concern and should prompt the surgeon for extra vigilance in looking for another missed hole.
Since much of the colon is retroperitoneal and covered with omentum and pericolic fat, it is easy to miss small colonic perforations. Therefore, any seemingly superficial subserosal hematoma on the colon must always be carefully unroofed because it may hide a transmural injury. When the trajectory of the wounding agent passes anywhere near a ureter, the relevant side of the colon should be mobilized and the ureter fully visualized.
![]() The Medial Visceral Rotations
The Medial Visceral Rotations
Rapid access to the abdominal great vessels (aorta and vena cava) and their major branches, as well as other retroperitoneal structures (such as the duodenum and kidneys), hinges on two maneuvers of medial visceral rotation. The Mattox maneuver and the Cattell–Braasch maneuver have become part of the folklore of trauma surgery, but there is still much confusion and misunderstanding regarding their proper use and precise anatomical details. Both maneuvers are based on the same anatomical principle of lifting the intraperitoneal viscera off the posterior abdominal wall and rolling them medially to expose the midline retroperitoneal structures. However, there are important differences in their utility. The Mattox maneuver is used for the specific purpose of gaining access to the suprarenal aorta, the most inaccessible segment of the abdominal aorta. The Cattell–Braasch maneuver provides access to the entire inframesocolic (or infrarenal) retroperitoneum and has therefore much wider use for different purposes and in various operative situations.
The left-sided medial visceral rotation (Fig. 27-6) is known in trauma surgery as the Mattox maneuver.11 The surgeon begins the maneuver by mobilizing the lowermost descending colon. Incising the white line of Toldt enables the surgeon to bluntly dissect behind the left colon and rapidly mobilize it from below toward the splenic flexure. Continuing the blunt dissection in an upward and medial direction in the same avascular plane immediately anterior to the muscles of the posterior abdominal wall allows the surgeon to gradually rotate the left kidney spleen, pancreas, and stomach medially and expose the entire length of the abdominal aorta all the way up to the diaphragmatic hiatus.4 In most situations requiring this maneuver, a large supramesocolic retroperitoneal hematoma greatly facilitates the dissection by lifting the peritoneal organs off the posterior abdominal wall. The surgeon can achieve proximal control of the supraceliac aorta immediately below the diaphragm. Another option is to incise the left diaphragmatic crus laterally, bluntly dissect around the aorta, and clamp it in the lower chest through the incised hole in the diaphragm.

FIGURE 27-6 The full left-sided medial visceral rotation for trauma (Mattox maneuver) always includes the left kidney. It provides access to the entire length of the abdominal aorta and is the only way to rapidly expose the suprarenal aortic segment in the presence of a central retroperitoneal hematoma. The inset shows the correct plane of dissection that is immediately on the muscles of the posterior abdominal wall. The presence of a large retroperitoneal hematoma greatly facilitates the maneuver. (Copyright © Kenneth L. Mattox, MD.)
One crucial anatomical detail distinguishes the Mattox maneuver from the previously described left-sided medial visceral rotation used for aortic exposure in elective vascular surgery.12 The Mattox maneuver always includes the left kidney because leaving it in place interposes the anterior renal fascia between the plane of dissection and the aorta. This obscures the aorta and requires division of the fascia to get to it. Furthermore, by leaving the left kidney in place, the left renal vein restricts access to the anterior aspect of the aorta and the left ureter is vulnerable to injury.
Splenic injury is by far the most common iatrogenic complication of left-sided medial visceral rotation,12 and is usually addressed by removing the injured spleen. Other sources of bleeding are avulsion of the descending lumbar vein from the left renal vein and clamp injury to the right lateral aortic wall when the aorta is clamped with insufficient circumferential dissection. Pancreatitis and gut ischemia related to retraction are rare and late complications that emphasize the need for gentle handling and protection of the rotated viscera.
The right-sided medial visceral rotation consists of three successive steps4 (Fig. 27-7). The surgeon begins with a full Kocher maneuver, mobilizing the duodenal loop (and head of pancreas) from the common bile duct superiorly to the superior mesenteric vein inferiorly. This exposes the second part of the duodenum but does not provide free access to the underlying vascular structures (inferior vena cava and right renal hilum), which is so important in a trauma situation.

FIGURE 27-7 The three successive stages of the right-sided medial visceral rotation. The first step (Kocher maneuver) mobilizes the pancreaticoduodenal complex and is of limited utility in trauma. The second step (mobilization of the right colon) provides wide exposure of the inferior vena cava and right kidney and hilum. The third step (Cattell–Braasch maneuver) is completed by separating the small bowel mesentery from the posterior peritoneum.
The second step improves this limited exposure by carrying the incision in the posterior peritoneum caudad along the white line of Toldt and mobilizing the right colon.13 Reflecting the right colon medially provides wide exposure of the right-sided retroperitoneal organs, including the entire infrahepatic vena cava, the right kidney and renal vasculature, as well as the right iliac vessels. In fact, it is possible (but not convenient) to gain control of the suprarenal aorta and both renal arteries from the right side using this exposure.14
The third and final step of the right-sided medial visceral rotation is the complete Cattell–Braasch maneuver.15 Gathering the small bowel to the right enables the surgeon to clearly visualize and incise the avascular line of fusion of the small bowel mesentery to the posterior peritoneum. The incision is carried around the cecum and then all the way up along the “white line” between the cecum and the ligament of Treitz. This enables the surgeon to swing the small bowel and right colon out of the abdomen and onto the patient’s anterior chest, exposing the entire inframesocolic retroperitoneum (Fig. 27-8).
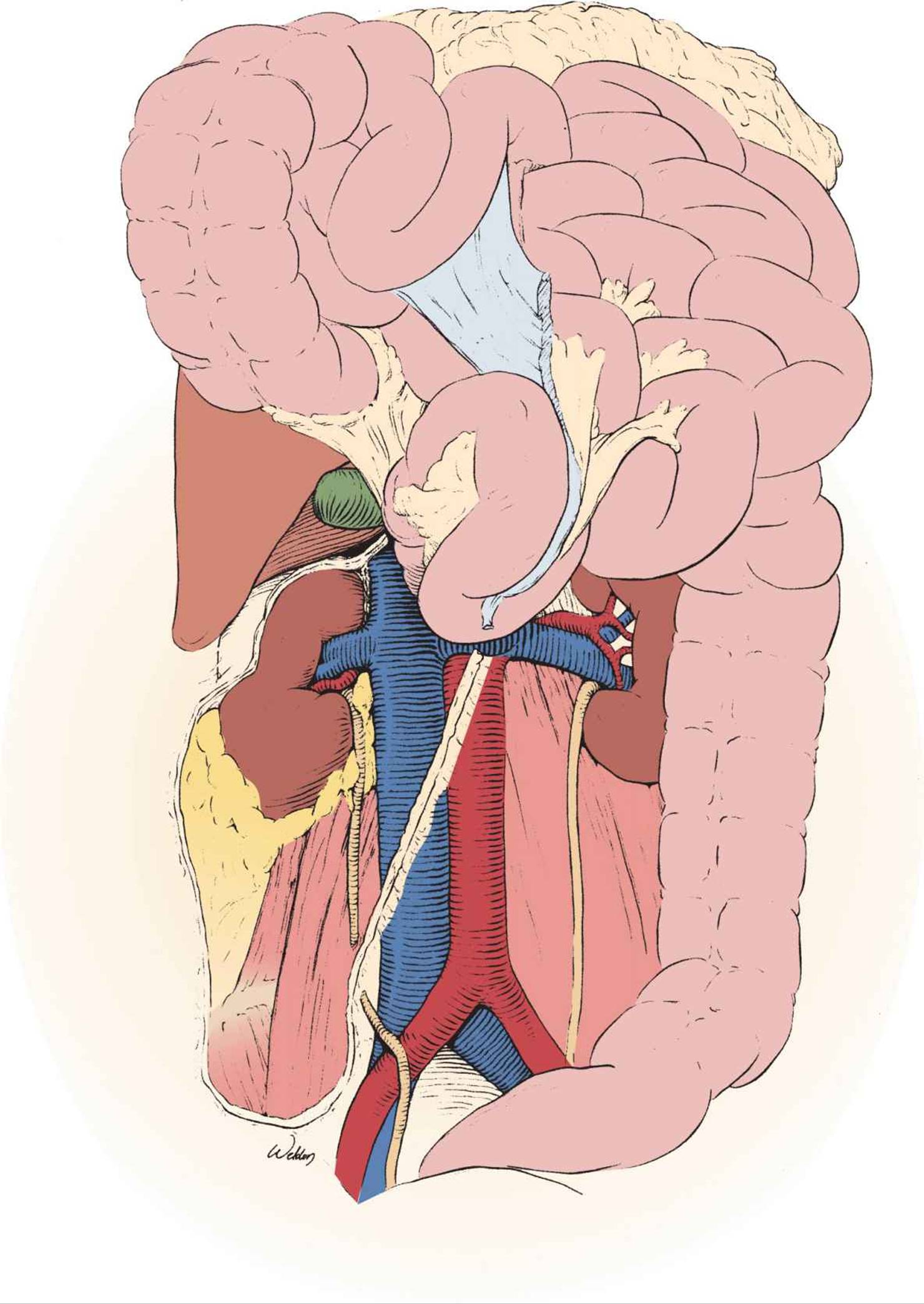
FIGURE 27-8 The full Cattell–Braasch maneuver provides panoramic exposure of the retroperitoneum. This maneuver is useful in a wide range of operative situations where wide exposure is needed to explore the retroperitoneum or access specific structures. The only two areas of the retroperitoneum that remain inaccessible are the retrohepatic vena cava and the suprarenal aorta. The latter is accessible with the Mattox maneuver.
Right-sided medial visceral rotation has fewer iatrogenic complications than its left-sided counterpart, the most serious being an injury to the superior mesenteric vein at the root of the mesentery. Once the right colon has been mobilized, it is hanging only by its mesentery and is vulnerable to avulsion of the middle colic vein off the superior mesenteric vein, resulting in unexpected fierce hemorrhage from the base of the mesentery. The ensuing rush to gain control may result in inadvertent clamping and division of the superior mesenteric vein.
Originally described as a technique for exposing the third and fourth parts of the duodenum in nontrauma situations,15 the Cattell–Braasch maneuver has found many applications in abdominal surgery. The panoramic retroperitoneal exposure afforded by it is useful in a range of operative situations where wide access to the retroperitoneum can help the surgeon figure out a complex bullet trajectory, approach an inframesocolic or pelvic retroperitoneal hematoma, or expose inaccessible retroperitoneal structures.
Mastering the medial visceral rotations adds very powerful weapons to the surgeon’s armamentarium. But they are precision weapons, not bludgeons. For example, the only indication to perform the Mattox maneuver is a central supramesocolic retroperitoneal hematoma. Yet the phrase “I Mattoxed the patient” is too often heard after a battle with abdominal bleeding where there was no indication to use the maneuver. It is therefore crucial to gain ownership of these techniques by studying them in detail, and if possible practicing them in the lab. It is also equally important to fully understand the indications for and potential benefit of each medial visceral rotation.
![]() Approach to Intra-Abdominal Bleeding
Approach to Intra-Abdominal Bleeding
The goal of a crash laparotomy is to achieve effective control of intra-abdominal bleeding before the patient exsanguinates or sustains an irreversible physiological insult (see the next section). A peritoneal cavity filled with blood or a large retroperitoneal hematoma can be an intimidating sight, but the surgeon must keep in mind that panic or reflexive action often leads to errors in bleeding control, failure to achieve hemostasis, and death on the operating table. The patient’s best chance is a cool-headed and systematic approach. The key to success is not technical brilliance but rather the ability to select effective techniques from a menu of hemostatic options and apply them in a methodical fashion until the bleeding stops.
Full evisceration allows the surgeon to organize the abdominal cavity and gain a general impression of the source and magnitude of bleeding. The single crucial decision that the surgeon must make during these first few hectic minutes inside the blood-filled abdomen is whether the bleeding problem is simple or complex, based on the bleeding rate and the accessibility of the bleeder.
Simple bleeding problems are easy to solve because they are accessible and can be attacked directly. For example, even fierce hemorrhage from organs that have an accessible vascular pedicle (such as a shattered spleen or kidney) can be rapidly controlled by mobilizing the organ into the midline, controlling the pedicle, and then deciding whether to repair the injury or resect the organ. Bleeding from the majority of liver injuries stops either with packing or with lesser hemostatic options such as suture or electrocoagulation.
A central retroperioneal hematoma is typically a manifestation of a major abdominal vascular injury and an example of a complex bleeding problem where a direct attack on the bleeder often proves a lethal mistake. Instead, the complex problem requires a disciplined approach consisting of proximal control, exposure of the injury using the appropriate exposure maneuver, and a formal vascular repair. Other examples of complex hemostatic problems are bleeding from a high-grade liver injury that cannot be controlled with packing and simultaneous vigorous hemorrhage from several sites (also known as multifocal exsanguination).
When addressing a complex bleeding problem, the surgeon must first achieve temporary hemostasis, and then pause the operation to mobilize the resources of the surgical support envelope and organize the attack on the injury. Pausing the operation at this point (sometimes for as long as 30 minutes or more) is absolutely vital because many resources that are required to successfully deal with a complex bleeding problem are located outside the OR, in the outer tiers of the support envelope. The surgeon may need another assistant or more experienced help, or may wish to use a special retraction system to better expose an inaccessible liver injury. The anesthesia team needs a stock of blood products in the room and an autotransfuser. The circulating nurse needs competent help and may decide that the situation also merits a more experienced scrub nurse in the operative field. Mobilizing all these resources takes time, but greatly improves the chances of success in accomplishing the core mission of the operation because it converts hectic fumbling into an organized, coordinated team effort.
When facing a complex bleeding problem, the surgeon and team have only a narrow time window to achieve definitive hemostasis and conclude the operation with a live patient. Battle with an exsanguinating injury is surprisingly short. For example, when facing a high-grade liver injury, the surgeon has roughly 30–45 minutes and something in the order of 10–15 U of blood to achieve hemostasis and bail out of the abdomen before the patient develops severe hypothermia, coagulopathic bleeding, and an irreversible physiological insult. If more than one visceral compartment is open (e.g., abdomen and chest), the time window is even narrower because heat loss is accelerated. An effective operative support envelope may extend this window—but not by much, since even the best support cannot slow down heat loss from the open abdomen. However, a well-organized attack has a higher chance of making effective use of the available time window.
![]() Choosing an Operative Profile
Choosing an Operative Profile
After having achieved definitive control of hemorrhage, the surgeon must decide how to proceed with the rest of the operation or, in other words, choose an operative profile. The options are either definitive repair of the injuries with formal abdominal closure (the correct choice for the majority of cases) or a rapid bailout using damage control techniques and temporary abdominal closure (see Chapter 38). Contrary to popular notions, the decision to use damage control does not rely on identifying the “lethal triad” of hypothermia, coagulopathy, and acidosis simply because by then it may be too late. Experienced surgeons decide to bail out early and preemptively,16 sometimes within minutes of entering the abdomen. They typically consider three factors (Table 27-3).
TABLE 27-3 Factors to Consider in Choosing an Operative Profile
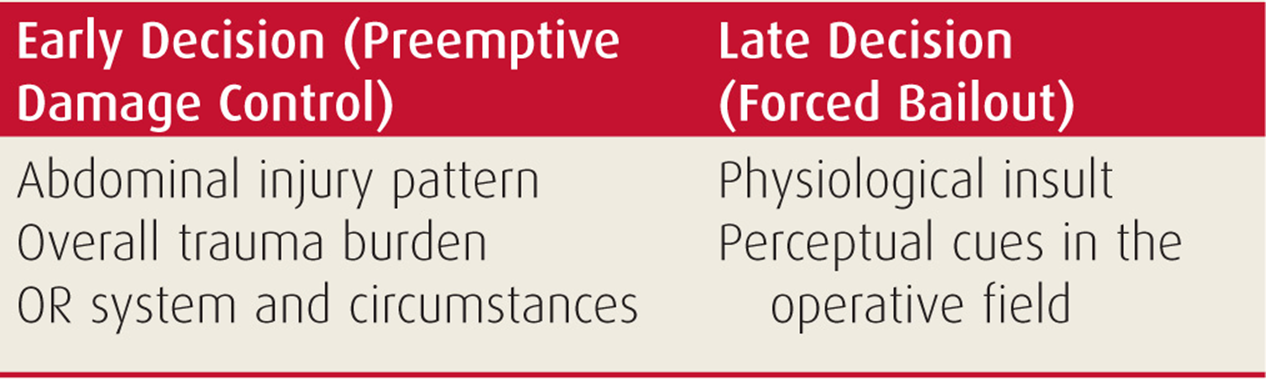
The pattern of injury often dictates a damage control approach. For example, a packed liver will have to be revisited and the combination of a major vascular trauma and hollow organ perforations will usually require a definitive repair for the former but a bailout solution for the latter.
The patient’s overall trauma burden is another consideration. Serious injury to anatomical regions outside the operative field (e.g., ongoing bleeding from a chest tube) or even the clinical suspicion of such trauma (e.g., unequal pupils) is a strong indication to abbreviate the laparotomy to address the other injuries as soon as possible.
A realistic assessment of the OR system and circumstances may also point to damage control as the safest option. Working in a suboptimal environment (such as a small rural facility or a military forward surgery team), having limited trauma experience, or an inadequate support envelope are all situations where a rapid bailout is often in the patient’s best interest. Damage control is rightfully known as the “great equalizer” of trauma surgery, allowing surgeons to successfully address complex life-threatening injuries in austere conditions or with limited trauma experience.
In gauging the magnitude of the physiological insult, the surgeon must focus on variables that reflect the cumulative insult over time rather than “snapshot” values of blood pressure or oxygen saturation. A realistic estimate of the overall blood volume lost, transfusion requirements, and the duration of hypotension are such cumulative variables. The degree of metabolic acidosis (pH <7.30) and hypothermia (<35°C) are useful but late indicators, since the surgeon should ideally make the decision to bail out well before they occur.
Occasionally, the surgeon proceeds with a definitive repair profile but recognizes at some point that the circumstances have changed and there is a need to abbreviate the operation. Subtle perceptual cues to make this switch are signs such as edema of the bowel wall, tissues cold to the touch, noncompliant and grossly edematous abdominal wall, and diffuse oozing from the surgical incision. Such a “forced” bailout actually means that the surgeon is trying to correct a previous error in judgment and should have bailed out long ago.
![]() Temporary Abdominal Closure
Temporary Abdominal Closure
The goals of temporary abdominal closure at the end of a damage control laparotomy are to provide rapid atraumatic containment of the abdominal viscera, protect the bowel, and spare the fascia for the definitive closure. Effective temporary closure also reduces the catabolic effects of the open abdominal wound. The specific technique chosen is largely a matter of personal preference and is heavily influenced by institutional traditions and practices.
The vacuum pack, introduced by Barker et al. in 1995, has revolutionized abdominal closure not only because it allowed rapid sutureless containment17 but also because it inadvertently modified the healing process of the abdominal wound.
The vacuum pack is a sandwich of three layers. The visceral block is wrapped with a polyethylene sheet that is carefully tucked between the viscera and the abdominal wall all the way down into the paracolic gutters. The sheet is covered with a surgical towel to absorb peritoneal fluid. Two suction drains are placed on the surgical towel and brought out through the lower or upper corner of the abdominal wound. Their role is to evacuate the peritoneal fluid in a controlled fashion. The third and final layer is a large polyester adhesive drape that seals the sandwich.
The unexpected beneficial effect of the vacuum pack on the wound healing process of the open abdomen is brought about by the innermost layer of the sandwich, since the polyethylene sheet serves as a physical barrier between the visceral block and the abdominal wall. It prevents adhesion formation between the visceral block and the inner aspect of the abdominal wall, so that they granulate separately. As a result, the lateral mobility of the abdominal wall is preserved for much longer than the 10–14 days it takes the granulating bowel to adhere to the inner aspect of the wall with other temporary closure techniques. This, in turn, extends the window of opportunity to achieve delayed primary closure of the open abdomen to as much as a month after the initial operation.18
The popularization and commercial success of vacuum-assisted wound management technology in the last decade rapidly led to its application to the open abdomen. Replacing the middle layer of the vacuum pack (the surgical towel that absorbs fluids) with a sponge has been advertised as facilitating wound healing and preventing lateral retraction of the fascia. While these claims have never been subject to a scientific comparison with the original vacuum pack, there is a growing number of reports that associate the use of vacuum-assisted wound management in the open abdomen with the formation of exposed (enteroatmospheric) fistulae,19 especially when there is a suture line in the bowel or if the gut is otherwise compromised. These have led to an FDA alert in November 2009 cautioning against using vacuum-assisted management systems in the open abdomen.
RELAPAROTOMY
![]() Urgent Reoperation for Bleeding
Urgent Reoperation for Bleeding
Reexploration of the injured abdomen for bleeding is a dramatic operation with high mortality: roughly one in four operations will end with a dead patient.20 The decision to operate may be difficult in critically injured patients since during the first few hours in the SICU they are often hemodynamically unstable and coagulopathic so a certain amount of postoperative bleeding and equilibration is to be expected. The surgeon must decide not whether the patient is bleeding but whether the amount or rate of hemorrhage exceeds what is expected in this specific clinical situation, representing a likely surgical bleeder.
Most patients with ongoing postoperative bleeding are hemodynamically unstable and some of them also develop intra-abdominal hypertension and occasionally a full-blown abdominal compartment syndrome. The operative profile is therefore that of a crash laparotomy. Nowhere is the quality of the surgeon’s support envelope more important than during this high-stakes procedure where the patient’s physiology, already compromised by the original injury and laparotomy, is extremely shaky from the start.
Reoperation for bleeding follows the same generic sequence described in the first part of this chapter. The only goal of the procedure is to achieve hemostasis and decompress the abdomen. All else is irrelevant. The patient’s only hope is a rapid focused effort to identify and control a surgical bleeder.
The abdomen is entered through the previous incision (or open wound). After rapid evisceration and evacuation of blood and clots, the surgeon proceeds to look for the source of bleeding. In roughly half the cases, the source of bleeding is incomplete (or failed) hemostasis at a site that was addressed during the original operation.20 This is most likely to occur in packed areas such as the liver and, less frequently, the retroperitoneum. Other sources of hemorrhage are missed injuries or iatrogenic damage (typically to the spleen). In reexploring the injured abdomen the surgeon must therefore be aware that the source of rebleeding is most likely found in the immediate vicinity of a previous repair or hemostatic effort.
Not uncommonly, the surgeon faces a peritoneal cavity filled with blood and clots but cannot identify a discrete bleeder anywhere. The best option in this situation is to perform a full systematic exploration with emphasis on the location of previously addressed injuries, until the surgeon is satisfied that there is no active bleeder to address. The peritoneal cavity is irrigated with warm saline and a decision made whether to use temporary abdominal closure with a vacuum pack or to reclose the abdomen.
Nothing is more frustrating than to find only diffuse oozing in the abdomen of a patient in extremis with no surgical bleeder. This situation is not amenable to surgical control and the reoperation only worsens the patient’s already precarious physiology. When encountered, diffuse coagulopathic oozing should be recognized early for what it is and the operation rapidly terminated. However, even with rapid packing and bailout, the survival after such procedures is very low.20
![]() Urgent Laparotomy for Abdominal Infection
Urgent Laparotomy for Abdominal Infection
Abdominal infection after laparotomy for trauma may be either controlled or uncontrolled. The former is more common and consists of single or multiple localized abscesses that can be drained percutaneously under CT guidance. Uncontrolled infection is much less frequent but more ominous, with an active ongoing source of infection that cannot simply be drained and therefore requires surgical control.21 Diffuse peritonitis from a duodenal missed injury, an exposed (enteroatmospheric) fistula draining through an open abdomen, and a septic dehiscence of the laparotomy wound due to a leaking colonic staple line are all examples of uncontrolled abdominal infection.
Severe uncontrolled abdominal infection results in an extremely hostile peritoneal cavity: the bowel is edematous and very friable, the mesentery is foreshortened, and the abdominal wall is swollen and noncompliant. Granulation tissue and dense vascular adhesions fuse adjacent bowel loops and glue the visceral block to the abdominal wall, impeding access to the leak.
The only goal of relaparotomy for abdominal infection is to achieve source control. The peritoneal cavity is typically entered through the previous incision, all purulent and necrotic material is removed, and the source of ongoing bacterial contamination is identified. Suture or staple lines created during the original operation are carefully inspected for leaks, and if none are found a diligent search is then undertaken for missed injuries along the presumed trajectory of the injuring missile and at locations prone to hide missed injuries (as described above).
The technical options to achieve source control are given in Table 27-4. The simplest way is to resect or exteriorize the leaking segment.22 As a general rule, creating new unprotected suture lines in a hostile and severely inflamed peritoneal cavity is a hazardous undertaking even when dealing with “forgiving” viscera such as the stomach or the small bowel. When considering how to control the leak, the surgeon must take into account not only the local conditions in the abdomen but also the patient’s overall physiological reserves and ability to survive another leak. An example of this dilemma is the decision whether to perform a pyloric exclusion for a duodenal leak that is addressed in a delayed fashion in a severely inflamed abdomen. The benefits of the pyloric exclusion (as opposed to creating a tube duodenostomy and accepting a controlled side fistula) must be weighed against the added risk of another suture line failure in the hostile peritoneal cavity.
TABLE 27-4 Technical Options for Source Control during Relaparotomy for Uncontrolled Abdominal Infection

When relaparotomy is delayed and the leaking bowel segment cannot be mobilized, exteriorized, or safely resected, the surgeon may still be able to achieve source control by proximal diversion, which can be lifesaving.22 Tube drainage alone typically does not provide effective source control since the leak continues around it, and the tube itself serves only to bore down and enlarge the hole in the gut. Vacuum-assisted wound management is an important adjunct that helps the surgeon achieve source control of exposed fistulae by continuous suction of the intestinal content. However, this modality must be used with caution in the presence of compromised bowel or intact suture lines in the gut as described above.23
Relaparotomy for uncontrolled abdominal infection usually commits the surgeon to a series of planned reoperations to wash out the abdomen, assess progress, optimize source control, and tailor the technical solutions (such as the size and shape of the vacuum-assisted dressing) to the granulating and gradually healing open abdominal wound.
![]() Planned Reoperation
Planned Reoperation
Planned relaparotomy after an initial damage control procedure is performed on a patient whose physiological envelope has been restored by rewarming and resuscitation. The patient often remains critically ill but is no longer facing imminent death from exsanguinations or irreversible shock. The operation is typically undertaken within 24–48 hours of the initial operation. The precise timing depends on clinical and logistical considerations. For example, the presence of a “closed loop” of bowel that has been stapled off at both ends is an argument for an earlier reoperation, while a high-grade liver injury that has been successfully packed will rarely be addressed before 48 hours, to allow the hemostatic clots time to stabilize and mature.21
Planned relaparotomy begins with meticulous exploration of the abdominal cavity. Significant injuries that have been overlooked or only partially defined in the heat of the original battle must now be identified and addressed. The next step is reconstruction. Major vascular reconstruction (e.g., replacing a temporary shunt in the common iliac artery with a synthetic graft) typically precedes GI tract repairs.
The guiding principle for GI reconstructions during reoperation is to be conservative and avoid risky suture lines. Reconstructions that could have been considered safe during the original operation (such as a colocolostomy or pancreaticojejunostomy) may no longer be acceptable after 48 hours of an open abdomen in an edematous patient who will not tolerate a leak.
The removal of packs and other temporary hemostatic devices (such as balloon catheter tamponade) is always the last step before abdominal closure, since there is always the risk that unpacking may result in recurrent bleeding and require repacking and another bailout. Packing should be removed after soaking the packs and the surrounding organs with saline and allowing time for the saline to soak through. Topical hemostatics and other adjuncts (such as an argon beam coagulator) should be ready to be applied to the unpacked areas, and a full set of vascular instruments should be available in case a major venous injury is uncovered under the packs.
A safe technique for pack removal is to use a jet of water from a large bulb syringe directed at the interface between the packs and the tissue. The packs are then slowly removed. When bleeding is encountered, the surgeon stops unpacking to achieve hemostasis. The most common problem is slow persistent oozing from a raw surface, which is typically controlled by a combination of electrocoagulation, local hemostatic agents, and the application of gentle firm pressure. A key principle during any relaparotomy for trauma is that the surgeon must be mentally prepared to accept failure.21 When repeated attempts to achieve hemostasis fail and there is no indication for a formal vascular repair, admitting failure and rapidly repacking the area is a safer option than insisting on a definitive hemostatic solution.
Formal abdominal closure, if feasible, is the last step in the reoperative sequence. Before closure, the surgeon should consider creating a route for early enteral feeding either via a nasojejunal tube passed by the anesthesiologist and guided by the surgeon past the pylorus or through a feeding jejunostomy.
Simple delayed primary closure of the open abdomen may not be possible if the abdominal wall has retracted laterally, creating a large gap. In these situations, the surgeon’s technical options depend on whether the peritoneal space has been preserved, as explained above (Table 27-5).
TABLE 27-5 Technical Options for Delayed Abdominal Closure at Reoperation
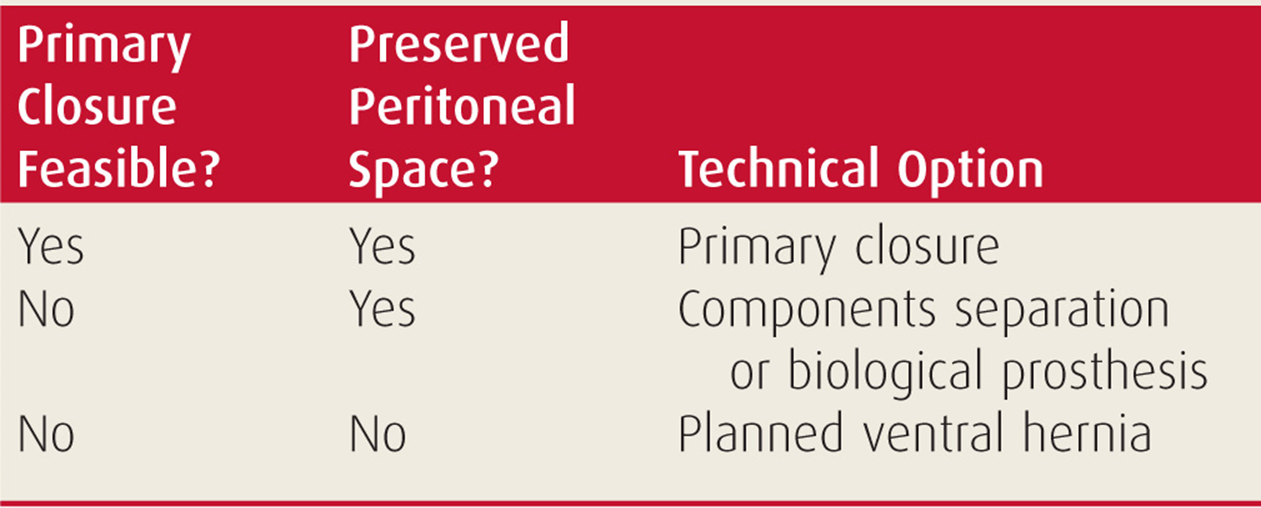
An obliterated peritoneal space means that the abdominal wound has granulated and healed by secondary intention. The wound edges therefore lack lateral-to-medial mobility and the fascia can neither be closed nor bridged. The only option is to cover the wound with a skin graft, accepting a giant planned ventral hernia that will be repaired many months later.24
With a preserved peritoneal space, the surgeon has two technical options to achieve delayed primary closure. The components separation technique25 achieves better long-term results, whereas the use of a biological prosthesis is technically more expedient but at the price of very high long-term failure rates.
BEDSIDE LAPAROTOMY
Bedside laparotomy in the SICU was originally introduced as a practical solution for patients who needed a relaparotomy but were simply too unstable for even a short trip to the OR.21 The classic examples are the patient with full-blown abdominal compartment syndrome in need of immediate decompression,26 or the crashing patient with postoperative bleeding in the open abdomen. For these and other patients on very high ventilator settings and high-dose pressor support, the journey to the OR is much more perilous than the reoperation itself.
At the other end of the severity spectrum is a hemodynamically stable (yet still critically ill) patient who needs a “routine washout” of the open abdomen but whose procedure is repeatedly postponed because of OR availability issues. As bedside laparotomy has gained popularity, the indications have expanded and also include planned and unplanned reoperation for abdominal infection (both controlled and uncontrolled) to drain infected fluid collections and optimize control of exposed fistulae.
The great advantage of a bedside laparotomy is expediency. Since the abdomen is usually open, access is not an issue and the suboptimal sterility of the operative field is not a concern. The surgeon must, however, bear in mind the obvious limitations of a bedside laparotomy: working conditions (such as lighting and the physical distance of the surgeon from the abdominal wound) are inferior to those in the OR, the surgeon’s support envelope is weaker, and the scope of the procedures that can be safely undertaken at the bedside is necessarily limited.
Bedside laparotomy in the SICU cannot be improvised on a whim. It requires planning, a standardized institutional protocol, and clear understanding of what can or cannot be achieved at the bedside.27 Most importantly, the procedure must be ingrained in the institutional culture as a routine feature of SICU care rather than a rare dramatic occurrence. In other words, a large number of “routine washouts” performed regularly in the SICU are needed to develop and maintain the capability for an occasional dramatic save of a patient with intra-abdominal hemorrhage.
Preparation for a bedside laparotomy consists of creating a sterile surgical perimeter on the patient’s bed as well as deploying a simplified support envelope around it. A basic laparotomy instrument tray and all necessary sterile adjuncts are brought in from the OR suite, including warm saline and a temporary abdominal closure device. A team member is assigned to continuously monitor the patient’s vital signs and lines throughout the procedure. In some institutions, OR nursing staff participates in bedside laparotomies as both scrub nurse and circulating nurse (Fig. 27-9). If this is not feasible, members of the SICU or trauma teams are assigned to fulfill these roles.
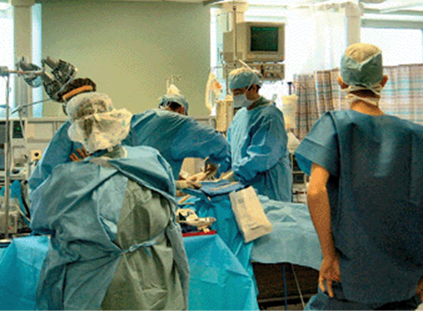
FIGURE 27-9 Bedside laparotomy in the Surgical Intensive Care Unit. A scrub nurse and circulating nurse from the OR staff are preparing a sterile instrument tray (left) while a surgical resident is assigned to monitor the patient’s vital signs (right). Bedside laparotomy should be a protocol-driven routine activity that is ingrained in the culture of the trauma service and SICU.
The sequence of a bedside laparotomy is tailored to the specific clinical circumstances. In a routine washout, the surgeon removes the temporary abdominal closure device, performs a limited exploration of the peritoneal cavity, manually separates the granulating visceral block from the overlaying abdominal wall to preserve the peritoneal space, and ascertains that no undrained sources of sepsis are left behind. Copious irrigation of the peritoneal cavity with warm saline is then followed by rapid temporary reclosure with a new temporary device.
An urgent bedside laparotomy for bleeding follows the same sequence as a similar procedure in the OR. However, the safe course of action is to only evacuate the blood and rapidly pack the abdomen to achieve temporary hemostasis and then transfer the patient to the OR for a formal exploration and definitive hemostasis. Unless the source of bleeding is trivial and immediately accessible, definitive control of surgical bleeding should be done in the optimal work environment of the OR.
In the patient with uncontrolled abdominal infection, bedside laparotomy can be used to search for an obvious source (such as a new hole in the gut or breakdown of an intestinal suture line), to ascertain bowel and stoma viability, and to optimize drainage by repositioning old drains or placing new ones. As a general rule, bowel resection and stoma formation should be done in the OR.
ROLE OF LAPAROSCOPY
Following a brief period of initial enthusiasm, trauma surgeons were quick to discard laparoscopy as a useful modality in abdominal trauma roughly a decade ago. This was based on several retrospective studies from the early days of laparoscopy in general surgery that led to the perception that laparoscopy is not sensitive enough because it misses significant injuries, especially in patients with penetrating abdominal injuries.28 The inability to “run the bowel” properly was also often cited as a shortcoming of the technology.
As a result, the current role of diagnostic laparoscopy in trauma is limited to ruling out diaphragmatic penetration in patients with asymptomatic left-sided thoracoabdominal stab wounds.29 Insufflating the abdomen and inserting a camera through a single umbilical port allows the surgeon to visualize the entire left hemidiaphragm and thus rule out a diaphragmatic penetration. If an injury is found, the procedure is typically converted to an open laparotomy since laparoscopy is considered unreliable in ruling out intra-abdominal visceral damage. Liberal use of laparoscopy led to the discovery of an occult diaphragmatic injury in roughly one in four of these patients.
Another useful application of laparoscopy in trauma is as a useful adjunct to the conservative management of hepatic trauma.30 In some of these patients, bile leak combined with perihepatic hematoma leads to a septic clinical picture of smoldering bile peritonitis with a prominent systemic inflammatory response. In these situations, laparoscopic washout of the right upper quadrant is an elegant minimally invasive approach to the problem. Using the port arrangement of laparoscopic cholecystectomy, the surgeon can wash out the bile from the right upper quadrant, use the blunt dissector and suction irrigation to remove superficial fibrinous rinds on the liver capsule or infected clots around the liver, and place a suction drain in Morisson’s pouch under direct vision.
The current very restricted role of laparoscopy in trauma does not do justice to the great potential of the technology. The early studies were done in an era of first-generation equipment and basic laparoscopic skills. Both have dramatically improved in the past decade. The technique for a detailed laparoscopic exploration of the abdominal cavity, including fully “running the bowel” using a four-port approach, has been published31 but has not caught on in the trauma community. Of the two modes of laparotomy in trauma described in Section “Introduction,” the procedure in a stable patient with a hole in the gut and limited or no bleeding is clearly suitable for at least a renewed evaluation of the role of laparoscopy. In an era when an increasing number of perforated gastric and duodenal ulcers are repaired laparoscopically, there is no good reason not to revisit the role of this technology in selected patients with abdominal trauma.
REFERENCES
1. Hirshberg A, Mattox KL. The crash laparotomy. In: Hirshberg A, Mattox KL, eds. Top Knife: The Art and Craft of Trauma Surgery. Shrewsbury: tfm Publishing; 2005:53–70:chap 4.
2. Como JJ, Bokhari F, Chiu WC, et al. Practice management guidelines for selective nonoperative management of penetrating abdominal trauma. J Trauma. 2010;68:721–733.
3. Hirshberg A, Wall MJ Jr, Mattox KL. Bullet trajectory predicts the need for “damage control”—an artificial neural network model. J Trauma. 2002;52:852–858.
4. Mattox KL, Hirshberg A. Access, control and repair techniques. In: Rich N, Mattox KL, Hirshberg A, eds. Vascular Trauma. 2nd ed. Philadelphia, PA: Elsevier Saunders; 2004:137–164:chap 7.
5. Rotondo MF, Zonies DH. The damage control sequence and underlying logic. Surg Clin North Am. 1997;77:761–777.
6. Hansen KS, Uggen PE, Brattebo G, et al. Team-oriented training for damage control surgery in rural trauma: a new paradigm. J Trauma. 2008;64:949–953.
7. Awad SS, Fagan SP, Bellows C, et al. Bridging the communication gap in the operating room with medical team training. Am J Surg. 2005;190: 770–774.
8. Chassin JL. Operative technique for midline incision. In: Chassin JL, ed. Operative Strategy in General Surgery: An Expositive Atlas. New York, NY: Springer-Verlag; 1980:40–43.
9. Veith FJ, Gupta S, Daly V. Technique for occluding the supraceliac aorta through the abdomen. Surg Gynecol Obstet. 1980;151:426–428.
10. Hirshberg A, Wall MJ Jr, Allen MK, et al. Causes and patterns of missed injuries in trauma. Am J Surg. 1994;168:299–303.
11. Mattox KL, McCollum WB, Beall AC, et al. Management of penetrating injuries of the suprarenal aorta. J Trauma. 1975;15:808–813.
12. Reilly LM, Ramos TK, Murray SP, et al. Optimal exposure of the proximal abdominal aorta: a critical appraisal of transabdominal medical visceral rotation. J Vasc Surg. 1994;19:375–390.
13. Buscaglia LC, Blaisdell FW, Lim RC. Penetrating abdominal vascular injuries. Arch Surg. 1969;99:764–769.
14. Lauter DM. Midline laparotomy and right retroperitoneal dissection is an alternative exposure for routine aortic surgery. Am J Surg. 2003;186:20–22.
15. Cattell RB, Braasch JW. A technique for the exposure of the third and fourth portions of the duodenum. Surg Gynecol Obstet. 1960;111: 378–379.
16. Hoey BA, Schwab CW. Damage control surgery. Scand J Surg. 2002;91: 92–103.
17. Barker DE, Kaufman HJ, Smith LA, et al. Vacuum pack technique of temporary abdominal closure: a 7-year experience with 112 patients. J Trauma. 2000;48:201–206.
18. Miller PR, Thompson JT, Faler BJ, et al. Late fascial closure in lieu of ventral hernia: the next step in open abdomen management. J Trauma. 2002;53:843–849.
19. Fischer JE. A cautionary note: the use of vacuum-assisted closure systems in the treatment of gastrointestinal cutaneous fistula may be associated with higher mortality from subsequent fistula development. Am J Surg. 2008;196:1–2.
20. Hirshberg A, Wall MJ Jr, Ramchandani MK, et al. Reoperation for bleeding in trauma. Arch Surg. 1993;128:1163–1167.
21. Hirshberg A, Stein M, Adar R. Reoperation: planned and unplanned. Surg Clin North Am. 1997;77:897–907.
22. Schein M, Decker GA. Gastrointestinal fistulas associated with large abdominal wall defects: experience with 43 patients. Br J Surg. 1990;77: 97–100.
23. Schecter WP, Hirshberg A, Chang DS, et al. Enteric fistulas: principles of management. J Am Coll Surg. 2009;209:484–489.
24. Jerrigan TW, Fabian TC, Croce MA, et al. Staged management of giant abdominal wall defects: acute and long-term results. Ann Surg. 2003; 238:349–357.
25. Shestak KC, Edington HJ, Johnson RR. The separation of anatomic components technique for the reconstruction of massive midline abdominal wall defects: anatomy, surgical technique, applications, and limitations revisited. Plast Reconstr Surg. 2000;105:731–738.
26. Morris JA Jr, Eddy VA, Blinman TA, et al. The staged celiotomy for trauma. Issues in unpacking and reconstruction. Ann Surg. 1993;217: 576–584.
27. Diaz JJ, Mejia V, Subhawong AP, et al. Protocol for bedside laparotomy in trauma and emergency general surgery: a low return to the operating room. Am Surg. 2005;71:986–991.
28. Villavicencio RT, Aucar JA. Analysis of laparoscopy in trauma. J Am Coll Surg. 1999;189:11–20.
29. Murray JA, Demetriades D, Asensio J, et al. Occult injuries to the diaphragm: prospective evaluation of laparoscopy in penetrating injuries to the left lower chest. J Am Coll Surg. 1998;187:626–630.
30. Franklin GA, Richardson JD, Brown AL, et al. Prevention of bile peritonitis by laparoscopic evacuation and lavage after nonoperative treatment of liver injuries. Am Surg. 2007;73:611–616.
31. Gorecki PJ, Cottam D, Angus LD, et al. Diagnostic and therapeutic laparoscopy for trauma: a technique for safe and systematic exploration. Surg Laparosc Endosc Percutan Tech. 2002;12:195–198.